Abstract
The radiation-based sterile insect technique (SIT) has successfully suppressed field populations of several insect pest species, but its effect on mosquito vector control has been limited. The related incompatible insect technique (IIT)—which uses sterilization caused by the maternally inherited endosymbiotic bacteria Wolbachia—is a promising alternative, but can be undermined by accidental release of females infected with the same Wolbachia strain as the released males. Here we show that combining incompatible and sterile insect techniques (IIT–SIT) enables near elimination of field populations of the world’s most invasive mosquito species, Aedes albopictus. Millions of factory-reared adult males with an artificial triple-Wolbachia infection were released, with prior pupal irradiation of the released mosquitoes to prevent unintentionally released triply infected females from successfully reproducing in the field. This successful field trial demonstrates the feasibility of area-wide application of combined IIT–SIT for mosquito vector control.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Source Data for the main and Extended Data figures are provided in the online version of this paper. Any other relevant data are available from the corresponding authors upon reasonable request.
References
Dyck, V. A., Hendrichs, J. & Robinson, A. S. Sterile Insect Technique: Principles and Practice in Area-Wide Integrated Pest Management (Springer Netherlands, 2005).
Dame, D. A., Curtis, C. F., Benedict, M. Q., Robinson, A. S. & Knols, B. G. Historical applications of induced sterilisation in field populations of mosquitoes. Malar. J. 8 (Suppl 2), S2 (2009).
Helinski, M. E., Parker, A. G. & Knols, B. G. Radiation biology of mosquitoes. Malar. J. 8 (Suppl 2), S6 (2009).
Lees, R. S., Gilles, J. R., Hendrichs, J., Vreysen, M. J. & Bourtzis, K. Back to the future: the sterile insect technique against mosquito disease vectors. Curr. Opin. Insect Sci. 10, 156–162 (2015).
Laven, H. Eradication of Culex pipiens fatigans through cytoplasmic incompatibility. Nature 216, 383–384 (1967).
LePage, D. P. et al. Prophage WO genes recapitulate and enhance Wolbachia-induced cytoplasmic incompatibility. Nature 543, 243–247 (2017).
Yen, J. H. & Barr, A. R. New hypothesis of the cause of cytoplasmic incompatibility in Culex pipiens L. Nature 232, 657–658 (1971).
Chambers, E. W., Hapairai, L., Peel, B. A., Bossin, H. & Dobson, S. L. Male mating competitiveness of a Wolbachia-introgressed Aedes polynesiensis strain under semi-field conditions. PLoS Negl. Trop. Dis. 5, e1271 (2011).
Zhang, D., Zheng, X., Xi, Z., Bourtzis, K. & Gilles, J. R. Combining the sterile insect technique with the incompatible insect technique: I impact of Wolbachia infection on the fitness of triple- and double-infected strains of Aedes albopictus. PLoS ONE 10, e0121126 (2015).
Atyame, C. M. et al. Comparison of irradiation and Wolbachia based approaches for sterile-male strategies targeting Aedes albopictus. PLoS ONE 11, e0146834 (2016).
Curtis, C. F. et al. A field trial on control of Culex quinquefasciatus by release of males of a strain integrating cytoplasmic incompatibility and a translocation. Entomol. Exp. Appl. 31, 181–190 (1982).
Dobson, S. L., Fox, C. W. & Jiggins, F. M. The effect of Wolbachia-induced cytoplasmic incompatibility on host population size in natural and manipulated systems. Proc. R. Soc. Lond. B 269, 437–445 (2002).
Pal, R. in The Use of Genetics in Insect Control (eds Pal, R. & Whitten, M. J.) 73–95 (Elsevier, 1974).
Curtis, C. F. Testing systems for the genetic control of mosquitoes. In Proceedings of XV International Congress of Entomology (eds White, D. & Packer, J. S.) 106–116 (1976)
Arunachalam, N. & Curtis, C. F. Integration of radiation with cytoplasmic incompatibility for genetic control in the Culex pipiens complex (Diptera: Culicidae). J. Med. Entomol. 22, 648–653 (1985).
Sharma, V. P., Subbarao, S. K., Adak, T. & Razdan, R. K. Integration of gamma irradiation and cytoplasmic incompatibility in Culex pipiens fatigans (Diptera: Culicidae). J. Med. Entomol. 15, 155–156 (1979).
Curtis, C. F. & Shahid, M. A. Radiation sterilization and cytoplasmic incompatibility in a “tropicalized” strain of the Culex pipiens complex (Diptera: Culicidae). J. Med. Entomol. 24, 273–274 (1987).
Mains, J. W., Brelsfoard, C. L., Rose, R. I. & Dobson, S. L. Female adult Aedes albopictus suppression by Wolbachia-infected male mosquitoes. Sci. Rep. 6, 33846 (2016).
Atyame, C. M. et al. Cytoplasmic incompatibility as a means of controlling Culex pipiens quinquefasciatus mosquito in the islands of the south-western Indian Ocean. PLoS Negl. Trop. Dis. 5, e1440 (2011).
Brelsfoard, C. L., Séchan, Y. & Dobson, S. L. Interspecific hybridization yields strategy for South Pacific filariasis vector elimination. PLoS Negl. Trop. Dis. 2, e129 (2008).
Xi, Z., Dean, J. L., Khoo, C. & Dobson, S. L. Generation of a novel Wolbachia infection in Aedes albopictus (Asian tiger mosquito) via embryonic microinjection. Insect Biochem. Mol. Biol. 35, 903–910 (2005).
Xi, Z., Khoo, C. C. & Dobson, S. L. Wolbachia establishment and invasion in an Aedes aegypti laboratory population. Science 310, 326–328 (2005).
Brelsfoard, C. L., St Clair, W. & Dobson, S. L. Integration of irradiation with cytoplasmic incompatibility to facilitate a lymphatic filariasis vector elimination approach. Parasit. Vectors 2, 38 (2009).
Zhang, D., Lees, R. S., Xi, Z., Gilles, J. R. & Bourtzis, K. Combining the sterile insect technique with Wolbachia-based approaches: II a safer approach to Aedes albopictus population suppression programmes, designed to minimize the consequences of inadvertent female release. PLoS ONE 10, e0135194 (2015).
Zhang, D., Lees, R. S., Xi, Z., Bourtzis, K. & Gilles, J. R. Combining the sterile insect technique with the incompatible insect technique: III robust mating competitiveness of irradiated triple Wolbachia-infected Aedes albopictus males under semi-field conditions. PLoS ONE 11, e0151864 (2016).
Bourtzis, K. et al. Harnessing mosquito-Wolbachia symbiosis for vector and disease control. Acta Trop. 132 (Suppl), S150–S163 (2014).
Fonseca, D. M. et al. Area-wide management of Aedes albopictus. Part 2: gauging the efficacy of traditional integrated pest control measures against urban container mosquitoes. Pest Manag. Sci. 69, 1351–1361 (2013).
Unlu, I., Farajollahi, A., Strickman, D. & Fonseca, D. M. Crouching tiger, hidden trouble: urban sources of Aedes albopictus (Diptera: Culicidae) refractory to source-reduction. PLoS ONE 8, e77999 (2013).
Xi, Z., Khoo, C. C. & Dobson, S. L. Interspecific transfer of Wolbachia into the mosquito disease vector Aedes albopictus. Proc. R. Soc. Lond. B 273, 1317–1322 (2006).
Fu, Y., Gavotte, L., Mercer, D. R. & Dobson, S. L. Artificial triple Wolbachia infection in Aedes albopictus yields a new pattern of unidirectional cytoplasmic incompatibility. Appl. Environ. Microbiol. 76, 5887–5891 (2010).
Blagrove, M. S., Arias-Goeta, C., Failloux, A. B. & Sinkins, S. P. Wolbachia strain wMel induces cytoplasmic incompatibility and blocks dengue transmission in Aedes albopictus. Proc. Natl Acad. Sci. USA 109, 255–260 (2012).
Suh, E., Mercer, D. R., Fu, Y. & Dobson, S. L. Pathogenicity of life-shortening Wolbachia in Aedes albopictus after transfer from Drosophila melanogaster. Appl. Environ. Microbiol. 75, 7783–7788 (2009).
Calvitti, M., Moretti, R., Lampazzi, E., Bellini, R. & Dobson, S. L. Characterization of a new Aedes albopictus (Diptera: Culicidae)–Wolbachia pipientis (Rickettsiales: Rickettsiaceae) symbiotic association generated by artificial transfer of the wPip strain from Culex pipiens (Diptera: Culicidae). J. Med. Entomol. 47, 179–187 (2010).
Laven, H. & Aslamkhan, M. Control of Culex pipiens pipiens and C. p. fatigans with integrated genetical systems. Pak. J. Sci. 22, 303–312 (1970).
Bian, G., Xu, Y., Lu, P., Xie, Y. & Xi, Z. The endosymbiotic bacterium Wolbachia induces resistance to dengue virus in Aedes aegypti. PLoS Pathog. 6, e1000833 (2010).
Moreira, L. A. et al. A Wolbachia symbiont in Aedes aegypti limits infection with dengue, Chikungunya, and Plasmodium. Cell 139, 1268–1278 (2009).
Dutra, H. L. et al. Wolbachia blocks currently circulating Zika virus isolates in Brazilian Aedes aegypti mosquitoes. Cell Host Microbe 19, 771–774 (2016).
Aliota, M. T., Peinado, S. A., Velez, I. D. & Osorio, J. E. The wMel strain of Wolbachia reduces transmission of Zika virus by Aedes aegypti. Sci. Rep. 6, 28792 (2016).
Zhang, D. et al. Establishment of a medium-scale mosquito facility: optimization of the larval mass-rearing unit for Aedes albopictus (Diptera: Culicidae). Parasit. Vectors 10, 569 (2017).
Zhang, D. et al. Establishment of a medium-scale mosquito facility: tests on mass production cages for Aedes albopictus (Diptera: Culicidae). Parasit. Vectors 11, 189 (2018).
Li, Y. et al. Comparative evaluation of the efficiency of the BG-Sentinel trap, CDC light trap and Mosquito-oviposition trap for the surveillance of vector mosquitoes. Parasit. Vectors 9, 446 (2016).
Halasa, Y. A. et al. Quantifying the impact of mosquitoes on quality of life and enjoyment of yard and porch activities in New Jersey. PLoS ONE 9, e89221 (2014).
Schmidt, T. L. et al. Genome-wide SNPs reveal the drivers of gene flow in an urban population of the Asian tiger mosquito, Aedes albopictus. PLoS Negl. Trop. Dis. 11, e0006009 (2017).
Sanogo, Y. O. & Dobson, S. L. Molecular discrimination of Wolbachia in the Culex pipiens complex: evidence for variable bacteriophage hyperparasitism. Insect Mol. Biol. 13, 365–369 (2004).
Zhou, W., Rousset, F. & O’Neil, S. Phylogeny and PCR-based classification of Wolbachia strains using wsp gene sequences. Proc. R. Soc. Lond. B 265, 509–515 (1998).
Heddi, A., Grenier, A. M., Khatchadourian, C., Charles, H. & Nardon, P. Four intracellular genomes direct weevil biology: nuclear, mitochondrial, principal endosymbiont, and Wolbachia. Proc. Natl Acad. Sci. USA 96, 6814–6819 (1999).
Das, S., Garver, L., Ramirez, J. R., Xi, Z. & Dimopoulos, G. Protocol for dengue infections in mosquitoes (A. aegypti) and infection phenotype determination. J. Vis. Exp. (5), PMC2557096 (2007).
Lu, P., Bian, G., Pan, X. & Xi, Z. Wolbachia induces density-dependent inhibition to dengue virus in mosquito cells. PLoS Negl. Trop. Dis. 6, e1754 (2012).
Zhang, M. et al. Quantitative analysis of replication and tropisms of Dengue virus type 2 in Aedes albopictus. Am. J. Trop. Med. Hyg. 83, 700–707 (2010).
McGraw, E. A., Merritt, D. J., Droller, J. N. & O’Neill, S. L. Wolbachia-mediated sperm modification is dependent on the host genotype in Drosophila. Proc. R. Soc. Lond. B 268, 2565–2570 (2001).
Bian, G., Zhou, G., Lu, P. & Xi, Z. Replacing a native Wolbachia with a novel strain results in an increase in endosymbiont load and resistance to dengue virus in a mosquito vector. PLoS Negl. Trop. Dis. 7, e2250 (2013).
Bian, G. et al. Wolbachia invades Anopheles stephensi populations and induces refractoriness to Plasmodium infection. Science 340, 748–751 (2013).
Carvalho, D. O. et al. Mass production of genetically modified Aedes aegypti for field releases in Brazil. J. Vis. Exp. 3579, e3579 (2014). 10.3791/3579
Focks, D. A. An improved separator for the developmental stages, sexes, and species of mosquitoes (Diptera: Culicidae). J. Med. Entomol. 17, 567–568 (1980).
Methods in Anopheles research. Malaria Research and Reference Reagent Resource Center http://www.mr4.org/Publications/MethodsinAnophelesResearch.aspx (2014).
Fried, M. Determination of sterile-insect competitiveness. J. Econ. Entomol. 64, 869–872 (1971).
Acknowledgements
This work was supported by Guangdong Innovative Research Team Program (No. 2011S009), Scientific and Technological Leading Talents of Guangzhou Development District (No. 2013L-P116), Science and Technology Planning Project of Guangdong Province (2016A020251001), a grant from the Foundation for the NIH through the Grand Challenges in Global Health Initiative of the Bill and Melinda Gates Foundation, the joint Food and Agricultural Organization (FAO) of the United Nations and International Atomic Energy Agency (IAEA) Division of Nuclear Techniques in Food and Agriculture and the IAEA Department of Technical Cooperation (RAS5066, RAS5082, D42016 and D44002), the 111 Project (grant no. B12003), Key Project of NNSF of China (11631005), China Postdoctoral Innovation Program (BX20180394), and a grant-in-aid for joint research (2017-AH-04) from the NJAU-MSU Asia-Hub Project. A.A.H. was supported by an NHMRC Fellowship. We thank X. Zhou, S. O’Neill, S. L. Dobson, G. Bian and E. Walker for their support, suggestions and technical assistance.
Author information
Authors and Affiliations
Contributions
Z.X., X.Z., D.Z., Y. Li, C.Y., Y. Wu, A.G.P., J.R.L.G., K.B., Z.W., L.A.B. and A.A.H. developed the concept and methodology; D.Z. performed radiation and male mating-competitiveness assay; Y. Liang and C.Y. performed population suppression and population replacement in laboratory cages; Y. Li and X.Z. performed human-landing assay; C.Y. performed mosquito quality control; Y. Li, Y. Wu, X.L. and X.P. performed vector competence assays; A.G.P designed the X-ray irradiator; D.Z., K.B. and J.R.L.G. performed the population-suppression experiment in semi-field cages; X.Z., Z.Y., Y. Wu and J. Zhuang performed community engagement; X.L., X.P., Q.S., J.-T.G. and M.Z. performed cell culture, virus titration and Wolbachia density quantification; Z.Y., Zhigang Hu, Z.Z., L.L. and Q.L. identified the field sites; B.Z., L.H. M.T. and J.Y. developed the mathematical model and performed spatial analyses; X.W. and J. Zhu performed mosquito mass rearing; Y. Wei and W.Q. performed release and field surveillance; J. Zhu, W.Q., X.-Y.H., Zhiyong Hu and Z.W. performed coordination for the project; W.Q. obtained regulatory approvals for mosquito releases; J.L. performed mosquito crosses and maintenance of mosquito lines; J.B. and Z.X. performed cost-effectiveness analysis; Z.X. provided oversight of the project and contributed to all experimental designs, data analysis and data interpretation; Z.X., L.A.B., X.Z., D.Z., Y.L. and A.A.H. wrote the manuscript. All authors participated in manuscript editing and final approval.
Corresponding author
Ethics declarations
Competing interests
Y. Li, X.W., Y. Wei, J. Zhu, W.Q., J.L. and Z.X. are affiliated with Guangzhou Wolbaki Biotech Co., Ltd. This does not alter our adherence to all Nature policies.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Peer review information Nature thanks William Sullivan and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
Extended Data Fig. 1 Illustration of the procedures used to establish A. albopictus HC line by embryonic microinjection and for PCR verification of the Wolbachia strains in HC.
a, The Wolbachia strain wPip from C. pipiens molestus (donor) was transinfected into the wAlbA/wAlbB superinfected HOU line of A. albopictus (recipient) by embryonic microinjection, to generate a mosquito line infected with three Wolbachia strains (HC). A circle containing a cross indicates that the line was discarded, and a tick indicates that the line was maintained. Red indicates wPip-positive, and white indicates wPip-negative individuals. b, Wolbachia infection status was verified by PCR in both male and female HC mosquitoes. Results indicate that HC contains both the native Wolbachia strains (wAlbA and wAlbB) and the new transinfected strain wPip. The experiments were repeated at least three times independently with similar results.
Extended Data Fig. 2 Inhibition of both horizontal and vertical transmission of Zika and dengue viruses in the A. albopictus HC line.
a, b, ZIKV (a) and DENV-2 (b) were significantly decreased in the saliva of HC compared to wild type, GUA and HOU, respectively. Fourteen days post-infection, mosquito saliva samples were collected, ZIKV copies were quantified by RT-qPCR, and the titre of DENV-2 was measured by plaque assay. Horizontal lines indicate the median value (two-sided Mann–Whitney test: ZIKV, n = 16 for both HC and GUA, P = 0.0049; DENV-2, n = 39 for HC and n = 36 for HOU, P < 0.0001). c, Experimental design to measure the horizontal transmission of ZIKV. d, Viral positive rate in mosquitoes at day 7 after feeding on Zika-infected suckling mice (two-sided Fisher’s exact test, n = 19 for GUA and 20 for HC, P = 0.047). e, Experimental design to measure the vertical transmission of ZIKV in mosquitoes. f, The minimum ZIKV vertical transmission rate in HC and GUA lines (two-sided Fisher’s exact test, n = 35 biologically independent samples, P = 0.004). g, h, ZIKV replication and dissemination in HC were both significantly decreased. Mosquitoes were infected with ZIKV by oral feeding. ZIKV replication was determined by viral genome copy numbers in mosquito abdomens at 7 dpi (n = 20), and dissemination was measured by ZIKV infection status in one mosquito hind leg at 14 dpi (n = 20). The observations showed that ZIKV replication (g) and dissemination (h) were both significantly inhibited in HC. The infection prevalence is shown as a percentage. Horizontal lines indicate the median number of viral copies (two-sided Mann–Whitney test: abdomen, P = 0.018; hind legs, P = 0.002).
Extended Data Fig. 3 Sensitivity analysis of the robustness of the 5:1 HC:GUA male release ratio to induce population suppression.
a–c, The mathematical model of the semi-field cage experiments shown in Fig. 1e, f and described in the Supplementary Information provides an accurate approximation to the semi-field data when r = 5 and one of three parameters listed in (5) in the Supplementary Information are varied across a wide range of values. a, R2 ∈ [0.9259, 0.9355] for ξ0 ∈ [0.6, 0.9]. b, R2 ∈ [0.9301, 0.9329] for μ ∈ [0.75, 0.95]. c, R2 ∈ [0.9325, 0.9573] for λ ∈ [0.5, 1]. d, The effect of mosquito migration and the efficiency of population suppression as measured by egg hatch rate. We fixed λ = 0.6, μ = 0.85, ξ0 = 0.80 and b0 = 75. The 5:1 ratio is sufficient to offset 20% migration with a suppression efficiency 92.20% as compared to 98.71% suppression efficiency without migration.
Extended Data Fig. 4 The proportion of egg-positive ovitraps, the average number of eggs per ovitrap, and the average percentage of eggs hatching per ovitrap in release and control sites before release of HC males.
a–c, Site 1: the proportion of egg-positive ovitraps (a), average number of eggs per ovitrap (b) and the average percentage egg hatch per ovitrap (c) in 2014, compared to the control site. d–f, Site 2: the proportion of egg-positive ovitraps (d), average number of eggs per ovitrap (e) and the average percentage egg hatch per ovitrap (f) in 2015, compared to the control site. The proportion of egg-positive ovitraps was calculated from the number of ovitraps with eggs divided by the total number of ovitraps used. The average number of eggs per ovitrap was calculated as the total number of eggs collected divided by the number of ovitraps used. The average percentage of eggs hatching per ovitrap was calculated as the mean of the percentage of hatched eggs per individual ovitrap for all the ovitraps that collected eggs. Data were collected weekly. The proportion of egg-positive ovitraps (two-sided Mann–Whitney test: site 1, n = 26, P = 0.591; site 2, n = 32, P = 0.3239), the average number of eggs per ovitrap (two-sided Mann–Whitney test: site 1, n = 26, P = 0.4516; site 2, n = 32, P = 0.6940), and the average percentage of eggs hatching per ovitrap (two-sided Mann–Whitney test: site 1, n = 26, P = 0.3186; site 2, n = 32, P = 0.8232) did not differ significantly between the control and release sites. In addition, there were significant and strong correlations across time between the release and their respective control sites for these three parameters, demonstrating similar temporal fluctuations in them: the proportion of egg-positive ovitraps (Pearson correlation: site 1, r = 0.88, n = 26, P < 0.0001; site 2, r = 0.85, n = 32, P < 0.0001), the average number of eggs per ovitrap (Pearson correlation: site 1, r = 0.77, n = 26, P < 0.0001; site 2, r = 0.96, n = 32, P < 0.0001), and the average percentage of eggs hatching per ovitrap (Pearson correlation: site 1, r = 0.67, n = 26, P = 0.0002; site 2, r = 0.70, n = 32, P < 0.0001).
Extended Data Fig. 5 Map of the ovitraps and BG traps distributed in the two release sites.
a, b, There were 110 ovitraps (grey circles) and 44 BG traps (blue circles) in release site 1 (a), and 40 ovitraps and 16 BG traps in release site 2 (b). Release site 1 was divided into 22 zones and release site 2 contained 8 zones. On average, there were five ovitraps and two BG traps in each zone, and collections from all traps were carried out weekly.
Extended Data Fig. 6 The proportion of egg-positive ovitraps, the average number of eggs per ovitrap, and the average percentage of eggs hatching per ovitrap in release and control sites after release of HC males.
a–c, Site 1: the proportion of egg-positive ovitraps (a), average number of eggs per ovitrap (b) and the average percentage egg hatch per ovitrap (c) in 2016 and 2017, compared to the control site. d–f, Site 2: the proportion of egg-positive ovitraps (d), average number of eggs per ovitrap (e) and the average percentage egg hatch per ovitrap (f) in 2016 and 2017, compared to the control site. Significant declines were observed for all three parameters in the two release sites compared to their control sites: the proportion of egg-positive ovitraps (two-sided Mann–Whitney test: site 1 2016, n = 36, P < 0.0001; site 2017, n = 35, P < 0.0001; site 2 2016, n = 32, P < 0.0001; site 2 2017, n = 35, P < 0.0001), the average number of eggs per ovitrap (two-sided Mann–Whitney test: site 1 2016, n = 36, P < 0.0001; site 1 2017, n = 35, P < 0.0001; site 2 2016, n = 32, P < 0.0001; site 2 2017, n = 35, P < 0.0001), and the average percentage of eggs hatching per ovitrap (two-sided Mann–Whitney test: site 1 2016, n = 36, P < 0.0001; site 1 2017, n = 35, P < 0.0001; site 2 2016, n = 32, P < 0.0001; site 2 2017, n = 35, P < 0.0001).
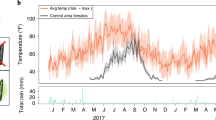
Extended Data Fig. 7 The total number of wPip-positive adult females collected monthly in release sites 1 and 2.
Females were collected weekly using BG traps and tested for wPip infection by PCR. The wPip-positive females were recorded monthly in site 1 and site 2 during the release period. No significant difference was observed in the number of wPip-positive females between 2015 (n = 3), 2016 (n = 7) and 2017 (n = 9) in site 1 (Kruskal–Wallis test, P = 0.6536), or between 2016 (n = 7) and 2017 (n = 9) in site 2 (two-sided Mann–Whitney test, P = 0.1164). No evidence of an increase in the number of wPip-positive females with time was apparent, but would have been expected if population replacement had started in the field.
Extended Data Fig. 8 Temporal and spatial distribution of wPip-positive ovitraps in the two release sites between 2015 and 2017.
a, b, Among 110 ovitraps in site 1 (a) and 40 ovitraps in site 2 (b), those from which wPip-positive larvae were detected are shown as red circles. The specific time points at which wPip-positive larvae were detected are also indicated (year.month). Overall, a total of 15 ovitraps with wPip-positive larvae were detected on 13 separate, spatially and/or temporally isolated, occasions in release site 1, whereas only one ovitrap with wPip-positive larvae was detected on a single occasion in release site 2. The first six of the ovitraps with wPip-positive larvae were detected in 2015, before the use of irradiation, while in 2017 only two were found in site 1 and none in site 2. c, Overall, wPip-positive rates of 0.9% (15/1,678 pooled larval samples taken weekly from individual ovitraps, referred to as ‘ovitrap weeks’) and 0.6% (1/166) were found during the release period in the 3 or 2 years of HC releases in sites 1 and 2, respectively. No evidence of an increase in the number of wPip-positive ovitraps with time was apparent, but would have been expected if population replacement had started in the field.
Extended Data Fig. 9 Induction of sterility in HC females after mating with irradiated HC males.
a, b, The effect of irradiating HC males on the egg hatch rate (a) and the level of induced sterility (b) in mated females. For each cross shown in the figure (x axis), a single treatment cage (30 × 30 × 30 cm) was set up containing males and females, at a 1:1 ratio, of the mosquito line with irradiation status indicated. The two control crosses (HC:HC and wild-type:wild-type) were set up with 100 individuals of each sex. All other treatment crosses used 300 individuals of each sex. IHC45Gy are HC males irradiated at the pupal stage with an X-ray dose of 45 Gy, as described in the Methods. None of the other mosquitoes used were irradiated (HC and GUA). Induced sterility was calculated as follows56: 100 − [(egg hatch rate of treatment cages)/(egg hatch rate of control cages) × 100]. Complete sterility (100%) was induced when wild-type females mated with either non-irradiated or irradiated HC males, whereas high levels of partial sterility (86.4%) were induced when HC females mated with IHC45Gy males, showing that irradiation causes sterility between the otherwise-compatible HC males and females. During HC release in the field, there is a high probability that HC females would mate with irradiated HC males owing to their high abundance relative to wild-type males.
Supplementary information
Supplementary Information
This PDF file contains Supplementary Methods, Supplementary Tables, Supplementary Discussion, and Supplementary Equations.
Rights and permissions
About this article
Cite this article
Zheng, X., Zhang, D., Li, Y. et al. Incompatible and sterile insect techniques combined eliminate mosquitoes. Nature 572, 56–61 (2019). https://doi.org/10.1038/s41586-019-1407-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1407-9
This article is cited by
-
Response of the mosquito immune system and symbiotic bacteria to pathogen infection
Parasites & Vectors (2024)
-
Mass irradiation of adult Aedes mosquitoes using a coolable 3D printed canister
Scientific Reports (2024)
-
Leucine aminopeptidase1 controls egg deposition and hatchability in male Aedes aegypti mosquitoes
Nature Communications (2024)
-
Modeling the dengue control dynamics based on a delay stochastic differential system
Computational and Applied Mathematics (2024)
-
Biological, ecological and trophic features of invasive mosquitoes and other hematophagous arthropods: What makes them successful?
Biological Invasions (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.