Abstract
Slow-wave sleep and rapid eye movement (or paradoxical) sleep have been found in mammals, birds and lizards, but it is unclear whether these neuronal signatures are found in non-amniotic vertebrates. Here we develop non-invasive fluorescence-based polysomnography for zebrafish, and show—using unbiased, brain-wide activity recording coupled with assessment of eye movement, muscle dynamics and heart rate—that there are at least two major sleep signatures in zebrafish. These signatures, which we term slow bursting sleep and propagating wave sleep, share commonalities with those of slow-wave sleep and paradoxical or rapid eye movement sleep, respectively. Further, we find that melanin-concentrating hormone signalling (which is involved in mammalian sleep) also regulates propagating wave sleep signatures and the overall amount of sleep in zebrafish, probably via activation of ependymal cells. These observations suggest that common neural signatures of sleep may have emerged in the vertebrate brain over 450 million years ago.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data that support the findings of this study are available as Supplementary Information and Source Data. The supplementary videos are linked in the Supplementary Information and are available for downloading at https://drive.google.com/open?id=1CQGSFzxm39KCvN9D_XwJR2rOY6hS7oC5. Any other relevant data are available from the corresponding author upon reasonable request.
Code availability
Code for custom Viewpoint protocols, Arduino sketches, FIJI macros and MatLab programs are available from https://github.com/louiscleung.
References
Campbell, S. S. & Tobler, I. Animal sleep: a review of sleep duration across phylogeny. Neurosci. Biobehav. Rev. 8, 269–300 (1984).
Leung, L. C. & Mourrain, P. Sleep: short sleepers should keep count of their hypocretin neurons. Curr. Biol. 28, R558–R560 (2018).
Pieron, H. Le Probleme Physiologique su Sommeil (Masson, 1913).
Shein-Idelson, M., Ondracek, J. M., Liaw, H. P., Reiter, S. & Laurent, G. Slow waves, sharp waves, ripples, and REM in sleeping dragons. Science 352, 590–595 (2016).
Prober, D. A. et al. Hypocretin/orexin overexpression induces an insomnia-like phenotype in zebrafish. J. Neurosci. 26, 13400–13410 (2006).
Yokogawa, T. et al. Characterization of sleep in zebrafish and insomnia in hypocretin receptor mutants. PLoS Biol. 5, e277 (2007).
Zhdanova, I. V. Sleep in zebrafish. Zebrafish 3, 215–226 (2006).
Aho, V. et al. Homeostatic response to sleep/rest deprivation by constant water flow in larval zebrafish in both dark and light conditions. J. Sleep Res. 26, 394–400 (2017).
Appelbaum, L. et al. Sleep-wake regulation and hypocretin-melatonin interaction in zebrafish. Proc. Natl Acad. Sci. USA 106, 21942–21947 (2009).
Mueller, T., Dong, Z., Berberoglu, M. A. & Guo, S. The dorsal pallium in zebrafish, Danio rerio (Cyprinidae, Teleostei). Brain Res. 1381, 95–105 (2011).
Berman, J. R., Skariah, G., Maro, G. S., Mignot, E. & Mourrain, P. Characterization of two melanin-concentrating hormone genes in zebrafish reveals evolutionary and physiological links with the mammalian MCH system. J. Comp. Neurol. 517, 695–710 (2009).
Chen, T. W. et al. Ultrasensitive fluorescent proteins for imaging neuronal activity. Nature 499, 295–300 (2013).
Madelaine, R. et al. MicroRNA-9 couples brain neurogenesis and angiogenesis. Cell Reports 20, 1533–1542 (2017).
Chauvette, S., Crochet, S., Volgushev, M. & Timofeev, I. Properties of slow oscillation during slow-wave sleep and anesthesia in cats. J. Neurosci. 31, 14998–15008 (2011).
Borbély, A. A. A two process model of sleep regulation. Hum. Neurobiol. 1, 195–204 (1982).
Borbély, A. A., Daan, S., Wirz-Justice, A. & Deboer, T. The two-process model of sleep regulation: a reappraisal. J. Sleep Res. 25, 131–143 (2016).
Ikeda-Sagara, M. et al. Induction of prolonged, continuous slow-wave sleep by blocking cerebral H1 histamine receptors in rats. Br. J. Pharmacol. 165, 167–182 (2012).
Marzanatti, M., Monopoli, A., Trampus, M. & Ongini, E. Effects of nonsedating histamine H1-antagonists on EEG activity and behavior in the cat. Pharmacol. Biochem. Behav. 32, 861–866 (1989).
Saitou, K., Kaneko, Y., Sugimoto, Y., Chen, Z. & Kamei, C. Slow wave sleep-inducing effects of first generation H1-antagonists. Biol. Pharm. Bull. 22, 1079–1082 (1999).
Niethard, N. et al. Sleep-stage-specific regulation of cortical excitation and inhibition. Curr. Biol. 26, 2739–2749 (2016).
Dissel, S. et al. Sleep restores behavioral plasticity to Drosophila mutants. Curr. Biol. 25, 1270–1281 (2015).
Che Has, A. T. et al. Zolpidem is a potent stoichiometry-selective modulator of α1β3 GABAA receptors: evidence of a novel benzodiazepine site in the α1–α1 interface. Sci. Rep. 6, 28674 (2016).
Sitaram, N., Wyatt, R. J., Dawson, S. & Gillin, J. C. REM sleep induction by physostigmine infusion during sleep. Science 191, 1281–1283 (1976).
Callaway, C. W., Lydic, R., Baghdoyan, H. A. & Hobson, J. A. Pontogeniculooccipital waves: spontaneous visual system activity during rapid eye movement sleep. Cell. Mol. Neurobiol. 7, 105–149 (1987).
Datta, S. Cellular basis of pontine ponto-geniculo-occipital wave generation and modulation. Cell. Mol. Neurobiol. 17, 341–365 (1997).
Baghdoyan, H. A., Lydic, R., Callaway, C. W. & Hobson, J. A. The carbachol-induced enhancement of desynchronized sleep signs is dose dependent and antagonized by centrally administered atropine. Neuropsychopharmacology 2, 67–79 (1989).
Coleman, C. G., Lydic, R. & Baghdoyan, H. A. M2 muscarinic receptors in pontine reticular formation of C57BL/6J mouse contribute to rapid eye movement sleep generation. Neuroscience 126, 821–830 (2004).
Datta, S., Quattrochi, J. J. & Hobson, J. A. Effect of specific muscarinic M2 receptor antagonist on carbachol induced long-term REM sleep. Sleep 16, 8–14 (1993).
Árnason, B. B., Þorsteinsson, H. & Karlsson, K. A. E. Absence of rapid eye movements during sleep in adult zebrafish. Behav. Brain Res. 291, 189–194 (2015).
Roessmann, U., Velasco, M. E., Sindely, S. D. & Gambetti, P. Glial fibrillary acidic protein (GFAP) in ependymal cells during development. An immunocytochemical study. Brain Res. 200, 13–21 (1980).
Conductier, G. et al. Control of ventricular ciliary beating by the melanin concentrating hormone-expressing neurons of the lateral hypothalamus: a functional imaging survey. Front. Endocrinol. 4, 182 (2013).
Dale, N. Purinergic signaling in hypothalamic tanycytes: potential roles in chemosensing. Semin. Cell Dev. Biol. 22, 237–244 (2011).
Rizzoti, K. & Lovell-Badge, R. Pivotal role of median eminence tanycytes for hypothalamic function and neurogenesis. Mol. Cell. Endocrinol. 445, 7–13 (2017).
Peyron, C., Sapin, E., Leger, L., Luppi, P. H. & Fort, P. Role of the melanin-concentrating hormone neuropeptide in sleep regulation. Peptides 30, 2052–2059 (2009).
Logan, D. W., Burn, S. F. & Jackson, I. J. Regulation of pigmentation in zebrafish melanophores. Pigment Cell Res. 19, 206–213 (2006).
Torterolo, P., Lagos, P., Sampogna, S. & Chase, M. H. Melanin-concentrating hormone (MCH) immunoreactivity in non-neuronal cells within the raphe nuclei and subventricular region of the brainstem of the cat. Brain Res. 1210, 163–178 (2008).
Conductier, G. et al. Melanin-concentrating hormone regulates beat frequency of ependymal cilia and ventricular volume. Nat. Neurosci. 16, 845–847 (2013).
Hassani, O. K., Lee, M. G. & Jones, B. E. Melanin-concentrating hormone neurons discharge in a reciprocal manner to orexin neurons across the sleep–wake cycle. Proc. Natl Acad. Sci. USA 106, 2418–2422 (2009).
Jego, S. et al. Optogenetic identification of a rapid eye movement sleep modulatory circuit in the hypothalamus. Nat. Neurosci. 16, 1637–1643 (2013).
Kawauchi, H., Kawazoe, I., Tsubokawa, M., Kishida, M. & Baker, B. I. Characterization of melanin-concentrating hormone in chum salmon pituitaries. Nature 305, 321–323 (1983).
Kwan, K. M. et al. The Tol2kit: a multisite gateway-based construction kit for Tol2 transposon transgenesis constructs. Dev. Dyn. 236, 3088–3099 (2007).
Hieber, V., Dai, X., Foreman, M. & Goldman, D. Induction of α1-tubulin gene expression during development and regeneration of the fish central nervous system. J. Neurobiol. 37, 429–440 (1998).
Muto, A., Ohkura, M., Abe, G., Nakai, J. & Kawakami, K. Real-time visualization of neuronal activity during perception. Curr. Biol. 23, 307–311 (2013).
Bernardos, R. L. & Raymond, P. A. GFAP transgenic zebrafish. Gene Expr. Patterns 6, 1007–1013 (2006).
Tabor, K. M. et al. Direct activation of the Mauthner cell by electric field pulses drives ultrarapid escape responses. J. Neurophysiol. 112, 834–844 (2014).
Appelbaum, L. et al. Circadian and homeostatic regulation of structural synaptic plasticity in hypocretin neurons. Neuron 68, 87–98 (2010).
Renier, C. et al. Genomic and functional conservation of sedative-hypnotic targets in the zebrafish. Pharmacogenet. Genomics 17, 237–253 (2007).
Rihel, J. et al. Zebrafish behavioral profiling links drugs to biological targets and rest/wake regulation. Science 327, 348–351 (2010).
Pitrone, P. G. et al. OpenSPIM: an open-access light-sheet microscopy platform. Nat. Methods 10, 598–599 (2013).
Acknowledgements
We thank all members of the Mourrain laboratory for helpful feedback and discussion during the project and preparation of the manuscript; L. de Lecea, J. Zeitzer, C. Heller, D. Grahn, D. Colas and A. Adamantidis for important feedback; P. Raymond, K. Kwan and H. Burgess for sharing constructs and lines; S. Nishino for the kind gift of zolpidem; L. Alexandre and S. Murphy for their diligent care of our zebrafish; Stanford Cell Sciences Imaging Facility for imaging assistance (funded by NCRR award S10RR02557401); and J. Goldberg (Research To Prevent Blindness) and Stanford Vision Research Core (NIH P30-EY0268771). Funding support was provided by Stanford School of Medicine Dean’s fellowship (L.C.L.); JP18H04988, NBRP from AMED (K.K.); NIMH, NIDA, DARPA, NSF, Wiegers Family Fund, AE Foundation, Tarlton Foundation, and Gatsby Foundation (K.D.); NIMH, NINDS, Tashia and John Morgridge Fund (A.E.U.); and NIDDK (5R01DK090065-05), NINDS, NIMH, NIA, Bright-Focus Foundation, Simons Foundation and John Merck Fund (P.M.).
Reviewer information
Nature thanks Herwig Baier and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
L.C.L., G.X.W. and P.M. conceived the study. L.C.L. and P.M. designed experiments. L.C.L. and G.X.W. built the imaging platform and computing hardware. L.C.L., R.M., K.K. and G.S. performed cloning and generated transgenic lines and mutants. L.C.L. performed all experiments except for two-photon imaging (G.X.W.). L.C.L. and G.X.W. developed and wrote the computational pipeline. L.C.L. performed analysis and produced the figures. K.D. provided project mentorship. A.E.U. provided genome analysis and mentorship. L.C.L. and P.M. wrote the paper with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Sleep deprivation by the continuous-swim paradigm.
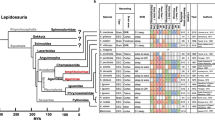
a, Left, evolutionary tree depicting current description of SWS and PS/REM sleep in amniotes. Right, schematic showing lateral view of major brain regions and neurochemistry that is implicated in sleep. Ob, olfactory bulbs; Am, amygdala; Hi, hippocampus; Te, telencephalon; Mi, midbrain; Cb, cerebellum; Md, medulla. b, Schematic showing the sleep deprivation (SD) setup (see Methods). c, d, Behavioural actimetry verifies that the sleep rebound induced by sleep deprivation causes decreasing wake and increasing sleep amounts as a function of number of days of sleep deprivation. Trial-averaged traces across 24 h after zebrafish were released following 1 day (n = 11, purple (c) or red (d)), 2 days (n = 11, blue (c) or orange (d)) or 3 days (n = 11, green (c) or yellow (d)) of sleep deprivation, against non-sleep-deprived sibling controls (n = 12, black). Mean activity is expressed as seconds of activity per 10-min bin (bold line) ± s.e.m. (shaded), per condition. Mean sleep amount is expressed as minutes of sleep per 10-min bin (bold) ± s.e.m. (shaded), per condition. The black triangle depicts the day-to-night transition, the white triangle depicts the night-to-day transition and the orange triangle shows the time of feeding. Red box in c indicates the equivalent time at which Ca2+ imaging was conducted. e, Box plots showing the spread of total amounts of activity and sleep between non-sleep-deprived and sleep-deprived conditions, separated out by day and night. Blue lines denote median and black circles present the mean value for the group. *P < 0.05, one-way ANOVA with 0.05 alpha level and Tukey’s post hoc multi-comparison testing. f, Scatter plot of average ITI from desynchronized and synchronized brain activities. *P < 0.05, two-sided Wilcoxon rank-sum test, n = 10 fish per condition. g, Bar chart showing proportion of samples that displayed SBS within 2 h of release from non-sleep-deprived, 1-day, 2-day and 3-day sleep deprived conditions.
Extended Data Fig. 2 Hypnotics produce distinct neural dynamics, including synchronous activity, travelling waves and broad silencing.
a–e, Left, representative activity from 3-h recordings in which drugs were added after 1 h (red triangle) to freely swimming zebrafish. Activity traces are expressed as the number of seconds of activity per 10-min time bin, and plotted as mean (solid line) ± s.e.m. (shaded) per dose of promethazine (a, n = 24 fish), MS222 (b, n = 24 fish), gaboxadol (c, n = 24 fish), zolpidem (d, n = 24 fish) and eserine (e, n = 24 fish) from low to high doses (black < purple < blue < green; see Supplementary Table 1 for concentrations), showing the induction of dose-responsive behavioural sleep. Middle, representative time-aligned ΔF/F traces of 20 randomly selected dorsal pallium neurons (red triangle indicates the addition of drug after 10 min). Promethazine induced mild synchronization of dorsal pallium neurons, MS222 and gaboxadol induce total silencing, and zolpidem and eserine induced travelling waves. Right, neural signature across all time points represented by the first independent component (top) or replotted on two-dimensional phase space following PCA (bottom, grey lines and black lines depict wake versus sleep trajectories, respectively). See Supplementary Information for reproducibility information.
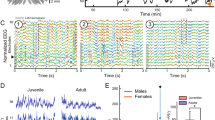
Extended Data Fig. 3 fPSG of SBS and PWS.
a, b, Dorsal pallium signature of wake (a) versus SBS (b) with time-aligned ΔF/F traces of 20 randomly selected masks. Neural signature plotted in two-dimensional phase space following PCA. c–e, Typical light-sheet images of a single z-slice through brain and trunk muscle during wake (c) and during SBS (d, e). Scale bar, 100 μm. f, g, Dorsal pallium signature of wake (f) versus PWS (g) with time-aligned ΔF/F traces of the 20 masks depicted in the left image. Pre- versus post-PMT-wave synchronicity indices: ITI, 2.88 ± 2.14 s versus 2.96 ± 1.35 s, P = 0.792, Wilcoxon rank-sum test, n = 5 fish; coherence index, 1.32% (n = 1,024 spikes) versus 1.08% (n = 1,065 spikes); P > 0.05, χ2 test. Neural signature plotted in two-dimensional phase space following PCA. Scale bars, 50 μm. h, Subpanels h1–h6 show selected frames from a typical fPSG volume acquisition of PWS in which z-planes have been compressed into a maximum projection. Frames depict preceding events up until, and through, the PMT-wave transition to sleep. Arrow in h1 indicates muscle rostrocaudal wave; arrows in h2 indicates coordinated ependymal cell activation; arrow in h3 indicates anterior hindbrain activation; arrow in h4 indicates wave initiation; arrow in h6 indicates propagation to the dorsal pallium (see Supplementary Information for detailed description). Scale bar, 100 μm. See Supplementary Information for reproducibility information.
Extended Data Fig. 4 Heart rate analysis.
a–c, Heart rate analysis before and after carbachol treatment (a) (*P < 0.05, two-sided paired t-test, n = 4 fish), mepyramine treatment (b) (*P < 0.05; two-sided paired t-test, n = 4 fish) and with or without sleep deprivation (c) (*P < 0.05; two-sided Student’s t-test, n = 7 non-sleep-deprived and 7 sleep-deprived fish). d–f, Histograms comparing the distribution of interbeat interval before (black bars) and after (red bars) treatment with carbachol (d) or mepyramine (e), and with (red bars) or without (black bars) sleep deprivation (f). Three representative histograms are shown for each condition. g, Representative images from heart rate capture showing ventricular contraction and the drawing of a mask (red) to measure fluorescence changes from the green fluorescent heartbeat. h, An illustrative plot of fluorescence variation measured by the mask, overlaid with the result of running peak analysis (red triangles) from which the heart rate and interbeat interval are derived.
Extended Data Fig. 5 Brain-wide imaging with additional hypnotics.
a, Brain-wide single-plane imaging of promethazine-stimulated zebrafish. Promethazine induces bilaterally synchronous burst dynamics in the dorsal pallium (red bar). Left, masks used to extract Ca2+ transient traces shown in the middle panel. Dynamics are summarized by PCA (right). b, Brain-wide single-plane imaging of eserine-stimulated zebrafish. Eserine induces PMT waves. Left, masks used to extract Ca2+ transient traces shown in the middle panel. Dynamics are summarized by PCA (right). c, Brain-wide single-plane imaging of carbachol-stimulated zebrafish pre-incubated with methoctramine. Methoctramine prevents carbachol-induction of the PMT wave. Left, masks used to extract Ca2+ transient traces shown in the middle panel. Dynamics are summarized by PCA (right). d, Brain-wide single-plane imaging of eserine-stimulated zebrafish pre-incubated with methoctramine. Methoctramine prevents the eserine-induced PMT dynamics. Left, masks used to extract Ca2+ transient traces shown in the middle panel. Dynamics are summarized by PCA (right). e, Brain-wide single-plane imaging of zolpidem-stimulated zebrafish. Zolpidem induced multiple PMT waves. Left, masks used to extract Ca2+ transient traces shown in the middle panel. Dynamics are summarized by PCA (right). Red triangles show the point at which the drug was added. f, PCA by singular value decomposition of the fPSG PMT signature seen in Fig. 3e. Distinct steady states for wake (grey lines) and PWS sleep (black lines) can be separated, as can marked trajectories of muscle and brain travelling waves (blue lines). See Supplementary Information for reproducibility information. Scale bars, 100 μm.
Extended Data Fig. 6 Ependymal cell activation is tightly correlated to the PMT wave and can be induced by the neuropeptide melanin concentrating hormone.
a, Dorsal view of a single slice through the optic tectum of Et(EP:mCherry) fish. Inset dashed box indicates location along the anteroposterior axis. Scale bar, 50 μm. b, Slice through the optic tectum of a 7-dpf Et(EP:mCherry);Tg(α-tubulin:gal4;UAS:GFP) triple transgenic fish. Arrows, mature ependymal cells (magenta) co-expressing α-tubulin (green). c, Slice through the optic tectum of a 7-dpf Et(EP:mCherry);Tg(GFAP:gal4;UAS:GFP) triple transgenic fish. Mature ependymal cells largely do not overlap with GFAP-positive cells. d–g, Ventricular cells activated before the PMT wave are EP:mCherry-positive ependymal cells. Confocal z-slice through a 7-dpf zPSG;EP:mCherry double transgenic fish before stimulation (d), ependymal cell activation (e) and PMT wave (f). g, Mean ± s.e.m. ΔF/F trace of five randomly selected double-positive cells showing the transient activation of ependymal cells before the PMT wave. Arrows indicate the time from which images shown in d–f are taken. Scale bar, 50 μm. h–m, MCH peptide induces activation of ependymal cells. Representative maximum projection through SPIM volume acquisition before (h, j) and 5 min after intracerebroventricular injection with carrier (i) or MCH peptide (k). l, Mean normalized fluorescence between carrier (n = 9 fish) and MCH (n = 12 fish) groups from the peritectoventricular zone (dashed lines) before and after injection. m, Box plot of normalized fluorescence percentage change values between carrier- and MCH-injected groups with black circle depicting mean. *P < 0.05; two-sided Student’s t-test. Scale bar, 100 μm. n, Pigment contraction is tightly coupled to discrete activations during PMT-wave dynamics. Representative frames selected from a single z-recording of a pigmented zebrafish during a PMT wave; (1) colour-coded selection of all pigments covering the optic tectum as a representative sample; (2) during darkness and baseline activity, pigments are spread out and cover more pixel area; (3, 4) pigments contract with ependymal cell activation and the PMT wave; (5) when the wave abates, pigments re-spread. Scale bar, 100 μm. o, Quantification of pixel area plotted with representative colour from subpanel (1) in n at the different time points shown in subpanels (1) to (5) in n. *P < 0.05, repeated-measures ANOVA, n = 10 pigments. See Supplementary Information for pigment dynamics description and reproducibility information.
Extended Data Fig. 7 Characterization of GFAP:epNTR and EP-mCherry transgenic fish.
a, Dorsal view of maximum projection of a 7-dpf Tg(GFAP:epNTR-tagRFPT) zebrafish used to ablate GFAP-positive cells. Scale bar, 100 μm. b, Maximum projection of a 7-dpf Et(EP:mCherry) zebrafish expressing mCherry in putative ependymal cells. Scale bar, 50 μm. c, Single z-slice of dorsal pallium of Et(EP:mCherry) fish. mCherry cells located on the ventricular surface of the telencephalic neuroepithelium and present in the midline. Scale bar, 50 μm. d, Single z-slice of optic tectum parenchyma of Et(EP:mCherry) fish. mCherry cells are located all along the ventricular surface comprising the ependyma. Long thin processes invade the mesencephalic parenchyma, which suggests the morphology of non-neuronal glial cells and (in particular) the specialized ependymal cell type, tanycytes. e, Single z-slice of hindbrain of Et(EP:mCherry) fish. mCherry cells are located all along the midline ventricular surface, representing the ependyma stretching to spinal cord central canal. Long thin processes suggest the morphology of non-neuronal cells. f, g, Actimetry shows GFAP-cell-ablated fish are irreversibly immobile without recovering activity during daytime. Mean activity is expressed as seconds of activity per 10-min bin (bold) ± s.e.m. (shaded), per condition. Mean sleep amount is expressed as minutes of sleep per 10-min bin (bold) ± s.e.m. (shaded), per condition. The black triangle depicts the day-to-night transition, the white triangle depicts the night-to-day transition and the orange triangle shows the time of feeding. Box plots showing total activity and sleep for sibling controls versus GFAP-cell-ablated larva. Blue line denotes median and black circles indicate the mean value (g). *P < 0.05, two-sided Student’s t-test, n = 18 sibling control and 18 GFAP:epNTR fish from two independent experiments. h, Photograph at 4× magnification of a restrained 7-dpf zebrafish receiving an intracerebroventricular injection of MCH solution with phenol red solution (pale red colour in the photograph). The ventricle is slightly expanded as a result of injection. Scale bar, 500 μm. i, j, Behavioural actimetry shows MCH-injected larvae are less mobile and sleep more during the first day and night after injection, but mostly recover by the second day. Box plots showing total activity and sleep for carrier- versus MCH-injected larva. Blue line denotes median and black asterisks indicate the mean value (j). P > 0.05, two-sided Student’s t-test, n = 6 carrier- and 6 MCH-injected fish.
Extended Data Fig. 8 Characterization of MCH:epNTR, H6408 and mchr1a mchr1b double mutants.
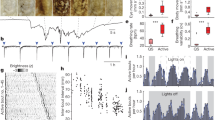
a, Schematic of the location of MCH-positive cells in the lateral hypothalamus of the zebrafish larvae. b, Confocal maximum projection shows MCH cells from the Tg(−5kbMCH:epNTR-tagRFPT) transgenic fish before treatment with the ablation agent metronidazole (MTZ). Faint signal of projections to the pituitary are seen (dashed region and asterisk). Scale bar, 50 μm. c, MCH cells are ablated in Tg(−5kbMCH:epNTR-tagRFPT) larva treated with metronidazole. d–f, Tg(−5kbMCH:epNTR-tagRFPT) larva stained with MCH antibody show lateral hypothalamic location and projection to the pituitary gland (d) (green) co-localizing to tagRFP-T expression (e, magenta; f, white). g, Alignment of wild-type MCHR1a protein sequence and predicted truncated mutant protein resulting from CRISPR deletion. h, Sequencing confirmation of loss of TATG sequence from exon 2 of mchr1a gene from genotyping of homozygous MCHR1a mutant. i, Alignment of wild-type MCHR1b protein sequence and predicted truncated mutant protein resulting from TILLING mutant with early stop codon. j, Sequencing confirmation of TGG to TGA substitution from exon 1 of mchr1b gene from genotyping of homozygous MCHR1b mutant. k–m, MCHR-antagonist (H6408)-treated fish display increased activity and disrupted onset of ependymal cell and PMT-wave dynamics. k, Top, timeline of experiment and addition of pharmacological agents during the 2-h Ca2+ imaging experiment (red triangle indicates the addition of carbachol). Bottom, proportion of activation of ependymal cells (open circle) and/or PMT wave (filled circle), witnessed in the imaging session between carrier control (n = 8 fish) and H6408 (n = 7 fish) is plotted against the mean latency to activation (in min) ± s.e.m. *P < 0.05 χ2 test. l, Top, actimetry of wild-type zebrafish (n = 20) treated with carrier versus MCHR antagonist (H6408; n = 20) in 10-min bins over a 24-h period, showing mean seconds of activity per bin (bold) ± s.e.m. (shaded). Bottom, sleep depicted as minutes of sleep per 10-min bin (bold) ± s.e.m. across a 24-h period. Black triangle depicts the day-to-night transition, white triangle depicts the night-to-day transition and the orange triangle shows the time of feeding. m, Box plots showing spread of total activities and sleep between between carrier (E3)-stimulated versus H6408-treated groups, separated further by day and night. Blue lines denote median and black circles indicate the mean value. *P < 0.05, two-sided Student’s t-test. See Supplementary Information for reproducibility information.
Extended Data Fig. 9 fPSG comparison of induced SBS in non-sleep-deprived and sleep-deprived fish.
a, b, Representative fPSG recording of non-sleep deprived (a) and sleep-deprived zebrafish (b) during normal night phase. Activity traces from fEOG (top), fEEG (middle) and fEMG (middle). fEEG and fEMG are ΔF/F traces from 14 masks covering broad brain regions (as labelled), with integration of neuronal activity across 1-s bins. Heart rate was measured immediately before imaging, and interbeat interval distribution is shown with coefficient of variation (CoV) (bottom). Transient synchronous bursting was detected (red boxes for example) between infrequent muscle twitches and broad brain arousal events. See Supplementary Information for reproducibility information.
Extended Data Fig. 10 SPIM of wake-maintained larvae reveal SBS and PWS signatures during normal night.
a, Schematic of wake-maintenance apparatus adopting randomized mechano-acoustical stimulus during the day phase to consolidate wake, and thus sleep in the night phase for imaging. b, Representative actimetry of wild-type zebrafish not treated with the stimulus (n = 12 fish) with actimetry of wild-type fish treated with the stimulus (n = 12 fish) in 10-min bins over a 24-h period, showing mean seconds of activity per bin (bold) ± s.e.m. (shaded). Bottom, sleep depicted as minutes of sleep per 10-min bin (bold) ± s.e.m. (shaded) across the 24-h period. The black triangle depicts the day-to-night transition, the white triangle depicts the night-to-day transition and the orange triangle shows the time of feeding. c, d, fPSG recording of SBS (c) and PWS (d) during normal night phase. Red box shows transient synchrony in dorsal pallium, indicative of SBS. Activity traces from fEOG (top), fEEG (middle) and fEMG (bottom). fEEG and fEMG are ΔF/F traces from 14 masks covering broad brain regions (as labelled), with integration of neuronal activity across 1-s bins. See Supplementary Information for reproducibility information.
Extended Data Fig. 11 SPIM imaging reveals spontaneous SBS and PWS during the night phase.
a, Schematic of protocol to sham mount versus extended agarose mount for 1–3 days to test the degree of wake stimulation resulting from agarose restraint. b, Behavioural actimetry shows that while initially agarose-mounted animals display sleep behaviour upon release, sleep–wake cycles return to normal; this indicates that long-term restraint is not traumatic or lethal to zebrafish larvae. Actimetry of sham-mounted (n = 36 fish) versus mounted (n = 31 fish) wild-type zebrafish, in 10-min bins over a 24-h period showing mean seconds of activity per bin (bold) ± s.e.m. (shaded). Bottom, sleep depicted as minutes of sleep per 10-min bin (bold) ± s.e.m. (shaded), across the 24-h period. The black triangles depict the day-to-night transition, the white triangles depict the night-to-day transition and the orange triangle shows the time of feeding. c, d, fPSG recording of SBS (c) and PWS (d) during normal night phase. Red box shows transient synchrony in dorsal pallium, indicative of SBS between muscle twitches. Activity traces from fEOG (top), fEEG (middle) and fEMG (bottom). fEEG and fEMG are ΔF/F traces from 14 masks covering broad brain regions (as labelled), with integration of neuronal activity across 1-s bins. See Supplementary Information for reproducibility information.
Supplementary information
Supplementary Information
This file contains supplementary text; which includes Supplementary Figures 1 and 2 and Supplementary Tables 1 and 2. Further details on this file can be found in the Supplementary Information Guide.
Supplementary Information
This file contains the Supplementary Information Guide. It contains more details on the supplementary text, and also includes the links to supplementary videos 1–19 and the video legends, which can also be downloaded from https://drive.google.com/open?id=1CQGSFzxm39KCvN9D_XwJR2rOY6hS7oC5.
Source data
Rights and permissions
About this article
Cite this article
Leung, L.C., Wang, G.X., Madelaine, R. et al. Neural signatures of sleep in zebrafish. Nature 571, 198–204 (2019). https://doi.org/10.1038/s41586-019-1336-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1336-7
This article is cited by
-
Neuronal dynamics direct cerebrospinal fluid perfusion and brain clearance
Nature (2024)
-
Understanding zebrafish sleep and wakefulness physiology as an experimental model for biomedical research
Fish Physiology and Biochemistry (2024)
-
Phylogenetic conservation of the interdependent homeostatic relationship of sleep regulation and redox metabolism
Journal of Comparative Physiology B (2024)
-
Interhemispheric competition during sleep
Nature (2023)
-
Wake-like skin patterning and neural activity during octopus sleep
Nature (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.