Abstract
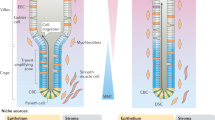
Adult intestinal stem cells are located at the bottom of crypts of Lieberkühn, where they express markers such as LGR51,2 and fuel the constant replenishment of the intestinal epithelium1. Although fetal LGR5-expressing cells can give rise to adult intestinal stem cells3,4, it remains unclear whether this population in the patterned epithelium represents unique intestinal stem-cell precursors. Here we show, using unbiased quantitative lineage-tracing approaches, biophysical modelling and intestinal transplantation, that all cells of the mouse intestinal epithelium—irrespective of their location and pattern of LGR5 expression in the fetal gut tube—contribute actively to the adult intestinal stem cell pool. Using 3D imaging, we find that during fetal development the villus undergoes gross remodelling and fission. This brings epithelial cells from the non-proliferative villus into the proliferative intervillus region, which enables them to contribute to the adult stem-cell niche. Our results demonstrate that large-scale remodelling of the intestinal wall and cell-fate specification are closely linked. Moreover, these findings provide a direct link between the observed plasticity and cellular reprogramming of differentiating cells in adult tissues following damage5,6,7,8,9, revealing that stem-cell identity is an induced rather than a hardwired property.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
RNA sequencing data that support the findings of this study have been deposited in the ArrayExpress database at EMBL-EBI (www.ebi.ac.uk/arrayexpress) under accession number E-MTAB-7660. Raw data used are provided in the Source Data files. All other data supporting the findings of this study are available from the corresponding author on reasonable request.
Code availability
Code for the biophysical modelling is provided as a Supplementary Information file.
References
Clevers, H. The intestinal crypt, a prototype stem cell compartment. Cell 154, 274–284 (2013).
Barker, N. et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449, 1003–1007 (2007).
Shyer, A. E., Huycke, T. R., Lee, C., Mahadevan, L. & Tabin, C. J. Bending gradients: how the intestinal stem cell gets its home. Cell 161, 569–580 (2015).
Nigmatullina, L. et al. Id2 controls specification of Lgr5+ intestinal stem cell progenitors during gut development. EMBO J. 36, 869–885 (2017).
Tetteh, P. W. et al. Replacement of lost Lgr5-positive stem cells through plasticity of their enterocyte-lineage daughters. Cell Stem Cell 18, 203–213 (2016).
van Es, J. H. et al. Dll1+ secretory progenitor cells revert to stem cells upon crypt damage. Nat. Cell Biol. 14, 1099–1104 (2012).
Buczacki, S. J. et al. Intestinal label-retaining cells are secretory precursors expressing Lgr5. Nature 495, 65–69 (2013).
Yui, S. et al. YAP/TAZ-dependent reprogramming of colonic epithelium links ECM remodeling to tissue regeneration. Cell Stem Cell 22, 35–49 (2018).
Nusse, Y. M. et al. Parasitic helminths induce fetal-like reversion in the intestinal stem cell niche. Nature 559, 109–113 (2018).
Guiu, J. & Jensen, K. B. From definitive endoderm to gut-a process of growth and maturation. Stem Cells Dev. 24, 1972–1983 (2015).
Sumigray, K. D., Terwilliger, M. & Lechler, T. Morphogenesis and compartmentalization of the intestinal crypt. Dev. Cell 45, 183–197 (2018).
Mustata, R. C. et al. Identification of Lgr5-independent spheroid-generating progenitors of the mouse fetal intestinal epithelium. Cell Reports 5, 421–432 (2013).
Moor, A. E. et al. Spatial reconstruction of single enterocytes uncovers broad zonation along the intestinal villus axis. Cell 175, 1156–1167 (2018).
Merlos-Suarez, A. et al. The intestinal stem cell signature identifies colorectal cancer stem cells and predicts disease relapse. Cell Stem Cell 8, 511–524 (2011).
Shyer, A. E. et al. Villification: how the gut gets its villi. Science 342, 212–218 (2013).
Walton, K. D. et al. Hedgehog-responsive mesenchymal clusters direct patterning and emergence of intestinal villi. Proc. Natl Acad. Sci. USA 109, 15817–15822 (2012).
Itzkovitz, S., Blat, I. C., Jacks, T., Clevers, H. & van Oudenaarden, A. Optimality in the development of intestinal crypts. Cell 148, 608–619 (2012).
Sato, T. et al. Single Lgr5 stem cells build crypt–villus structures in vitro without a mesenchymal niche. Nature 459, 262–265 (2009).
Fordham, R. P. et al. Transplantation of expanded fetal intestinal progenitors contributes to colon regeneration after injury. Cell Stem Cell 13, 734–744 (2013).
Yui, S. et al. Functional engraftment of colon epithelium expanded in vitro from a single adult Lgr5+ stem cell. Nat. Med. 18, 618–623 (2012).
Ritsma, L. et al. Intestinal crypt homeostasis revealed at single-stem-cell level by in vivo live imaging. Nature 507, 362–365 (2014).
Tian, H. et al. A reserve stem cell population in small intestine renders Lgr5-positive cells dispensable. Nature 478, 255–259 (2011).
Chan, C. J., Heisenberg, C. P. & Hiiragi, T. Coordination of morphogenesis and cell-fate specification in development. Curr. Biol. 27, R1024–R1035 (2017).
Hannan, N. R. et al. Generation of multipotent foregut stem cells from human pluripotent stem cells. Stem Cell Reports 1, 293–306 (2013).
Spence, J. R. et al. Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature 470, 105–109 (2011).
Watson, C. L. et al. An in vivo model of human small intestine using pluripotent stem cells. Nat. Med. 20, 1310–1314 (2014).
Sun, X. et al. Directed differentiation of human embryonic stem cells into thymic epithelial progenitor-like cells reconstitutes the thymic microenvironment in vivo. Cell Stem Cell 13, 230–236 (2013).
McCracken, K. W. et al. Modelling human development and disease in pluripotent stem-cell-derived gastric organoids. Nature 516, 400–404 (2014).
Kroon, E. et al. Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nat. Biotechnol. 26, 443–452 (2008).
Muzumdar, M. D., Tasic, B., Miyamichi, K., Li, L. & Luo, L. A global double-fluorescent Cre reporter mouse. Genesis 45, 593–605 (2007).
Means, A. L., Xu, Y., Zhao, A., Ray, K. C. & Gu, G. A CK19CreERT knockin mouse line allows for conditional DNA recombination in epithelial cells in multiple endodermal organs. Genesis 46, 318–323 (2008).
Snippert, H. J. et al. Intestinal crypt homeostasis results from neutral competition between symmetrically dividing Lgr5 stem cells. Cell 143, 134–144 (2010).
el Marjou, F. et al. Tissue-specific and inducible Cre-mediated recombination in the gut epithelium. Genesis 39, 186–193 (2004).
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 13, 133–140 (2010).
Walton, K. D. & Kolterud, A. Mouse fetal whole intestine culture system for ex vivo manipulation of signaling pathways and three-dimensional live imaging of villus development. J. Vis. Exp. 91, e51817 (2014).
Zheng, G. X. et al. Massively parallel digital transcriptional profiling of single cells. Nat. Commun. 8, 14049 (2017).
Fan, J. et al. Characterizing transcriptional heterogeneity through pathway and gene set overdispersion analysis. Nat. Methods 13, 241–244 (2016).
Butler, A., Hoffman, P., Smibert, P., Papalexi, E. & Satija, R. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat. Biotechnol. 36, 411–420 (2018).
Acknowledgements
We thank members of the Jensen and Simons laboratories for comments and suggestion; H. Clevers (Rosa26-lsl-Confetti and Lgr5-eGFP-ires-creERT2), F. de Sauvage (Genentech) (Lgr5-iDTR-eGFP), A. McMahon (Krt20-T2A-creERT2) and G. Gu (Krt19-creERT) for gifts of mice; and Y. Antoku in Imaging core facilities at BRIC and the Center for Advanced Bioimaging at University of Copenhagen for experimental support. This work was supported by Lundbeck Foundation (R105-A9755 to K.B.J.; R190-2014-3904 to T.H.P.), the Novo Nordisk Foundation (NNF14OC0012927 to K.B.J. and NNF16OC0019920 to K.K.), the Carlsberg Foundation, EMBO Young Investigator programme (to K.B.J.), the Marie Curie fellowship programme (S.Y. and J.G.; 625238/FP7-PEOPLE-2013-IIF, 656099/H2020-MSCA-IF-2014) and the Wellcome Trust (098357/Z/12/Z to B.D.S., 110326/Z/15/Z to E.H.). B.D.S. also acknowledges funding from the Royal Society E. P. Abraham Research Professorship (RP\R1\180165). This project has received funding from the European Union’s Horizon 2020 research and innovation programme (grant agreements STEMHEALTH ERCCoG682665 and INTENS 668294 to K.B.J.). The Novo Nordisk Foundation Center for Stem Cell Biology and the Novo Nordisk Foundation Center for Basic Metabolic Research are supported by Novo Nordisk Foundation grants (NNF17CC0027852 and NNF18CC0034900, respectively).
Reviewer information
Nature thanks Hans Clevers, Fred de Sauvage and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
J.G. and K.B.J. conceived the project. J.G., E.H., B.D.S. and K.B.J. designed experiments. J.G., S.Y., S.U. and M.M. performed experiments. A.J., S.P., L.L., L.S.M., C.Y.A., L.L.T., K.J.H., A.L. and R.H.O. collected and processed human fetal material. S.D., T.H.P. and K.K. analysed single-cell sequencing data. E.H. and B.D.S. designed and performed the mathematical modelling. J.G. wrote the manuscript with input from K.B.J., E.H. and B.D.S. J.G., S.Y., B.D.S. and K.B.J. acquired funding. B.D.S. and K.B.J. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 LGR5+ cells and proliferative cells are restricted to the intervillus regions.
a, Detection of E-cadherin (red) and GFP (Lgr5-DTR-eGFP) at E13.5 and E16.5 in proximal and distal small intestine. Tissue is counterstained with DAPI (cyan). A representative image from each time point and intestinal segment is shown (n = 3 mice analysed). b, In situ hybridization of endogenous Lgr5 at E13.5 and E16.5 in proximal and distal small intestine. Tissue is counterstained with DAPI (cyan). A representative image from each time point and intestinal segment is shown (n = 3 mice analysed). c, Detection of E-cadherin (magenta) and GFP (Lgr5-DTR-eGFP) in intestinal whole mount at E16.5 in proximal small intestine. A representative image from n = 3 independent samples is shown. d, Detection of Ki67 and EdU (red) following a 1-h pulse at the indicated time points (left and right, respectively). Three mice were analysed at each time point and a representative image is shown. Scale bars, 50 μm. e, f, Quantification of number and location of EdU+ cells as depicted (e) from at least ten intervillus regions per mouse at E16.5, P5, P11 and adult, and 20 intervillus regions per mouse at P0 up to position 10 and position 17 (adult). Samples from three mice were analysed at each time point. Data are mean ± s.e.m.
Extended Data Fig. 2 LGR5-derived clones are located in intervillus regions and qualitative and quantitative morphological analysis of the intestine from fetal to adult stages.
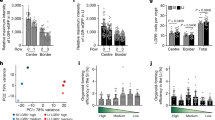
a, Quantification of localization of labelled clones at P0 following labelling at E16.5 in Rosa26-lsl-Confetti;Lgr5-eGFP-ires-creERT2 mice. Villi containing clones were divided into three equal regions (T, top; M, middle; B, bottom) based on the z projections in 3D to determine the clone localization at P0 (n = 24 clones). b, Detection of E-cadherin (red) in whole mounts at the indicated time points. Scale bars, 100 μm. A minimum of three mice were analysed at each time point and representative images are shown (E16.5 n = 3, P0 n = 9, P5 n = 9, P11 n = 9, adult n = 9 mice). c, Measurements of the total epithelial volume per unit area based on detection of E-cadherin relative to the area of the intestine (samples from b). Dots represent independent biological samples; data are mean ± s.e.m. d, Length of the small intestine at E16.5 (n = 12), P0 (n = 3), P5 (n = 3), P11 (n = 8) and adult (n = 4). Dots represent individual mice and lines represent the mean ± s.e.m. e, Luminal perimeter of the small intestine at E16.5, P0, P5, P11 and adult. Dots represent individual mice (n = 3) and lines represent the mean ± s.e.m. f–h, Quantification of GFP as a proxy for LGR5 in proximal and distal small intestine at E13.5 (n = 3 mice both proximal and distal) and E16.5 (n = 7 mice proximal, n = 5 mice distal) small intestines (f). g, Fluorescence minus one (GFP) controls used to establish the positive gates. h, Representative FACS dot plot illustrating the gating strategy to quantify the size of the LGR5–DTR–eGFP+ population (gate, DAPI−CD31−CD45−EpCAM+). Dots represent measurement in individual mice; data are mean ± s.e.m.
Extended Data Fig. 3 Characterization of the fetal small intestinal epithelium.
a, t-distributed stochastic neighbor embedding (t-SNE) plots from scRNA-seq of epithelial cells from the proximal small intestine showing expression of intestinal stem-cell (Lgr5) and differentiation markers (Muc2, Lyz1, ChgA and Alpi). Darker colour indicates higher normalized gene expression. Each dot represents a cell; a total of 3,509 cells is shown. b, Detection of differentiation markers in E16.5 and adult small intestine. Tissue is counterstained with haematoxylin and eosin. Scale bar, 250 μm. Samples from n = 3 mice were analysed at each time point and representative images are shown. c, Cartoon depicting that adult villi are transcriptionally zonated in five regions numbered from bottom to top (z, zone). d, t-SNE plots showing enrichment of villi clusters in the scRNA-seq from E16.5 small intestine. e, t-SNE plot showing the enrichment of a proliferation signature in specific cell populations (left) and fraction of cells in each subpopulation scoring positive for the proliferation gene signature (right). A darker colour in the t-SNE plot indicates higher expression levels of the proliferation gene signature. f, t-SNE plot showing expression of keratin 19 (Krt19). Darker colour indicates higher normalized gene expression levels. g, Detection of KRT19 (red) and GFP (Lgr5-DTR-eGFP) at E16.5 in proximal small intestine. Tissue is counterstained with DAPI (cyan). Scale bars, 50 μm. Samples from three mice were analysed and a representative image is shown. h, Detection of KRT19 (red) at different time points in tissue from the small intestine. Tissue is counterstained with DAPI (cyan). Scale bars, 50 μm. Samples from three mice were analysed per time point and representative images are shown. i, Relative volume (projected) of KRT19 clones and epithelium based on E-cadherin. E-cadherin is also shown in Fig. 1c and KRT19 clones are also shown in Fig. 2. KRT19 clones, number of biologically independent samples: P0 n = 1; P5 n = 3, P11 n = 6, adult n = 3; E-cadherin, number of biologically independent samples: P0 n = 9, P5 n = 9, P11 n = 9, adult n = 9. j, Quantification of the localization of labelled clones at P0 following administration of 4-hydroxytamoxifen at E16.5 in Rosa26-lsl-Confetti;Krt19-creERT mice. Villi containing clones were divided in three equal regions (T, top; M, middle; B, bottom) based on the z projection in 3D to determine where clones were located at P0 (n = 27). k, Detection of GFP (green) and RFP (red) in whole mounts from the proximal part of the small intestine isolated from mT/mG Krt19-creERT mice at P0 following induction at E16.5. A representative image of n = 3 biologically independent samples is shown. Scale bars, 25 μm. l, Apoptotic cells were detected by cleaved caspase-3. Arrowheads demarcate positive cells in inserts. Samples from three mice were analysed per time point and representative pictures are shown. Scale bars, 250 μm.
Extended Data Fig. 4 Villi formation and parameter description of villi fissions.
a, Total number of villi (projected) in the proximal half of the small intestine based on equal density along the length. Samples from three mice were analysed per time point. Each dot represents a mouse; data are mean ± s.e.m. b, The fold change in villi numbers between the indicated time points based on three samples analysed per time point. Each dot represents an independent sample and lines represent the mean ± s.e.m. c, Villus height at the different time points. The demarcated red lines indicate the interval containing villi with sharing stroma. The length was assessed in 25 villi per mouse and in 3 mice per time point. Dots represent independent measurements and lines represent the mean. d, Quantification of the number of villi sharing stroma in three mice per indicated time point (E16.5 n = 233, P0 n = 412, P5 n = 406, P11 n = 412, adult n = 129 villi were counted). Dots represent the percentage of villi sharing stroma in each independent mouse and the line represents the mean. e, Detection of E-cadherin (red) and PDGFRA (yellow) in whole mounts indicating villi with sharing stroma (arrowhead). Samples from three mice were analysed per time point and representative images are shown. Scale bars, 100 μm. f, Detection of E-cadherin (magenta) and GFP (Lgr5-DTR-eGFP) in E16.5 intestinal whole mount. Boxed area (1) indicates a villus undergoing fission, which is shown at higher magnification (middle). Transverse sections (2 and 3) illustrating villi surrounded by LGR5-expressing cells (2) and villus with shared mesenchyme (3). The arrowhead indicates that pockets formed in a fissioning villus are LGR5-negative, the dashed line outlines the epithelium. Samples from three mice were analysed and a representative image is shown. g, Detection of EdU incorporation (green) in the epithelium (E-cadherin, red) following a 1-h chase in E16.5 intestinal whole mounts. Arrowheads indicate proliferative cells at the edge of putative villi undergoing fission. These are detected in 11 out of 16 structures. Representative pictures from three mice analysed are shown. Scale bars, 50 μm. h, Quantification of EdU intensity in the fissioning areas compared to the surrounding villi at the same height quantified as depicted in the cartoon on the basis of thresholded intensity in the demarcated boxes. n = 16 independent villi sharing stroma were quantified. Box plots show the median, box edges represent the first and third quartiles, and the whiskers extend to minimum and maximum values. Dots represent fluorescent ratio of the independent villi sharing stroma. Paired t-test. i, Height of the proliferative fissioning areas compared to the surrounding intervillus regions were quantified as depicted in the cartoon. n = 11 independent villi sharing stroma were quantified. j, Images showing the start and end points from live imaging of villi undergoing fission (Supplementary Video 6). Image from mT/mG;Villin-cre mice, where the epithelium is shown Green (mG) and remaining cells in red (mT). A representative fission event from five analysed mice is shown. Scale bar, 50 μm.
Extended Data Fig. 5 Outline of the model and typical outputs from simulations.
a, Schematics of the model for the renewal of intervillus LGR5+ cells. On the basis of proliferation data, we assume that the classical model of symmetrically dividing and competing LGR5+ cells holds embryonically, with a division rate once a day. The ‘losing’ cell is expelled into the transit-amplifying (TA) compartment, displacing all cells above it by one position. b, Schematics of the model for the dynamics of differentiated cells on the villus. The model dynamics are separated into two phases. The first phase occurs from E16.5 to P5: LGR5+ cells are the only proliferative cells, and villi fission occurs as a stochastic event, resulting in the duplication of a villus subregion into a whole new intervillus–villus grid, and a resulting shift of cells along the existing villus. A second phase occurs after P5: the dynamics are similar to adulthood, with rapidly dividing transit-amplifying cells (occupying 1/5th of the villus at the bottom) and cell loss at the top. Proliferation of transit-amplifying cells is again exclusively along the top–bottom axis, resulting in unidirectional displacement of all cells above the dividing cell. c, Three snapshots from a numerical simulation of the epithelium as a growing elastic sheet on a growing elastic medium. The growth is assumed to be quasi-static, so that the epithelium maintains a deformation at a given wavelength, minimizing the elastic energy of the sheet and substrate. Where the system grows (top to bottom panels), this results in de novo villi formation from local deformations of the epithelial sheet, resulting in villus and intervillus regions shifting places via tissue bending (the dashed lines serve as a guide to represent how a cell in a given position x can change height z). d, e, Two sets of snapshots from two numerical simulations of an E16.5 LGR5 tracing, according to the rules outlined in a, b. Each black box represents an intervillus–villus grid, with the number increasing in time owing to random villi fission. Red squares indicate the labelled cells at E16.5 (initially in the bottom-most layer of LGR5+ cells). An example of a clone that becomes lost in time, despite having participated in villi fission between E18.5 and P1 is shown in d. An example of a clone that becomes fixed within one of the villi having formed de novo during the simulation (while the labelled cell in the original intervillus–villus region of induction was shifted away from the intervillus by a villi fission event between E16.5 and E17.5) is shown in e.
Extended Data Fig. 6 Clone size distributions based on simulation using the cell-repositioning model.
a, Total number of intestinal villi over time (purple, same as Extended Data Fig. 4a) normalized against the fitted trend for the villi formation rate (green line), which we use in the numerical simulations. Samples from three mice were analysed per time point. Data are mean ± s.e.m. b, Cumulative distribution of clone sizes (induced at E16.5) at P0 in the LGR5 (red) compared to the KRT19 (blue) tracing (also depicted in Fig. 2d). c, Cumulative distribution of clone sizes (induced at E16.5) at P5 in the LGR5 (red) compared to the KRT19 (blue) tracing also depicted in Fig. 2d. d, Comparison between experimental (dots) and theoretical cumulative distribution of LGR5 clone sizes (induced at E16.5) at P0 (purple), P5 (red) and P11 (cyan). e, Comparison between experimental (dots) and theoretical cumulative distribution of KRT19 clone sizes (induced at E16.5) at P0 (purple), P5 (red) and P11 (cyan). For all panels, clone size was inferred from clonal volume (Supplementary Table 2), using average single-cell measurements of volume as a conversion factor.
Extended Data Fig. 7 Theoretical controls and sensitivity analysis.
a–c, Comparison between experimental data (dots and error bars) and theoretical predictions (thick lines) for the time evolution of the clonal rootedness (a), average clone size (b) and clonal persistence (c), for both KRT19 (cyan) and the LGR5 (green) tracings from E16.5. Here, the theoretical prediction corresponds to the case of villi fission, following exactly the same model in Extended Data Fig. 6, but stopping at P1 instead of at P5. This shows that fetal fission is enough to explain the bulk of the equipotency between LGR5 and KRT19 clones. d–f, Comparison between experimental data (dots and error bars) and theoretical predictions (thick lines) for the time evolution of the clonal rootedness (d), average clone size (e) and clonal persistence (f), for both KRT19 (cyan) and LGR5 (green) tracings from E16.5. Here, the theoretical prediction corresponds to the case of no new villi formation occurring, showing a very poor fit for the clonal persistence and size to the data. g–i, Comparison between experimental data (dots and error bars) and theoretical predictions (thick lines) for the time evolution of the clonal rootedness (g), average clone size (h) and clonal persistence (i), for both KRT19 (cyan) and LGR5 (green) tracings from E16.5. Here, the theoretical prediction corresponds to the case of villi fission occurring as in the model shown in Fig. 3, but without a shift of cells upon villi fission (see schematics in Extended Data Fig. 5), showing a very poor fit for the clonal persistence to the data. j, Cartoon illustrating that the current villification model suggests that villi emerge from the intervillus region. k–m, Comparison between experimental data (dots and error bars) and theoretical predictions (thick lines) for the time evolution of the clonal rootedness (k), average clone size (l) and clonal persistence (m), for both KRT19 (cyan) and LGR5 (green) tracings from E16.5. Here, the theoretical prediction corresponds to the case of villi formation occurring only from existing crypts, showing a very poor fit for the clonal persistence to the data. n, Volume of single villus cells and intervillus–crypts cells. Total E-cadherin volume of villi and intervilli–crypts, subsequently divided by the number of DAPI+ nuclei to estimate single-cell volume. E16.5 villi n = 8, E16.5 intervilli n = 10, P0 villi n = 6, P0 intervilli n = 3, P5 villi n = 11, P5 intervilli n = 4, P11 villi n = 4, P11 intervilli n = 4, adult villi n = 7, adult intervilli n = 4 independent images were analysed. o, Sensitivity analysis on the influence of differential LGR5− and LGR5+ cellular volume on the model prediction—considering either that all cells have the same volume (continuous lines and a–n) or the differential volume measured at E16.5 (see Supplementary Theory Note, dashed lines). For a–i, k–m: P0 n = 3, P5 n = 3, P11 n = 3, adult n = 6 independent samples analysed for LGR5; P0 n = 1, P5 n = 3, P11 n = 6, adult n = 3 independent samples analysed for KRT19. Data are mean ± s.e.m.
Extended Data Fig. 8 Postnatal lineage tracing.
a, Detection of E-cadherin (cyan) and clones (red) induced at P0 either randomly (Krt19-creERT) or in the intervillus region (Lgr5-eGFP-ires-creERT2). Samples from three mice were analysed per time point and representative images are shown. b, Relative volume of clones (projected) induced either randomly (Krt19-creERT) or in the intervillus region (Lgr5-eGFP-ires-creERT2) as assessed by quantitative clonal analysis following induction at P0. Samples from three mice were analysed per time point and dots represent individual samples and lines indicate the mean ± s.e.m. c, Theoretical labelled-cell fraction of the LGR5 clones induced at P0, using the same model dynamics as the E16.5 induction. Expansion of the LGR5 clones is represented by the thick red line, normalized to the Krt19 tracing (blue, indicative of global tissue growth) and to its P5 value. This displays a net twofold increase between P5 and P11, consistent with the results of the experimental P0 tracing.
Extended Data Fig. 9 The contribution of KRT20-expressing cells during development and homeostasis.
a, Detection of keratin 20 (KRT20, magenta), lysozyme1 (LYZ1, red) and GFP (Lgr5-DTR-eGFP) in adult mouse intestine. Tissue is counterstained with DAPI (cyan). Representative image of n = 3 biologically independent samples is shown. b, Detection of E-cadherin (cyan), GFP (green) and RFP (red) in tissue whole mounts from the proximal part of the small intestine isolated from Rosa26-lsl-Confetti;Krt20-T2A-creERT2 mice at 3 and 40 days after label induction with 4-hydroxytamoxifen. Samples from three mice were analysed per time point and representative images are shown. c, In situ hybridization of Lgr5 (green) and cre (red) in proximal E16.5 small intestine. Tissue is counterstained with DAPI (cyan). Samples from three mice were analysed per time point and representative images are shown. d, Detection of E-cadherin (cyan) and tdTomato (red) in tissue whole mounts from the proximal small intestine isolated from Rosa26-lsl-tdTomato;Krt20-T2A-creERT2 mice at E18.5 and adulthood following administration of 4-hydroxytamoxifen at E16.5. Representative images of n = 2 samples are shown.
Extended Data Fig. 10 Testing intestinal epithelial cells for equipotency.
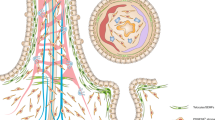
a, Detection of LGR5–eGFP (green) and CD44 (red) in the E16.5 small intestine isolated from Lgr5-eGFP-ires-creERT2 E16.5 mice. Tissue was counterstained with DAPI (blue). Samples from three mice were analysed and a representative image is shown. Scale bars, 50 μm. b, Cartoon showing the positions used to quantify the pattern of CD44 and LGR5 expression. Quantification of the localization of CD44 and LGR5 positive cells. A total of 14 intervillus regions (x axis) were quantified up to position ±10 (y axis). c, d, Representative FACS dot plot illustrating the gating strategy used to quantify CD44. Dots represent the quantification in individual mice (n = 4). e, Spheroids forming from cells isolated based on DAPI−EpCAM+LGR5–eGFP− and DAPI−EpCAM+LGR5–eGFP− from the proximal half of the small intestine from mice at E16.5. Representative images of n = 3 biologically independent samples. f, Quantification of spheroid seeding efficiency following isolation based on either CD44 or LGR5 (n = 3 mice per condition). g–i, Gating strategy for purification of villus and intervillus cells for transplantation. The gating hierarchy of the panels is numbered i–vi (g), an example of purity is indicated (h) and an mT/mG-derived organoid is shown (i). Spheroids were generated from a pool of n = 6 biologically independent samples. j, k, Outline for transplantation experiment. In brief, experimental colitis was induced in RAG2−/− mice by administration of DSS in the drinking water. Organoids from the different cultures were subsequently infused into lumen of the mice (j) and engrafted regions were immunostained for lineage and stem-cell markers (k). l, Scheme summarizing the main findings of this work.
Supplementary information
Supplementary Information
This file contains the Supplementary Theory Note, Supplementary References, Supporting Clonal Data and Supplementary Tables 1-2.
Supplementary Data
This zipped file contains the biophysical modeling code and an example to reproduce the model-data comparison.
Video 1: related to Extended Figure 1b.
Video showing a 3D reconstruction of RFP and GFP in Lgr5-eGFP-ires-CreERT2;Rosa26-lsl-Confetti at P11, pregnant mouse was treated with 4-Hydroxytamoxifen at E16.5. Representative video of n=3 biologically independent samples analyzed.
Video 2: related to Extended Figure 3g.
Video showing a 3D reconstruction of RFP and GFP in Krt19-CreERT/mT/mG P0 newborns, treated with 4-Hydroxytamoxifen at E16.5. A representative video of n=3 biologically independent samples is shown.
Video 3: related to Figure 3a.
Video showing a 3D reconstruction of E-Cadherin and PDGFRA stained E16.5 fetal intestine. A representative video of n=3 biologically independent samples is shown.
Video 4: related to Figure 3a.
Video showing a transversal section of 3D reconstruction of E-Cadherin and PDGFRA stained E16.5 fetal intestine to illustrate an example of villi with shared stroma. A representative video of n=3 biologically independent samples is shown.
Video 5: related to Figure 3b.
Video showing a transversal section of 3D reconstruction of E-Cadherin, PDGFRA and EdU (1 hour after EdU injection) stained E16.5 fetal intestine to illustrate proliferative cells villi with shared stroma. A representative video of n=3 biologically independent samples is shown.
Video 6: related to Extended Figure 4j.
Video showing a villus fission event VillinCre;mT:mG intestine. A fission event of n=5 independent biological samples is shown.
Video 7: related to Figure 3d.
Video showing a villus fission event in a bright field. A fission event of n=3 independent biological samples is shown.
Source data
Rights and permissions
About this article
Cite this article
Guiu, J., Hannezo, E., Yui, S. et al. Tracing the origin of adult intestinal stem cells. Nature 570, 107–111 (2019). https://doi.org/10.1038/s41586-019-1212-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1212-5
This article is cited by
-
Mechanical state transitions in the regulation of tissue form and function
Nature Reviews Molecular Cell Biology (2024)
-
Mechanically enhanced biogenesis of gut spheroids with instability-driven morphomechanics
Nature Communications (2023)
-
Cell competition in development, homeostasis and cancer
Nature Reviews Molecular Cell Biology (2023)
-
Advanced Progression for the Heterogeneity and Homeostasis of Intestinal Stem Cells
Stem Cell Reviews and Reports (2023)
-
3D and organoid culture in research: physiology, hereditary genetic diseases and cancer
Cell & Bioscience (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.