Abstract
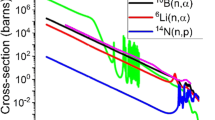
Highly efficient neutron detectors are critical in many sectors, including national security1,2, medicine3, crystallography4 and astronomy5. The main neutron detection technologies currently used involve 3He-gas-filled proportional counters6 and light scintillators7 for thermalized neutrons. Semiconductors could provide the next generation of neutron detectors because their advantages could make them competitive with or superior to existing detectors. In particular, solids with a high concentration of high-neutron-capture nuclides (such as 6Li, 10B) could be used to develop smaller detectors with high intrinsic efficiencies. However, no promising materials have been reported so far for the construction of direct-conversion semiconductor detectors. Here we report on the semiconductor LiInP2Se6 and demonstrate its potential as a candidate material for the direct detection of thermal neutrons at room temperature. This compound has a good thermal-neutron-capture cross-section, a suitable bandgap (2.06 electronvolts) and a favourable electronic band structure for efficient electron charge transport. We used α particles from an 241Am source as a proxy for the neutron-capture reaction and determined that the compact two-dimensional (2D) LiInP2Se6 detectors resolved the full-energy peak with an energy resolution of 13.9 per cent. Direct neutron detection from a moderated Pu–Be source was achieved using 6Li-enriched (95 per cent) LiInP2Se6 detectors with full-peak resolution. We anticipate that these results will spark interest in this field and enable the replacement of 3He counters by semiconductor-based neutron detectors.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
Change history
29 February 2020
A Correction to this paper has been published: https://doi.org/10.1038/s41586-020-2065-7
References
Detection of Radioactive Materials at Borders (International Atomic Energy Agency, 2002).
Runkle, R. C., Bernstein, A. & Vanier, P. E. Securing special nuclear material: recent advances in neutron detection and their role in nonproliferation. J. Appl. Phys. 108, 111101 (2010).
Barth, R. F., Coderre, J. A., Vicente, M. G. H. & Blue, T. E. Boron neutron capture therapy of cancer: current status and future prospects. Clin. Cancer Res. 11, 3987–4002 (2005).
Wollan, E. O. & Shull, C. G. The diffraction of neutrons by crystalline powders. Phys. Rev. 73, 830–841 (1948).
Ruffolo, D. et al. Monitoring short-term cosmic-ray spectral variations using neutron monitor time-delay measurements. Astrophys. J. 817, 38–50 (2016).
Kouzes, R. T. The 3He Supply Problem. Report No. PNNL-18388 (Pacific Northwest National Laboratory, 2009).
Breukers, R. D., Bartle, C. M. & Edgar, A. Transparent lithium loaded plastic scintillators for thermal neutron detection. Nucl. Instrum. Methods Phys. Res. A 701, 58–61 (2013).
Dunning, J. R., Pegram, G. B., Fink, G. A. & Mitchell, D. P. Interaction of neutrons with matter. Phys. Rev. 48, 265–280 (1935).
Knoll, G. F. in Radiation Detection and Measurement 505–535 (John Wiley & Sons, 2000).
Ochs, T. R. et al. Present status of the microstructured semiconductor neutron detector-based direct helium-3 replacement. IEEE Trans. Nucl. Sci. 64, 1846–1850 (2017).
Simpson, A. P., Jones, S., Clapham, M. J. & Mcelhaney, S. A. A review of neutron detection technology alternatives to helium-3 for safeguards applications. In INMM 52nd Annual Meeting Vol. 8 (2011).
Caruso, A. N. The physics of solid-state neutron detector materials and geometries. J. Phys. Condens. Matter 22, 443201 (2010).
Shultis, J. K. & Mcgregor, D. S. Design and performance considerations for perforated semiconductor thermal-neutron detectors. Nucl. Instrum. Methods Phys. Res. A 606, 608–636 (2009).
Tupitsyn, E. et al. Lithium containing chalcogenide single crystals for neutron detection. J. Cryst. Growth 393, 23–27 (2014).
Guo, L. et al. Stoichiometric effects on the photoelectric properties of LiInSe2 crystals for neutron detection. Cryst. Growth Des. 18, 2864–2870 (2018).
Hamm, D. S. et al. Semiconducting lithium indium diselenide: charge-carrier properties and the impacts of high flux thermal neutron irradiation. Appl. Phys. Lett. 112, 242104 (2018).
Maity, A., Grenadier, S. J., Li, J., Lin, J. Y. & Jiang, H. X. High-efficiency and high-sensitivity thermal neutron detectors based on hexagonal BN epilayers. In Proc. SPIE Hard X-Ray, Gamma-Ray, and Neutron Detector Physics XIX, 10392 (2017).
Pfeiff, R. & Kniep, R. Quaternary selenodiphosphates(IV): MIMIII[P2Se6], (MI = Cu, Ag; MIII = Cr, Al, Ga. J. Alloys Compd. 186, 111–133 (1992).
Seidlmayer, S. Strukturchemische Untersuchungen an Hexachalkogenohypodiphosphaten und Verwandten Verbindungen. PhD Thesis, University of Regensburg (2009).
Wang, F. et al. New frontiers on van der Waals layered metal phosphorous trichalcogenides. Adv. Funct. Mater. 28, 1–24 (2018).
Binnewies, M., Glaum, R., Schmidt, M. & Schmidt, P. in Chemical Vapor Transport Reactions 360–369 (De Gruyter, 2012).
Knoll, G. F. Radiation Detection and Measurement 757–775 (John Wiley & Sons, 2000).
Feher, F. Handbuch der Präparativen Anorganischen Chemie (F. Enke, 1954).
Aitken, J. A., Cowen, J. A. & Kanatzidis, M. G. Metamagnetic transition in EuSe2: a new, metastable binary rare-earth polychalcogenide. Chem. Mater. 10, 3928–3935 (1998).
Sheldrick, G. M. SHELXT – integrated space-group and crystal- structure determination. Acta Crystallogr. A 71, 3–8 (2014).
Sheldrick, G. M. Crystal structure refinement with SHELXL. Acta Crystallogr. C 71, 3–8 (2015).
Dolomanov, O. V., Bourhis, L. J., Gildea, R., Howard, J. A. K. & Puschmann, H. OLEX2: a complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 42, 339–341 (2009).
Momma, K. & Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 44, 1272–1276 (2011).
Klimeš, J., Bowler, D. R. & Michaelides, A. Chemical accuracy for the van der Waals density functional. J. Phys. Condens. Matter 22, 022201 (2010).
Setyawan, W. & Curtarolo, S. High-throughput electronic band structure calculations: challenges and tools. Comput. Mater. Sci. 49, 299–312 (2010).
Larsen, A. et al. The atomic simulation environment - a Python library for working with atoms. J. Phys. Condens. Matter 29, 273002 (2017).
Ong, S. P. et al. Python Materials Genomics (pymatgen): a robust, open-source python library for materials analysis. Comput. Mater. Sci. 68, 314–319 (2013).
Fonari, A. & Sutton, C. Effective mass calculator https://github.com/afonari/emc (2012).
Owens, A. & Peacock, A. Compound semiconductor radiation detectors. Nucl. Instrum. Methods Phys. Res. A 531, 18–37 (2004).
Binnewies, M., Glaum, R., Schmidt, M. & Schmidt, P. in Chemical Vapor Transport Reactions 452–457 (De Gruyter, 2012).
Binnewies, M., Glaum, R., Schmidt, M. & Schmidt, P. in Chemical Vapor Transport Reactions 165–166 (De Gruyter, 2012).
McCall, K. M., Stoumpos, C. C., Kostina, S. S., Kanatzidis, M. G. & Wessels, B. W. Strong electron–phonon coupling and self-trapped excitons in the defect halide perovskites A3M2I9(A = Cs, Rb; M = Bi, Sb). Chem. Mater. 29, 4129–4145 (2017).
Schmidt, T., Lischka, K. & Zulehner, W. Excitation-power dependence of the near-band-edge photoluminescence of semiconductors. Phys. Rev. B 45, 8989–8994 (1992).
Pankove, J. I. Optical processes in semiconductors (Dover, 1971).
Pelant, I. & Valenta, J. Luminescence Spectroscopy of Semiconductors (Oxford Univ. Press, 2012).
Veale, M. C. et al. X-ray spectroscopy and charge transport properties of CdZnTe grown by the vertical Bridgman method. Nucl. Instrum. Methods Phys. Res. A 576, 90–94 (2007).
Watanabe, S. et al. Stacked CdTe gamma-ray detector and its application to a range finder. Nucl. Instrum. Methods Phys. Res. A 505, 118–121 (2003).
Acknowledgements
The exploratory synthesis and materials characterization work was supported by the National Science Foundation through grant DMR-1708254. The device fabrication and neutron response measurements were supported by Laboratory Directed Research and Development (LDRD) funding from Argonne National Laboratory, provided by the Director, Office of Science of the US Department of Energy under contract number DE-AC02-06CH11357. PL measurements were supported by the Murphy Fellowship from Northwestern University. This work made use of the SPID and EPIC facilities of Northwestern University’s NUANCE Center, which has received support from the Soft and Hybrid Nanotechnology Experimental (SHyNE) Resource (NSF ECCS-1542205); the MRSEC programme (NSF DMR-1720139) at the Materials Research Center; the International Institute for Nanotechnology (IIN); the Keck Foundation; and the State of Illinois, through IIN. This work used the Northwestern University’s Keck Biophysics Facility, which is funded by a Cancer Center Support Grant (NCI CA060553). This work made use of IMSERC at Northwestern University, which has received support from the Soft and Hybrid Nanotechnology Experimental (SHyNE) Resource (NSF ECCS-1542205), the State of Illinois and IIN.
Author information
Authors and Affiliations
Contributions
The idea for this project was conceived by M.G.K., D.Y.C., D.G.C. and Y.H. The manuscript was written by D.G.C., Y.H. and M.G.K. with input from all authors. D.G.C. developed the synthetic and crystal growth methods. The crystal structure and physical properties, excluding the optical properties, were measured by D.G.C. The band structure and effective-mass calculations were conducted by G.T. Optical transmission measurements were performed by K.M.M. and Z.L. K.M.M. measured the variable-temperature and power-dependent PL and the Raman spectra. Y.H. and D.G.C. fabricated the devices and performed pulse-height spectrometry of the 241Am and 57Co sources. Neutron measurements were performed by R.O.P., Y.H., D.G.C., D.Y.C. and P.M.D.L. M.G.K. and B.W.W. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare the U.S. patent application 62/701,144.
Additional information
Peer review information Nature thanks Paul Sellin and Mariya Zhuravleva for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 X-ray powder diffraction patterns of LiInP2Se6 prepared under different conditions.
a, b, Experimental versus simulated powder diffraction pattern of LiInP2Se6 scaled to the largest peak (a) and scaled by a factor of 20 (b). The simulated pattern has a March–Dollase parameter of 0.3. The X-ray powder diffraction pattern of LiInP2Se6 in a shows a considerable preference to orientation along \(\langle 0\,0\,1\rangle \), that is, the peaks with the largest intensity correspond to the (0 0 2), (0 0 4) and (0 0 8) planes. This preferred orientation stems from the layered nature of this compound, which causes the layers to lie parallel to the sample holder. The simulated pattern with a March–Dollase parameter of 0.3 was the reference pattern used to compare with the experimental pattern, and accurately accounts for the sample’s preferred orientation. When the patterns are zoomed-in closer to the baseline, the reflections with contributions from the h and k directions can be seen and match well with the simulated pattern (b). c, Experimental versus simulated powder diffraction pattern of LiInP2Se6 obtained using a stoichiometric amount of reagents. The unknown secondary phases are marked by asterisks. d, Sink side of the reaction tube used for CVT with no iodine charged into the tube. e, Sink side of the reaction tube used for CVT with iodine as the transporting agent. This reaction did not employ an initial reverse transport step. f, Experimental versus simulated powder diffraction pattern of LiInP2Se6 grown using CVT. The simulated pattern has a March–Dollase coefficient of 0.3. g, Experimental powder diffraction pattern of bulk LiInP2Se6 before (red) and after (black) DTA up to 760 °C. h, Experimental diffraction pattern of LiInP2Se6 grown by CVT before (red) and after (black) DTA up to 760 °C.
Extended Data Fig. 2 Thermal analysis, morphology and compositional analysis for LiInP2Se6.
a, b, DTA measurement of bulk-synthesized (a) and CVT-grown (b) LiInP2Se6 up to 760 °C. The vertical axis is a measure of the voltage difference of the thermocouples on the sample and the alumina standard as the temperature is varied; thus, the spikes correspond to a transfer of heat from the sample to the environment and vice versa. The melting point of LiInP2Se6 is considerably lower than that of LiInSe2 (~915 °C)14, which yields several practical benefits, including the reduction of thermally induced defects and improved stoichiometry from reduced evaporative losses of the volatile Li, P and Se. c, d, Scanning electron microscopy images of bulk LiInP2Se6 in the basal plane (c) and a cross-section of the layers (d). The lamellar crystal morphology of the bulk ingot can be clearly seen in the images. A large flake with a smooth surface was selected to examine the surface of the basal plane. c shows a typical image of the smooth basal plane. The composition obtained from EDS from both orientations yielded the semiquantitative formula In1.1P2.0 Se6.0, which is consistent with the expected ratio of 1:2:6 determined from the crystal structure. The lithium content could not be determined from EDS because of the insensitivity of EDS to lithium. e, EDS mapping of the surface of LiInP2Se6 grown using CVT without reverse transport. The growth front of the layer appears rich in iodine.
Extended Data Fig. 3 PL characterization of LiInP2Se6.
a, Dependence of the PL spectra of LiInP2Se6 at 12.5 K on laser power. b, Power-law fits of the integrated PL intensity versus power for each peak. c, Temperature dependence of the PL spectra of LiInP2Se6 at 2 mW. d, PL peak energy versus temperature. e, Arrhenius plots of integrated PL intensity versus inverse temperature. The solid lines are the least-squares best fit for each peak according to Eq. (1). f, Tabulated PL peak properties for LiInP2Se6 at 12.5 K. See Methods for further discussion.
Extended Data Fig. 4 Electrical properties and spectroscopic response of LiInP2Se6 device.
a, Current–voltage plot (from –100 V to 100 V) of a pristine LiInP2Se6 device showing linear behaviour. b, Pulse-height spectra of α particles from an 241Am source at 700 V with a Gaussian fit (blue curve). c, Binned pulse-height spectrum of a 7 × 7 mm2 6LiInP2Se6 device (90 μm thickness) exposed to a moderated Pu–Be source while unshielded under 300 V bias for 30 min, and the corresponding Gaussian fit (blue curve). The tables in b, c show the Gaussian equation used to fit the full-energy peak and values of the peak parameters. y0, vertical offset; xc, centre; w, width; A, area; FWHM, full-width at half-maximum.
Extended Data Fig. 5 Channel number versus voltage for LiInP2Se6 devices collecting electrons.
The figure of merit for charge transport in semiconductor radiation detectors is the mobility–lifetime product, μτ. For reliable neutron detection, μτ must be sufficient so that counts from neutron capture are at a sufficiently high channel number to be distinct from the background to allow accurate discrimination. Here the channel number is plotted versus voltage for a typical device, and surprisingly the curve exhibits two slopes in different voltage ranges. The Hecht equation cannot model these data because the model assumes linear behaviour at low voltages, which then saturate towards a maximum channel number as the CCE reaches unity. The observed behaviour has the opposite concavity to the model, with a slight rise in slope as voltage increases. This change in slope can be explained by the reduction of the effective voltage from an internal polarization antiparallel to the applied field. Similarly atypical behaviour has been observed in the α-particle response of cadmium zinc telluride at T = 200 K, where this effect was attributed to polarization from deep-level traps41.
Extended Data Fig. 6 Pulse-height spectra of four other samples.
a–h, Pulse-height spectra are shown for electron (a–d) and hole (e–h) collection. The growth conditions of these samples were the same as those used for the samples that gave the spectra shown in the main-text figures. For hole collection, the spectra had lower channel numbers than the electron-collection spectra, indicating lower CCE for hole transport.
Extended Data Fig. 7 Pulse-height spectrum for 6Li-enriched LiInP2Se6 devices.
a–e, Data are shown for 6Li-enriched LiInP2Se6 devices with areas of 7 × 7 mm2 (a, b), 5 × 5 mm2 (c) and 3 × 3 mm2 (d, e). Devices with larger electrode areas had lower energy resolutions for electron collection, possibly owing to property variation among different samples. f, As-made LiInP2Se6 detectors with a diameter of 2 mm (‘1’) and 5 mm (‘2’) on the same single crystal. g, Time-dependent α-particle spectra collection by LiInP2Se6 detector 2 in f. We note that the peak channel number and count rate per second were constant over time. h, Collection of α-particle spectra by detector 1 in f with a pulse generator. i, Collection of α-particle spectra by detector 2 in f with a pulse generator. We note that the positions of the pulser peak in h and i are the same and the corresponding energy resolution is (3.5 ± 0.1)%, indicating the same electronics noise during collection.
Extended Data Fig. 8 Absorption probability versus 6LiInP2Se6 thickness for thermalized neutrons.
In addition to charge transport properties, the neutron-capture cross-section of LiInP2Se6 must be considered, as this directly correlates with the intrinsic maximum of the detector efficiency. A linear attenuation coefficient can be used to quantify the percentage of incident neutrons absorbed in a given material through the equation: fraction of neutrons captured (%) = (1 – e−lα) × 10%, where l is the thickness of the active region and α is the linear attenuation coefficient, which is calculated using the capture cross-section of thermalized neutrons for each element and its molar density. The calculated mass attenuation coefficient for thermalized neutrons is 5.1 cm−1 and 1.4 cm−1 for LiInP2Se6 fully enriched in 6Li and natural-abundance Li, respectively. A detector with a thickness of about 9 mm would be able to absorb 99% of the incident neutrons. Here we achieved the successful growth of crystals with thickness of ~1 mm, which would absorb ~40% of incident neutrons. Increases in efficiency would be achievable through scaled-up growth of thicker crystals or stacking of several thinner detectors42. When 115In (the most common isotope in natural indium) absorbs a neutron, the nuclide produces a γ-ray instead of a highly energetic charged particle. Thus, about 20% of the neutrons absorbed do not contribute to the signal, which sets the maximum theoretical detector efficiency of 6LiInP2Se6 to approximately 80%.
Rights and permissions
About this article
Cite this article
Chica, D.G., He, Y., McCall, K.M. et al. Direct thermal neutron detection by the 2D semiconductor 6LiInP2Se6. Nature 577, 346–349 (2020). https://doi.org/10.1038/s41586-019-1886-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1886-8
This article is cited by
-
Heavy-to-light electron transition enabling real-time spectra detection of charged particles by a biocompatible semiconductor
Nature Communications (2024)
-
Robust 2D layered MXene matrix–boron carbide hybrid films for neutron radiation shielding
Nature Communications (2023)
-
Latest developments in room-temperature semiconductor neutron detectors: Prospects and challenges
Science China Physics, Mechanics & Astronomy (2023)
-
The performance of a high-resistance semiconductor detector based on h-\(^{10}\)BN with thermal neutron detection capability
Journal of Materials Science (2023)
-
Discovery of chalcogenides structures and compositions using mixed fluxes
Nature (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.