Abstract
Although glucose-sensing neurons were identified more than 50 years ago, the physiological role of glucose sensing in metazoans remains unclear. Here we identify a pair of glucose-sensing neurons with bifurcated axons in the brain of Drosophila. One axon branch projects to insulin-producing cells to trigger the release of Drosophila insulin-like peptide 2 (dilp2) and the other extends to adipokinetic hormone (AKH)–producing cells to inhibit secretion of AKH, the fly analogue of glucagon. These axonal branches undergo synaptic remodelling in response to changes in their internal energy status. Silencing of these glucose-sensing neurons largely disabled the response of insulin-producing cells to glucose and dilp2 secretion, disinhibited AKH secretion in corpora cardiaca and caused hyperglycaemia, a hallmark feature of diabetes mellitus. We propose that these glucose-sensing neurons maintain glucose homeostasis by promoting the secretion of dilp2 and suppressing the release of AKH when haemolymph glucose levels are high.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Raw mass spectrometry files have been deposited in the MassIVE database; with MassIVE accession ID: MSV000083796. All other raw data are available from the corresponding author on reasonable request.
References
Mayer, J. Glucostatic mechanism of regulation of food intake. N. Engl. J. Med. 249, 13–16 (1953).
Oomura, Y. et al. Reciprocal activities of the ventromedial and lateral hypothalamic areas of cats. Science 143, 484–485 (1964).
Parton, L. E. et al. Glucose sensing by POMC neurons regulates glucose homeostasis and is impaired in obesity. Nature 449, 228–232 (2007).
Levin, B. E. Neuronal glucose sensing: still a physiological orphan? Cell Metab. 6, 252–254 (2007).
Dus, M. et al. Nutrient sensor in the brain directs the action of the brain–gut axis in Drosophila. Neuron 87, 139–151 (2015).
López-Gambero, A. J., Martínez, F., Salazar, K., Cifuentes, M. & Nualart, F. Brain glucose-sensing mechanism and energy homeostasis. Mol. Neurobiol. 56, 769–796 (2019).
Dus, M., Min, S., Keene, A. C., Lee, G. Y. & Suh, G. S. B. Taste-independent detection of the caloric content of sugar in Drosophila. Proc. Natl Acad. Sci. USA 108, 11644–11649 (2011).
Pfeiffer, B. D. et al. Tools for neuroanatomy and neurogenetics in Drosophila. Proc. Natl Acad. Sci. USA 105, 9715–9720 (2008).
Tayler, T. D., Pacheco, D. A., Hergarden, A. C., Murthy, M. & Anderson, D. J. A neuropeptide circuit that coordinates sperm transfer and copulation duration in Drosophila. Proc. Natl Acad. Sci. USA 109, 20697–20702 (2012).
Kapan, N., Lushchak, O. V., Luo, J. & Nässel, D. R. Identified peptidergic neurons in the Drosophila brain regulate insulin-producing cells, stress responses and metabolism by coexpressed short neuropeptide F and corazonin. Cell. Mol. Life Sci. 69, 4051–4066 (2012).
Kim, S. K. & Rulifson, E. J. Conserved mechanisms of glucose sensing and regulation by Drosophila corpora cardiaca cells. Nature 431, 316–320 (2004).
Lee, G. & Park, J. H. Hemolymph sugar homeostasis and starvation-induced hyperactivity affected by genetic manipulations of the adipokinetic hormone-encoding gene in Drosophila melanogaster. Genetics 167, 311–323 (2004).
Sweeney, S. T., Broadie, K., Keane, J., Niemann, H. & O’Kane, C. J. Targeted expression of tetanus toxin light chain in Drosophila specifically eliminates synaptic transmission and causes behavioral defects. Neuron 14, 341–351 (1995).
Chen, T.-W. et al. Ultrasensitive fluorescent proteins for imaging neuronal activity. Nature 499, 295–300 (2013).
Miyamoto, T., Slone, J., Song, X. & Amrein, H. A fructose receptor functions as a nutrient sensor in the Drosophila brain. Cell 151, 1113–1125 (2012).
Ashcroft, F. M. & Rorsman, P. KATP channels and islet hormone secretion: new insights and controversies. Nat. Rev. Endocrinol. 9, 660–669 (2013).
Bryan, J. & Aguilar-Bryan, L. The ABCs of ATP-sensitive potassium channels: more pieces of the puzzle. Curr. Opin. Cell Biol. 9, 553–559 (1997).
Masuyama, K., Zhang, Y., Rao, Y. & Wang, J. W. Mapping neural circuits with activity-dependent nuclear import of a transcription factor. J. Neurogenet. 26, 89–102 (2012).
Zhang, Y. Q., Rodesch, C. K. & Broadie, K. Living synaptic vesicle marker: synaptotagmin–GFP. Genesis 34, 142–145 (2002).
Rulifson, E. J., Kim, S. K. & Nusse, R. Ablation of insulin-producing neurons in flies: growth and diabetic phenotypes. Science 296, 1118–1120 (2002).
Feinberg, E. H. et al. GFP reconstitution across synaptic partners (GRASP) defines cell contacts and synapses in living nervous systems. Neuron 57, 353–363 (2008).
Lima, S. Q. & Miesenböck, G. Remote control of behavior through genetically targeted photostimulation of neurons. Cell 121, 141–152 (2005).
Macpherson, L. J. et al. Dynamic labelling of neural connections in multiple colours by trans-synaptic fluorescence complementation. Nat. Comm. 6, 10024 (2015).
Cao, G. et al. Genetically targeted optical electrophysiology in intact neural circuits. Cell 154, 904–913 (2013).
Lee, K.-S. et al. Drosophila short neuropeptide F signalling regulates growth by ERK-mediated insulin signalling. Nat. Cell Biol. 10, 468–475 (2008).
Shang, Y. et al. Short neuropeptide F is a sleep-promoting inhibitory modulator. Neuron 80, 171–183 (2013).
Garczynski, S. F., Brown, M. R., Shen, P., Murray, T. F. & Crim, J. W. Characterization of a functional neuropeptide F receptor from Drosophila melanogaster. Peptides 23, 773–780 (2002).
Thorens, B. Neural regulation of pancreatic islet cell mass and function. Diabetes Obes. Metab. 16 (Suppl 1), 87–95 (2014).
Pipeleers, D. G. Heterogeneity in pancreatic β-cell population. Diabetes 41, 777–781 (1992).
Bonner-Weir, S. & Aguayo-Mazzucato, C. Pancreatic β-cell heterogeneity revisited. Nature 535, 365–366 (2016).
Nern, A., Pfeiffer, B. D. & Rubin, G. M. Optimized tools for multicolor stochastic labeling reveal diverse stereotyped cell arrangements in the fly visual system. Proc. Natl Acad. Sci. USA 112, E2967–E2976 (2015).
Jourjine, N., Mullaney, B. C., Mann, K. & Scott, K. Coupled sensing of hunger and thirst signals balances sugar and water consumption. Cell 166, 855–866 (2016).
Ai, M. et al. Acid sensing by the Drosophila olfactory system. Nature 468, 691–695 (2010).
Burgess, A. et al. Loss of human Greatwall results in G2 arrest and multiple mitotic defects due to deregulation of the cyclin B–Cdc2/PP2A balance. Proc. Natl Acad. Sci. USA 107, 12564–12569 (2010).
Liu, Q. et al. Branch-specific plasticity of a bifunctional dopamine circuit encodes protein hunger. Science 356, 534–539 (2017). https://doi.org/10.1126/science.aal3245.
Park, S. et al. A genetic strategy to measure circulating Drosophila insulin reveals genes regulating insulin production and secretion. PLoS Genet. 10, e1004555 (2014).
Chen, W., Hwang, Y. Y., Gleaton, J. W., Titus, J. K. & Hamlin, N. J. Optimization of a peptide extraction and LC–MS protocol for quantitative analysis of antimicrobial peptides. Future Sci. 5, FSO348 (2018).
Rappsilber, J., Mann, M. & Ishihama, Y. Protocol for micro-purification, enrichment, pre-fractionation and storage of peptides for proteomics using StageTips. Nat. Protoc. 2, 1896–1906 (2007).
Cox, J. et al. Andromeda: a peptide search engine integrated into the MaxQuant environment. J. Proteome Res. 10, 1794–1805 (2011).
MacLean, B. et al. Skyline: an open source document editor for creating and analyzing targeted proteomics experiments. Bioinformatics 26, 966–968 (2010).
Johnson, R. S., Martin, S. A., Biemann, K., Stults, J. T. & Watson, J. T. Novel fragmentation process of peptides by collision-induced decomposition in a tandem mass spectrometer: differentiation of leucine and isoleucine. Anal. Chem. 59, 2621–2625 (1987).
Roepstorff, P. & Fohlman, J. Proposal for a common nomenclature for sequence ions in mass spectra of peptides. Biol. Mass Spectrom. 11, 601 (1984).
Gáliková, M. et al. Energy homeostasis control in Drosophila adipokinetic hormone mutants. Genetics 201, 665–683 (2015).
Saadipour, K. et al. The transmembrane domain of the p75 neurotrophin receptor stimulates phosphorylation of the TrkB tyrosine kinase receptor. J. Biol. Chem. 292, 16594–16604 (2017).
Nicolaï, L. J. J. et al. Genetically encoded dendritic marker sheds light on neuronal connectivity in Drosophila. Proc. Natl Acad. Sci. USA 107, 20553–20558 (2010).
Acknowledgements
We thank M. Kanai and J.-Y. Park for providing expertise on the two-choice behaviour assay and physiology. We also thank S. K. Kim, J. Veenstra, J. H. Park and E. Hafen for providing antibodies used in this study; B. Levin, G. Schwartz, J. W. Sohn, H. Kim and the Suh laboratory members for stimulating discussion. This work is supported by NIH R01 grants (R01DK116294, R01DK106636 to G.S.B.S. and NINDS P30NS050276 to T.A.N.), KAIST Chancellor’s fund and a grant from Samsung Science & Technology Foundation (G.S.B.S).
Author information
Authors and Affiliations
Contributions
Y.O. performed nearly all immunohistochemistry, calcium imaging, behaviour testing, haemolymph glycaemia measurement, statistical analyses and figure design. J.S.-Y.L., H.J.M. and B.G. carried out the Gal4 screen using the two-choice assay. H.E.B. and T.A.N. conducted mass spectrometry analyses to measure dilp2 and AKH levels in haemolymph. K.S. performed dot blot assays to measure dilp2 and AKH levels in haemolymph. J.G.W. performed feeding assays and assisted in measuring haemolymph glycaemia. F.A. assisted with the CaLexA experiment. G.S.B.S. supervised the project and provided intellectual support. Y.O. and G.S.B.S. wrote the manuscript with inputs from other authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Peer review information Nature thanks Mark L. Evans and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
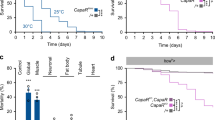
Extended Data Fig. 1 Identification of neurons that are required for the starvation-induced nutrient selection.
a, The result of a screen of VT-Gal4 lines when bearing UAS-Kir2.1, tub-Gal80ts that failed to select d-glucose after 24 h of starvation. b, Expression patterns of 18 candidate Gal4 lines isolated from the screen. Scale bar, 100 µm. Brain images from FlyLight Gal4 LexA collection are shown. White boxes denote a pair of the dorsolateral cells. c, Expression patterns of VT58471-Gal4>UAS-mCD8::GFP line, VT43147-Gal4>UAS-mCD8::GFP line and Crz-Gal4>UAS-mCD8::GFP line in the brain, the VNC and a part of the foregut, stained with anti-GFP (green) antibody. Neuropil is stained with nc82 (bruchpilot, magenta) antibody. Cell bodies of candidate neurons (arrowheads) and their projections to the foregut (arrows) are shown. Scale bar, 100 µm. Images are z-stacked projections. d, Inactivation of the candidate neurons by expressing UAS-Kir2.1, tub-Gal80ts under the control of VT58471-Gal4, VT43147-Gal4 or Crz-Gal4 at 30 °C blunts a preference for d-glucose in starved flies. ***P < 0.001; one-way ANOVA with Tukey post hoc test. See Supplementary Table 1 for the sample sizes and statistical analyses.
Extended Data Fig. 2 Axons and dendrites of CN neurons and a LexA line that labels a pair of CN neurons.
a, Axons and dendrites of CN neurons in the brain visualized by an axonal marker, UAS-Syt::eGFP, and a dendritic marker, UAS-DenMark, under the control of CN-Gal4, stained with anti-GFP (green) and anti-dsRed (DenMark, magenta, see Methods) antibodies. Arrowheads denote CN cell bodies and arrows indicates CN axons (left) and dendrites (middle). b, The brain of a fly carrying R20F11-LexA and LexAop-GFP, stained with anti-GFP (green) antibody. Cell bodies of CN neurons are stained with anti-Crz (magenta) antibody. Arrowheads indicate cell bodies of the dorsolateral CN neurons. Scale bar, 20 µm. c, Inactivation of CN neurons by expressing LexAop-TNT under the control of R20F11-LexA blunts a preference for d-glucose in starved flies. SEZ, subesophageal zone. Scale bar, 50 µm. Z-stacked projections are shown. ***P < 0.001; one-way ANOVA with Tukey post hoc test. See Supplementary Table 1 for the sample sizes and statistical analyses.
Extended Data Fig. 3 The activity of CN neurons is stimulated by glucose or pyruvate, but not by sucrose or a mixture of essential amino acids.
a, Representative traces of CN neuronal activity in response to 2 mM, 10 mM and 15 mM d-glucose. b–e, Quantifications of CN responses to 1 mM, 2 mM, 5 mM, 10 mM, 15 mM and 20 mM d-glucose. When 1 to 5 mM d-glucose were applied, all parameters increased. When higher concentrations (10 to 20 mM d-glucose) were applied, the oscillation number (c) and oscillation frequency (e) decreased, while the peak amplitude (b) and duration (d) increased. f, Representative traces of CN neuronal activity in response to 20 mM sucrose, 20 mM pyruvate and a mixture of ten EAAs. g–j, Quantifications of CN neuronal responses to 20 mM sucrose, 20 mM pyruvate and a mixture of 10 EAAs. *P < 0.05 and **P < 0.01; one-way ANOVA with Tukey post hoc. See Supplementary Table 1 for the sample sizes and statistical analyses.
Extended Data Fig. 4 Glut1, Hex-C, KATP channel and voltage-gated calcium channel are required in CN neurons to respond to glucose, and the activity of CN neuron is controlled by the internal energy state in live animals.
a, Inactivation of Glut1 by expressing UAS-Glut1 RNAi under the control of CN-Gal4 (but not other glucose transporters) blunts a preference for d-glucose in starved flies. b, Inactivation of Hex-C by expressing UAS-HexC RNAi under the control of CN-Gal4 blunts a preference for d-glucose, but not other hexokinases, in starved flies (Hex-T2 and Hex-A). c, Inactivation of KATP channel or voltage-gated calcium channel by expressing UAS-SUR1 RNAi or UAS-Ca-α1D RNAi by CN-Gal4 blunts a preference for d-glucose in starved flies. d–h, Representative traces (d) and quantifications (e–h) of calcium responses to 1 mM glucose by CN neurons of flies in which Glut1, SUR1 or voltage-gated calcium channel subunit (Ca-α1D) was knocked down by RNAi, or those of control flies. i, j, Representative images (i) and quantifications of native CaLexA-driven GFP intensity from CN cell bodies (j) of fed, starved or refed flies carrying CN-Gal4 and UAS-CaLexA (UAS-mLexA-VP16-NFAT, LexAop-GFP). Cell bodies of CN neurons are stained with anti-Crz (magenta) antibody. Scale bar, 20 µm. Images are z-stacked projections. *P < 0.05, **P < 0.01 and ***P < 0.001; one-way ANOVA with Tukey post hoc test. See Supplementary Table 1 for the sample sizes and statistical analyses.
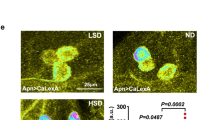
Extended Data Fig. 5 Nutrient-dependent plasticity does not occur in the majority of Crz neurons except a pair of the dorsolateral CN neurons.
a–d, The relative fluorescence intensities of Syt–GFP signals (a, b) and the branch lengths (c, d) of axon 1 (A1) (neurite to IPCs, a, c) and axon 2 (A2) (neurite to CC, b, d) in fed, starved or refed flies carrying Crz-Gal4 and UAS-Syt::GFP. e–h, Representative images (e) and quantifications (f–h) of the number of Syt–GFP+ puncta in fed, starved or refed flies carrying Crz-Gal4 and UAS-Syt::GFP in the dorsal lateral area (DL, f), the medial lateral area (ML, g) and the SEZ. In h, Arrowheads and arrows indicate Syt–GFP+ puncta on A1 and A2, respectively. Scale bar, 50 µm. Images are z-stacked projections. *P < 0.05, **P < 0.01 and ***P < 0.001; one-way ANOVA with Tukey post hoc test. See Supplementary Table 1 for the sample sizes and statistical analyses.
Extended Data Fig. 6 CN neurons expressing P2X2 receptor can be readily activated by ATP application, and Dh44 neurons and CN neurons are not functionally coupled.
a, b, Average GCaMP traces (a) and their ΔF/F (max) quantifications (b) of CN neurons when exposed to 2.5 mM ATP. c, Average GCaMP trace of flies carrying Dh44-Gal4, UAS-GCaMP6s in response to ATP. d, Average GCaMP trace of flies carrying Dh44-Gal4, UAS-GCaMP6s, Dh44-LexA and LexAop-P2X2 in response to ATP. e, Average GCaMP trace of flies carrying Crz-Gal4, UAS-GCaMP6s, Dh44-LexA and LexAop-P2X2 in response to ATP. f, Average GCaMP trace of flies carrying Dh44-Gal4, UAS-GCaMP6s, R20F11-LexA and LexAop-P2X2 in response to ATP. ***P < 0.001; one-way ANOVA with Tukey post hoc test. See Supplementary Table 1 for the sample sizes and statistical analyses.
Extended Data Fig. 7 The circulating levels of dilp2 and AKH in haemolymph are measured by mass spectrometry and dot blot assay.
a, Sequences of dilp2, AKH, tryptic peptide of dilp2 B chain and tryptic peptide of AKH. We detected dilp2 B chain at m/z (mass to charge ratio): 369.1785 (TLCSEK, M2H+: 369.1785) and AKH at m/z: 497.2374 (QLTFSPDW, M2H+: 497.2374). b, Nomenclature and m/z values of fragment ions (N-terminal directed ‘a’ and ‘b’ ions, as well as, C-terminal directed ‘y’ ions) which is driven by dilp2 B chain and AKH. c, d, Relative extracted ion intensities for the dilp2 B chain and its fragment ions in each trial (c), and AKH and its fragment ions in each trial (d) generated from the haemolymph of fed flies in which CN neurons were inactivated, or those of control flies; see Methods. e, f, A dot blot (e) and its quantification (f) show the levels of dilp2 in the haemolymph of wild type (w1118), UAS-Kir2.1/CN-Gal4;dilp2-HA and UAS-Kir2.1/+;dilp2-HA flies, probed with anti-HA antibody to detect dilp2. Because w1118 flies do not express dilp2–HA, they were used as a negative control. g, h, A dot blot (g) and its quantification (h) show the levels of AKH in the haemolymph of CN-Gal4/+, UAS-Kir2.1/+ and UAS-Kir2.1/CN-Gal4 flies, probed with anti-AKH antibody. The intensity of black dots in the red dashed circle represents the quantity of dilp2 or AKH that was later normalized to Ponceau staining. For gel source data, see Supplementary Fig. 1. **P < 0.01 and ***P < 0.001; unpaired two-tailed t-test (f) and one-way ANOVA with Tukey post hoc test (h). See Supplementary Table 1 for the sample sizes and statistical analyses.
Extended Data Fig. 8 Calcium responses to D-glucose or KATP channel blocker by IPCs of flies in which CN neurons were silenced or sNPF receptor was rendered nonfunctional.
a, b, Individual traces of IPCs in control flies carrying R20F11-LexA, dilp2-Gal4 and UAS-GCaMP6s (a) and in experimental flies carrying R20F11-LexA, LexAop-TNT, dilp2-Gal4 and UAS-GCaMP6s (b) responding to d-glucose. c, Average GCaMP traces and ΔF/F (max) quantifications from IPCs of fed flies in which CN neurons were inactivated by TNT in response to KATP channel blocker glibenclamide, or those of control flies. d, IPCs partition into three subpopulations depending on their response to glibenclamide; see Methods. e–f, Individual traces of IPCs in control flies (e) and in flies in which CN neurons were inactivated by TNT (f) responding to glibenclamide. Because a saturating concentration of d-glucose (20 mM) was used to quantify the populations of IPCs, we used 100 µM glibenclamide, a saturating concentration for glibenclamide according to our control experiments. g, IPCs partition into three subpopulations according to their response to glucose with or without sNPF receptor; see Methods. h, i, Individual traces of IPCs in control flies (h) and in flies in which sNPF receptor was rendered non-functional by expressing a dominant negative allele of sNPF receptor in the IPCs (i) responding to d-glucose. ***P < 0.001; unpaired two-tailed t-test (c). See Supplementary Table 1 for the sample sizes and statistical analyses.
Extended Data Fig. 9 Crz is not required in CN neurons for the two-choice behaviour and sugar-evoked Crz secretion.
a, b, Expression of GFP in the brains of flies carrying Crz-Gal4, UAS-mCD8::GFP (a) or Crz receptor (CrzR)-Gal4, UAS-mCD8::GFP (b). Scale bar, 100 µm. c, d, Knockdown of Crz or Crz receptor in flies carrying Crz-Gal4 and UAS-Crz RNAi (c) or CrzR-Gal4 and UAS-CrzR RNAis (d), respectively, does not impair the selection of d-glucose in starved flies. e, Immunoreactivity of intracellular Crz in CN neurons, probed with anti-Crz antibody, when the brains were incubated in 80 mM sucrose, 80 mM d-glucose (d-Glc), 80 mM d-glucose mixed with 0.5 µM TTX (d-Glc/TTX) or 80 mM l-glucose (l-Glc) in AHL. Scale bar, 5 µm. Images are z-stacked projections. One-way ANOVA with Tukey post hoc test (c, e) or unpaired two-tailed t-test (d). See Supplementary Table 1 for the sample sizes and statistical analyses.
Extended Data Fig. 10 Activity of IPCs was stimulated by sNPF application, and the circulating trehalose level in which CN neurons had beeninactivated was reduced.
a, b, Average GCaMP traces (a) and ΔF/F (max) quantifications (b) from the IPCs in response to 80 µM sNPF in AHL or DMSO in AHL. c, An increase in circulating trehalose level in fed or starved flies in which CN neurons were inactivated. The levels of circulating trehalose in flies carrying CN-Gal4 and UAS-Kir2.1 and its heterozygote controls (CN-Gal4/+ and UAS-Kir2.1/+). d, A working model of the anatomic connectivities between CN neurons and IPCs (green), and between CN neurons and AKH-producing cells (red). CN neurons regulate glucose homeostasis by counter-balancing the activities of IPCs and AKH-producing cells through sNPF neurotransmitter that activates IPCs and inactivates AKH-producing cells. *P < 0.05, **P < 0.01 and ***P < 0.001; unpaired two-tailed t-test (b) and one-way ANOVA with Tukey post hoc test (c). See Supplementary Table 1 for the sample sizes and statistical analyses.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-3 and Supplementary Figure 1.
Source data
Rights and permissions
About this article
Cite this article
Oh, Y., Lai, J.SY., Mills, H.J. et al. A glucose-sensing neuron pair regulates insulin and glucagon in Drosophila. Nature 574, 559–564 (2019). https://doi.org/10.1038/s41586-019-1675-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1675-4
This article is cited by
-
Postprandial sodium sensing by enteric neurons in Drosophila
Nature Metabolism (2024)
-
Descending GABAergic pathway links brain sugar-sensing to peripheral nociceptive gating in Drosophila
Nature Communications (2023)
-
A rapid and bidirectional reporter of neural activity reveals neural correlates of social behaviors in Drosophila
Nature Neuroscience (2023)
-
Dietary cysteine drives body fat loss via FMRFamide signaling in Drosophila and mouse
Cell Research (2023)
-
Intermittent fasting and Alzheimer's disease—Targeting ketone bodies as a potential strategy for brain energy rescue
Metabolic Brain Disease (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.