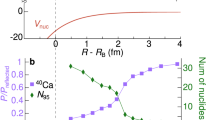
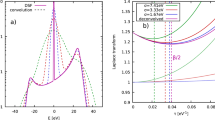
Abstract
The 1989 claim of ‘cold fusion’ was publicly heralded as the future of clean energy generation. However, subsequent failures to reproduce the effect heightened scepticism of this claim in the academic community, and effectively led to the disqualification of the subject from further study. Motivated by the possibility that such judgement might have been premature, we embarked on a multi-institution programme to re-evaluate cold fusion to a high standard of scientific rigour. Here we describe our efforts, which have yet to yield any evidence of such an effect. Nonetheless, a by-product of our investigations has been to provide new insights into highly hydrided metals and low-energy nuclear reactions, and we contend that there remains much interesting science to be done in this underexplored parameter space.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Fleischmann, M. & Pons, S. Electrochemically induced nuclear fusion of deuterium. J. Electroanal. Chem. Interfacial Electrochem. 261, 301–308 (1989). Article that introduced the possibility of room-temperature fusion during electrolysis with a palladium cathode in a LiOD/D 2 O electrolyte solution, publicly revealed at a press conference at the University of Utah on 23 March 1989.
Cookson, C. Scientists claim nuclear fusion produced in test tube. Financial Times 1 (23 March 1989).
Bishop, J. Breakthrough in fusion may be announced. Wall Street J. B1 (23 March 1989).
Browne, M. W. Fusion in a jar: chemists’ claim ignites an uproar. New York Times 1 (28 March 1989).
Miskelly, G. M. et al. Analysis of the published calorimetric evidence for electrochemical fusion of deuterium in palladium. Science 246, 793–796 (1989).
Albagli, D. et al. Measurement and analysis of neutron and gamma-ray emission rates, other fusion products, and power in electrochemical cells having Pd cathodes. J. Fusion Energy 9, 133–148 (1990).
Williams, D. E. et al. Upper bounds on ‘cold fusion’ in electrolytic cells. Nature 342, 375–384 (1989).
Maddox, J. Farewell (not fond) to cold fusion. Nature 344, 365–366 (1990).
USDOE Office of Energy Research, Energy Research Advisory Board. Cold Fusion Research. Report DOE/S-0073 https://doi.org/10.2172/5144772 (USDOE, 1989).
Mullins, J. Cold fusion back from the dead. IEEE Spectrum 41, 22–23 (2004).
Ritter, S. K. Cold fusion died 25 years ago, but the research lives on. Chem. Eng. News 94, 34–39 (2016).
Krivit, S. B. & Marwan, J. A new look at low-energy nuclear reaction research. J. Environ. Monit. 11, 1731–1746 (2009).
Sanderson, K. Cold fusion is back at the American Chemical Society. Nature https://doi.org/10.1038/news070326-12 (2007).
Goodstein, D. Whatever happened to cold fusion? Account. Res. 8, 59–75 (2000).
Krivit, S. B. in Low-Energy Nuclear Reactions Sourcebook Vol. 998 (eds Marwan, J. & Krivit, S. B.) 3–16 (American Chemical Society, 2008).
Brumfiel, G. US review rekindles cold fusion debate. Nature https://www.nature.com/news/2004/041129/full/news041129-11.html (2004).
Mohtadi, R. & Orimo, S.-I. The renaissance of hydrides as energy materials. Nat. Rev. Mater. 2, 16091 (2016).
Montoya, J. H. et al. Materials for solar fuels and chemicals. Nat. Mater. 16, 70–81 (2016).
Seh, Z. W. et al. Combining theory and experiment in electrocatalysis: insights into materials design. Science 355, eaad4998 (2017).
Stamenkovic, V. R., Strmcnik, D., Lopes, P. P. & Markovic, N. M. Energy and fuels from electrochemical interfaces. Nat. Mater. 16, 57–69 (2016).
Teschner, D. et al. The roles of subsurface carbon and hydrogen in palladium-catalyzed alkyne hydrogenation. Science 320, 86–89 (2008).
Liu, N., Tang, M. L., Hentschel, M., Giessen, H. & Alivisatos, A. P. Nanoantenna-enhanced gas sensing in a single tailored nanofocus. Nat. Mater. 10, 631–636 (2011).
Li, G. et al. Hydrogen storage in Pd nanocrystals covered with a metal–organic framework. Nat. Mater. 13, 802–806 (2014).
Li, G. et al. Shape-dependent hydrogen-storage properties in Pd nanocrystals: which does hydrogen prefer, octahedron (111) or cube (100)? J. Am. Chem. Soc. 136, 10222–10225 (2014).
Huke, A. et al. Enhancement of deuteron-fusion reactions in metals and experimental implications. Phys. Rev. C 78, 015803 (2008).
Assenbaum, H. J., Langanke, K. & Rolfs, C. Effects of electron screening on low-energy fusion cross sections. Z. Phys. A 327, 461–468 (1987).
Laurence, W. L. Cold fusion of hydrogen atoms; a fourth method pulling together. New York Times E7 (30 December 1956).
Rafelski, J. & Jones, S. E. Cold nuclear fusion. Sci. Am. 257, 84–89 (1987).
Jones, S. E. et al. Observation of cold nuclear fusion in condensed matter. Nature 338, 737–740 (1989).
Rolfs, C. E. & Rodney, W. S. Cauldrons in the Cosmos: Nuclear Astrophysics (Univ. Chicago Press, 1988).
Miley, G. H., Towner, H. & Ivich, N. Fusion Cross Sections and Reactivities (Illinois Univ., 1974).
Raiola, F. et al. Enhanced electron screening in d(d,p)t for deuterated metals. Eur. Phys. J. A 19, 283–287 (2004).
Bertulani, C. A. & Kajino, T. Frontiers in nuclear astrophysics. Prog. Part. Nucl. Phys. 89, 56–100 (2016).
Spitaleri, C., Bertulani, C. A., Fortunato, L. & Vitturi, A. The electron screening puzzle and nuclear clustering. Phys. Lett. B 755, 275–278 (2016).
Czerski, K. et al. Screening and resonance enhancements of the 2H(d, p)3H reaction yield in metallic environments. Europhys. Lett. 113, 22001 (2016).
Coraddu, M., Lissia, M. & Quarati, P. Anomalous enhancements of low-energy fusion rates in plasmas: the role of ion momentum distributions and inhomogeneous screening. Cent. Eur. J. Phys. 7, 527–533 (2009).
US Department of Energy. Report of the Review of Low Energy Nuclear Reactions (USDOE, 2004). Report from a review conducted in 2004 by the US Department of Energy to revisit the question of scientific evidence for low-energy nuclear reactions.
Packham, N. J. C., Wolf, K. L., Wass, J. C., Kainthla, R. C. & Bockris, J. O. Production of tritium from D2O electrolysis at a palladium cathode. J. Electroanal. Chem. Interfacial Electrochem. 270, 451–458 (1989).
McKubre, M. C. H. Cold fusion: comments on the state of scientific proof. Curr. Sci. 108, 495–498 (2015). Discussion of early experiments performed to assess the Fleischmann–Pons claim.
Focardi, S., Habel, R. & Piantelli, F. Anomalous heat production in Ni-H systems. Nuovo Cim. A 107, 163–167 (1994).
Claytor, T. N., Jackson, D. D. & Tuggle, D. G. Tritium Production from a Low Voltage Deuterium Discharge on Palladium and Other Metals. https://doi.org/10.2172/102234 (LANL, 1995). Plasma loading method claimed to produce an unexpected amount of tritium from palladium wires.
Wicke, E., Brodowsky, H. & Züchner, H. in Hydrogen in Metals II (eds Alefield, G. & Völki, J.) 73–155 (Springer, 1978).
Schirber, J. E. & Morosin, B. Lattice constants of β – PdHx and β – PdDx with x near 1.0. Phys. Rev. B 12, 117–118 (1975).
Fukai, Y. & Ōkuma, N. Evidence of copious vacancy formation in Ni and Pd under a high hydrogen pressure. Jpn. J. Appl. Phys. 32, L1256–L1259 (1993).
Fukai, Y. & Okuma, N. Formation of superabundant vacancies in Pd hydride under high hydrogen pressures. Phys. Rev. Lett. 73, 1640–1643 (1994).
Skoskiewicz, T. Superconductivity in the palladium-hydrogen and palladium-nickel-hydrogen systems. Phys. Status Solidi A 11, K123–K126 (1972).
Miller, R. J. & Satterthwaite, C. B. Electronic model for the reverse isotope effect in superconducting Pd-H(D). Phys. Rev. Lett. 34, 144–148 (1975).
Harper, J. M. E. Effect of hydrogen concentration on superconductivity and clustering in palladium hydride. Phys. Lett. A 47, 69–70 (1974).
Pröbst, F., Wagner, F. E. & Karger, M. Magnetic properties of solute 57Fe in β phase palladium hydride. J. Less Common Met. 88, 201–209 (1982).
Möller, W., Besenbacher, F. & Bottiger, J. Saturation and isotope mixing during low-temperature implantations of hydrogen into metals. Appl. Phys. A 27, 19–29 (1982).
Fukai, Y. et al. Superabundant vacancy–hydrogen clusters in electrodeposited Ni and Cu. J. Alloys Compd. 356–357, 270–273 (2003).
Murray, J. B. et al. Apparatus for combined nanoscale gravimetric, stress, and thermal measurements. Rev. Sci. Instrum. 89, 085106 (2018).
Benck, J. D., Jackson, A., Young, D., Rettenwander, D. & Chiang, Y.-M. Producing high concentrations of hydrogen in palladium via electrochemical insertion from aqueous and solid electrolytes. Chem. Mater. https://doi.org/10.1021/acs.chemmater.9b01243 (2019). Electrochemical insertion of hydrogen into palladium electrodes using aqueous, polymer and ceramic electrolytes at room temperature and ambient pressure to produce PdH x as high as x = 0.96 ± 0.02.
Felici, R., Bertalot, L., DeNinno, A., LaBarbera, A. & Violante, V. In situ measurement of the deuterium (hydrogen) charging of a palladium electrode during electrolysis by energy dispersive x-ray diffraction. Rev. Sci. Instrum. 66, 3344–3348 (1995).
Knies, D. L. et al. In-situ synchrotron energy-dispersive X-ray diffraction study of thin Pd foils with Pd:D and Pd:H concentrations up to 1:1. J. Appl. Phys. 112, 083510 (2012).
Maoka, T. & Enyo, M. Hydrogen absorption by palladium electrode polarized in sulfuric acid solution containing surface active substances—I. The cathodic region. Electrochim. Acta 26, 607–614 (1981).
Sherbo, R. S. et al. Accurate coulometric quantification of hydrogen absorption in palladium nanoparticles and thin films. Chem. Mater. 30, 3963–3970 (2018).
Benck, J. D., Rettenwander, D., Jackson, A., Young, D. & Chiang, Y.-M. Apparatus for operando X-ray diffraction of fuel electrodes in high temperature solid oxide electrochemical cells. Rev. Sci. Instrum. 90, 023910 (2019).
Lawson, D. R. et al. Use of a coulometric assay technique to study the variables affecting deuterium loading levels within palladium electrodes. Electrochim. Acta 36, 1515–1522 (1991).
Czerwiński, A. C., Kiersztyn, I., Grdeń, M. & Czapla, J. The study of hydrogen sorption in palladium limited volume electrodes (Pd-LVE) I. Acidic solutions. J. Electroanal. Chem. 471, 190–195 (1999).
Levi, G. et al. Indication of anomalous heat energy production in a reactor device. Preprint at https://arXiv.org/abs/1305.3913 (2013).
Young, D. et al. An operando calorimeter for high temperature electrochemical cells. Preprint at http://arXiv.org/abs/1808.02596 (2018).
MacLeod, B. P. et al. High-temperature high-pressure calorimeter for studying gram-scale heterogeneous chemical reactions. Rev. Sci. Instrum. 88, 084101 (2017).
MacLeod, B. P., Fork, D. K., Lam, B. & Berlinguette, C. P. Calorimetry under non-ideal conditions using system identification. J. Therm. Anal. Calorim. https://doi.org/10.1007/s10973-019-08271-z (2019).
Fork, D. K., MacLeod, B. P. & Parlane, F. G. L. Calorimetry System Identification (CSID). https://github.com/calorimetry/CSID (2017).
Broggini, C., Bemmerer, D., Caciolli, A. & Trezzi, D. LUNA: status and prospects. Prog. Part. Nucl. Phys. 98, 55–84 (2018).
Kaczmarski, M. et al. New accelerator facility for measurements of nuclear reactions at energies below 1 keV. Acta Phys. Pol. B 45, 509–518 (2014).
Schenkel, T. et al. Investigation of light ion fusion reactions with plasma discharges. Preprint at http://arXiv.org/abs/1905.03400 (2019). Observation of D–D reaction yields are about 100-fold higher than expected for bare nuclei in a plasma discharge experiment designed to demonstrate screening effects in light-ion fusion.
Best, A. et al. Underground nuclear astrophysics: why and how. Eur. Phys. J. A 52, 72 (2016).
Casey, D. T. et al. Thermonuclear reactions probed at stellar-core conditions with laser-based inertial-confinement fusion. Nat. Phys. 13, 1227–1231 (2017).
Arcones, A. et al. White paper on nuclear astrophysics and low energy nuclear physics. Part 1: Nuclear astrophysics. Prog. Part. Nucl. Phys. 94, 1–67 (2017).
ITER Organization. ITER Organization 2016 Annual Report. https://www.iter.org/org/team/odg/comm/AnnualReports (ITER, 2017).
Fountain, H. A dream of clean energy at a very high price. New York Times D1 (27 March 2017).
Sherbo, R. S., Delima, R. S., Chiykowski, V. A., MacLeod, B. P. & Berlinguette, C. P. Complete electron economy by pairing electrolysis with hydrogenation. Nature Catal. 1, 501–507 (2018).
Johnson, N. J. J. et al. Facets and vertices regulate hydrogen uptake and release in palladium nanocrystals. Nat. Mater. 18, 434–458 (2019). In situ X-ray diffraction experiments reveal the rate of hydrogen absorption by palladium nanocrystals increases with the number of vertices, and the rate of hydrogen desorption is approximately tenfold faster at (100) facets than at (111) facets.
Palm, K. J., Murray, J. B., Narayan, T. C. & Munday, J. N. Dynamic optical properties of metal hydrides. ACS Photonics 5, 4677–4686 (2018). Use of a strain-compensated microbalance for the in situ determination of gaseous hydrogen loading for various palladium, magnesium, zirconium, titanium and vanadium hydrides.
Koningstein, R. & Fork, D. Energy’s creative destruction. IEEE Spectrum 51, 30–35 (2014).
Hemminger, J., Fleming, G. & Ratner, M. Directing Matter and Energy: Five Challenges for Science and the Imagination. https://doi.org/10.2172/935427 (USDOE Basic Energy Sciences Advisory Committee, 2007).
Hemminger, J. C., Sarrao, J., Crabtree, G., Flemming, G. & Ratner, M. Challenges at the Frontiers of Matter and Energy: Transformative Opportunities for Discovery Science. https://doi.org/10.2172/1283188 (USDOE Basic Energy Sciences Advisory Committee, 2015).
Chadwick, M. B. et al. ENDF/B-VII.0: next generation evaluated nuclear data library for nuclear science and technology. Nucl. Data Sheets 107, 2931–3060 (2006).
Bosch, H. S. & Hale, G. M. Improved formulas for fusion cross-sections and thermal reactivities. Nucl. Fusion 32, 611–631 (1992).
Caughlan, G. R. & Fowler, W. A. Thermonuclear reaction rates V. Atom. Data Nucl. Data Tables 40, 283–334 (1988).
Salpeter, E. E. Electron screening and thermonuclear reactions. Aust. J. Phys. 7, 373–388 (1954).
Kasagi, J. et al. Strongly enhanced DD fusion reaction in metals observed for keV D+ bombardment. J. Phys. Soc. Jpn 71, 2881–2885 (2002).
Breunlich, W. H., Kammel, P., Cohen, J. S. & Leon, M. Muon-catalyzed fusion. Annu. Rev. Nucl. Part. Sci. 39, 311–356 (1989).
Williams, D. R. Sun fact sheet https://nssdc.gsfc.nasa.gov/planetary/factsheet/sunfact.html (2018).
Tumino, A. et al. An increase in the 12C + 12C fusion rate from resonances at astrophysical energies. Nature 557, 687–690 (2018); erratum 561, E3 (2018).
Ichimaru, S. Nuclear fusion in dense plasmas. Rev. Mod. Phys. 65, 255–299 (1993).
Acknowledgements
Students, postdoctoral researchers and staff responsible for advancing and supporting this project are listed as co-signatories in the Supplementary Information. P.A. Schauer, B. Lam, B.P. MacLeod, F.G.L. Parlane and A.K. Brown are gratefully acknowledged for their substantial contributions to the construction of this manuscript. The research supporting this Perspective was conducted with funding from Google LLC. Some aspects of the materials studies conducted at the University of British Columbia (UBC) were partially supported by the Canadian Natural Science and Engineering Research Council (CRD-502052-16) and made use of resources and facilities provided by the Canadian Foundation for Innovation (229288). A portion of the UBC research was conducted at: the 4D LABS shared facilities supported by the Canada Foundation for Innovation, British Columbia Knowledge Development Fund, Western Economic Diversification Canada, and Simon Fraser University; and the Centre for High-Throughput Phenogenomics at UBC, a facility supported by the Canadian Foundation for Innovation, British Columbia Knowledge Development Fund, and the UBC Faculty of Dentistry. Work at Lawrence Berkeley National Laboratory (Berkeley Lab) was performed under the auspices of the US Department of Energy (DE-AC02-05CH11231). Work at the Massachusetts Institute of Technology (MIT) used instrumentation in the Center for Materials Science and Engineering, a MRSEC Shared Experimental Facility supported by the National Science Foundation under award number DMR-14-19807. Some of the work at the University of Maryland (UMD) employed the shared facilities of the Maryland NanoCenter and its FabLab. The views and conclusions of authors expressed herein do not necessarily state or reflect those of the United States or Canadian Governments, or any agencies thereof.
Author information
Authors and Affiliations
Contributions
All authors outlined the manuscript. C.P.B. and M.D.T. wrote the manuscript. All authors reviewed and edited the entire manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
This file contains a list of co-signatories to this paper.
Rights and permissions
About this article
Cite this article
Berlinguette, C.P., Chiang, YM., Munday, J.N. et al. Revisiting the cold case of cold fusion. Nature 570, 45–51 (2019). https://doi.org/10.1038/s41586-019-1256-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1256-6
This article is cited by
-
Photon-induced low-energy nuclear reactions
Pramana (2022)
-
Start-up fusion
Nature Physics (2021)
-
Google revives controversial cold-fusion experiments
Nature (2019)
-
Coming in from the cold
Nature Materials (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.