Abstract
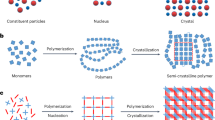
Inorganic materials have essential roles in society, including in building construction, optical devices, mechanical engineering and as biomaterials1,2,3,4. However, the manufacture of inorganic materials is limited by classical crystallization5, which often produces powders rather than monoliths with continuous structures. Several precursors that enable non-classical crystallization—such as pre-nucleation clusters6,7,8, dense liquid droplets9,10, polymer-induced liquid precursor phases11,12,13 and nanoparticles14—have been proposed to improve the construction of inorganic materials, but the large-scale application of these precursors in monolith preparations is limited by availability and by practical considerations. Inspired by the processability of polymeric materials that can be manufactured by crosslinking monomers or oligomers15, here we demonstrate the construction of continuously structured inorganic materials by crosslinking ionic oligomers. Using calcium carbonate as a model, we obtain a large quantity of its oligomers (CaCO3)n with controllable molecular weights, in which triethylamine acts as a capping agent to stabilize the oligomers. The removal of triethylamine initiates crosslinking of the (CaCO3)n oligomers, and thus the rapid construction of pure monolithic calcium carbonate and even single crystals with a continuous internal structure. The fluid-like behaviour of the oligomer precursor enables it to be readily processed or moulded into shapes, even for materials with structural complexity and variable morphologies. The material construction strategy that we introduce here arises from a fusion of classic inorganic and polymer chemistry, and uses the same cross-linking process for the manufacture the materials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Meyers, M. A., Chen, P. Y., Lin, A. Y. M. & Seki, Y. Biological materials: structure and mechanical properties. Prog. Mater. Sci. 53, 1–206 (2008).
Riley, F. L. Silicon nitride and related materials. J. Am. Ceram. Soc. 83, 245–265 (2000).
Xu, H. H. K. et al. Calcium phosphate cements for bone engineering and their biological properties. Bone Res. 5, 17056 (2017).
Hu, C. L. & Li, Z. J. A review on the mechanical properties of cement-based materials measured by nanoindentation. Constr. Build. Mater. 90, 80–90 (2015).
Thanh, N. T. K., Maclean, N. & Mahiddine, S. Mechanisms of nucleation and growth of nanoparticles in solution. Chem. Rev. 114, 7610–7630 (2014).
Gebauer, D., Kellermeier, M., Gale, J. D., Bergstrom, L. & Colfen, H. Pre-nucleation clusters as solute precursors in crystallisation. Chem. Soc. Rev. 43, 2348–2371 (2014).
Gebauer, D., Volkel, A. & Colfen, H. Stable prenucleation calcium carbonate clusters. Science 322, 1819–1822 (2008).
Kellermeier, M. et al. Colloidal stabilization of calcium carbonate prenucleation clusters with silica. Adv. Funct. Mater. 22, 4301–4311 (2012).
Wolf, S. E. et al. Carbonate-coordinated metal complexes precede the formation of liquid amorphous mineral emulsions of divalent metal carbonates. Nanoscale 3, 1158–1165 (2011).
Smeets, P. J. M. et al. A classical view on nonclassical nucleation. Proc. Natl Acad. Sci. USA 114, E7882–E7890 (2017).
Gower, L. B. & Odom, D. J. Deposition of calcium carbonate films by a polymer-induced liquid-precursor (PILP) process. J. Cryst. Growth 210, 719–734 (2000).
Cheng, X. G. & Gower, L. B. Molding mineral within microporous hydrogels by a polymer-induced liquid-precursor (PILP) process. Biotechnol. Prog. 22, 141–149 (2006).
Wolf, S. E. et al. Strong stabilization of amorphous calcium carbonate emulsion by ovalbumin: gaining insight into the mechanism of ‘polymer-induced liquid precursor’ processes. J. Am. Chem. Soc. 133, 12642–12649 (2011).
De Yoreo, J. J. et al. Crystallization by particle attachment in synthetic, biogenic, and geologic environments. Science 349, aaa6760 (2015).
Koltzenburg, S., Maskos, M. & Nuyken, O. (eds) Polymer Chemistry Ch. 14 (Springer, 2017).
Lewis, J. A. Colloidal processing of ceramics. J. Am. Ceram. Soc. 83, 2341–2359 (2000).
Eckel, Z. C. et al. Additive manufacturing of polymer-derived ceramics. Science 351, 58–62 (2016).
Liu, H. L., Gong, Q. H., Yue, Y. H., Guo, L. & Wang, X. Sub-1 nm nanowire based superlattice showing high strength and low modulus. J. Am. Chem. Soc. 139, 8579–8585 (2017).
Truby, R. L. & Lewis, J. A. Printing soft matter in three dimensions. Nature 540, 371–378 (2016).
Feinle, A., Elsaesser, M. S. & Husing, N. Sol-gel synthesis of monolithic materials with hierarchical porosity. Chem. Soc. Rev. 45, 3377–3399 (2016).
Caminade, A. M., Hey-Hawkins, E. & Manners, I. Smart inorganic polymers. Chem. Soc. Rev. 45, 5144–5146 (2016).
Drake, K., Mukherjee, I., Mirza, K., Ji, H. F. & Wei, Y. Phenylethynyl and phenol end-capping studies of polybiphenyloxydiphenylsilanes for cross-linking and enhanced thermal stability. Macromolecules 44, 4107–4115 (2011).
Sautter, A., Thalacker, C., Heise, B. & Wurthner, F. Hydrogen bond-directed aggregation of diazadibenzoperylene dyes in low-polarity solvents and the solid state. Proc. Natl Acad. Sci. USA 99, 4993–4996 (2002).
Liu, Z. M. et al. Ionization controls for biomineralization-inspired CO2 chemical looping at constant room temperature. Phys. Chem. Chem. Phys. 17, 10080–10085 (2015).
Franke, D. & Svergun, D. I. DAMMIF, a program for rapid ab-initio shape determination in small-angle scattering. J. Appl. Crystallogr. 42, 342–346 (2009).
Demichelis, R., Raiteri, P., Gale, J. D., Quigley, D. & Gebauer, D. Stable prenucleation mineral clusters are liquid-like ionic polymers. Nat. Commun. 2, 590 (2011).
Michel, F. M. et al. Structural characteristics of synthetic amorphous calcium carbonate. Chem. Mater. 20, 4720–4728 (2008).
Weaver, J. C. et al. Analysis of an ultra hard magnetic biomineral in chiton radular teeth. Mater. Today 13, 42–52 (2010).
Broz, M. E., Cook, R. F. & Whitney, D. L. Microhardness, toughness, and modulus of Mohs scale minerals. Am. Mineral. 91, 135–142 (2006).
Hirano, S., Yogo, T. & Kikuta, K. Synthetic calcite single-crystals for optical-device. Prog. Cryst. Growth Charact. Mater. 23, 341–367 (1992).
Nielsen, S. S. et al. BioXTAS RAW, a software program for high-throughput automated small-angle X-ray scattering data reduction and preliminary analysis. J. Appl. Crystallogr. 42, 959–964 (2009).
Franke, D. et al. ATSAS 2.8: a comprehensive data analysis suite for small-angle scattering from macromolecular solutions. J. Appl. Crystallogr. 50, 1212–1225 (2017).
Juhás, P., Davis, T., Farrow, C. L. & Billinge, S. J. L. PDFgetX3: a rapid and highly automatable program for processing powder diffraction data into total scattering pair distribution functions. J. Appl. Crystallogr. 46, 560–566 (2013).
Van Der Spoel, D. et al. GROMACS: fast, flexible, and free. J. Comput. Chem. 26, 1701–1718 (2005).
Van der Spoel, D. et al. Gromacs User Manual version 4.5 (2010).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996).
Berendsen, H. J., Postma, J. V., van Gunsteren, W. F., DiNola, A. & Haak, J. Molecular dynamics with coupling to an external bath. J. Chem. Phys. 81, 3684–3690 (1984).
Darden, T., York, D. & Pedersen, L. Particle mesh Ewald: an N·log(N) method for Ewald sums in large systems. J. Chem. Phys. 98, 10089–10092 (1993).
Shen, J. W., Li, C., van der Vegt, N. F. & Peter, C. Understanding the control of mineralization by polyelectrolyte additives: simulation of preferential binding to calcite surfaces. J. Phys. Chem. C 117, 6904–6913 (2013).
Raiteri, P., Gale, J. D., Quigley, D. & Rodger, P. M. Derivation of an accurate force-field for simulating the growth of calcium carbonate from aqueous solution: a new model for the calcite−water interface. J. Phys. Chem. C 114, 5997–6010 (2010).
Jorgensen, W. L. & Tiradorives, J. The Opls potential functions for proteins. Energy minimizations for crystals of cyclic peptides and crambin. J. Am. Chem. Soc. 110, 1657–1666 (1988).
Nielsen, M. H., Aloni, S. & De Yoreo, J. J. In situ TEM imaging of CaCO3 nucleation reveals coexistence of direct and indirect pathways. Science 345, 1158–1162 (2014).
Smeets, P. J., Cho, K. R., Kempen, R. G., Sommerdijk, N. A. & De Yoreo, J. J. Calcium carbonate nucleation driven by ion binding in a biomimetic matrix revealed by in situ electron microscopy. Nat. Mater. 14, 394–399 (2015).
Brinker, C. J. & Scherer, G. W. Sol-gel-glass. 1. Gelation and gel structure. J. Non-Cryst. Solids 70, 301–322 (1985).
Oshida, K. et al. Analysis of pore structure of activated carbon-fibers using high-resolution transmission electron-microscopy and image-processing. J. Mater. Res. 10, 2507–2517 (1995).
Ihli, J., Kulak, A. N. & Meldrum, F. C. Freeze-drying yields stable and pure amorphous calcium carbonate (ACC). Chem. Commun. 49, 3134–3136 (2013).
Konrad, F., Gallien, F., Gerard, D. E. & Dietzel, M. Transformation of amorphous calcium carbonate in air. Cryst. Growth Des. 16, 6310–6317 (2016).
Ihli, J. et al. Dehydration and crystallization of amorphous calcium carbonate in solution and in air. Nat. Commun. 5, 3169 (2014).
Xu, X., Han, J. T., Kim, D. H. & Cho, K. Two modes of transformation of amorphous calcium carbonate films in air. J. Phys. Chem. B 110, 2764–2770 (2006).
Tao, J. H., Zhou, D. M., Zhang, Z. S., Xu, X. R. & Tang, R. K. Magnesium-aspartate-based crystallization switch inspired from shell molt of crustacean. Proc. Natl Acad. Sci. USA 106, 22096–22101 (2009).
Kim, Y. Y. et al. An artificial biomineral formed by incorporation of copolymer micelles in calcite crystals. Nat. Mater. 10, 890–896 (2011).
Sánchez-Iglesias, A. et al. High-yield seeded growth of monodisperse pentatwinned gold nanoparticles through thermally induced seed twinning. J. Am. Chem. Soc. 139, 107–110 (2017).
Politi, Y., Arad, T., Klein, E., Weiner, S. & Addadi, L. Sea urchin spine calcite forms via a transient amorphous calcium carbonate phase. Science 306, 1161–1164 (2004).
Li, L. et al. Bio-inspired enamel repair via Glu-directed assembly of apatite nanoparticles: an approach to biomaterials with optimal characteristics. Adv. Mater. 23, 4695–4701 (2011).
Koeppe, B. et al. Polar solvent fluctuations drive proton transfer in hydrogen bonded complexes of carboxylic acid with pyridines: NMR, IR and ab initio MD study. Phys. Chem. Chem. Phys. 19, 1010–1028 (2017).
Shawoosh, A. S. & Kutub, A. A. An investigation of the electrical, optical and DSC properties of a copper phosphate-glass composition. J. Mater. Sci. 28, 5060–5064 (1993).
Acknowledgements
We thank J. J. De Yoreo, H. Cölfen, P. Fratzl, N. A. J. M. Sommerdijk, D. Joester and L. B. Gower for discussions; Y. Li and Y. Qiu for help with SAXS data analysis; C. Yang and B. Wang for assistance with synchrotron SAXS at the Shanghai Synchrotron Radiation Facility; C. Jin, F. Chen, W. Wang and Y. Wang for assistance with electron microscopy; J. Liu for assistance with XRD; and M. Yu and Y. Liu for assistance with NMR. This work was supported by the National Natural Science Foundation of China (21625105 and 21805241) and the China Postdoctoral Science Foundation (2017M621909 and 2018T110585). We thank W. Liu for his help and inspiration.
Author information
Authors and Affiliations
Contributions
Z.L. and R.T. initiated the project. Z.L. performed the syntheses and the FTIR, MS, NMR and XRD experiments, the calcite repair and conductivity experiments; C.S. carried out the nanoindentation and SEM experiments; B.J. performed the TEM experiments; Z.Z. performed the computer simulations; Y.Z. acquired the synchrotron SAXS and SEM data; R.T. and Z.L. supervised and supported the project; and Z.L. and X.X. analysed the data. The manuscript was written by Z.L. and R.T. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Peer review information Nature thanks Kazuo Onuma and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
Extended Data Fig. 1 ESI-MS analysis of (CaCO3)n oligomers.
a, Negative-ion mode analysis of CaCl2 and TEA in ethanol solution. b, Negative-ion mode analysis of (CaCO3)n oligomers after 5 days, indicating that TEA has a long-term stabilization effect. n represents the number of Ca2+:CO32− units in one (CaCO3)n oligomer. c, d, Positive ion mode analysis of CaCl2 and TEA mixed ethanol solution (c) and (CaCO3)n oligomers (d). e, ESI tandem MS analysis of the oligomers with m/z 368, showing only one fragmentation peak at around m/z 260 with negative charges. Because no TEA could be detected in the MS–MS peak (the intensity below m/z 200 is zero), this indicates that no TEA was present in the species with m/z 368.
Extended Data Fig. 2 Synchrotron SAXS, liquid-cell TEM, dynamic light scattering and viscosity analyses of (CaCO3)n oligomers and their aggregates.
a, Scattering plots of different concentrations of (CaCO3)n measured by synchrotron SAXS. The red curve is the fitting result from DAMMIF. At a concentration of 57.0 mg l−1 an increase in the scattered intensity was detected, and this broad shoulder peak indicated the aggregation of (CaCO3)n with a large distribution of sizes. A pronounced maxima of the scattered intensity could be observed at a concentration of 285 mg l−1, demonstrating inter-oligomer correlations induced by the formation of larger (CaCO3)n aggregates. b, Size distribution function of oligomer aggregates at 57.0 mg l−1. The inset shows the simulated shape of aggregates, indicating the existence of chain-like structures, including branches at high concentrations. c, Liquid-cell TEM of the aggregates of the (CaCO3)n oligomers showing a chain-like contrast; the boundary between the background (blue) and sample (yellow) is enhanced in the false colour image on the right, and the chain-like aggregates are marked with arrows. d, Viscosity of (CaCO3)n oligomers measured at 5 °C (n ≥ 3). The change in the viscosity with concentration is better fitted with the Huggins equation (R2 = 0.98) than with the Einstein equation (R2 = 0.94), indicating that the shape of the aggregates in solution was more chain-like than spherical. e, The hydrodynamic diameter of (CaCO3)n oligomers or their aggregates at different oligomer concentrations measured by dynamic light scattering with a Ca:TEA ratio of 1:20. The increase (decrease) of the hydrodynamic diameter is reversible by condensing (diluting) the (CaCO3)n oligomer solution. f, Reversible aggregations and disaggregations of the (CaCO3)n oligomers revealed by synchrotron SAXS. g, Scheme of the reversible aggregations and disaggregations of the oligomer unit are controllable by the concentration changes. h, In situ liquid-cell TEM observation of (CaCO3)n oligomers. The chain aggregates remain in dynamical change (aggregation–disaggregation) states.
Extended Data Fig. 3 FTIR spectra of gel-like (CaCO3)n oligomers.
a, FTIR spectra showing the peak corresponding to the carbonate group in (CaCO3)n. b, The C–N bond in TEA. c, The N···H–O bond between TEA and protonated carbonate55. d, The spectra between 400 cm−1 and 4,000 cm−1 are almost the same for ethanol and gel-like oligomers, which confirms that ethanol is the major component in the gel-like (CaCO3)n oligomers. e, The peak at 3,340 cm−1 was contributed by the –OH group of ethanol, which obstructed the signal from H in N···H–O. f, The spectrum of ethyl acetate and ethyl acetate-based gel-like (CaCO3)n oligomers. The ethanol had been completely removed from the ethyl acetate-based gel-like (CaCO3)n oligomers, because the specific peaks of ethanol were not detected; by contrast, the signal at 3,000–4,000 cm−1 was detected. g, The peak at 3,555 cm−1 was contributed by H in the N···H–O bond.
Extended Data Fig. 4 Molecular dynamics simulation of the TEA-stabilized (CaCO3)n.
a, Simulated size distribution of (CaCO3)n clusters in the presence and absence of TEA. b, The branch structure in a (CaCO3)5 cluster without TEA. The circle shows the branching site. c, The branch structure in the large cluster aggregates without TEA. d, There is no branch structure in a cluster of (CaCO3)7 with TEA stabilization. e, A branch structure in (CaCO3)15 even with TEA stabilization. These results demonstrate that the stabilization effect of TEA promotes linear growth of (CaCO3)n.
Extended Data Fig. 5 (CaCO3)n oligomers in ethanol, DMSO and water.
a, d, g, Conductivity of dispersed oligomer solution with ethanol, DMSO or water as solvent. After high-speed centrifugation and redispersion of (CaCO3)n oligomers in ethanol, DMSO or water, the conductivity of the oligomer solution remained steady at 1.2 μS cm−1 in ethanol (a); the conductivity increased to 4.6 μS cm−1 in DMSO and then decreased to 3.8 μS cm−1 within 1 h in DMSO (d); the conductivity increased to 161.2 μS cm−1 but rapidly decreased to 72.0 μS cm−1 within 6 min in water (g). b, e, h, 1H NMR measurement of the ethyl group of TEA (pure or bound with oligomer) in ethanol (b), DMSO (e) and water (h). The results revealed that the chemical shift of 1H of the ethyl group of pure TEA was 2.56 ppm in ethanol and 2.67 ppm in the oligomer solution (b), and this change in chemical shift can be attributed to the interaction between the TEA and the carbonates in the oligomers. After two days, the unchanged chemical shift of 2.68 ppm corroborated the stable capping effects of the TEA in ethanol. In DMSO (e), two signals—2.57 and 2.42 ppm—were attributed to bonded and de-bonded TEA, respectively. In water (h), only the peak for de-bonded TEA in the form of [H-TEA]+ could be detected at 2.83 ppm. c, f, i, Photographs of the (CaCO3)n materials, which exhibit gel-like features in ethanol and powder-like features in DMSO and water. j–l, 1H NMR measurement of the ethyl group and hydroxyl group of ethanol (pure or bound with oligomer) in ethanol (j), DMSO (k) and water (l). The negligible change in chemical shift proved that no distinct interactions occurred between ethanol and the (CaCO3)n oligomers.
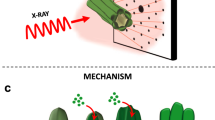
Extended Data Fig. 6 Cross-linking of (CaCO3)n oligomers.
a, In situ XRD analysis during the drying of (CaCO3)n oligomers, showing the evolution from gels to an ACC monolith. b, Evolution of the RDF from gels to an ACC monolith. The peak at around 2.4 Å is attributed to Ca–O, and its intensity increased during crosslinking. c, Additional high-resolution TEM images of the crosslinking of (CaCO3)n upon the removal of TEA, showing the stepwise chain growth and branch formation. d, Statistical results of the lengths and fractal dimensions of grown (CaCO3)n oligomers. The scheme shows the anisotropic chain growth with branches after TEA removal. e, Cross-linked (CaCO3)n with a network structure over an ultra-thin carbon support film. f, EDS analysis of the crosslinked (CaCO3)n in e. g, EDS mapping analysis of the crosslinked CaCO3 oligomers.
Extended Data Fig. 7 Monolithic ACC and other inorganic materials.
a, FTIR analysis of the monolithic CaCO3, showing ACC as the only component. b, Thermal gravimetric analysis of the monolithic CaCO3, showing a CaCO3:H2O molar ratio in the ACC of 3:1. c–f, Photographs and XRD patterns of multiple amorphous monoliths, including calcium phosphate (c), calcium sulfate (d), cupric phosphate (e) and manganous phosphate (f) monoliths. The subsequent crystallization manners of these samples are different. The crystallizations of amorphous cupric phosphate and amorphous manganous phosphate cannot be induced by our thermal treatments (700 °C)56. Amorphous calcium phosphate and calcium sulfate can transform to hydroxyapatite (c) and gypsum (d), respectively, by using humidity or water treatments. Although large single crystals without cracks have not been obtained, we believe that future improvements in crystallization control may provide a suitable solution. In conclusion, crosslinking oligomers provides a general strategy for the construction of amorphous monoliths with continuous structures, but an appropriate crystallization treatment is also required to extend the application of this method to the production of crystalline monoliths.
Extended Data Fig. 8 Crystallization of ACC under different conditions.
a, FTIR spectra of the monolithic CaCO3 at different time periods under 100% relative humidity at 25 °C. The peaks at 866 cm−1 belong to the amorphous phase, and those at 875 cm−1 belong to the crystalline phase. The change of the peaks with time indicates a humidity-induced crystallization process. b, Kinetics of phase transformation from ACC to calcite under different conditions: <5% relative humidity treatment at 25 °C, 100% relative humidity treatment at 25 °C, 300 °C thermal treatment, and 320 °C thermal treatment. The results show the treatment condition controls for the crystallization. It should be noted that although single crystals can be generated by humidity treatment, the size limitation is stricter than that for single crystals generated by thermal treatment. c, X-ray diffraction pattern of thermal induced calcite sample measured along <100>, <010>, <001> and <111> zone axes. The indexing rate of our sample was 79%, while that of geological single-crystal calcite was 87%.
Extended Data Fig. 9 Control of thermal-induced crystallization.
a, Scheme of thermal-induced single-crystalline calcite formation. The infrared thermal images show the thermal gradients from the proximal heating site to the distal site during the treatment, which is the key to ensure the oriented crystallization for single-crystal formation. b, POM image of a single crystalline calcite. c–i, Thermal treatment at different conditions demonstrating the importance of selecting an appropriate protocol . H, T and C represent the heating rate, transformation temperature, and cooling rate, respectively. c, d, The continuous movement of the complete amorphous-crystalline crystallization frontier induced by the well-organized thermal gradient with the recommend protocol of H = 1 °C min−1, T = 320 °C and C = 5 °C min−1. e, XRD pattern of ACC with a protocol of T = 300 °C, showing no crystallization at the low heating temperature. f, A protocol of H = 1 °C min−1, T = 325 °C and C = 5 °C min−1 results in a break of the thermal gradient; a spontaneous calcite crystallization is induced at a distal site rather than the amorphous–crystalline crystallization frontier, resulting in polycrystalline formation. g, A protocol of H = 2 °C min−1, T = 320 °C and C = 5 °C min−1 also results in a thermal gradient break so that the random crystallizations occur at the amorphous–crystalline interface. h, A protocol of H = 1 °C min−1, T = 320 °C and C = 10 °C min−1 causes the formation of cracks in the resulting calcite owing to the fast cooling rate. i, Using the ACC bulk produced by the conventional particle packing, polycrystalline formation is induced after a thermal treatment of H = 1 °C min−1, T = 320 °C and C = 5 °C min−1, which is attributed to its discontinuous internal structure.
Extended Data Fig. 10 Repair of sea-urchin spine and enamel by a combination of the crosslinking oligomers.
a, POM image of natural sea-urchin spine. b, c, SEM images of natural sea-urchin spine, showing the complicated structure with a smooth surface. Inset in c is a cross-sectional view of CaCO3 on the spine. d, ATR-FTIR characterization of the sea-urchin spine, which is inorganic calcite. e, POM image of the sea-urchin spine grown using the (CaCO3)n oligomers; the crystallization is induced by a simulated sea water at 25 °C. f, g, SEM images of the repaired sea-urchin spine, which maintains the original complicated structure. A magnified image (g) shows the surface of freshly grown CaCO3. The inset of g is a cross-sectional view of the CaCO3 grown on the spine, demonstrating the continuous interfacial structure between the native sea-urchin spine and the grown calcite. h, ATR-FTIR characterization of the repaired layer on the sea-urchin spine, which is also calcite. i, SEM image of the repaired sea-urchin spine using a conventional solution crystallization method. The random precipitation of numerous calcite particles rather than the expected oriented growth is resulted, demonstrating a failure in the biomineral repair. j, Regrowth of enamel structure by using calcium phosphate oligomers and crystallization is induced by simulated oral fluid at 37 °C. Epitaxial growth of the enamel rods is observed, which is due to the continuous construction by the crosslinking oligomers.
Rights and permissions
About this article
Cite this article
Liu, Z., Shao, C., Jin, B. et al. Crosslinking ionic oligomers as conformable precursors to calcium carbonate. Nature 574, 394–398 (2019). https://doi.org/10.1038/s41586-019-1645-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1645-x
This article is cited by
-
Inorganic ionic polymerization: From biomineralization to materials manufacturing
Nano Research (2024)
-
Elastic ceramic plastic made using molecules with organic and inorganic parts
Nature (2023)
-
Mechanistic formulation of inorganic membranes at the air–liquid interface
Nature (2023)
-
Organic–inorganic covalent–ionic molecules for elastic ceramic plastic
Nature (2023)
-
Nonclassical mechanisms to irreversibly suppress β-hematin crystal growth
Communications Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.