Abstract
Humoral immune responses to immunization and infection and susceptibilities to antibody-mediated autoimmunity are generally lower in males1,2,3. However, the mechanisms underlying such sexual dimorphism are not well understood. Here we show that there are intrinsic differences between the B cells that produce germinal centres in male and female mice. We find that antigen-activated male B cells do not position themselves as efficiently as female B cells in the centre of follicles in secondary lymphoid organs, in which germinal centres normally develop. Moreover, GPR174—an X-chromosome-encoded G-protein-coupled receptor—suppresses the formation of germinal centres in male, but not female, mice. This effect is intrinsic to B cells, and correlates with the GPR174-enhanced positioning of B cells towards the T-cell–B-cell border of follicles, and the distraction of male, but not female, B cells from S1PR2-driven follicle-centre localization. Biochemical fractionation of conditioned media that induce B-cell migration in a GPR174-dependent manner identifies CCL21 as a GPR174 ligand. In response to CCL21, GPR174 triggers a calcium flux and preferentially induces the migration of male B cells; GPR174 also becomes associated with more Gαi protein in male than in female B cells. Male B cells from orchidectomized mice exhibit impaired GPR174-mediated migration to CCL21, and testosterone treatment rescues this defect. Female B cells from testosterone-treated mice exhibit male-like GPR174–Gαi association and GPR174-mediated migration. Deleting GPR174 from male B cells causes more efficient positioning towards the follicular centre, the formation of more germinal centres and an increased susceptibility to B-cell-dependent experimental autoimmune encephalomyelitis. By identifying GPR174 as a receptor for CCL21 and demonstrating its sex-dependent control of B-cell positioning and participation in germinal centres, we have revealed a mechanism by which B-cell physiology is fine-tuned to impart sexual dimorphism to humoral immunity.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data generated here are included within the paper (and its Supplementary Information files) or available from the corresponding author upon reasonable request. Source Data for Figs. 1–4 and Extended Data Figs. 1–10 are provided with the paper.
References
Whitacre, C. C. Sex differences in autoimmune disease. Nat. Immunol. 2, 777–780 (2001).
Klein, S. L. & Flanagan, K. L. Sex differences in immune responses. Nat. Rev. Immunol. 16, 626–638 (2016).
Ghosh, S. & Klein, R. S. Sex drives dimorphic immune responses to viral infections. J. Immunol. 198, 1782–1790 (2017).
Ansel, K. M. et al. A chemokine-driven positive feedback loop organizes lymphoid follicles. Nature 406, 309–314 (2000).
Förster, R. et al. A putative chemokine receptor, BLR1, directs B cell migration to defined lymphoid organs and specific anatomic compartments of the spleen. Cell 87, 1037–1047 (1996).
Nakano, H. et al. A novel mutant gene involved in T-lymphocyte-specific homing into peripheral lymphoid organs on mouse chromosome 4. Blood 91, 2886–2895 (1998).
Förster, R. et al. CCR7 coordinates the primary immune response by establishing functional microenvironments in secondary lymphoid organs. Cell 99, 23–33 (1999).
Green, J. A. et al. The sphingosine 1-phosphate receptor S1P2 maintains the homeostasis of germinal center B cells and promotes niche confinement. Nat. Immunol. 12, 672–680 (2011).
Pereira, J. P., Kelly, L. M., Xu, Y. & Cyster, J. G. EBI2 mediates B cell segregation between the outer and centre follicle. Nature 460, 1122–1126 (2009).
Gatto, D., Paus, D., Basten, A., Mackay, C. R. & Brink, R. Guidance of B cells by the orphan G protein-coupled receptor EBI2 shapes humoral immune responses. Immunity 31, 259–269 (2009).
Pereira, J. P. P., Kelly, L. M. & Cyster, J. G. Finding the right niche: B-cell migration in the early phases of T-dependent antibody responses. Int. Immunol. 22, 413–419 (2010).
Inoue, A. et al. TGFα shedding assay: an accurate and versatile method for detecting GPCR activation. Nat. Methods 9, 1021–1029 (2012).
Barnes, M. J. et al. The lysophosphatidylserine receptor GPR174 constrains regulatory T cell development and function. J. Exp. Med. 212, 1011–1020 (2015).
Bangasser, D. A. et al. Sex differences in corticotropin-releasing factor receptor signaling and trafficking: potential role in female vulnerability to stress-related psychopathology. Mol. Psychiatry 15, 896–904 (2010).
Lyons, J. A., San, M., Happ, M. P. & Cross, A. H. B cells are critical to induction of experimental allergic encephalomyelitis by protein but not by a short encephalitogenic peptide. Eur. J. Immunol. 29, 3432–3439 (1999).
Genain, C. P., Cannella, B., Hauser, S. L. & Raine, C. S. Identification of autoantibodies associated with myelin damage in multiple sclerosis. Nat. Med. 5, 170–175 (1999).
Mulero, P., Midaglia, L. & Montalban, X. Ocrelizumab: a new milestone in multiple sclerosis therapy. Ther. Adv. Neurol. Disorder. https://doi.org/10.1177/1756286418773025 (2018).
Wang, Y. et al. Germinal-center development of memory B cells driven by IL-9 from follicular helper T cells. Nat. Immunol. 18, 921–930 (2017).
Xu, H. et al. Follicular T-helper cell recruitment governed by bystander B cells and ICOS-driven motility. Nature 496, 523–527 (2013).
Acknowledgements
We thank the Protein Preparation and Characterization Core Facility at the Tsinghua University Branch of the China National Center for Protein Sciences for chromatography support; and T. Li, K. He and H. Wang at the National Center of Biomedical Analysis for expert advice and assistance with mass spectrometry. This work was funded in part by the National Natural Science Foundation of China (grants 81621002, 31830023, 81761128019 and 81425011), the Tsinghua-Peking Center for Life Sciences and the Beijing Municipal Science and Technology Commission. This work was also funded in part by the Bill and Melinda Gates Foundation and the Howard Hughes Medical Institute. The findings and conclusions within are those of the authors and do not necessarily reflect the positions or policies of the Bill and Melinda Gates Foundation or the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
R.Z. conducted a majority of the experiments and designed parts of the study. X.C. made the initial observation of GPR174-mediated positioning effects, developed conditioned media for ligand identification and, together with W.M., conducted baseline characterization of GPR174-deficient mice. X.C., J.G. and X. Zhou generated GPR174–GFP BAC transgenic mice. J.Z. conducted antibody titre analyses. J.Y., J.S. and J.W. conducted RNA-sequencing analyses. X. Zhong and J.R. helped with transwell and immunohistochemistry analyses, respectively. H.Q. conceptualized the study, supervised the work and wrote the paper with R.Z.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Andrew Luster, Charles Mackay and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
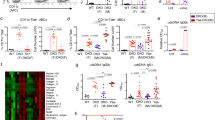
Extended Data Fig. 1 Intrinsic differences between male and female B cells in forming geminal centres.
a, Representative contour plots and frequencies of dsRed-expressing and GFP-expressing MD4-derived cells of the indicated sexes, found in the initial transfer inoculum (left panels) or in the germinal-centre compartment in draining lymph nodes five days after subcutaneous immunization (middle and right panels) of male recipient mice. b, Summary statistics of the dsRed/GFP ratio in MD4 germinal centres (GCs), normalized against the dsRed/GFP ratio in transfer inoculum. Each symbol represents one mouse (n = 17 and 16); data were pooled from three independent experiments with similar results. c, Representative distribution patterns of dsRed-expressing and GFP-expressing MD4 cells of the indicated sexes in follicles 90 h after immunization. Scale bar, 50 μm. d, Definition of the centre concentration index (CCI) and summary statistics of the CCI of dsRed+ tester cells of the indicated sexes, normalized against the CCI of GFP+ male control cells in the same follicle. Each symbol denotes one follicle (n = 38 and 46). Data pooled from two independent experiments with similar results. Scatter plots have horizontal lines denoting mean (b, d); two-tailed unpaired Student’s t-tests were used to calculate the P values at the top of each graph.
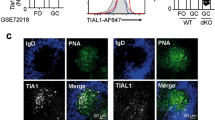
Extended Data Fig. 2 Expression of guidance GPCRs in male and female cells, differential GPR174 expression by naive and germinal-centre B cells, and lack of influence on CCR7 expression.
a, Relative mRNA expression of Cxcr5, S1pr2, CCR7 and Gpr183 in male or female dsRed-expressing MD4 B cells, normalized against that in cotransferred male GFP-expressing MD4 B cells (Extended Data Fig. 1), 90 h after immunization. Data are shown as pairs of three individual experiments. b, Relative Gpr174 mRNA levels in indicated splenic B cell subsets, normalized against Gapdh expression. The results are shown as scatter plots of three biological replicates, with lines indicating mean, and are representative of two independent experiments with similar results. c, Surface CCR7 levels on B cells that were retrovirally transduced with a control or GPR174-expressing vector. GFP+, infected cells after transduction; GFP−, uninfected cells after transduction; representative of three independent experiments. Two-tailed paired (a) or unpaired (b) Student’s t-tests were used to calculate P values, given at the top of each graph; ****P < 0.0001.
Extended Data Fig. 3 GPR174 suppresses germinal-centre formation in male, but not female, mice.
a, Representative cytometric profiles of FAShiGL7hi frequencies of transduced MD4 B cells in B6 recipients five days after immunization. b, c, Representative cytometric profiles of germinal centres (b) and SPPCs (c) in mice of the indicated genotypes and sexes seven days after SRBC immunization, matching the summary statistics presented in Fig. 1d, e, respectively. d, Representative cytometric profiles (left panels) and summary statistics (right panel) of germinal centres in littermate mice of the indicated genotypes and sexes, eight days after infection with LCMV Armstrong (105 plaque-forming units per mouse). The scatter plot (right) shows individual mice with lines denoting mean (n = 15, 15, 14 and 14 mice, from left to right). Data pooled from three independent experiments with similar results. A two-tailed unpaired Student’s t-test was used. e, f, Representative cytometric profiles of germinal centres (e) and SPPCs (f) in mixed bone-marrow (BM) chimeric mice of indicated types seven days after SRBC immunization, matching the summary statistics presented in Fig. 1g, h, respectively.
Extended Data Fig. 4 Endogenous GPR174 does not affect the follicular distribution of naive B cells or the accumulation of freshly activated B cells to the T-cell–B-cell border.
a, Splenic distribution of GPR174-sufficient and -deficient naive B cells 24 h after adoptive transfer. WT, wild type; KO, knockout. Data are representative of two independent experiments involving either male or female cells. Scale bars, 50 μm. b, Left, splenic distribution of GPR174-sufficient or -deficient B cells that were stimulated with anti-IgM, 6 h after adoptive transfer. Scale bar, 50 μm. Middle, definitions of follicular area, the T-cell–B-cell border and the T-cell zone, based on IgD and CD3 staining. Right, relative abundance of transferred B cells in the indicated regions. Data (n = 24 biological replicates) represent two independent experiments with similar results. Lines in scatter plots denote mean. We used two-way ANOVA with Bonferroni’s multiple comparisons; ns, not significant.
Extended Data Fig. 5 Minimal LysoPS effects on GPR174-dependent B-cell migration and GPR174 ligand identification.
a, Transwell migration of lipopolysaccharide-activated GPR174-sufficient or -deficient B cells in response to LysoPS of indicated concentrations. Data are plotted as mean ± s.e.m. of the percentage of cells that migrated in triplicated wells at each concentration, fitted with three-parameter log dose–response curves; two-way ANOVA was used to compare the two groups. One of two independent experiments with similar results is shown. b–d, Transwell migration of lipopolysaccharide-activated B cells that were transduced with a control or GPR174-expressing vector, in response to stroma-conditioned medium (stroma), dimethylsulfoxide (DMSO) control or LysoPS of indicated concentrations (b); or in response to stroma-conditioned medium (stroma), conditioned medium heated at 100 °C for 10 min (heated), conditioned medium treated with the proteinase K inhibitor PMSF alone (PMSF), conditioned medium treated with proteinase K and then PMSF (PK + PMSF), or conditioned media treated as above and then further supplemented with 300 ng ml−1 CXCL13 (PK + PMSF + CXCL13) to exclude cell damage owing to remaining proteinase activity (c); or in response to blank culture medium, or mouse or porcine stroma-conditioned media (d). All data are plotted as the mean percentages of cells that migrated in triplicated wells, from one experiment representative of three with similar results. Two-way ANOVA with Bonferroni’s multiple comparison tests were used to compare vector and GPR174 groups, with P values given in the graphs. ****P < 0.0001; ns, not significant. e, Workflow showing the six-step biochemical fractionation method for identifying GPR174 ligands in splenic stroma-conditioned medium. See Methods for details. CV, column volume. f–k, Chromatography traces and putative ligand activities as detected by the transwell assay in d in relevant fractions during the six steps of e. AU, arbitrary units; mS, millisiemens. l, Left, silver staining of the indicated fractions from k resolved on 12% SDS–PAGE; the black box on fraction 5 marks bands subjected to liquid chromatography-mass spectrometry (LC-MS)/MS analysis. Right, identified unique porcine CCL21-derived peptides (solid underlines) aligned against porcine, human and murine CCL21 protein sequences. For gel source data, see Supplementary Fig. 1. Data shown in f–l are from one of two independent experiments with similar results. FT, flow-through.
Extended Data Fig. 6 CCL21 and CCL19 are chemoattractant ligands of GPR174.
a, b, Transwell migration of lipopolysaccharide-activated B cells that were transduced with a control or GPR174-expressing vector: a, in response to stroma-conditioned medium (stroma) or 100 ng ml−1 recombinant CCL21 or CCL19, in the presence of control IgG or 5 μg ml−1 CCL21- or CCL19-blocking antibody, individually or in combination; b, in response to recombinant mouse CCL21 (left) or CCL19 (right). Data represent triplicated wells for each condition from one of three independent experiments with similar results. Two-tailed unpaired Student’s t-tests; ****P < 0.0001. c, Cytometric profiles and MFI of CCL21 binding to HEK293T cells transfected with control GFP, GPR174–GFP or CCR7–GFP fusion constructs, with background staining on the GFP− fraction subtracted from the corresponding GFP+ fraction. Left, gating of GFP− and GFP+ cells in each group (top) and histograms of anti-His staining (bottom) of cells that were incubated with different doses of His-tagged recombinant CCL21. PE, phycoerythrin. Right, data represent three biological replicates for each condition, from one of three independent experiments with similar results. Two-way ANOVA tests were used to compare groups, with P values given in the graph; red asterisks, GFP versus GPR174–GFP; blue asterisks, GFP versus CCR7–GFP; green asterisks, GPR174–GFP versus CCR7–GFP; ****P < 0.0001. d, e, Exemplary calcium responses of HEK293T cells transfected with vector, GPR174 or CCR7, following stimulation with CCL21 or the calcium ionophore ionomycin at the indicated concentrations (d); and the magnitude of CCL21-triggered calcium responses as a fraction of the ionomycin-triggered maximum of the same cells, overlaid with three-parameter log dose–response curves on top (e). The two different symbols indicate data from two independent experiments. P values obtained from an extra sum-of-squares F-test of the null hypothesis that the GPR174 and CCR7 curves have the same EC50.
Extended Data Fig. 7 Testosterone dependence of the GPR174-mediated migratory response to CCL21 does not involve modulation of GPR174 or CCR7 expression.
a, CCL21-induced transwell migration of B cells from GPR174-sufficient (WT) or -deficient (KO) male littermate mice that were sham-operated (sham) or orchidectomized (ORX), allowed to recover for six weeks, and then treated with either vehicle or testosterone for two weeks. Data represent three biological replicates from one of two independent experiments with similar results. Blue asterisks, male sham wild type plus vehicle versus sham GPR174-knockout plus vehicle; red asterisks, male ORX wild type plus vehicle versus male ORX wild type plus testosterone; green asterisks, male sham wild type plus vehicle versus male ORX wild type plus testosterone. b, Relative Gpr174 mRNA levels normalized to Gapdh expression in naive and activated B cells from male or female mice that were either sham-operated or gonadectomized. Data (n = 3 biological replicates) represent three independent experiments with similar results. c, Surface CCR7 levels on naive or activated B cells isolated from mice of the indicated GPR174 genotypes, sexes and gonadectomies. Data shown are histogram overlays (coloured lines, CCR7; grey lines, isotype staining control) and CCR7 MFIs after isotype staining background subtraction. Each symbol in the MFI data represents one mouse, and data represent three independent experiments with similar results. d, e, Relative Gpr174 mRNA levels (d) and surface CCR7 levels (e) on activated B cells from male mice that were sham-operated or orchidectomized and then treated with either vehicle or testosterone. Data represent three mice per group from one of three independent experiments with similar results. f, g, Relative Gpr174 mRNA levels (f) and surface CCR7 levels (g) on activated B cells from male or female mice that were treated with either vehicle or testosterone. Data represent three mice per group from one of three independent experiments with similar results. h, Relative expression of Gpr174 mRNA in male or female dsRed-expressing MD4 B cells normalized against that in cotransferred male GFP-expressing MD4 B cells 90 h after activation by HEL–OVA immunization (as in Extended Data Fig. 1); three line-connected pairs indicate three independent experiments. Two-way ANOVA with Bonferroni’s multiple comparisons (a) or two-tailed unpaired (b–g) or paired (h) Student’s t-tests were used for statistical comparisons, with P values given in the graph.
Extended Data Fig. 8 Characterization of GPR174–GFP transgenic mice.
a, Diagrams of the GPR174–GFP fusion (top) and GPR174–GFP BAC construct (bottom) in the context of the Gpr174 genomic locus, with the five exons numbered in italics. b, Transwell migration of lipopolysaccharide-activated B cells that were transduced with a control, GPR174-expressing or GPR174-GFP-expressing vector in response to stroma-conditioned medium (stroma) or 10 ng ml−1 or 50 ng ml−1 recombinant CCL21. Data represent three biological replicates from one of two independent experiments with similar results. c, d, Histograms (c) and geometric MFI (geo.mean) (d) of GFP fluorescence from naive and germinal-centre B cells in B6 (grey histograms) or GPR174–GFP BAC (open histograms) transgenic mice. Each symbol in the MFI plot represents one mouse, and data represent two independent experiments with similar results. Two-way ANOVA with Bonferroni’s multiple comparisons (b) or two-tailed unpaired Student’s t-test (d) was used for comparisons between groups. ****P < 0.0001; ns, not significant.
Extended Data Fig. 9 GPR174 retards follicular-centre localization and germinal-centre formation by male B cells.
a, Representative distribution patterns of dsRed-expressing and GFP-expressing MD4 cells of the indicated genotypes in follicles of the draining lymph nodes 90 h after immunization of male recipient mice. Scale bar, 50 μm. b, Summary statistics of ratios between CCIs, as defined in Extended Data Fig. 1d, of dsRed+ cells and GFP+ cells of the indicated genotypes in the same follicles. Each symbol denotes one follicle (n = 42 and 43), with lines denoting mean. Data are pooled from two independent experiments with similar results. c, Representative contour plots and frequencies of dsRed-expressing and GFP-expressing cells of the indicated genotypes in the transferred MD4 (left panels) or in the germinal-centre compartment five days after immunization (middle and right panels). d, Summary statistics of the dsRed/GFP ratio in MD4 germinal centres normalized against the dsRed/GFP ratio in transferred MD4. Each symbol denotes one mouse (n = 19 and 19), with lines denoting mean. Data are pooled from three independent experiments with similar results. Two-tailed unpaired Student’s t-tests were used to compare groups, with P values given in the graph.
Extended Data Fig. 10 GPR174 does not affect germinal-centre formation by female B cells.
a, Representative contour plots and frequencies of dsRed-expressing and GFP-expressing cells of the indicated genotypes in the transferred MD4 (left panels) or in the germinal-centre compartment five days after immunization (middle and right panels) of male recipient mice. b, Summary statistics of the dsRed/GFP ratio in MD4 germinal centres, normalized against the dsRed/GFP ratio in transferred MD4 cells. Each symbol denotes one mouse, with lines showing mean. Data (n = 8 and 8) are pooled from two independent experiments with similar results. Two-tailed unpaired Student’s t-test was used to compare groups.
Supplementary information
Supplementary Figure
Supplementary Figure 1: uncropped gel scans with size marker indications
Source data
Rights and permissions
About this article
Cite this article
Zhao, R., Chen, X., Ma, W. et al. A GPR174–CCL21 module imparts sexual dimorphism to humoral immunity. Nature 577, 416–420 (2020). https://doi.org/10.1038/s41586-019-1873-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1873-0
This article is cited by
-
The conneXion between sex and immune responses
Nature Reviews Immunology (2024)
-
Hallmarks of sex bias in immuno-oncology: mechanisms and therapeutic implications
Nature Reviews Cancer (2024)
-
Advances in sex disparities for cancer immunotherapy: unveiling the dilemma of Yin and Yang
Biology of Sex Differences (2022)
-
TRPM8 contributes to sex dimorphism by promoting recovery of normal sensitivity in a mouse model of chronic migraine
Nature Communications (2022)
-
GPR174 knockdown enhances blood flow recovery in hindlimb ischemia mice model by upregulating AREG expression
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.