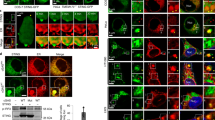
Abstract
Cyclic GMP-AMP (cGAMP) synthase (cGAS) detects infections or tissue damage by binding to microbial or self DNA in the cytoplasm1. Upon binding DNA, cGAS produces cGAMP that binds to and activates the adaptor protein STING, which then activates the kinases IKK and TBK1 to induce interferons and other cytokines2,3,4,5,6. Here we report that STING also activates autophagy through a mechanism that is independent of TBK1 activation and interferon induction. Upon binding cGAMP, STING translocates to the endoplasmic reticulum–Golgi intermediate compartment (ERGIC) and the Golgi in a process that is dependent on the COP-II complex and ARF GTPases. STING-containing ERGIC serves as a membrane source for LC3 lipidation, which is a key step in autophagosome biogenesis. cGAMP induced LC3 lipidation through a pathway that is dependent on WIPI2 and ATG5 but independent of the ULK and VPS34–beclin kinase complexes. Furthermore, we show that cGAMP-induced autophagy is important for the clearance of DNA and viruses in the cytosol. Interestingly, STING from the sea anemone Nematostella vectensis induces autophagy but not interferons in response to stimulation by cGAMP, which suggests that induction of autophagy is a primordial function of the cGAS–STING pathway.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The authors declare that all relevant data supporting the findings of this study are available within the paper and its supplementary information files. Additional information including raw data are available from the corresponding author upon reasonable request.
References
Sun, L., Wu, J., Du, F., Chen, X. & Chen, Z. J. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science 339, 786–791 (2013).
Wu, J. et al. Cyclic GMP-AMP is an endogenous second messenger in innate immune signaling by cytosolic DNA. Science 339, 826–830 (2013).
Ishikawa, H. & Barber, G. N. STING is an endoplasmic reticulum adaptor that facilitates innate immune signalling. Nature 455, 674–678 (2008).
Wu, J. & Chen, Z. J. Innate immune sensing and signaling of cytosolic nucleic acids. Annu. Rev. Immunol. 32, 461–488 (2014).
Zhong, B. et al. The adaptor protein MITA links virus-sensing receptors to IRF3 transcription factor activation. Immunity 29, 538–550 (2008).
Crowl, J. T., Gray, E. E., Pestal, K., Volkman, H. E. & Stetson, D. B. Intracellular nucleic acid detection in autoimmunity. Annu. Rev. Immunol. 35, 313–336 (2017).
Kranzusch, P. J. et al. Ancient origin of cGAS–STING reveals mechanism of universal 2′,3′ cGAMP signaling. Mol. Cell 59, 891–903 (2015).
Ge, L., Melville, D., Zhang, M. & Schekman, R. The ER–Golgi intermediate compartment is a key membrane source for the LC3 lipidation step of autophagosome biogenesis. eLife 2, e00947 (2013).
Dooley, H. C. et al. WIPI2 links LC3 conjugation with PI3P, autophagosome formation, and pathogen clearance by recruiting Atg12-5-16L1. Mol. Cell 55, 238–252 (2014).
Brandizzi, F. & Barlowe, C. Organization of the ER–Golgi interface for membrane traffic control. Nat. Rev. Mol. Cell Biol. 14, 382–392 (2013).
Ge, L., Baskaran, S., Schekman, R. & Hurley, J. H. The protein–vesicle network of autophagy. Curr. Opin. Cell Biol. 29, 18–24 (2014).
D’Souza-Schorey, C. & Chavrier, P. ARF proteins: roles in membrane traffic and beyond. Nat. Rev. Mol. Cell Biol. 7, 347–358 (2006).
Choy, A. et al. The Legionella effector RavZ inhibits host autophagy through irreversible Atg8 deconjugation. Science 338, 1072–1076 (2012).
Tanaka, Y. & Chen, Z. J. STING specifies IRF3 phosphorylation by TBK1 in the cytosolic DNA signaling pathway. Sci. Signal. 5, ra20 (2012).
Liu, S. et al. Phosphorylation of innate immune adaptor proteins MAVS, STING, and TRIF induces IRF3 activation. Science 347, aaa2630 (2015).
Zhang, X. et al. Cyclic GMP–AMP containing mixed phosphodiester linkages is an endogenous high-affinity ligand for STING. Mol. Cell 51, 226–235 (2013).
Seth, R. B., Sun, L., Ea, C. K. & Chen, Z. J. Identification and characterization of MAVS, a mitochondrial antiviral signaling protein that activates NF-κB and IRF3. Cell 122, 669–682 (2005).
Gao, D. et al. Cyclic GMP-AMP synthase is an innate immune sensor of HIV and other retroviruses. Science 341, 903–906 (2013).
Shalem, O. et al. Genome-scale CRISPR-Cas9 knockout screening in human cells. Science 343, 84–87 (2014).
Li, X. D. et al. Pivotal roles of cGAS-cGAMP signaling in antiviral defense and immune adjuvant effects. Science 341, 1390–1394 (2013).
Sauer, J. D. et al. The N-ethyl-N-nitrosourea-induced Goldenticket mouse mutant reveals an essential function of Sting in the in vivo interferon response to Listeria monocytogenes and cyclic dinucleotides. Infect. Immun. 79, 688–694 (2011).
Yang, H., Wang, H., Ren, J., Chen, Q. & Chen, Z. J. cGAS is essential for cellular senescence. Proc. Natl Acad. Sci. USA 114, E4612–E4620 (2017).
Gao, D. et al. Activation of cyclic GMP-AMP synthase by self-DNA causes autoimmune diseases. Proc. Natl Acad. Sci. USA 112, E5699–E5705 (2015).
Acknowledgements
We thank C. Roy (Yale University) for providing the RavZ expression plasmids, B. Levine (UT Southwestern) for providing tissues from beclin 1-knockout mice, and A. Darehshouri at UT Southwestern Electron Microscopy Core Facility for training in electron microscopy sample preparation and image processing. We are grateful for suggestions and technical supports from Chen Laboratory members, especially L. Sun, Y. Wu and S. Hu. This work was supported by grants from the Cancer Prevention and Research Institute of Texas (RP120718 and RP150498) and the Welch Foundation (I-1389). M.L. was supported by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health (T32AI005284). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. T. L. and X.T. are Cancer Research Institute Irvington Postdoctoral Fellows. Z.J.C. is an Investigator of Howard Hughes Medical Institute.
Reviewer information
Nature thanks Kate Fitzgerald and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
X.G. and H.Y. planned the study under the guidance of Z.J.C., performed all experiments, and analysed the data. T.L. performed the mass spectrometry experiments. P.S. and F.D. generated several cell lines used in this study. Z.J.C. designed and supervised this study. X.G., H.Y., X.T., M.L. and Z.J.C. prepared and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 cGAMP-induced LC3 lipidation requires vesicle trafficking but not TBK1 or IKK.
a, DNA and cGAMP, but not RNA, trigger LC3 lipidation. BJ cells were stimulated with cGAMP by digitonin permeabilization or transfected with ISD or poly(I:C). Cell lysates were analysed by immunoblotting with the indicated antibodies. b, DNA virus but not RNA virus induces LC3 conversion. BJ cells were infected with wild-type (WT) HSV-1, HSV-1(ΔICP34.5) or Sendai virus (SeV) at the indicated MOI for 6 h followed by immunoblotting. c, cGAMP induces STING degradation in the lysosome. HeLa cells that stably express STING–Flag were treated with cGAMP or starved in the presence or absence of chloroquine, followed by immunoblotting. d, Inhibition of TBK1 or IKK does not impair LC3 lipidation. Inhibitors of TBK1 (BX-795 or MRT 67307) or IKK (TPCA1) were incubated with BJ cells before stimulation of the cells with cGAMP. Cell lysates were analysed by immunoblotting. e, Control experiment showing that TPCA1 inhibits IκBα degradation (by inhibiting IKK). f, Quantification of double-membrane autophagosomes in wild-type and TBK1−/− BJ cells. The cells were stimulated with cGAMP as indicated. The number of double-membrane autophagosomes per cell was counted in BJ cells (n = 13, 12, 17, 11). Mean ± s.e.m. is shown. ****P < 0.0001 (two-tailed Student’s t-test); NS, not significant (significance level, α = 0.01). g, STING S366 phosphorylation by TBK1 is essential for IRF3 phosphorylation but not LC3 conversion. HEK293T cells that stably express wild-type or mutant STING(S366A) or STING(R238/Y240A) were transfected with a cGAS expression plasmid followed by immunoblotting. h, HeLa cells stably expressing LC3–GFP and different STING mutants (R238A, Y240A or V155M) were stimulated with cGAMP followed by confocal immunofluorescence microscopy. i, Quantification of the cells with colocalization of LC3 and STING puncta. The percentage of cells with colocalized LC3 and STING was quantified from 100 cells (n = 2). N.D., not detectable. j, k, HeLa cells that stably express LC3GFP and STING–Flag were treated with bafilomycin A1 (BafA1) or BFA followed by stimulation with cGAMP and confocal immunofluorescence microscopy. The percentage of cells with colocalized LC3 and STING was quantified from 100 cells (n = 3, mean ± s.d., two-tailed Student’s t-test). l, BJ cells were treated with BFA, lysosome inhibitors (BafA1 or chloroquine) or proteasome inhibitors (MG132 or Velcade) before stimulation with cGAMP. Cell lysates were analysed by immunoblotting.
Extended Data Fig. 2 Delineation of the STING region (residues 330–334) required for LC3 lipidation.
a, Expression plasmids that encode truncated STING mutants were transiently transfected into HEK293T cells for 24 h, followed by stimulation with cGAMP for 4 h. Cell lysates were analysed by immunoblotting. b, Expression plasmids of indicated STING truncation mutants were transfected into HeLa LC3–GFP cells for 24 h. The cells were stimulated with cGAMP followed by immunostaining and fluorescence microscopy. c, d, Mutations of STING at L333 and R334 impaired LC3 lipidation and TBK1 activation. c, Expression plasmids that encode full-length STING containing the indicated mutations were transiently transfected into HEK293T cells, followed by stimulation with cGAMP. Cell lysates were analysed by immunoblotting. FL, full length. d, Indicated STING mutants were transfected and stimulated as described in c followed by immunostaining and fluorescence microscopy. e, Schematic of functional domains and residues of human (huSTING) and sea anemone STING (nvSTING), highlighting the evolutionary conservation of the cyclic dinucleotide (CDN) binding domain but not the C-terminal activation domain.
Extended Data Fig. 3 STING-induced LC3 conversion is a primordial function of the cGAS-STING pathway.
a, Sea anemone STING (NvSTING) induces LC3 conversion, but not TBK1 activation, in response to 2′3′-cGAMP. HEK293T cells were transfected with expression plasmids encoding full-length human STING (hu-STING), human STING(1–340) or NvSTING for 24 h and then treated with 2′3′-cGAMP for 3 h. Cell lysates were analysed by immunoblotting. b–d, Sea anemone cGAS (NvcGAS) produces 2′3′-cGAMP. b, Expression vectors encoding human cGAS (hu-cGAS), NvcGAS or DncV were transfected into HEK293T cells for 24 h. Small molecules were extracted from cells for analysis of cGAMP isomers by tandem mass spectrometry. Mass spectra of cGAMP from NvcGAS-expressing cells match those from hu-cGAS-expressing cells but not those from DncV-expressing cells. c, Similar to b, except that the small-molecule extracts were fractionated using a monoQ column, and each fraction was delivered into THP1–ISG luciferase reporter cells for measuring the CDN activity. d, CDNs produced by hu-cGAS and NvcGAS, but not that by DncV, is resistant to digestion by RNase T1. Small-molecule extracts from HEK293T cells expressing the indicated CDN synthases were treated with RNase T1 or RNaseT2 or left untreated before delivery into THP1–ISG luciferase reporter cells to measure the CDN activity. Mean ± s.e.m. was shown (n = 3) in the group treated with RNase T1. e, Domain organization of STING from human (hu), Danio rerio (Dr) and Xenopus tropicalis (Xt). f, Xenopus STING stimulates LC3 lipidation but not IRF3 phosphorylation. HEK293T cells were transfected with expression plasmids for STING–Flag from human, Danio or Xenopus for 24 h and then stimulated with cGAMP for 1 h. Anti-Flag antibody was used to immunoprecipitate STING from cell lysates followed by immunoblotting with the indicated antibodies. g, cGAMP stimulates formation of perinuclear puncta of STING from different species. HeLa cells transiently expressing human, Danio or Xenopus STING–Flag were stimulated with cGAMP for 3 h. Cells were immunostained with a Flag antibody followed by fluorescence microscopy. h, Quantification of the percentage of cells with STING peri-nuclear foci formation. The percentage of cells with STING foci formation was quantified from 100 cells (n = 2). All results in this figure are representative of at least two independent experiments.
Extended Data Fig. 4 cGAMP stimulates STING translocation to ERGIC vesicles that promote LC3 lipidation.
a, STING colocalizes with the ERGIC and autophagosomes in response to cGAMP stimulation. HeLa cells that stably express GFP–LC3 and STING–Flag were stimulated with cGAMP for the indicated time followed by immunofluorescence microscopy. b, STING trafficking to the ERGIC is blocked by GCA. BJ cells were stimulated with cGAMP for 3 h in the presence or absence of GCA. Cells were stained with DAPI or the indicated antibodies and examined by confocal microscopy. c, Procedures for in vitro reconstitution of cGAMP-induced LC3 lipidation and membrane fractionation. d, ATG5−/− HEK293T cells that stably express STING–Flag were transfected with a cGAS expression plasmid or an empty vector for 24 h. Membrane pelleted at 25,000g (P25) from these cells was incubated with cytosolic extracts (S100) from starved or untreated 293T cells in the presence of GTP and an ATP regenerating system. After incubation at 30 °C for 90 min, the reaction mixtures were analysed by immunoblotting. e, Similar to c, except that different organelle membranes enriched by differential centrifugation were prepared and incubated with cytosol (S100) from HEK293T cells to detect LC3 lipidation. f, Similar to c, except that P25 membranes were further fractionated by sucrose ultracentrifugation to generate P25P (pellet) and P25L (light) and incubated with cytosol (S100) from HEK293T cells to detect LC3 lipidation.
Extended Data Fig. 5 cGAMP-bound STING traffics through the Golgi and endosomes or the ERGIC and autophagosomes before reaching lysosomes.
a, BJ cells were stimulated with cGAMP for the indicated time. Cells were immunostained with a STING antibody together with an antibody against GM130 (cis-Golgi), TGN38 (trans-Golgi), GGA3 (post-Golgi vesicles), CD63 (late endosomes) or LAMP1 (lysosomes), followed by immunofluorescence microscopy. b, Quantification of the percentage of cells in which STING colocalized with different organelle markers in a. The percentage was quantified from 50 cells (n = 2). All results in this figure are representative of at least two independent experiments. c, cGAMP induces trafficking of STING to the ERGIC, COP-I vesicles and LC3 autophagosomes. HeLa cells that stably express STING–Flag and LC3–GFP were stimulated with cGAMP for the indicated time. Cells were immunostained with antibodies specific for Flag (to detect STING), ERGIC53 (ERGIC) or beta-COP (COP1 vesicles), followed by fluorescence microscopy. d, Quantification of the percentage of cells in which STING colocalized with different organelle markers in c. The percentage was quantified from 50 cells (n = 2). e, BJ cells were stimulated with cGAMP in the presence or absence of bafilomycin A1 (BafA1). Cells were immunostained with an antibody specific for STING or RAB7A followed by microscopy. f, Quantification of the percentage of cells in which STING colocalized with RAB7A in e. The percentage was quantified from 50 cells (n = 3, mean ± s.d., two-tailed Student’s t-test). g, BJ cells were transfected with an siRNA targeting RAB7A or a control siRNA for three days before stimulation with cGAMP for the indicated time. Cell lysates were analysed by immunoblotting.
Extended Data Fig. 6 ULK kinases are dispensable for cGAMP-induced LC3 conversion and autophagosome formation.
a, ATG5 is required for LC3 lipidation but dispensable for STING degradation induced by cGAMP. ATG5−/− or ATG5-reconstituted BJ cells were stimulated with cGAMP for the indicated time followed by immunoblotting of cell lysates. b, ATG9 is dispensable for LC3 lipidation and STING degradation. ATG9−/− or ATG9-reconstituted BJ cells were stimulated with cGAMP for the indicated time followed by immunoblotting of cell lysates. c, STING activation does not induce mTOR inhibition. BJ cells were treated with cGAMP, HT-DNA, torin 1 or rapamycin for the indicated time followed by immunoblotting of cell lysates. d, Loss of ULK1 and ULK2 impairs LC3 conversion and p62 degradation induced by mTOR inhibition. Wild-type and Ulk1−/−Ulk2−/− MEF cells were treated with torin 1 at the indicated time followed by immunoblotting of cell lysates. e, STING-induced LC3 conversion is independent of ULK1 and ULK2. Wild-type and Ulk1−/−Ulk2−/− MEF cells were treated with cGAMP, HT-DNA or DMXAA for the indicated time followed by immunoblotting of cell lysates. f, Electron micrographs of wild-type and Ulk1−/−Ulk2−/− MEF cells stimulated with cGAMP or torin 1. Boxed areas are enlarged to show double-membrane organelles that represent autophagosomes. Red arrows highlight double-membrane characteristic of autophagosomes in stimulated cells. Scale bar, 1 µm (original image), 200 nm (magnified image).
Extended Data Fig. 7 STING-induced LC3 conversion does not require beclin 1 (BECN1) or VPS34.
a, BECN1 is dispensable for LC3 conversion triggered by cGAMP. Wild-type and BECN1−/− bone-marrow-derived macrophages were stimulated with cGAMP or HT-DNA at the indicated time followed by immunoblotting of cell lysates. b, BECN1 is not essential for LC3 conversion in conventional autophagy. Wild-type and BECN1−/− bone-marrow-derived macrophages were stimulated with cGAMP or torin 1 or cultured in EBSS starvation medium at the indicated time, followed by immunoblotting of cell lysates. c, VPS34 depletion delayed cGAMP-induced STING degradation but not LC3 lipidation. VPS34-knockout BJ cells were treated with cGAMP for the indicated time followed by immunoblotting of cell lysates. d, Electron micrographs of HEK293T STING–Flag and HEK293T STING–Flag BECN1−/− cells, stimulated with cGAMP or torin 1. Boxed areas are enlarged to show double-membrane organelles that represent autophagosomes. Red arrow highlights double-membrane characteristic of autophagosomes in stimulated cells. Scale bar, 1 µm (original image), 200 nm (magnified image). e, f, ULK1 and VPS34 are essential for LC3 puncta formation induced by torin 1 but not by cGAMP. ULK1−/−, VPS34−/− or ATG5−/− HeLa LC3–GFP cells were treated with torin 1 or cGAMP for the indicated time. LC3–GFP puncta formation was visualized by fluorescence microscopy (e) and the percentage of cells with LC3–GFP peri-nuclear foci formation was quantified (f). The percentage of cells with LC3–GFP puncta was quantified from 100 cells (n = 2).
Extended Data Fig. 8 STING membrane trafficking and signalling requires SAR1A, SEC24C and ARF GTPases.
a, b, HeLa STING–GFP cells were transfected with siRNAs targeting SAR1A, SEC24C or luciferase (control) for 3 days before stimulation with cGAMP (75 nM) for 1 h. STING–GFP puncta were detected by confocal microscopy (a) and quantified (b). The percentage of cells with STING–GFP puncta was quantified from three random fields (n = 3, mean ± s.d.). c, BJ cells were transfected with siRNAs targeting SAR1A for three days before transfection with HT-DNA or poly(I:C) for the indicated time. Total RNA was isolated to measure the expression of indicated genes by RT–qPCR. d, Similar to c, except that HeLa cells were transfected with siRNAs targeting SEC24C, and cells were stimulated with cGAMP or poly(I:C). Mean ± s.d. was shown. Data represent two independent experiments with three replicates. e, Membrane trafficking is essential for cytosolic DNA but not RNA signalling. BJ cells were stimulated with BFA or GCA before transfection with HT-DNA or poly(I:C) or Lipofectamine (Lipo) alone. Cell lysates were analysed by native gel (for IRF3 dimerization) or SDS–PAGE followed by immunoblotting with the indicated antibodies. f, g, BJ cells were transfected with two different siRNAs targeting ARF1 before transfection with HT-DNA or poly(I:C) for the indicated time. Total RNA was isolated to measure the expression of the indicated genes by RT-qPCR. Mean ± s.d. is shown. Data represent two independent experiments with three replicates. h, i, HeLa STING–GFP cells were transfected with siRNAs targeting different ARF family members for three days and then stimulated with cGAMP (75 nM) for 1 h. STING–GFP foci were detected by confocal microscopy (h) and quantified (i). The percentage of cells with STING–GFP puncta was quantified from three random fields (n = 3, mean ± s.d.).
Extended Data Fig. 9 cGAMP induces anti-viral defence through autophagy.
a, Cytosolic DNA colocalizes with LC3 vesicles in STING-expressing cells. Cy3–ISD was delivered into HeLa LC3–GFP cells (lacking endogenous STING) or cells that stably express STING in the presence of PFO, followed by fluorescence microscopy. Single-cell images are shown, representing >90% of the cells under examination. b, c, cGAMP-induced activation of STING(1–340) enhances clearance of HIV-1 and HSV-1. HEK293T cells reconstituted with wild-type or mutant STING were stimulated with cGAMP and then infected with the pseudotyped HIV1–GFP virus for 24 h (b) or HSV1–GFP for 18 h (c). GFP+ cells were analysed by FACS. The results are representative of two independent experiments. d, STING(1–340) does not induce IFNβ or TNF. HEK293T cells that stably express full-length STING, STING(S336A), STING(1–340) or STING(1–340, R238A) were stimulated with cGAMP (2 μM) for 8 h and mRNA was extracted for RT–qPCR analysis of expression of the genes for IFNβ or TNF. Representative data are shown from two independent experiments; n = 2. Data are presented as mean ± s.d. e, RavZ catalyses LC3 deconjugation. HEK293T-STING stable cells were transfected with RavZ expression plasmids (wild-type or C258A mutant) for 36 h and then stimulated with cGAMP for the indicated time. Cell lysates were analysed by immunoblotting with the indicated antibodies. f, ATG5 knockout partially reverses cGAMP-mediated repression of HSV-1. ATG5 or TBK1 were knocked out using CRISPR in STING-expressing HEK293T cells. The cells were then infected with HSV–GFP with or without cGAMP treatment. FACS was performed to quantify relative virus GFP intensity in each cell line. g, ATG5 deficiency partially abrogated cGAMP-mediated suppression of HSV-1. One of BECN1, ATG5 or TBK1 was knocked out using CRISPR in STING-expressing HEK293T cells. The cells were then infected with HSV-1(ΔICP34.5) with or without cGAMP stimulation. qPCR using HSV-1 primers was performed to quantify relative viral genome equivalent in each cell line; n = 3. Data are presented as mean ± s.d. **P < 0.01. NS, not significant (two-tailed Student’s t-test).
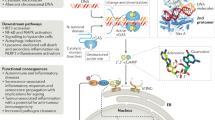
Extended Data Fig. 10 A model of DNA-induced autophagy and signalling through the cGAS–STING pathway.
Step 1, DNA from pathogens or damaged cells activates cGAS to synthesize cGAMP. cGAMP binds to STING and triggers STING translocation from the endoplasmic reticulum to the ERGIC and Golgi in a process that depends on SAR1, SEC24C and ARF family members. Step 2, the ERGIC, which contains cGAMP-bound STING, serves as a membrane source for LC3 recruitment and lipidation through a WIPI2-dependent mechanism. LC3-positive membranes target DNA and pathogens to autophagosomes, which are subsequently fused with lysosomes. Step 3, cGAMP-bound STING can also translocate through the trans-Golgi network and endosomes to lysosomes for degradation via the multi-vesicular body (MVB) pathway. Both the MVB and autophagosome fuse with lysosomes in a process that requires RAB7 GTPase.
Supplementary information
Supplementary Information
This file contains Supplementary Results, Supplementary References, Supplementary Tables 1-3 and Supplementary Video Legends.
Supplementary Figure 1
This file contains the uncropped blots.
Video 1: Live cell imaging of cGAMP-induced GFP-LC3 puncta formation.
Hela cells stably expressing STING-Flag and GFP-LC3 were stimulated with cGAMP (1 μM) after undergoing transient permeabilization with PFO at 0.1 μg/ml. As a control, cells were also grown in EBSS starvation media in the presence of Bafilomycin A1 (BafA1, 0.2 μM). Recording started at 20 min after adding cGAMP or changing to the starvation medium and lasted for 160 min with 2 min intervals.
Video 2: Live cell imaging of cGAMP-stimulated STING translocation to autophagosomes.
HeLa cells stably expressing STING-GFP and RFP-LC3 were stimulated with cGAMP (1 μM) in the presence or absence of Brefeldin A (BFA, 5 μM) or Bafilomycin A1 (BafA1, 0.2 μM). Recording started at 1 hr after addition of cGAMP and lasted 5 hr and 30 min with 2 min intervals.
Video 3: Live cell imaging of DNA-stimulated STING translocation to autophagosomes.
HeLa cells stably expressing STING-GFP and RFP-LC3 were transfected with HT-DNA (2 μg/ml) in the presence or absence of Brefeldin A (BFA, 5 μM) or bafilomycin A1 (BafA1, 0.2 μM). Recording started at 1 hr after DNA transfection and lasted approximately 11 hr with 4 min intervals.
Video 4: 3D imaging of STING translocation to the ERGIC and LC3 positive autophagosomes.
HeLa cells stably expressing STING-Flag and GFP-LC3 were stimulated with cGAMP (1 μM) for 2 hr before immunostaining with antibodies against Flag and ERGIC53. 3D fluorescence micrograph shows that, after cGAMP stimulation, STING (red) colocalizes with the ERGIC (cyan) and GFP-LC3 (green) in the peri-nuclear region.
Video 5: Live cell imaging of cytosolic DNA clearance by DNA-induced autophagy.
Cy3-ISD (0.5 μg/ml or 2 μg/ml) was delivered into HeLa cells stably expressing STING-Flag and GFP-LC3 that were transiently permeabilized with PFO (0.1 μg/ml) for 3 hr. After the medium was replaced with fresh DMEM (10% FBS), cells were recorded by live cell imaging for another 3 hr. Video was tracked to detect CY3-ISD translocation to GFP-LC3 puncta. Recording started at 20 min after adding CY3-ISD and lasted approximately 6 hr with 4 min intervals.
Video 6: Live cell imaging of cytosolic DNA clearance by cGAMP-induced autophagy.
Cy3-ISD was delivered together with different amounts of cGAMP (as indicated in the movie) into HeLa cells stably expressing STING-Flag and GFP-LC3 that were transiently permeabilized with PFO (0.1 μg/ml) for 1.5 hr. The medium was replaced with fresh DMEM (10% FBS) and cells were recorded by live cell imaging for another 5 hr. Video was tracked to detect CY3-ISD translocation to GFP-LC3 puncta. Recording started at 20 min after adding CY3-ISD and lasted approximately 7 hr with 4 min intervals.
Rights and permissions
About this article
Cite this article
Gui, X., Yang, H., Li, T. et al. Autophagy induction via STING trafficking is a primordial function of the cGAS pathway. Nature 567, 262–266 (2019). https://doi.org/10.1038/s41586-019-1006-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1006-9
This article is cited by
-
High glucose-induced p66Shc mitochondrial translocation regulates autophagy initiation and autophagosome formation in syncytiotrophoblast and extravillous trophoblast
Cell Communication and Signaling (2024)
-
The dual function of cGAS-STING signaling axis in liver diseases
Acta Pharmacologica Sinica (2024)
-
Cytosolic DNA sensors in neurodegenerative diseases: from physiological defenders to pathological culprits
EMBO Molecular Medicine (2024)
-
Second messenger 2'3'-cyclic GMP-AMP (2'3'-cGAMP): the cell autonomous and non-autonomous roles in cancer progression
Acta Pharmacologica Sinica (2024)
-
A new road to STING activation
Nature Chemical Biology (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.