Abstract
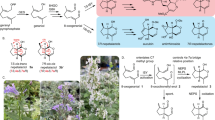
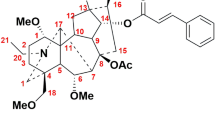
(+)-Perseanol is an isoryanodane diterpene that is isolated from the tropical shrub Persea indica1 and has potent antifeedant and insecticidal properties. It is structurally related to (+)-ryanodine, which is a high-affinity ligand for and modulator of ryanodine receptors—ligand-gated ion channels that are critical for intracellular Ca2+ signalling in most multicellular organisms2. Ryanodine itself modulates ryanodine-receptor-dependent Ca2+ release in many organisms, including mammals; however, preliminary data indicate that ryanodane and isoryanodane congeners that lack the pyrrole-2-carboxylate ester—such as perseanol—may have selective activity in insects3. Here we report a chemical synthesis of (+)-perseanol, which proceeds in 16 steps from commercially available (R)-pulegone. The synthesis involves a two-step annulation process that rapidly assembles the tetracyclic core from readily accessible cyclopentyl building blocks. This work demonstrates how convergent fragment coupling, when combined with strategic oxidation tactics, can enable the concise synthesis of complex and highly oxidized diterpene natural products.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Characterization data for all compounds produced in this study are available in Supplementary Information or on request from the corresponding author. Metrical parameters for the structures of 32 and S21 are available free of charge from the Cambridge Crystallographic Data Centre (CCDC, https://www.ccdc.cam.ac.uk/) under reference numbers 1909375 and 1914686, respectively.
References
González-Coloma, A., Terrero, D., Perales, A., Escoubas, P. & Fraga, B. M. Insect antifeedant ryanodane diterpenes from Persea indica. J. Agric. Food Chem. 44, 296–300 (1996).
Sutko, J. L., Airey, J. A., Welch, W. & Ruest, L. The pharmacology of ryanodine and related compounds. Pharmacol. Rev. 49, 53–98 (1997).
Gonzalez-Coloma, A., Reina, M., Gutierrez, C. & Fraga, B. M. in Studies in Natural Products Chemistry Vol. 26 (ed. Rahman, A.) 849–879 (Elsevier, 2002).
R.E.D. Facts Ryanodine. EPA-000-F-99-002 (United States Environmental Protection Agency, 1999).
Lahm, G. P. et al. in Pesticide Chemistry: Crop Protection, Public Health, Environmental Safety (eds Ohkawa, H. et al.) 111–120 (Wiley-VCH, 2007).
Cordova, D. et al. in Pesticide Chemistry: Crop Protection, Public Health, Environmental Safety (eds Ohkawa, H. et al.) 121–126 (Wiley-VCH, 2007).
Nohara, T. et al. Cinncassiol D1 and its glucoside, novel pentacyclic diterpenes from Cinnamomi cortex. Tetrahedr. Lett. 21, 2647–2648 (1980).
Chai, X. Y. et al. Six insecticidal isoryanodane diterpenoids from the bark and twigs of Itoa orientalis. Tetrahedron 64, 5743–5747 (2008).
Zeng, J. et al. Diterpenoids with immunosuppressive activities from Cinnamomum cassia. J. Nat. Prod. 77, 1948–1954 (2014).
Ling, S. Q., Xu, Y. N., Gu, Y. P., Liu, S. Y. & Tang, W. W. Toxicity and biochemical effects of itol A on the brown planthopper, Nilaparvata lugens (Stål) (Hemiptera: Delphacidae). Pestic. Biochem. Physiol. 152, 90–97 (2018).
Sattelle, D. B., Cordova, D. & Cheek, T. R. Insect ryanodine receptors: molecular targets for novel pest control chemicals. Invert. Neurosci. 8, 107–119 (2008).
Koshimizu, M., Nagatomo, M. & Inoue, M. Construction of a pentacyclic ring system of isoryanodane diterpenoids by SmI2-mediated transannular cyclization. Tetrahedron 74, 3384–3390 (2018).
Bélanger, A. et al. Total synthesis of ryanodol. Can. J. Chem. 57, 3348–3354 (1979).
Chuang, K. V., Xu, C. & Reisman, S. E. A 15-step synthesis of (+)-ryanodol. Science 353, 912–915 (2016).
Xu, C., Han, A., Virgil, S. C. & Reisman, S. E. Chemical synthesis of (+)-ryanodine and (+)-20-deoxyspiganthine. ACS Cent. Sci. 3, 278–282 (2017).
Luparia, M., Vadalà, A., Zanoni, G. & Vidari, G. 1,2-Bisanionic coupling approach to 2,3-disubstituted cyclopentenols and cyclopentenones. Org. Lett. 8, 2147–2150 (2006).
Marx, J. N. & Norman, L. R. Synthesis of (–)-acorone and related spirocyclic sesquiterpenes. J. Org. Chem. 40, 1602–1606 (1975).
Shen, Y. et al. Protecting-group-free total synthesis of (−)-jiadifenolide: development of a [4 + 1] annulation toward multisubstituted tetrahydrofurans. Org. Lett. 17, 5480–5483 (2015).
Yasuda, A., Yamamoto, H. & Nozaki, H. Highly stereospecific isomerization of oxiranes into allylic alcohols by means of organoaluminum amides. Bull. Chem. Soc. Jpn. 52, 1705–1708 (1979).
Steves, J. E. & Stahl, S. S. Copper(i)/ABNO-catalyzed aerobic alcohol oxidation: alleviating steric and electronic constraints of Cu/TEMPO catalyst systems. J. Am. Chem. Soc. 135, 15742–15745 (2013).
Iimura, S., Overman, L. E., Paulini, R. & Zakarian, A. Enantioselective total synthesis of guanacastepene N using an uncommon 7-endo Heck cyclization as a pivotal step. J. Am. Chem. Soc. 128, 13095–13101 (2006).
Mewshaw, R. E. Vilsmeier reagents: preparation of β-halo-α,β-unsaturated ketones. Tetrahedr. Lett. 30, 3753–3756 (1989).
Helal, C. J. & Meyer, M. P. in Lewis Base Catalysis in Organic Synthesis 1st edn (eds Vedejs, E. & Denmark, S. E.) 387–455 (Wiley-VCH, 2016).
Corey, E. J. & Link, J. O. A new chiral catalyst for the enantioselective synthesis of secondary alcohols and deuterated primary alcohols by carbonyl reduction. Tetrahedr. Lett. 30, 6275–6278 (1989).
Nwoye, E. O. & Dudley, G. B. Synthesis of para-methoxybenzyl (PMB) ethers under neutral conditions. Chem. Commun. 1436–1437 (2007).
Wu, L. P. et al. Palladium-catalyzed carbonylative transformation of C(sp3)–X bonds. ACS Catal. 4, 2977–2989 (2014).
Odell, L. R., Russo, F. & Larhed, M. Molybdenum hexacarbonyl mediated CO gas-free carbonylative reactions. Synlett 5, 685–698 (2012).
Ueda, T., Konishi, H. & Manabe, K. Palladium-catalyzed carbonylation of aryl, alkenyl, and allyl halides with phenyl formate. Org. Lett. 14, 3100–3103 (2012).
Ueda, T., Konishi, H. & Manabe, K. Palladium-catalyzed fluorocarbonylation using N-formylsaccharin as CO source: general access to carboxylic acid derivatives. Org. Lett. 15, 5370–5373 (2013).
Jiang, X. et al. Palladium-catalyzed formylation of aryl halides with tert-butyl isocyanide. Org. Lett. 16, 3492–3495 (2014).
Krasovskiy, A., Kopp, F. & Knochel, P. Soluble lanthanide salts (LnCl3·2LiCl) for the improved addition of organomagnesium reagents to carbonyl compounds. Angew. Chem. Int. Ed. 45, 497–500 (2006).
Baldwin, J. E. et al. 5-endo-trigonal reactions: disfavoured ring-closure. J. Chem. Soc. Chem. Commun. 736–738 (1976).
Wolleb, H. & Carreira, E. M. Total synthesis of (+)-dendrowardol C. Angew. Chem. Int. Ed. 56, 10890–10893 (2017).
Acknowledgements
We acknowledge S. Virgil and the Caltech Center for Catalysis and Chemical Synthesis for access to analytical equipment; L. Henling and J. Hofstra for X-ray data collection and data refinement, respectively, for the structures of 32 and S21; Y. Zhang for providing original spectral data of perseanol; and K. Chuang for contributions to the synthetic design. Fellowship support was provided by the National Institutes of Health (NIH; Nos. 5T32GM007616-37 and 1F31GM120821, to A.H.). S.E.R. is a Heritage Medical Research Investigator. Financial support from the NIH (Nos. NIGMS RGM097582-01 and R35GM118191-01), Eli Lilly and Novartis is acknowledged.
Author information
Authors and Affiliations
Contributions
A.H. and S.E.R. conceived this work; A.H., Y.T. and S.E.R. designed the experiments and analysed the data; A.H. and Y.T. conducted the experiments; and A.H. and S.E.R. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
This file contains: General Procedures; Positional Numbering; Synthetic Scheme of Inoue’s Isoryanodane Approach; Synthetic Procedures; 1H and 13C NMR Comparison Tables for Perseanol; Single Crystal X-Ray Diffraction Data; Supplementary References and 1H and 13C NMR Spectral Data.
Supplementary Data
This zipped file contains checkcif files for compounds 32 and S21.
Rights and permissions
About this article
Cite this article
Han, A., Tao, Y. & Reisman, S.E. A 16-step synthesis of the isoryanodane diterpene (+)-perseanol. Nature 573, 563–567 (2019). https://doi.org/10.1038/s41586-019-1580-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1580-x
This article is cited by
-
Palladium-catalyzed asymmetric carbamoyl-carbonylation of alkenes
Science China Chemistry (2021)
-
Constructing chiral bicyclo[3.2.1]octanes via palladium-catalyzed asymmetric tandem Heck/carbonylation desymmetrization of cyclopentenes
Nature Communications (2020)
-
The rise of preprints in chemistry
Nature Chemistry (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.