Abstract
The human gastrointestinal tract consists of a dense and diverse microbial community, the composition of which is intimately linked to health. Extrinsic factors such as diet and host immunity are insufficient to explain the constituents of this community, and direct interactions between co-resident microorganisms have been implicated as important drivers of microbiome composition. The genomes of bacteria derived from the gut microbiome contain several pathways that mediate contact-dependent interbacterial antagonism1,2,3. Many members of the Gram-negative order Bacteroidales encode the type VI secretion system (T6SS), which facilitates the delivery of toxic effector proteins into adjacent cells4,5. Here we report the occurrence of acquired interbacterial defence (AID) gene clusters in Bacteroidales species that reside within the human gut microbiome. These clusters encode arrays of immunity genes that protect against T6SS-mediated intra- and inter-species bacterial antagonism. Moreover, the clusters reside on mobile elements, and we show that their transfer is sufficient to confer resistance to toxins in vitro and in gnotobiotic mice. Finally, we identify and validate the protective capability of a recombinase-associated AID subtype (rAID-1) that is present broadly in Bacteroidales genomes. These rAID-1 gene clusters have a structure suggestive of active gene acquisition and include predicted immunity factors of toxins derived from diverse organisms. Our data suggest that neutralization of contact-dependent interbacterial antagonism by AID systems helps to shape human gut microbiome ecology.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data required to assess the conclusion of this research are available in the main text and Supplementary Information, have been deposited at the Sequence Read Archive (SRA) under BioProject accession PRJNA484981 or are available from https://github.com/borenstein-lab/T6SS).
Code availability
Python and R scripts used in this work are available for download (https://github.com/borenstein-lab/T6SS).
References
Whitney, J. C. et al. A broadly distributed toxin family mediates contact-dependent antagonism between Gram-positive bacteria. eLife 6, e26938 (2017).
Zhang, D., de Souza, R. F., Anantharaman, V., Iyer, L. M. & Aravind, L. Polymorphic toxin systems: comprehensive characterization of trafficking modes, processing, mechanisms of action, immunity and ecology using comparative genomics. Biol. Direct 7, 18 (2012).
Coyne, M. J., Roelofs, K. G. & Comstock, L. E. Type VI secretion systems of human gut Bacteroidales segregate into three genetic architectures, two of which are contained on mobile genetic elements. BMC Genomics 17, 58 (2016).
Russell, A. B. et al. A type VI secretion-related pathway in Bacteroidetes mediates interbacterial antagonism. Cell Host Microbe 16, 227–236 (2014).
Hood, R. D. et al. A type VI secretion system of Pseudomonas aeruginosa targets a toxin to bacteria. Cell Host Microbe 7, 25–37 (2010).
Cornforth, D. M. & Foster, K. R. Competition sensing: the social side of bacterial stress responses. Nat. Rev. Microbiol. 11, 285–293 (2013).
Hille, F. et al. The biology of CRISPR–Cas: backward and forward. Cell 172, 1239–1259 (2018).
Verster, A.J. et al. The landscape of type VI secretion across human gut microbiomes reveals its role in community composition. Cell Host Microbe 22, 411–419 (2017).
Wexler, A. G. et al. Human symbionts inject and neutralize antibacterial toxins to persist in the gut. Proc. Natl Acad. Sci. USA 113, 3639–3644 (2016).
Hecht, A. L. et al. Strain competition restricts colonization of an enteric pathogen and prevents colitis. EMBO Rep. 17, 1281–1291 (2016).
Kirchberger, P. C., Unterweger, D., Provenzano, D., Pukatzki, S. & Boucher, Y. Sequential displacement of type VI secretion system effector genes leads to evolution of diverse immunity gene arrays in Vibrio cholerae. Sci. Rep. 7, 45133 (2017).
Steele, M. I., Kwong, W. K., Whiteley, M. & Moran, N. A. Diversification of type VI secretion system toxins reveals ancient antagonism among bee gut microbes. mBio 8, e26938 (2017).
Ting, S. Y. et al. Bifunctional immunity proteins protect bacteria against Ftsz-targeting ADP-ribosylating toxins. Cell 175, 1380–1392 (2018).
Lloyd-Price, J. et al. Strains, functions and dynamics in the expanded Human Microbiome Project. Nature 550, 61–66 (2017).
Qin, J. et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65 (2010).
Manor, O. et al. Metagenomic evidence for taxonomic dysbiosis and functional imbalance in the gastrointestinal tracts of children with cystic fibrosis. Sci. Rep. 6, 22493 (2016).
Siguier, P., Gourbeyre, E. & Chandler, M. Bacterial insertion sequences: their genomic impact and diversity. FEMS Microbiol. Rev. 38, 865–891 (2014).
Zhao, S. et al. Adaptive evolution within gut microbiomes of healthy people. Cell Host Microbe 25, 656–667.e8 (2019).
Wozniak, R. A. & Waldor, M. K. Integrative and conjugative elements: mosaic mobile genetic elements enabling dynamic lateral gene flow. Nat. Rev. Microbiol. 8, 552–563 (2010).
Stevens, A. M., Shoemaker, N. B. & Salyers, A. A. The region of a Bacteroides conjugal chromosomal tetracycline resistance element which is responsible for production of plasmidlike forms from unlinked chromosomal DNA might also be involved in transfer of the element. J. Bacteriol. 172, 4271–4279 (1990).
Castillo, F., Benmohamed, A. & Szatmari, G. Xer site specific recombination: double and single recombinase systems. Front. Microbiol. 8, 453 (2017).
Abu-Ali, G. S. et al. Metatranscriptome of human faecal microbial communities in a cohort of adult men. Nat. Microbiol. 3, 356–366 (2018).
Foster, K. R. & Bell, T. Competition, not cooperation, dominates interactions among culturable microbial species. Curr. Biol. 22, 1845–1850 (2012).
Poole, S. J. et al. Identification of functional toxin/immunity genes linked to contact-dependent growth inhibition (CDI) and rearrangement hotspot (Rhs) systems. PLoS Genet. 7, e1002217 (2011).
Drider, D., Fimland, G., Héchard, Y., McMullen, L. M. & Prévost, H. The continuing story of class IIa bacteriocins. Microbiol. Mol. Biol. Rev. 70, 564–582 (2006).
Coyte, K. Z., Schluter, J. & Foster, K. R. The ecology of the microbiome: networks, competition, and stability. Science 350, 663–666 (2015).
Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature 486, 207–214 (2012).
Qin, J. et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 490, 55–60 (2012).
Li, J. et al. An integrated catalog of reference genes in the human gut microbiome. Nat. Biotechnol. 32, 834–841 (2014).
Truong, D. T. et al. MetaPhlAn2 for enhanced metagenomic taxonomic profiling. Nat. Methods 12, 902–903 (2015).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Luo, R. et al. SOAPdenovo2: an empirically improved memory-efficient short-read de novo assembler. Gigascience 1, 18 (2012).
Besemer, J., Lomsadze, A. & Borodovsky, M. GeneMarkS: a self-training method for prediction of gene starts in microbial genomes. Implications for finding sequence motifs in regulatory regions. Nucleic Acids Res. 29, 2607–2618 (2001).
Bacic, M. K. & Smith, C. J. Laboratory maintenance and cultivation of bacteroides species. Curr. Protoc. Microbiol. 9, 13C.1.1–13C.1.21 (2008).
Koropatkin, N. M., Martens, E. C., Gordon, J. I. & Smith, T. J. Starch catabolism by a prominent human gut symbiont is directed by the recognition of amylose helices. Structure 16, 1105–1115 (2008).
Degnan, P. H., Barry, N. A., Mok, K. C., Taga, M. E. & Goodman, A. L. Human gut microbes use multiple transporters to distinguish vitamin B12 analogs and compete in the gut. Cell Host Microbe 15, 47–57 (2014).
Hoffman, L. R. et al. Escherichia coli dysbiosis correlates with gastrointestinal dysfunction in children with cystic fibrosis. Clin. Infect. Dis. 58, 396–399 (2014).
Wick, R. R., Judd, L. M., Gorrie, C. L. & Holt, K. E. Unicycler: resolving bacterial genome assemblies from short and long sequencing reads. PLOS Comput. Biol. 13, e1005595 (2017).
Martens, E. C., Chiang, H. C. & Gordon, J. I. Mucosal glycan foraging enhances fitness and transmission of a saccharolytic human gut bacterial symbiont. Cell Host Microbe 4, 447–457 (2008).
García-Bayona, L. & Comstock, L. E. Bacterial antagonism in host-associated microbial communities. Science 361, eaat2456 (2018).
Lim, B., Zimmermann, M., Barry, N. A. & Goodman, A. L. Engineered regulatory systems modulate gene expression of human commensals in the gut. Cell 169, 547–558 (2017).
Paik, J. et al. Potential for using a hermetically-sealed, positive-pressured isocage system for studies involving germ-free mice outside a flexible-film isolator. Gut Microbes 6, 255–265 (2015).
Bailey, T. L. et al. MEME SUITE: tools for motif discovery and searching. Nucleic Acids Res. 37, W202–W208 (2009).
Silverman, J. M. et al. Haemolysin coregulated protein is an exported receptor and chaperone of type VI secretion substrates. Mol. Cell 51, 584–593 (2013).
Cardona, S. T. & Valvano, M. A. An expression vector containing a rhamnose-inducible promoter provides tightly regulated gene expression in Burkholderia cenocepacia. Plasmid 54, 219–228 (2005).
Bookout, A. L., Cummins, C. L., Mangelsdorf, D. J., Pesola, J. M. & Kramer, M. F. High-throughput real-time quantitative reverse transcription PCR. Curr. Protoc. Mol. Biol. 73, 15.8.1–15.8.28 (2006).
Caro-Quintero, A. & Ochman, H. Assessing the unseen bacterial diversity in microbial communities. Genome Biol. Evol. 7, 3416–3425 (2015).
Acknowledgements
We thank the UW GNAC for assistance with gnotobiotic experiments. We thank C. Sears, A. Goodman, T. Kuwahara and E. Martens for providing Bacteroides strains. This work was supported by National Institutes of Health (NIH) grants AI080609 (to J.D.M.), P30DK089507 (to L.R.H. as pilot study PI), R01DK095869 (to L.R.H.), K99GM129874 (to B.D.R.), R01GM124312 (to E.B.), and New Innovator Award DP2AT00780201 (to E.B.), and the Burroughs Wellcome Fund (to J.D.M.). A.J.V. was supported by a postdoctoral fellowship from the Natural Sciences and Engineering Research Council of Canada. B.D.R. was supported by a Simons Foundation-sponsored Life Sciences Research Foundation postdoctoral fellowship. E.B. is a Faculty Fellow of the Edmond J. Safra Center for Bioinformatics at Tel Aviv University. J.D.M. is an HHMI Investigator.
Author information
Authors and Affiliations
Contributions
B.D.R., A.J.V., A.M.H., S.B.P., E.B. and J.D.M. designed the study. B.D.R. and D.T.S. performed in vitro growth experiments; A.J.V., B.D.R. and M.C.R. performed bioinformatic analyses; B.D.R., C.E.P. and L.R.H. isolated and sequenced genomes of gut bacteria; B.D.R., D.T.S., A.M.H. and S.B.P. performed gnotobiotic mouse experiments; B.D.R., A.J.V., M.C.R., D.T.S., A.M.H., S.B.P., E.B. and J.D.M. analysed data; and B.D.R., A.J.V., S.B.P., E.B. and J.D.M. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Peer review information Nature thanks Melanie Blokesch, Kevin Foster and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
Extended Data Fig. 1 Prevalence of B. fragilis-specific orphan immunity genes in adult and infant microbiomes.
a, Number of adult human gut microbiome samples in which the indicated immunity genes (1–14, GA3_i1–14 from ref. 8) can be detected at an 80% nucleotide identity threshold and an abundance more than tenfold that of B. fragilis marker genes. Bars coloured as in Fig. 1a, and asterisks indicate immunity genes without orphan representation. b, Comparison of abundance of B. fragilis-specific T6SS immunity genes with B. fragilis species-specific marker genes in infant microbiome samples16 (Supplementary Table 4). Abundances are calculated as in Fig. 1a. Samples in which immunity gene abundance exceeds that of Bacteroides by over tenfold (blue) are highlighted.
Extended Data Fig. 2 Diversity and genomic context of orphan immunity genes in human gut microbiomes and diverse Bacteroides species.
a, Representative AID-1 gene clusters containing homologues of the indicated B. fragilis T6S immunity genes from the indicated reference genomes. b, Data points indicate the amino acid identity of unique genes homologous to indicated B. fragilis-specific T6SS cognate immunity genes identified through BLAST analysis of the IGC29 (n = 88 genes, maximum E = 1 × 10−40; minimum percentage identity, 60%).
Extended Data Fig. 3 Orphan immunity genes specifically enhance the fitness of Bacteroides strains in vitro and in vivo.
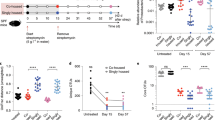
a, b, T6SS-dependent competitiveness of parental strains of B. ovatus 3725 and the indicated mutant and complemented derivatives during in vitro growth competition experiments with B. fragilis 9343. Relative recipient fitness was determined by calculating the ratio of final to initial c.f.u. and normalizing to the corresponding experiment with B. fragilis 9343 lacking tssC (T6S-inactive). Data are mean ± s.d. of three independent biological replicates. *P < 0.01, unpaired two-tailed t-test. c, T6SS-dependent competitiveness of a parental strain of B. ovatus 3725 or a strain bearing in-frame deletions of indicated orphan immunity genes, during in vitro growth competition experiments with an orthogonal effector-bearing B. fragilis 638R parental strain or a derivative strain lacking tssC (T6S-inactive). Relative recipient fitness and statistics were calculated as in a and b. n = 3 independent biological replicates. d, e, Recovery of B. fragilis 9343 (d) or 638R (e) and the indicated orphan immunity mutant derivative from pairwise competitions of the strains in germ-free mice. Lines indicate the mean at each time point (n = 8 mice per group for each of two independent experiments). Alternating time points of these data are included in ratio form in Fig. 3c. f, Schematic depicting genomic loci for the B. fragilis ATCC 43859 parental strain, the B. fragilis 638R AID-1 donor strain, the AID-1 system, and the ATCC 43859 AID-1 recipient. Grey shading indicates homology; red arrows indicate the position of PCR primers used to infer insertion of the AID-1 element at the tRNALys insertion site. g, Abundance of B. ovatus in samples lacking detected orphan immunity genes (−) and samples in which the indicated orphan immunity genes were assigned to B. ovatus (+). Abundances are calculated as in Fig. 1a. *P < 0.001, Wilcoxon rank-sum test. n = 128 non-orphan samples, n = 24 samples containing orphan immunity. For box plots, the middle line denotes the median; the box denotes the interquartile range (IQR); and the whiskers denote 1.5× the IQR.
Extended Data Fig. 4 The GA2 system of Bacteroidales mediates interbacterial antagonism.
Recovery of Bacteroides dorei DSM 17855 cells lacking GA2_e14-i14 (BACDOR_RS22955-17020) from two-strain in vitro growth competition experiments with the indicated donor strains. n = 3 technical replicates representative of three biological replicates. **P < 0.01, unpaired two-tailed t-test.
Extended Data Fig. 5 rAID-1 systems include conserved and repetitive intergenic sequences and bear hallmarks of horizontal gene transfer.
a, Left, motif enrichment analysis from the intergenic sequences immediately 3′ of the recombinase stop codon to the start codon of the first downstream open reading frame within 16 randomly selected rAID-1 gene clusters. This region is highlighted in blue in three representative rAID-1 systems shown above. Right, motif enrichment analysis from all 86 intergenic sequences between the ORFs of six rAID-1 clusters (B. fragilis NCTC 9343, B. cellulosilyticus WH2, B. ovatus 3725, Paraprevotella clara YIT 11840, Parabacteroides goldsteinii dnLKV18, and Parabacteroides gordonii MS-1)44. This region is highlighted in red in three representative rAID-1 systems shown above. b, Average G + C nucleotide content of rAID-1-associated recombinase versus rAID-1 predicted ORFs (n = 226). ***P < 0.0001, unpaired two-tailed t-test. c, Schematic depicting the G + C and A + T nucleotide content across a representative rAID-1 system from B. fragilis 9343. d, Frequency distribution of gene number in rAID-1 clusters (n = 1,247 genes in 226 clusters). Bin width is five genes. e, Composition of genes in rAID-1 clusters (n = 226 clusters) as determined by profile HMM scans and BLAST analysis against a curated database of Bacteroidales T6SS immunity genes2,8. f, Comparison of the total abundances of rAID-1-associated predicted recombinases and the Bacteroides genus in adult microbiome samples derived from the HMP and MetaHIT studies (Supplementary Table 8). Abundance values are calculated as in Fig. 1; genus abundance corresponds to the sum of all Bacteroides spp. (calculated individually as the average of species-specific marker gene abundances). g, Results of qRT–PCR analyses for the indicated B. ovatus 3725 genes belonging to AID-1 (i6, M088_1971) or AID-1 clusters (Rec, recombinase, M088_1401; orf1, M088_1400) under conditions of growth in mono- or co-culture with B. fragilis 9343 for 2 h. Data are mean ± s.d. of three independent biological replicates. *P < 0.05, **P < 0.01, Wilcoxon two-tailed sign-rank test.
Supplementary information
Supplementary Figure 1
Source data (unedited gel images) associated with Fig. 3
Supplementary Table 1
Metagenomic results derived from the HMP and MetaHIT studies utilized in this study.
Supplementary Table 2
List of Bacteroidales T6SS cognate immunity genes.
Supplementary Table 3
Accession numbers and abbreviations of relevant Bacteroides strains.
Supplementary Table 4
Metagenomic results derived from infant stool samples utilized in this study.
Supplementary Table 5
Description of AID gene clusters in genomes depicted in Fig. 2.
Supplementary Table 6
Description of rAID-1 gene clusters depicted in Fig. 4.
Supplementary Table 7
Features associated with rAID-1 gene clusters in Bacteroidales genomes.
Supplementary Table 8
Metagenomic results of rAID-1 genes derived from the HMP and MetaHit studies utilized in this study.
Supplementary Table 9
Metagenomic and metatranscriptomic results from analysis of rAID-1 genes from ref. 19.
Supplementary Table 10
Strains, plasmids and primers used in this study.
Rights and permissions
About this article
Cite this article
Ross, B.D., Verster, A.J., Radey, M.C. et al. Human gut bacteria contain acquired interbacterial defence systems. Nature 575, 224–228 (2019). https://doi.org/10.1038/s41586-019-1708-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1708-z
This article is cited by
-
Bacterial defences: mechanisms, evolution and antimicrobial resistance
Nature Reviews Microbiology (2023)
-
Community composition and the environment modulate the population dynamics of type VI secretion in human gut bacteria
Nature Ecology & Evolution (2023)
-
Microbiota-mediated colonization resistance: mechanisms and regulation
Nature Reviews Microbiology (2023)
-
Rhs NADase effectors and their immunity proteins are exchangeable mediators of inter-bacterial competition in Serratia
Nature Communications (2023)
-
Bacterial type VI secretion system (T6SS): an evolved molecular weapon with diverse functionality
Biotechnology Letters (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.