Abstract
Angiocrine signals derived from endothelial cells are an important component of intercellular communication and have a key role in organ growth, regeneration and disease1,2,3,4. These signals have been identified and studied in multiple organs, including the liver, pancreas, lung, heart, bone, bone marrow, central nervous system, retina and some cancers1,2,3,4. Here we use the developing liver as a model organ to study angiocrine signals5,6, and show that the growth rate of the liver correlates both spatially and temporally with blood perfusion to this organ. By manipulating blood flow through the liver vasculature, we demonstrate that vessel perfusion activates β1 integrin and vascular endothelial growth factor receptor 3 (VEGFR3). Notably, both β1 integrin and VEGFR3 are strictly required for normal production of hepatocyte growth factor, survival of hepatocytes and liver growth. Ex vivo perfusion of adult mouse liver and in vitro mechanical stretching of human hepatic endothelial cells illustrate that mechanotransduction alone is sufficient to turn on angiocrine signals. When the endothelial cells are mechanically stretched, angiocrine signals trigger in vitro proliferation and survival of primary human hepatocytes. Our findings uncover a signalling pathway in vascular endothelial cells that translates blood perfusion and mechanotransduction into organ growth and maintenance.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Source Data for all quantifications are provided. Supplementary Video 1 is available at https://figshare.com/s/7f48df3583bff7ce0ea1; Supplementary Video 2 is available at https://figshare.com/s/361947a6f67b8b925042; and Supplementary Video 3 is available at https://figshare.com/s/f7ceb9d5980d10084ad8. Full scans of western blots are provided in Supplementary Fig. 1.
References
Cleaver, O. & Melton, D. A. Endothelial signaling during development. Nat. Med. 9, 661–668 (2003).
Rafii, S., Butler, J. M. & Ding, B. S. Angiocrine functions of organ-specific endothelial cells. Nature 529, 316–325 (2016).
Kostallari, E. & Shah, V. H. Angiocrine signaling in the hepatic sinusoids in health and disease. Am. J. Physiol. Gastrointest. Liver Physiol. 311, G246–G251 (2016).
Augustin, H. G. & Koh, G. Y. Organotypic vasculature: from descriptive heterogeneity to functional pathophysiology. Science 357, eaal2379 (2017).
Zaret, K. S. & Grompe, M. Generation and regeneration of cells of the liver and pancreas. Science 322, 1490–1494 (2008).
Si-Tayeb, K., Lemaigre, F. P. & Duncan, S. A. Organogenesis and development of the liver. Dev. Cell 18, 175–189 (2010).
Mouta Carreira, C. et al. LYVE-1 is not restricted to the lymph vessels: expression in normal liver blood sinusoids and down-regulation in human liver cancer and cirrhosis. Cancer Res. 61, 8079–8084 (2001).
Ding, B. S. et al. Inductive angiocrine signals from sinusoidal endothelium are required for liver regeneration. Nature 468, 310–315 (2010).
Planas-Paz, L. et al. Mechanoinduction of lymph vessel expansion. EMBO J. 31, 788–804 (2012).
Zhang, X., Groopman, J. E. & Wang, J. F. Extracellular matrix regulates endothelial functions through interaction of VEGFR-3 and integrin α5β1. J. Cell. Physiol. 202, 205–214 (2005).
Galvagni, F. et al. Endothelial cell adhesion to the extracellular matrix induces c-Src-dependent VEGFR-3 phosphorylation without the activation of the receptor intrinsic kinase activity. Circ. Res. 106, 1839–1848 (2010).
Schmidt, C. et al. Scatter factor/hepatocyte growth factor is essential for liver development. Nature 373, 699–702 (1995).
LeCouter, J. et al. Angiogenesis-independent endothelial protection of liver: role of VEGFR-1. Science 299, 890–893 (2003).
Wang, L. et al. Liver sinusoidal endothelial cell progenitor cells promote liver regeneration in rats. J. Clin. Invest. 122, 1567–1573 (2012).
Ingber, D. Integrins as mechanochemical transducers. Curr. Opin. Cell Biol. 3, 841–848 (1991).
Benedito, R. et al. The Notch ligands Dll4 and Jagged1 have opposing effects on angiogenesis. Cell 137, 1124–1135 (2009).
Licht, A. H., Raab, S., Hofmann, U. & Breier, G. Endothelium-specific Cre recombinase activity in flk-1-Cre transgenic mice. Dev. Dyn. 229, 312–318 (2004).
Potocnik, A. J., Brakebusch, C. & Fässler, R. Fetal and adult hematopoietic stem cells require β1 integrin function for colonizing fetal liver, spleen, and bone marrow. Immunity 12, 653–663 (2000).
Forbes, S. J. & Newsome, P. N. Liver regeneration – mechanisms and models to clinical application. Nat. Rev. Gastroenterol. Hepatol. 13, 473–485 (2016).
Borowiak, M. et al. Met provides essential signals for liver regeneration. Proc. Natl Acad. Sci. USA 101, 10608–10613 (2004).
Huh, C. G. et al. Hepatocyte growth factor/c-met signaling pathway is required for efficient liver regeneration and repair. Proc. Natl Acad. Sci. USA 101, 4477–4482 (2004).
Michalopoulos, G. K. Liver regeneration after partial hepatectomy: critical analysis of mechanistic dilemmas. Am. J. Pathol. 176, 2–13 (2010).
Böhm, F., Köhler, U. A., Speicher, T. & Werner, S. Regulation of liver regeneration by growth factors and cytokines. EMBO Mol. Med. 2, 294–305 (2010).
Mohammed, F. F. et al. Metalloproteinase inhibitor TIMP-1 affects hepatocyte cell cycle via HGF activation in murine liver regeneration. Hepatology 41, 857–867 (2005).
Kim, T. H., Mars, W. M., Stolz, D. B. & Michalopoulos, G. K. Expression and activation of pro-MMP-2 and pro-MMP-9 during rat liver regeneration. Hepatology 31, 75–82 (2000).
Zhou, B. et al. Matrix metalloproteinases-9 deficiency impairs liver regeneration through epidermal growth factor receptor signaling in partial hepatectomy mice. J. Surg. Res. 197, 201–209 (2015).
Olle, E. W. et al. Matrix metalloproteinase-9 is an important factor in hepatic regeneration after partial hepatectomy in mice. Hepatology 44, 540–549 (2006).
Wayner, E. A., Gil, S. G., Murphy, G. F., Wilke, M. S. & Carter, W. G. Epiligrin, a component of epithelial basement membranes, is an adhesive ligand for alpha 3 beta 1 positive T lymphocytes. J. Cell Biol. 121, 1141–1152 (1993).
Szendroedi, J. et al. Cohort profile: the German Diabetes Study (GDS). Cardiovasc. Diabetol. 15, 59 (2016).
Manavski, Y. et al. Endothelial transcription factor KLF2 negatively regulates liver regeneration via induction of activin A. Proc. Natl Acad. Sci. USA 114, 3993–3998 (2017).
Ventura, A. et al. Restoration of p53 function leads to tumour regression in vivo. Nature 445, 661–665 (2007).
Haiko, P. et al. Deletion of vascular endothelial growth factor C (VEGF-C) and VEGF-D is not equivalent to VEGF receptor 3 deletion in mouse embryos. Mol. Cell. Biol. 28, 4843–4850 (2008).
Zeeb, M. et al. Pharmacological manipulation of blood and lymphatic vascularization in ex vivo-cultured mouse embryos. Nat. Protocols 7, 1970–1982 (2012).
Sies, H. The use of perfusion of liver and other organs for the study of microsomal electron-transport and cytochrome P-450 systems. Methods Enzymol. 52, 48–59 (1978).
Mitchell, C. & Willenbring, H. A reproducible and well-tolerated method for 2/3 partial hepatectomy in mice. Nat. Protocols 3, 1167–1170 (2008).
Schmittgen, T. D. & Livak, K. J. Analyzing real-time PCR data by the comparative C T method. Nat. Protocols 3, 1101–1108 (2008).
Livingstone, R. S. et al. Initial clinical application of modified Dixon with flexible echo times: hepatic and pancreatic fat assessments in comparison with 1H MRS. MAGMA 27, 397–405 (2014).
Häussinger, D. in Metabolism of Human Diseases (eds Lammert, E. & Zeeb, M.) 173–180 (Springer, Vienna, 2014).
Acknowledgements
This work was supported by Deutsche Forschungsgemeinschaft (DFG) through the Collaborative Research Centres SFB 974 (‘Communication and Systems Relevance during Liver Damage and Regeneration’), SFB 1116 (‘Master switches in cardiac ischemia’), IRTG 1902 (‘Intra- and interorgan communication of the cardiovascular system’) as well as DFG LA1216/6-1 (‘Investigation of the role of vascular endothelium in blood glucose metabolism’), the German Center for Diabetes Research (DZD e.V.), the Federal Ministry of Health, the Ministry of Culture and Science of North Rhine-Westphalia, the Academy of Finland, the Novo Nordisk Foundation and Helsinki Institute of Life Science (HiLIFE). We are also grateful to Y. Koh for schematic illustrations, M. Astrachan and his team (XVIVO scientific animation) for the animation, L. S. Hilger for establishing VEGFR3 western blots, S. Jakob, B. Bartosinska, A. Köster and T. Zobel (Centre for Advanced Imaging) for technical support as well as B.-F. Belgardt, C. Bridges, M. Kelly-Goss and M. Gearing for critical reading of the manuscript.
Reviewer information
Nature thanks G. Michalopoulos, E. Tzima and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
L.L. and J.A. performed the majority of the experiments; J.A. performed most experiments on mouse embryos—many of which were repeated and/or finalized by L.L., C.H. and S.U.—and contributed to correlation analyses; L.L. performed the experiments on human cells and the analyses of ex vivo perfused livers; T.B. performed the first embryonic experiments, planned and designed the first ex vivo liver perfusion experiments along with N.E. and performed partial hepatectomy experiments; C.H. contributed to manipulation of mouse embryos and whole embryo culture. S.F. and H.N. were supervised by K.A., and provided VEGFR3 knockout embryos, genotyping and knockout efficiencies. D.H. planned, designed and supervised the ex vivo liver perfusion experiments performed by N.E. R.H. and J.S. performed and analysed the contrast-enhanced ultrasound measurements. K.M., K.B. and M.R. recruited, screened and phenotyped human individuals within the German Diabetes Study of which M.R. is the principal investigator; J.-H.H. and K.B. performed the magnetic resonance imaging and 1H magnetic resonance spectroscopy analyses along with M.R.; and O.K. performed the correlation analyses and adjustments. D.E. isolated hepatic ECs from mouse embryos and was involved in immunohistochemical analyses of mouse embryos, discussions, data management and statistical evaluations. E.L. supervised and guided J.A., T.B., C.H., S.U. and L.L. during their experiments and wrote the manuscript with the help from J.A. and L.L. All authors read and contributed to the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Hepatic cell proliferation in perfused liver periphery versus liver centre at E11.5.
a, b, c, Transversal section through a liver after injection of EdU into its lobes and WEC for 3 h. Magnified fields of view in the liver centre (a′, b′, c′) and the liver periphery (a′′, b′′, c′′), as indicated in c by white boxes. d, d′, e, e′, Transversal sections through E11.5 livers showing the liver centre (d, d′) and liver periphery (e, e′). f, Quantification of the Ki-67+ area in the liver centre versus liver periphery (n = 3 livers, *P = 0.0142 (119.40; 376.70)). Scale bars, 100 μm (a, b, c) and 50 μm (all other panels). Two-tailed paired Student’s t-test, 95% confidence interval (lower confidence limit; upper confidence limit).
Extended Data Fig. 2 Activation of β1 integrin, VEGFR3 and total β1 integrin expression in ECs of E11.5 livers.
a, a′, b, b′, Transversal sections through livers with quantified areas of activated endothelial β1 integrin (for quantification, see Fig. 1m). c, c′, d, d′, Transversal sections with VEGFR3 tyrosine phosphorylation indicated by PLA (red dots and white arrowheads; for quantification, see Fig. 1n). e, e′, f, f′, g, Transversal sections with total β1 integrin (e, f) and quantified area of total endothelial β1 integrin (e′, f′) in liver centre versus liver periphery (g, n = 4 livers, P = 0.316 (−44.98; 99.49)). Scale bars, 25 μm. Two-tailed paired Student’s t-tests, 95% confidence interval (lower confidence limit; upper confidence limit).
Extended Data Fig. 3 Endothelial β1 integrin is required for hepatic cell proliferation and survival in the liver periphery.
a, a′, b, b′, d, d′, e, e′, Transversal sections through E11.5 livers from EC-specific heterozygous (control) versus homozygous (β1 integrin-null) knockouts of β1 integrin, including magnifications (a′, b′, d′, e′) after 3 h EdU incorporation in WEC. c, Quantification of EdU-positive peripheral areas: n = 4 control versus n = 3 β1 integrin-null livers (*P = 0.0207 (19.06; 93.54)). f, Ki-67-positive cells in embryonic livers: n = 6 Cre-control versus n = 6 β1 integrin-null livers (P = 0.0666 (3.71; −86.50)). g, g′, g′′, h, h′, h′′, i, i′, i′′, Transversal sections through control (g, g′, g′′) and β1 integrin-null (h, h′, h′′) E13.5 livers with vascular-painted (RCA), perfused hepatic vessels, including magnified images (i, i′, i′′). Scale bars, 100 μm (a, b, d, e), 50 μm (a′, b′, d′, e′, g, g′, g′′, h, h′, h′′) and 25 μm (i, i′, i′′). Data are mean ± s.e.m. Two-tailed unpaired Student’s t-tests, 95% confidence interval (lower confidence limit; upper confidence limit).
Extended Data Fig. 4 Cdh5-creERT2-mediated depletion of β1 integrin reduces liver growth, survival, VEGFR3 activation and HGF production.
a, a′, b, b′, E13.5 mouse embryos (a′, b′), with their abdomen magnified (a, b). c, Calculated volumes of livers taken from mouse embryos with Cdh5-creERT2-mediated depletion of β1 integrin (labelled ‘Cdh5-β1−/−’) and control littermates (n = 3 embryos each, *P = 0.0446 (0.15; 5.03)). d, d′, e, e′, Transversal sections through E13.5 livers with magnifications (d′, e′). f, Quantification of cleaved caspase-3-positive areas (n = 3 embryos each, *P = 0.0462 (12.62; 634.90)). g, VEGFR3 tyrosine phosphorylation normalized to total VEGFR3 (n = 4 control embryos versus n = 4 embryos with Cdh5-creERT2-mediated depletion of β1 integrin, *P = 0.0445 (1.81; 91.90)). h, HGF protein concentrations normalized to total protein (n = 16 control embryos versus n = 9 embryos with Cdh5-creERT2-mediated depletion of β1 integrin, *P = 0.0030 (8.81; 36.46)). Scale bars, 500 μm (a, b), 1 mm (a′, b′), 200 μm (d, e) and 50 μm (d′, e′). Data are mean ± s.e.m. Two-tailed unpaired Student’s t-tests, 95% confidence interval (lower confidence limit; upper confidence limit).
Extended Data Fig. 5 β1 integrin acts upstream of perfusion-dependent activation of VEGFR3, which is required for liver growth and survival.
a, b, VEGFR3 tyrosine phosphorylation, normalized to total VEGFR3 protein, in liver lysates from E12.5 embryos with heterozygous depletion of endothelial β1 integrin (control) (a) and E12.5 endothelial β1 integrin-null embryos (b) in gain-of-perfusion experiments. n = 5 control embryos treated with control solution versus n = 9 control embryos treated with epinephrine and atropine (*P = 0.0072 (66.37;323.00)), and n = 7 control embryos treated with control solution versus n = 7 epinephrine- and atropine-treated endothelial β1 integrin-null embryos, (P = 0.3853 (−97.93; 40.64)). c, c′, d, d′, Abdominal region of an E13.5 control littermate (c′) and an E13.5 Vegfr3−/− mouse embryo (d′). e, Calculated liver volumes (n = 5 control embryos versus n = 4 Vegfr3−/− embryos, *P = 0.0462 (0.05; 3.86). f, f′, g, g′, Transversal sections through E13.5 livers with magnifications (f′, g′). h, Quantification of cleaved caspase-3-positive areas: n = 5 control livers versus n = 4 Vegfr3−/− livers (*P = 0.0103 (156.50; 591.60)). i, HGF protein concentrations in lysates from embryonic livers normalized to total protein: n = 7 control embryos versus n = 10 Vegfr3−/− embryos (*P = 0.0022 (15.55; 54.81)). Scale bars, 500 μm (c, d, f, g), 1 mm (c′, d′) and 50 μm (f′, g′). Data are mean ± s.e.m. Two-tailed unpaired Student’s t-tests, 95% confidence interval (lower confidence limit; upper confidence limit).
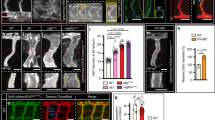
Extended Data Fig. 6 Vessel dilation, blood volume and blood flow velocity in liver sinusoids after a two-thirds partial hepatectomy.
a–c, Sections through livers isolated from adult mice after sham operation (a) versus partial hepatectomy (PHx) (b), and quantification (c) of vessel dilation by measurement of vascular lumen areas (n = 3 livers each, P = 0.0622 (−12.43; 280.20)). d, Contrast-enhanced ultrasound measurements on the liver of a mouse. e, f, Relative blood volume (calculated from peak enhancement of contrast agent, n = 10 mice, *P = 0.0014 (51.98; 154.40)) (e) and relative blood flow velocity (calculated from time to peak of contrast agent enhancement, n = 10 mice, P = 0.2264 (−25.41; 93.88)) (f) in the right liver lobe of adult mice normalized to the liver area before (‘Pre OP’) and after partial hepatectomy. Data in c are mean ± s.e.m. Scale bar, 20 μm. Student’s t-tests were two-tailed unpaired (c) or two-tailed paired (e, f), 95% confidence interval (lower confidence limit; upper confidence limit).
Extended Data Fig. 7 Mechanically induced release of angiocrine signals.
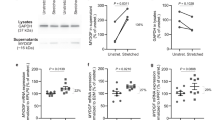
a, a′, b, b′, c, c′, d, d′, e, e′, f, f′, h, h′, i, i′, Unstretched versus mechanically stretched human hepatic ECs, stained for F-actin (a′, b′, c′, d′, e′, f′, h′, i′), activated β1 integrin (a, a′,b, b′) and shown as PLA dots: VEGFR3 tyrosine phosphorylation (c, c′, d, d′), co-localization of β1 integrin and VEGFR3 (e, e′, f, f′) and VEGFR2 tyrosine phosphorylation (h, h′,i, i′). g, j, Quantification of the interaction between VEGFR3 and β1 integrin (n = 3 unstretched chambers versus n = 4 stretched chambers, *P = 0.0091 (0.99; 4.16)), and VEGFR2 tyrosine phosphorylation (n = 4 stretch chambers each, P = 0.4000 (−0.37; 0.19)). k, IL-6 protein concentration (n = 4 stretch chambers each, *P = 0.0191 (14.51; 109.70)). l, TNF protein concentration (n = 4 unstretched chambers versus n = 5 stretched chambers, *P = 0.0493 (0.43; 192.40)). m, MMP9 activity (n = 4 stretch chambers each, *P = 0.0488 (0.44; 111.20)), in the supernatant of unstretched versus stretched hepatic ECs. Scale bars, 10 μm. Data are mean ± s.e.m. Two-tailed unpaired Student’s t-tests, 95% confidence interval (lower confidence limit; upper confidence limit).
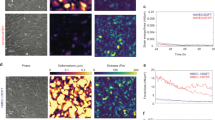
Extended Data Fig. 8 Mechanically induced angiocrine signals promote proliferation and survival of human hepatocytes.
a–d, Primary human hepatocytes treated for 6 h with supernatant from unstretched (a, c) versus stretched (b, d) human hepatic ECs. e–g, Unstretched ECs without (e) or with (f) activating β1 integrin antibody, including quantification (g) of β1 integrin activation (n = 4 wells each, *P = 0.0062 (73.97; 276.10)). h, i, Primary human hepatocytes treated for 6 h with supernatant from unstretched hepatic ECs treated without (h) or with (i) an activating β1 integrin antibody. j, Quantification of hepatocyte proliferation after incubation with supernatant from ECs without or with activating β1 integrin antibody (n = 4 wells each, *P = 0.0110 (91.40; 698.60)), with activating β1 integrin antibody alone without previous incubation with ECs (n = 4 wells, *P = 0.0299 (31.15; 638.40)) and with conditioned medium of ECs treated with an IgG1 isotype control (n = 3 wells, *P = 0.0436 (8.99; 664.80)). Scale bars, 50 μm (a, b, h, i) and 10 μm (c, d, e, f). Data are mean ± s.e.m. Two-tailed unpaired Student’s t-tests (g) and one-way ANOVA followed by Tukey’s test (j), 95% confidence interval (lower confidence limit; upper confidence limit).
Extended Data Fig. 9 Correlation of blood pressure and liver volume in metabolically healthy human individuals.
a, Baseline characteristics (mean ± standard deviation, minimum and maximum values) of the study cohort, which was comprised of individuals recruited from glucose-tolerant humans who served as controls in the prospective observational German Diabetes Study29. b, c, Correlation of blood pressure with liver volume as assessed by MRI taken between 07:00 and 10:30. Correlation of systolic blood pressure with liver volume (n = 42; r = 0.45; *P = 0.0022 (0.17; 0.66)), and correlation of diastolic blood pressure with liver volume (n = 42; r = 0.39; *P = 0.0104 (0.09; 0.62)). Reported correlations are Pearson correlation coefficients with 95% confidence intervals (lower confidence limit; upper confidence limit).
Extended Data Fig. 10 Model of mechanotransduced angiocrine signals in the liver.
Simplified drawings of liver sinusoids with liver sinusoidal ECs (LSECs), hepatic stellate cells (HSCs), extracellular matrix (ECM), Kupffer cells (KCs) and hepatocytes (with schematic elements taken from a previous publication38). a, Liver sinusoid under normal blood flow. b, Magnification of space of Disse. c, Liver sinusoid with enhanced blood perfusion. d, Magnification of space of Disse. When the vascular lumen widens owing to enhanced blood perfusion, circumferential stretching of liver sinusoidal ECs activates endothelial β1 integrin and its interaction with VEGFR3. The hepatic ECs (in concert with other cells such HSCs) subsequently release angiocrine signals—such as HGF, IL-6 and TNF—and activate MMP9, and thus enhance proliferation and survival of the adjacent hepatocytes.
Supplementary information
Supplementary Figure
This file contains Supplementary Figure 1: Western blot scans with size marker indications. Western blot bands in black-rimmed boxes were used for quantification. Molecular weights are in kilodalton (kDa).
Source data
Rights and permissions
About this article
Cite this article
Lorenz, L., Axnick, J., Buschmann, T. et al. Mechanosensing by β1 integrin induces angiocrine signals for liver growth and survival. Nature 562, 128–132 (2018). https://doi.org/10.1038/s41586-018-0522-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0522-3
Keywords
This article is cited by
-
Identification of myeloid-derived growth factor as a mechanically-induced, growth-promoting angiocrine signal for human hepatocytes
Nature Communications (2024)
-
Fetal liver development and implications for liver disease pathogenesis
Nature Reviews Gastroenterology & Hepatology (2023)
-
Liver endothelial cells in NAFLD and transition to NASH and HCC
Cellular and Molecular Life Sciences (2023)
-
Emerging roles of mechanosensitive ion channels in acute lung injury/acute respiratory distress syndrome
Respiratory Research (2022)
-
Sinusoidal and lymphatic vessel growth is controlled by reciprocal VEGF-C–CDH5 inhibition
Nature Cardiovascular Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.