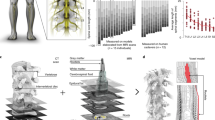
Abstract
Spinal cord injury leads to severe locomotor deficits or even complete leg paralysis. Here we introduce targeted spinal cord stimulation neurotechnologies that enabled voluntary control of walking in individuals who had sustained a spinal cord injury more than four years ago and presented with permanent motor deficits or complete paralysis despite extensive rehabilitation. Using an implanted pulse generator with real-time triggering capabilities, we delivered trains of spatially selective stimulation to the lumbosacral spinal cord with timing that coincided with the intended movement. Within one week, this spatiotemporal stimulation had re-established adaptive control of paralysed muscles during overground walking. Locomotor performance improved during rehabilitation. After a few months, participants regained voluntary control over previously paralysed muscles without stimulation and could walk or cycle in ecological settings during spatiotemporal stimulation. These results establish a technological framework for improving neurological recovery and supporting the activities of daily living after spinal cord injury.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Data that support the findings and software routines developed for the data analysis will be made available upon reasonable request to the corresponding author.
References
Behrman, A. L., Ardolino, E. M. & Harkema, S. J. Activity-based therapy: from basic science to clinical application for recovery after spinal cord injury. J. Neurol. Phys. Ther. 41, S39–S45 (2017).
Jones, M. L. et al. Activity-based therapy for recovery of walking in individuals with chronic spinal cord injury: results from a randomized clinical trial. Arch. Phys. Med. Rehabil. 95, 2239–2246 (2014).
Field-Fote, E. C., Lindley, S. D. & Sherman, A. L. Locomotor training approaches for individuals with spinal cord injury: a preliminary report of walking-related outcomes. J. Neurol. Phys. Ther. 29, 127–137 (2005).
Edgerton, V. R. et al. Training locomotor networks. Brain Res. Rev. 57, 241–254 (2008).
Côté, M. P., Murray, M. & Lemay, M. A. Rehabilitation strategies after spinal cord injury: inquiry into the mechanisms of success and failure. J. Neurotrauma 34, 1841–1857 (2017).
Borton, D., Micera, S., Millán, J. d. R. & Courtine, G. Personalized neuroprosthetics. Sci. Transl. Med. 5, 210rv2 (2013).
Field-Fote, E. C. & Roach, K. E. Influence of a locomotor training approach on walking speed and distance in people with chronic spinal cord injury: a randomized clinical trial. Phys. Ther. 91, 48–60 (2011).
Minassian, K., McKay, W. B., Binder, H. & Hofstoetter, U. S. Targeting lumbar spinal neural circuitry by epidural stimulation to restore motor function after spinal cord injury. Neurotherapeutics 13, 284–294 (2016).
Angeli, C. A. et al. Recovery of over-ground walking after chronic motor complete spinal cord injury. N. Engl. J. Med. 379, 1244–1250 (2018).
Gill, M. L. et al. Neuromodulation of lumbosacral spinal networks enables independent stepping after complete paraplegia. Nat. Med. https://doi.org/10.1038/s41591-018-0175-7 (2018).
Barolat, G., Myklebust, J. B. & Wenninger, W. Enhancement of voluntary motor function following spinal cord stimulation-case study. Appl. Neurophysiol. 49, 307–314 (1986).
Angeli, C. A., Edgerton, V. R., Gerasimenko, Y. P. & Harkema, S. J. Altering spinal cord excitability enables voluntary movements after chronic complete paralysis in humans. Brain 137, 1394–1409 (2014).
Danner, S. M. et al. Human spinal locomotor control is based on flexibly organized burst generators. Brain 138, 577–588 (2015).
Grahn, P. J. et al. Enabling task-specific volitional motor functions via spinal cord neuromodulation in a human with paraplegia. Mayo Clin. Proc. 92, 544–554 (2017).
Carhart, M. R., He, J., Herman, R., D’Luzansky, S. & Willis, W. T. Epidural spinal-cord stimulation facilitates recovery of functional walking following incomplete spinal-cord injury. IEEE Trans. Neural Syst. Rehabil. Eng. 12, 32–42 (2004).
Minassian, K. et al. Stepping-like movements in humans with complete spinal cord injury induced by epidural stimulation of the lumbar cord: electromyographic study of compound muscle action potentials. Spinal Cord 42, 401–416 (2004).
Rattay, F., Minassian, K. & Dimitrijevic, M. R. Epidural electrical stimulation of posterior structures of the human lumbosacral cord: 2. Quantitative analysis by computer modeling. Spinal Cord 38, 473–489 (2000).
Capogrosso, M. et al. A computational model for epidural electrical stimulation of spinal sensorimotor circuits. J. Neurosci. 33, 19326–19340 (2013).
Moraud, E. M. et al. Closed-loop control of trunk posture improves locomotion through the regulation of leg proprioceptive feedback after spinal cord injury. Sci. Rep. 8, 76 (2018).
Gerasimenko, Y., Roy, R. R. & Edgerton, V. R. Epidural stimulation: comparison of the spinal circuits that generate and control locomotion in rats, cats and humans. Exp. Neurol. 209, 417–425 (2008).
Wenger, N. et al. Spatiotemporal neuromodulation therapies engaging muscle synergies improve motor control after spinal cord injury. Nat. Med. 22, 138–145 (2016).
Wenger, N. et al. Closed-loop neuromodulation of spinal sensorimotor circuits controls refined locomotion after complete spinal cord injury. Sci. Transl. Med. 6, 255ra133 (2014).
Capogrosso, M. et al. A brain-spine interface alleviating gait deficits after spinal cord injury in primates. Nature 539, 284–288 (2016).
Dominici, N. et al. Versatile robotic interface to evaluate, enable and train locomotion and balance after neuromotor disorders. Nat. Med. 18, 1142–1147 (2012).
Asboth, L. et al. Cortico-reticulo-spinal circuit reorganization enables functional recovery after severe spinal cord contusion. Nat. Neurosci. 21, 576–588 (2018).
van den Brand, R. et al. Restoring voluntary control of locomotion after paralyzing spinal cord injury. Science 336, 1182–1185 (2012).
Mignardot, J. B. et al. A multidirectional gravity-assist algorithm that enhances locomotor control in patients with stroke or spinal cord injury. Sci. Transl. Med. 9, eaah3621 (2017).
Cappellini, G., Ivanenko, Y. P., Dominici, N., Poppele, R. E. & Lacquaniti, F. Migration of motor pool activity in the spinal cord reflects body mechanics in human locomotion. J. Neurophysiol. 104, 3064–3073 (2010).
Yakovenko, S., Mushahwar, V., VanderHorst, V., Holstege, G. & Prochazka, A. Spatiotemporal activation of lumbosacral motoneurons in the locomotor step cycle. J. Neurophysiol. 87, 1542–1553 (2002).
Asanuma, H. & Mackel, R. Direct and indirect sensory input pathways to the motor cortex; its structure and function in relation to learning of motor skills. Jpn. J. Physiol. 39, 1–19 (1989).
Gourab, K. & Schmit, B. D. Changes in movement-related β-band EEG signals in human spinal cord injury. Clin. Neurophysiol. 121, 2017–2023 (2010).
Capogrosso, M. et al. Configuration of electrical spinal cord stimulation through real-time processing of gait kinematics. Nat. Protoc. 13, 2031–2061 (2018).
Schieppati, M. The Hoffmann reflex: a means of assessing spinal reflex excitability and its descending control in man. Prog. Neurobiol. 28, 345–376 (1987).
Schindler-Ivens, S. & Shields, R. K. Low-frequency depression of H-reflexes in humans with acute and chronic spinal-cord injury. Exp. Brain Res. 133, 233–241 (2000).
Formento, E. et al. Electrical spinal cord stimulation must preserve proprioception to enable locomotion in humans with spinal cord injury. Nat. Neurosci. https://doi.org/10.1038/s41593-018-0262-6 (2018).
Moraud, E. M. et al. Mechanisms underlying the neuromodulation of spinal circuits for correcting gait and balance deficits after spinal cord injury. Neuron 89, 814–828 (2016).
Takeoka, A., Vollenweider, I., Courtine, G. & Arber, S. Muscle spindle feedback directs locomotor recovery and circuit reorganization after spinal cord injury. Cell 159, 1626–1639 (2014).
Holtmaat, A. & Svoboda, K. Experience-dependent structural synaptic plasticity in the mammalian brain. Nat. Rev. Neurosci. 10, 647–658 (2009).
Nishimura, Y., Perlmutter, S. I., Eaton, R. W. & Fetz, E. E. Spike-timing-dependent plasticity in primate corticospinal connections induced during free behavior. Neuron 80, 1301–1309 (2013).
Perez, M. A., Field-Fote, E. C. & Floeter, M. K. Patterned sensory stimulation induces plasticity in reciprocal Ia inhibition in humans. J. Neurosci. 23, 2014–2018 (2003).
Urbin, M. A., Ozdemir, R. A., Tazoe, T. & Perez, M. A. Spike-timing-dependent plasticity in lower-limb motoneurons after human spinal cord injury. J. Neurophysiol. 118, 2171–2180 (2017).
Dietz, V. Behavior of spinal neurons deprived of supraspinal input. Nat. Rev. Neurol. 6, 167–174 (2010).
West, C. R. et al. Association of epidural stimulation with cardiovascular function in an individual with spinal cord injury. JAMA Neurol. 75, 630–632 (2018).
Herrity, A. N., Williams, C. S., Angeli, C. A., Harkema, S. J. & Hubscher, C. H. Lumbosacral spinal cord epidural stimulation improves voiding function after human spinal cord injury. Sci. Rep. 8, 8688 (2018).
van Middendorp, J. J. et al. A clinical prediction rule for ambulation outcomes after traumatic spinal cord injury: a longitudinal cohort study. Lancet 377, 1004–1010 (2011).
Sharrard, W. J. The segmental innervation of the lower limb muscles in man. Ann. R. Coll. Surg. Engl. 35, 106–122 (1964).
Acknowledgements
See Supplementary Notes. Support: International Foundation for Research in Paraplegia (IRP), Wings for Life, Wyss Center for Neuroengineering, European Union’s Horizon 2020 No.785907 (Human Brain Project SGA2), Eurostars No. E10889, GTXmedical, National Center of Competence in Research (NCCR) Robotics of the Swiss National Foundation, the Commission of Technology and Innovation Innosuisse (CTI) No. 25761.1, Voirol Foundation, Firmenich Foundation, Pictet Group Charitable Foundation, Panacée Foundation, riders4riders, SOFMER (to P.S.), the Whitaker International Scholars Program (to I.S.) and the H2020-MSCA-COFUND-2015 EPFL Fellows program (No. 665667 to F.B.W.).
Reviewer information
Nature thanks C. Moritz, J. Henderson and K. Moxon for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
N.B. and T.D., Neural Research Programmer development. F.B.W., Mi.C., C.G.L.G.-M., R.H., V.D. and J.v.Z., technological framework. J.B., surgeries. F.B.W., J.-B.M., C.G.L.G.-M., Ma.C., E.Pi., K.M., R.D., S.K., I.S. and G.C. performed and analysed experiments. R.D., S.K. and Ma.C. contributed equally. F.B., muscle data. I.F., L.M., M.V., P.S., I.S., F.B.W., J.-B.M., C.G.L.G.-M., K.M., K.V.D.K. and G.E., neurorehabilitation. K.V.D.K., F.B., J.P., B.S., E.Pr., P.S. and S.C., clinical and neurological evaluations. A.R. and Ma.C., computational framework. A.R., E.Pa., E.N. and N.K., computational simulations. J.-B.M., C.G.L.G.-M., R.D., S.K. and F.B.W. generated figures. A.W., M.V., R.B., V.D. and H.L., regulatory affairs. K.M., J.B. and G.C., conception and supervision. G.C. wrote the paper with J.B., F.B.W. and K.M.
Corresponding author
Ethics declarations
Competing interests
G.C., J.B., Ma.C. and V.D. hold various patents in relation to the present work. T.D., R.B. and N.B. are Medtronic employees, and V.D., H.L., J.v.Z., A.W., Mi.C. and E.Pa. are GTXmedical employees. In review of the manuscript they contributed to technical accuracy but did not influence the results or the content of the manuscript. G.C., J.B., V.D., and H.L. are founders and shareholders of GTXmedical, a company with direct relationships to the presented intervention.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Surgical procedure and technological framework.
a, Surgery. Step 1: high-resolution MRI for pre-surgical planning. The entry point into the epidural space is based on the position of the conus. Step 2: placement of subdermal and intramuscular needle EMG electrodes for key leg muscles and paraspinal (PS) muscles. A subdermal needle is inserted over the sacrum and used as a return electrode for stimulation. Bottom, schematic of the 16-electrode paddle array. Step 3: surgical openings based on pre-surgical planning, typically between the L1 and L2 vertebrae, which are identified through intraoperative X-ray. The mediolateral positions of the paddle array are evaluated with X-ray and recordings of EMG responses following single pulses of EES delivered to the most rostral or most caudal midline electrodes. Step 4: the rostrocaudal position of the paddle array is optimized using EMG responses to single-pulse EES delivered to the electrodes located at each corner of the paddle array. The aim is to obtain strong ipsilateral responses in hip flexors with the most rostral electrodes and strong ipsilateral responses in ankle extensors with the most caudal electrodes. Step 5: implantable pulse generator (IPG) placed within the abdomen. Once connected to the paddle array, the impedance of the electrodes is evaluated to verify that all the components are properly connected. Step 6: post-surgical CT scan showing the location of the paddle array with respect to the vertebrae in each participant. b, Technological framework and surgical procedure. Step 1: participants wear reflective markers that are monitored using infrared cameras. An algorithm assigns the markers to the joints in real-time. Step 2: the spatiotemporal trajectory of the foot around a calculated centre of rotation (centroid, updated every 3 s) is converted into angular coordinates that trigger and terminate EES protocols when a user-defined threshold is crossed. Step 3: EES commands are transmitted to the IPG via Bluetooth (1) to a module that converts them into infrared signals (2), which are then transferred to the stimulation programmer device (2′). Step 4: the stimulation programmer transmits EES commands into the IPG (4) via induction telemetry, using an antenna (3) taped to the skin and aligned to the IPG. EES is delivered through the paddle array (5).
Extended Data Fig. 2 Identification of electrode configurations to target selected posterior roots.
Step 1: single-pulse EES and EMG recording setup. Step 2: motor neuron pools are located in specific segments, which provides information on the relative recruitment of each posterior root with EES. For example, electrodes targeting the L3 or L4 posterior roots will elicit the strongest EMG responses in the knee extensors. A personalized computational model of EES allows the performance of simulations that evaluate the relative activation of a given posterior root with a given electrode over the entire amplitude range. Each curve corresponds to an electrode. The highlighted curve corresponds to the electrode selected after steps 3–5. Step 3: single pulses of EES are delivered through the subset of electrodes identified by simulations. The EMG responses are recorded over a broad range of EES amplitudes. Step 4: the EMG responses are concatenated and averaged across n = 4 repetitions for each EMG amplitude, and the peak-to-peak amplitude of the average responses is calculated to elaborate a recruitment curve for each recorded leg muscle (black traces: targeted muscles). Step 5: the circular plots display the normalized EMG responses (greyscale) when delivering single-pulse EES at increasing amplitudes (radial axis), where the white circle highlights the optimal EES amplitude and the polygon quantifies the relative muscular selectivity at this amplitude (median response taken over n = 4 EES pulses). The motor neuron activation maps are shown for the optimal amplitudes. Step 6: decision tree to validate or optimize electrode configurations. The selected electrode is tested during standing as the position of the spinal cord with respect to the paddle array can change between supine and standing. In this example, the selectivity improves during standing. When the selectivity is deemed insufficient, the current is steered towards the targeted posterior roots using multipolar configurations. The example shows the increased selectivity of a multipolar configuration with two cathodes surrounded by three anodes, compared to the two corresponding monopolar configurations. These results were verified experimentally and with computer simulations.
Extended Data Fig. 3 Spatial selectivity of targeted electrode configurations.
Monopolar configurations (shown on paddle array schematics) experimentally selected to target the left and right posterior roots associated with hip flexion (L1), knee extension (L3), ankle flexion (L4) and ankle extension (S1) for the three participants. The circular plots and motor neuron activation maps use the same conventions as in Fig. 2 and Extended Data Fig. 2 (median of n = 4 pulses). The normalized selectivity index is reported above each motor neuron activation map. This index represents the percentage of posterior root selectivity for the electrode configuration selected experimentally, with respect to the maximum posterior root selectivity that can be achieved among all monopolar configurations (all selectivity indices obtained from computational simulations). Note that in P2, the electrode selected experimentally to target the right S1 root was located on the midline and resulted in bilateral activation within computational simulations, which resulted in a normalized selectivity index of zero.
Extended Data Fig. 4 Single-joint movements enabled by targeted EES.
Step 1: participants are placed in standardized positions to allow assessment of voluntary torque production at a single joint (isometric contractions) without and with targeted EES. Step 2: EES protocols elaborated from single-pulse experiments (Extended Data Figs. 2, 3) are optimized for each task using multipolar configurations and adjustments of EES amplitude and frequency. Step 3: sequence of each trial. Participants were asked to produce a maximal voluntary contribution, but failed in most cases, as evidenced by the absence of EMG activity during this period. While they continued trying to activate the targeted muscle, EES was switched on. After a few seconds, participants were instructed to stop their voluntary contribution. After a short delay, EES was switched off. For each sequence, the produced torque and EMG activity of the key agonist and antagonist muscles acting at the targeted joint were calculated over the four indicated phases of the trial. Plots report the measured torques and EMG activity during the various phase of the trial for the left legs of all participants for the four tested joints (cyan, flexor; magenta, extensor), together with EES parameters and electrode configurations. All measurements were performed before rehabilitation, except for hip extension in P1 and P2 (not tested before), and ankle extension in P3 (no capacity before rehabilitation), which were carried out after rehabilitation. Targeted EES enabled or augmented the specific recruitment of the targeted muscle, which resulted in the production of the desired torque at the targeted joint, except for ankle extension of P2. Plots show quantification of the EMG activity and torque for n = 3 trials per condition. Note that hip flexion can be enabled or augmented with EES targeting L1 and/or L4 posterior roots (heteronymous facilitation of flexor motor neuron pools).
Extended Data Fig. 5 Modulation of EEG activity during volitional contraction of leg muscles without and with EES.
a, Recordings of EEG activity while participants were asked to produce an isometric torque at the knee joint without and with continuous EES targeting motor neuron pools innervating knee extensors, as shown in b. b, Superimposed EEG responses (n = 40 repetitions) and temporal changes in the topography of average activity over the cortical surface after the onset of EES, as indicated above each map. The onset was calculated from the onset of EMG responses in the targeted vastus lateralis muscle (insets). The stimulation elicited a robust event-related response over the left sensorimotor cortex with a latency of 90 ± 40 ms for P1 and P3, and of 170 ± 40 ms for P2 (full range of the peaks and middle of this range indicated). c, Average normalized time–frequency plots (n = 40 trials) showing ERD and ERS over the Cz electrode (central top electrode) for each individual during the voluntary activation of knee extensor muscles without and with EES. Schematic drawings (left) indicate the motor scores of the tested legs, including the targeted muscles (*), at the time of enrolment in the study. Both legs were tested in P1 owing to his asymmetric deficits. d, Normalized average power (mean ± s.e.m.) of the β-band over the Cz electrode during ERS from 0 to 500 ms after termination of contraction without and with continuous EES (n = 40 repetitions for each condition, individual data points shown except for outliers more than 3 median absolute deviations away from the median). ***P < 0.001 (permutation tests, see Methods).
Extended Data Fig. 6 Configuration of spatiotemporal EES to enable walking.
a, Spatial configuration. Step 1: select electrode configurations from single-pulse experiments to target the three hotspots underlying the production of walking in healthy individuals (weight acceptance: L3; propulsion: S1; swing: L1/L4). Step 2: optimize EES amplitude and frequency while delivering EES during standing. Multipolar configurations can be used to refine selectivity of EES protocols. Example shows continuous EES targeting the right L3 posterior root to facilitate right knee extension during standing, and trains (500 ms) of EES targeting the right L1 posterior root stimulation to facilitate hip flexion. Two EES frequencies are shown (P3). b, Temporal configuration. Step 3: decision tree to select the best strategy to configure the temporal structure of EES protocols. If the participant is able to initiate leg movements consistently, use closed-loop EES based on real-time processing of foot trajectory. If the participant is not able to initiate consistent leg movements but can feel when EES is applied, use open-loop EES. If the participant is not able to generate movement and cannot feel EES, use closed-loop EES combined with physiotherapist assistance to move the legs. Step 4: real-time monitoring of the spatiotemporal trajectory of the feet. The trajectory is modelled as a foot rotating in space around the centroid of the movement (updated every 3 s). Angular thresholds determine the onset and end of EES protocols. Step 5: example showing the effect of three different angular thresholds on the onset of EES and resulting kinematics and EMG activity, including the quantification of kinematics for each step and condition that enables selecting the optimal onset of EES trains (P1). The same approach is used to optimize the duration of each train. c, Comparisons between closed-loop and open-loop EES. Plots show the vertical displacements of the left and right feet and successive step heights during walking with spatiotemporal EES delivered in closed loop versus open loop, showing the reduced variability of step height during pre-programmed EES sequences (P1). d, Resulting EMG patterns. Step 6: example of the progressive addition of EES protocols targeting specific hotspots. Plots show the quantification of EMG activity for the displayed muscles (n = 7 gait cycles for no EES and n = 9 gait cycles for each stimulation condition, P2). Step 7: EES amplitudes and frequencies are adjusted to avoid detrimental interactions between the different EES protocols and thus obtain the desired kinematic and EMG activity. Plots report the modulation of EMG activity and kinematics with increases in EES amplitude and frequency (mean ± s.e.m.; amplitude data: n = 10, 12, 12, 30, 19, 12, 11, 10 gait cycles for amplitudes in increasing order, P2; frequency data: n = 20, 15, 16, 17, 15, 16, 15 gait cycles for frequencies in increasing order, P3).
Extended Data Fig. 7 Targeted modulation of muscle activity during walking.
Each panel reports the same representative data and quantification for one participant. Left, EMG activity of leg muscles during walking on a treadmill without EES (EES OFF) and with spatiotemporal EES (EES ON) while applying 50%, 45% and 70% body weight support for participants P1, P2 and P3, respectively. Stance and swing phases are indicated by grey and white backgrounds, respectively. The personalized spatiotemporal EES sequence (open loop) is schematized at the top right. The colours of each EES protocol refer to the targeted hotspots: weight acceptance (salmon), propulsion (magenta) and swing (cyan). These colours are used in the EMG traces to indicate the temporal window over which each targeted EES protocol is active. The bar plots report the amplitude of muscle activity without EES and with spatiotemporal EES, for which the quantification was performed over the entire burst of EMG activity and during each temporal window with targeted EES. The temporal windows are labelled with a number that refers to the spatiotemporal EES sequence. These results show the pronounced increase in the EMG activity of the targeted muscles (P1, no EES: n = 7 gait cycles, EES: n = 11 gait cycles; P2, no EES: n = 9 gait cycles, EES: n = 9 gait cycles; P3, no EES: n = 10 gait cycles, EES: n = 57 gait cycles). The average spatiotemporal trajectories of both feet with respect to the hip in the sagittal plane are shown for walking without EES and with spatiotemporal EES. The presence of targeted EES is indicated with the same colour code. Plots at bottom right show the relationships between EES frequency and the modulation of the EMG activity of flexor (blue) and extensor (magenta or salmon) muscles and maximum amplitude of hip movements during walking (mean ± s.e.m.; P1: n = 14, 17, 15, 19 gait cycles for increasing frequencies; P2: n = 13, 16, 10, 17, 12 gait cycles for increasing frequencies; P3: n = 20, 15, 16, 17, 15, 16, 15 gait cycles for increasing frequencies). ***P < 0.001. Student’s t-test.
Extended Data Fig. 8 Volitional adaptations of walking during otherwise unchanged spatiotemporal EES.
a–c, Quantifications of experiments shown in Fig. 4a–c for each participant. a, Step height and TA EMG activity with and without EES during overground walking (P1, EES ON: n = 7 gait cycles; P2, EES ON: n = 16 gait cycles; P3, EES ON, n = 7 gait cycles). b, Step height and TA EMG activity during normal steps and when participants were requested to perform exaggerated step elevations during overground walking (P1, n = 15 normal gait cycles, n = 11 exaggerated gait cycles; P2, n = 31 normal gait cycles, n = 23 exaggerated gait cycles; P3, n = 14 normal gait cycles, n = 10 exaggerated gait cycles). c, Step height and TA EMG activity during the first and last 30 steps extracted from a sequence of 1 h of locomotion on a treadmill (n = 30 gait cycles for all conditions). ***P < 0.001; n.s., non-significant; Student’s t-test. d, EMG activity of representative leg muscles, vertical displacements of the foot and anteroposterior oscillations of the leg (virtual limb joining the hip to the foot) while P2 was walking continuously on the treadmill with spatiotemporal EES (open loop). The participant was asked to suppress the effects of EES and stand during one cycle of open-loop spatiotemporal EES sequence, highlighted in brown (SKIP), whereas he actively contributed to the production of movement the rest of the time. Plots report the quantification of step height and TA EMG activity during walking and when skipping steps for each participant (P1, n = 13 normal gait cycles, n = 1 skipped cycles; P2, n = 36 normal gait cycles, n = 3 skipped gait cycles; P3, n = 11 normal gait cycles, n = 2 skipped cycles). e, EMG activity of two representative muscles, vertical displacements of the foot and anteroposterior oscillations of the leg while P1 was walking on the treadmill and the speed of the belt increased progressively from 0.8 to 2 km h–1. Plots show relationships between treadmill speed and mean stride length and TA EMG activity in all participants (P1: n = 9, 9, 9, 9, 10, 18, 15, 9, 9 gait cycles for increasing speeds; P2: n = 13, 10, 7, 8, 10, 9 gait cycles for increasing speeds; P3: n = 8, 8, 10, 9, 9, 8 gait cycles for increasing speeds; s.e.m. shown). The range of tested speeds was adapted to the walking ability of each participant.
Extended Data Fig. 9 Comparison between continuous and spatiotemporal EES during overground walking.
Each panel represents one participant who is attempting to walk overground with gravity-assist without EES (left), with continuous EES (middle) and with spatiotemporal EES (right). EMG activity of representative leg muscles, vertical position of the foot and distance covered by the foot in the forward direction are displayed for each experimental condition. Continuous EES is applied throughout the trial (red). For P2 and P3, we optimized EES protocols that targeted the posterior roots on both sides, whereas EES was applied over the most rostral and most caudal midline electrodes for P1, as shown next to each plot. Spatiotemporal EES is represented using the same colour scheme as in Fig. 3 and Extended Data Fig. 7. The plots report quantification of EMG activity, step height and mean speed (based on distance covered) for the three experimental conditions (P1, n = 6, 7, 8 gait cycles for no EES, continuous EES and spatiotemporal EES; P2, n = 17, 7, 9 gait cycles for no EES, continuous EES and spatiotemporal EES; P3, n = 6, 10, 9 gait cycles for no EES, continuous EES and spatiotemporal EES). ***P < 0.001; **P < 0.01; n.s., non-significant. One-way ANOVA, post hoc Tukey’s HSD. These recordings were repeated on at least three different days for each participant.
Extended Data Fig. 10 Rehabilitation program and evolution of walking capacity.
a, Rehabilitation programs were continuously personalized on the basis of the current motor performance of participants. Walking capacities evolved in phases (b). For this reason, the relative percentage of training in the various tasks has been divided into clusters, which correspond to the evolution of walking capacities. To facilitate the sustained production of reproducible locomotor movements (Extended Data Fig. 6c), EES was delivered in open-loop mode during gait rehabilitation. b, Walking capacities evolved through stereotypical phases that are illustrated in the snapshots. c, Plots showing the progression of the three participants along the phases of recovery during the rehabilitation program, and during the subsequent 6 months for P1 and P2. P3 had just completed the rehabilitation program at the time of submission of this study. See also Supplementary Video 4.
Extended Data Fig. 11 Changes in muscle mass and quality and recovery of voluntary movements with and without EES in participant P3.
a, Skeletal muscle mass and quality were assessed at the pre- and post-rehabilitation time points using X-ray attenuation from CT images obtained at the abdomen (L3 vertebra) and mid-thigh (25 cm above femorotibial joint space). Muscle mass was determined by measuring the cross-sectional areas (CSAs) of muscle tissues, while muscle quality was reflected by CT attenuation numbers (in Hounsfield units, HU) within the CSAs. Muscle segmentations were performed semi-automatically using ImageJ and muscle-specific HU thresholds (–29 to 150 HU). Plots report the substantial changes in muscle mass at mid-thigh, for both flexor and extensor muscles, and of trunk muscles. Muscle quality was also improved at both levels: total mid-thigh, left: 52.9 to 56.1 HU, right: 51.9 to 56.7 HU; total L3, 45.9 to 48.3 HU. This increase in CT attenuation numbers between the baseline CT scan and the follow-up imaging reflected the decrease in muscle fibre lipid content at the mid-thigh and abdomen. These evaluations were part of a protocol amendment obtained when enrolling P3. b, Assessment of voluntary torque production at the ankle (extension) with targeted EES before and after rehabilitation. Conventions are as in Extended Data Fig. 4. c, Snapshots showing voluntary extension of the left leg against the direction of gravity together with the concomitant sequence of EMG activity in the extensor and flexor muscles of this leg. The zoomed window shows the relationship between the movement and the EMG activity, indicated with the numbers. This participant presented flaccid paralysis, and had thus no control over leg muscles before the surgery. This movement was observed repeatedly at the end of the rehabilitation period (at least two days per week for several weeks).
Extended Data Fig. 12 Performance of closed-loop spatiotemporal EES to enable walking and cycling outside the laboratory.
a, P1 and P2 were asked to walk freely overground with a walker (no body weight support) for 6 min. The concomitant vertical displacements of the foot show the consistency of EES triggering events despite variable foot kinematics and voluntary breaks. The trajectory of the centre of mass is shown from a top view to illustrate the ability to steer locomotion along any desired path. EES protocols took into account the deficits of each participant (cyan, EES targeting hip flexion; magenta, EES targeting knee and ankle extension). Histograms indicate the number of detected foot-off events for the represented leg as a function of the latency with respect to real foot-off events. The confusion matrix associated with these detections is represented below, as a percentage of the real events that were correctly or incorrectly classified. Detections were considered valid if they occurred between 400 ms before and 100 ms after real foot-off events, as highlighted in green on histograms (P1, n = 49 gait cycles; P2, n = 79 gait cycles). b, Closed-loop spatiotemporal EES was delivered in P3 using an electric trike powered by hand and foot pedals. Traces show EMG activities of the targeted hip flexor and knee extensor muscles on one leg together with the tangential acceleration of the pedal and power generated at the foot pedal. Plots report the quantification of flexor and extensor EMG activities, peak tangential accelerations and generated power without and with EES. Successive ankle trajectories during cycling are shown together with the timing of EES protocols targeting the hip flexor and knee extensor muscles. The histograms and confusion matrices report the performance of the controller following the same conventions as in a, except that the correct detection window was restricted to 50 ms before and 100 ms after the desired crank position (P3: n = 73 pedalling cycles). ***P < 0.001. Student’s t-test.
Supplementary information
Supplementary Information
This file contains Supplementary Notes and Methods.
Supplementary Video 1 Technological framework and surgical procedure
Description of the gait rehabilitation environment, together with the methods to construct the spatiotemporal maps of motoneuron activation underlying walking. An animation shows the surgical placement of the electrode paddle array, and the methods underlying the delivery of spatially-selective stimulation trains through real-time movement feedback during walking assisted with a multidirectional body weight support system.
Supplementary Video 2 Spatiotemporal EES enables voluntary walking
(1) Overground walking without and with spatiotemporal EES for all participants, where EES sequences are displayed in real-time and EMG traces are colored during stimulation targeting this muscle. (2) voluntary modulation of leg movements, including exaggerated foot elevation, stepping across speeds, and walk/stand/walk transition. Spatiotemporal EES is delivered in open-loop, without any change in EES parameters. All recordings were repeated at least on 5 different days for each participant. (3) Beginning and end of a one hour bout of walking (1.2 km). This recording was performed only once in each participant, but participants routinely walked longer distance during training.
Supplementary Video 3 Comparison between continuous and spatiotemporal stimulation
Example of overground locomotion without EES, with continuous EES, and with spatiotemporal EES. Conventions are the same as in Supplementary Video 2. These recordings were repeated at least on 3 different days for each participant.
Supplementary Video 4 Improvements with rehabilitation
Movies showing the evolution of walking capacities with EES over the course of the rehabilitation program for the three participants. Conventions are the same as in Supplementary Video 2. Similar observations were made on at least 5 different days for each participant and condition.
Supplementary Video 5 Improvements with rehabilitation
Movies showing the evolution of voluntary leg movements and walking capacities without EES over the course of the rehabilitation program for participants P1 and P2. Performance of participant P3 is shown during overground walking with an assistive device and EES, but without body weight support. Examples of voluntary hip flexion and sustained knee extension without EES are also shown. Conventions are the same as Supplementary Video 2. Similar observations were made on at least 3 different days for each participant and condition.
Supplementary Video 6 Integrated solution to use spatiotemporal EES in ecological setting
Movie showing the wearable, voice-controlled technology that supports closed-loop spatiotemporal EES to walk outside the laboratory environment and to ride a trike powered with the arms and legs.
Rights and permissions
About this article
Cite this article
Wagner, F.B., Mignardot, JB., Le Goff-Mignardot, C.G. et al. Targeted neurotechnology restores walking in humans with spinal cord injury. Nature 563, 65–71 (2018). https://doi.org/10.1038/s41586-018-0649-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0649-2
Keywords
This article is cited by
-
Noninvasive spinal stimulation improves walking in chronic stroke survivors: a proof-of-concept case series
BioMedical Engineering OnLine (2024)
-
Functional plasticity of glutamatergic neurons of medullary reticular nuclei after spinal cord injury in mice
Nature Communications (2024)
-
The future of brain circuit-targeted therapeutics
Neuropsychopharmacology (2024)
-
Biomimetic computer-to-brain communication enhancing naturalistic touch sensations via peripheral nerve stimulation
Nature Communications (2024)
-
Neuromorphic hardware for somatosensory neuroprostheses
Nature Communications (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.