Abstract
Current models of somatosensory perception emphasize transmission from primary sensory neurons to the spinal cord and on to the brain1,2,3,4. Mental influence on perception is largely assumed to occur locally within the brain. Here we investigate whether sensory inflow through the spinal cord undergoes direct top-down control by the cortex. Although the corticospinal tract (CST) is traditionally viewed as a primary motor pathway5, a subset of corticospinal neurons (CSNs) originating in the primary and secondary somatosensory cortex directly innervate the spinal dorsal horn via CST axons. Either reduction in somatosensory CSN activity or transection of the CST in mice selectively impairs behavioural responses to light touch without altering responses to noxious stimuli. Moreover, such CSN manipulation greatly attenuates tactile allodynia in a model of peripheral neuropathic pain. Tactile stimulation activates somatosensory CSNs, and their corticospinal projections facilitate light-touch-evoked activity of cholecystokinin interneurons in the deep dorsal horn. This touch-driven feed-forward spinal–cortical–spinal sensitization loop is important for the recruitment of spinal nociceptive neurons under tactile allodynia. These results reveal direct cortical modulation of normal and pathological tactile sensory processing in the spinal cord and open up opportunities for new treatments for neuropathic pain.
Similar content being viewed by others
Main
To investigate whether descending corticospinal projections regulate sensory processing in the spinal cord, we performed a bilateral pyramidotomy6,7 to lesion the CST in the brainstem of adult mice (Fig. 1a, b) and tested them for sensitivity to noxious and innocuous sensory stimuli (Fig. 1c–i), with histological verification (Extended Data Fig. 1a). Bilateral CST lesions caused a marked deficit in behavioural responses to dynamic and static light touch, as indicated by a reduced response to stroking with a soft paintbrush (Fig. 1h) and increased latency to detect tape on the hindpaw plantar surface (Fig. 1i). Similarly, in the von Frey test, mice with CST lesions exhibited reduced responses to innocuous low pressure punctate stimuli8 (0.16 g) but had normal sensitivity to higher mechanical forces (0.6–2 g), which activate nociceptors9 (Fig. 1g). The hindpaw withdrawal responses to noxious thermal and mechanical stimuli were intact in mice with a pyramidotomy (Fig. 1c–f), as was gross motor performance (Extended Data Fig. 1b, c). The selective deficits in light touch sensitivity (Extended Data Fig. 1a) corroborate the finding that CST axons co-terminate with low-threshold mechanosensory afferents in laminae III–V10,11 (Extended Data Fig. 2a, b).
a, Schematic of experimental paradigm. Py, pyramidotomy. b, Left, representative images of transverse sections of the brain stem at pyramids and dorsal spinal cord (L3) in sham (n = 8) or pyramidotomized (n = 12) mice used for sensory tests stained with anti-PKCΥ. Arrowheads and dashed lines indicate the location of the main CST. Scale bar, 500 μm. Right, quantification of PKCΥ immunofluorescence intensity (normalized to control) in the lumbar dorsal funiculus of sham or pyramidotomized mice. P < 0.0001. c–i, Measurement of thermal and mechanical sensitivities by hot plate (c, P = 0.59 and 0.35 for flinch and lick, respectively), laser heat (d, P = 0.50), acetone (e, P = 0.73), pinprick (f, P = 0.58), von Frey filaments (vF; g, P = 0.016 for 0.16 g), dynamic light brush (h, P = 0.001), and tape removal (i, P = 0.0003) in sham (n = 8) or pyramidotomized mice (n = 12). NS, not significant. b, c–f, h, i, Two-sided t-test; g, two-way repeated measures ANOVA followed by Bonferroni correction. Data shown as mean ± s.e.m.
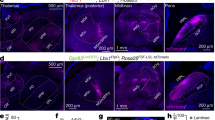
To determine which areas of the cerebral cortex12 contain CSNs that may be responsible for the sensory modulation, we used anterograde tracing to identify innervation of the dorsal horn of the spinal cord. CST axons from hindlimb primary sensory (S1) cerebral cortex, with a minor contribution from secondary sensory cortex (S2), terminate in laminae III–V (Fig. 2a, b), suggesting that they have a sensory role, whereas the hindlimb motor cortex projects more ventrally, to laminae VI–VII (Fig. 2a, b), consistent with its known motor functions. Thus, CST axons from S1 and S2 are more likely to execute control over tactile sensory processing in the spinal cord. To test this further, we used intersectional targeting12,13,14 to express human diphtheria toxin receptor (DTR) in S1/S2 CSNs, enabling selective ablation of these neurons. High-efficiency retrograde lentiviral vectors (HiRet) expressing Cre were injected into the lumbar spinal cord (thoracic (T)13–lumbar (L)6) of mice at postnatal day (P)14, a time when cortical pruning is complete15,16, followed by an injection of AAV-FLEX-DTR into S1/S2 (Fig. 2c). Administration of diphtheria toxin to these mice ablated about 85% of CSNs in S1/S2 (Extended Data Fig. 3), and mice with such selective ablation fully phenocopied the results of pyramidotomy (Fig. 2d–g), indicating that the CST axons from S1/S2 cortex contribute to tactile sensitivity.
a, Representative images showing projection patterns of CST axons originating from indicated cortical areas. AP, anteroposterior; ML, mediolateral. Scale bar, 500 μm. b, Quantification of relative CST termination percentile within different laminae from indicated cortical areas (n = 3). c, Schematic of regional CSN ablation where HiRet viruses were injected into the lumbar spinal cord (T13–L6) at P14. DT, diphtheria toxin; SNI, spared nerve injury. d–g, Measurements of sensitivity to laser heat (d, P = 0.40), pinprick (e, P = 0.28), von Frey filaments (f, P = 0.004 for 0.16g) and brush stimuli (g, P < 0.0001) in control mice (n = 7) and mice in which hindlimb somatosensory CSNs have been ablated (n = 8). h, i, Measurement of punctate and dynamic mechanical allodynia after SNI in control mice (n = 7) and mice in which hindlimb somatosensory CSNs have been ablated (n = 8). j, Schematic of silencing of S1/S2 CSNs by injection of HiRet-Cre into the lumbar spinal cord at P14. PSEM, pharmacologically selective effector molecule (a specific ligand to activate engineered GlyR expressed in the hindlimb S1/S2). k, l, Measurement of punctate and dynamic allodynia after SNI with PSEM administration in mice injected with AAV-FLEX-GlyR (n = 7) or AAV-GFP (n = 8). P = 0.0001 and 0.0006 for 14 and 21 d, respectively, in i; P = 0.002 and 0.016 for 20 and 40 min, respectively, in k; elsewhere, **P < 0.0001. d, e, g, Two-sided t-tests; f, h, i, k, l, two-way repeated measures ANOVA followed by Bonferroni correction. Data shown as mean ± s.e.m.
A prominent feature of peripheral neuropathic pain is mechanical allodynia, in which light touch triggers pain3,4. To assess whether the CST modulates mechanical allodynia, we subjected mice with ablation of S1/S2 CSNs to the spared nerve injury (SNI) model of neuropathic pain17. Mice in which S1/S2 CSNs had been ablated exhibited a profound reduction in both the punctate allodynia evoked by low-intensity static von Frey stimuli and in dynamic allodynia evoked by brushes at all time points tested (1 to 21 days after SNI) (Fig. 2h, i). Mice with a pyramidotomy also failed to develop mechanical allodynia induced by SNI (Extended Data Fig. 4a–c) or by intraplantar injection of complete Freund’s adjuvant (CFA), a model of peripheral inflammatory pain (Extended Data Fig. 4f, g). However, the CST-lesioned mice still exhibited cold allodynia and hyperalgesia in response to intense mechanical stimuli after SNI (Extended Data Fig. 4d, e). We conclude that CSNs from S1/S2 are required for the induction of mechanical allodynia but not other aspects of the pain hypersensitivity phenotype. In support of this, transiently silencing CSNs in the hindlimb S1/S2 cortex using an inducible chemogenetic method18 diminished established mechanical allodynia in mice with SNI (Fig. 2j–l). These results show that somatosensory CSNs projecting to the deep dorsal horn contribute to both innocuous tactile processing in normal conditions and the generation of mechanical allodynia in pathological states, something not suspected before.
To assess how CSNs in S1 respond to peripheral tactile stimulation, we used in vivo miniaturized microscopy12,19 to monitor calcium levels in CSNs expressing GCaMP6s, a genetically encoded fluorescent calcium indicator (Fig. 3a, Extended Data Fig. 5). S1 CSNs in intact freely moving mice showed sparse and temporally scattered activity (Fig. 3b, c). By contrast, brush or von Frey stimulation, but not laser heat, to the hindpaw elicited calcium activity in a subset of S1 CSNs, and such activation was significantly increased after SNI in the same mice (Fig. 3b, c). In addition, the proportion of cells that responded to both brush and von Frey stimulation was increased after SNI (Extended Data Fig. 5). Thus, CSNs in the somatosensory cortex of intact mice are activated by innocuous tactile stimuli and this is amplified under neuropathic pain conditions, suggesting a spinal–cortical–spinal sensitization loop.
a, Diagram of calcium activity imaging in S1 CSNs with a head-mounted miniaturized microscope. b, Trial-averaged activity of CSNs in response to indicated sensory stimuli before and after SNI. Active calcium event traces are aligned to the time when the brush (pre-SNI, n = 156 neurons from 4 mice; post-SNI, n = 103 neurons from 4 mice), von Frey filament (pre-SNI, n = 151 neurons from 4 mice; post-SNI, n = 94 neurons from 4 mice), or laser (pre-SNI, n = 156 neurons from 4 mice; post-SNI, n = 116 neurons from 4 mice) affected the hindpaw (green arrowheads). Trial averages (13–17 trials per mouse) were sorted by peak activity time. Arrowheads indicate onset of stimulation. c, Average response to brush, von Frey filament (0.04 g) and laser heat stimuli before and after SNI. Number of neurons as in b. *P = 0.03 and ***P = 0.0002 for brush and von Frey, respectively; rank sum test. d–g, Representative images (d, e) and quantification (f, g) of c-Fos and pERK (red) or NK1R (green) immunoreactivity in the dorsal horn of the spinal cord (L3–4) in mice under the following conditions: naive control (n = 3), brush only (n = 3), SNI only (n = 3), SNI and brush (n = 4), SNI and brush with pyramidotomy (n = 3). Scale bars, 500 and 100 μm in d, e, respectively. Arrowheads indicate co-localization of pERK and NK1R. f, g, One-way ANOVA with Bonferroni correction. P = 0.0008 for SNI + brush with or without pyramidotomy in f; otherwise, **P < 0.0001. Data shown as mean ± s.e.m.
We next examined whether descending CST axons modulate the neuronal activation evoked by low-intensity mechanical stimulation in the spinal cord. We detected few c-Fos+ cells in the dorsal horn of intact mice after brush stimulation or in mice with SNI without brush stimulation (Fig. 3d). However, brush stimulation in mice with SNI increased the number of c-Fos+ cells in all laminae (I–V) of the ipsilateral dorsal spinal cord (Fig. 3d, f). A CST lesion reduced this effect in both deep laminae (III–V) and superficial laminae (I/II), a region that normally processes only inputs from nociceptors and which is not contacted directly by CST (Fig. 3d, f). These results suggest that CST inputs contribute to the level of brush-evoked neuronal activation in all laminae of the dorsal horn. Neurons expressing the neurokinin 1 receptor NK1R in lamina I of the spinal cord are a major nociceptive output pathway to the brain20,21. To assess whether CSNs modulate the patterns of activation of these neurons, we monitored phospho-ERK (pERK) immunoreactivity as an activity marker22. Tactile stimulation in mice with SNI, but not in intact mice, induced a significant, CST-dependent increase in the number of pERK–NK1R+ neurons in lamina I (Fig. 3e, g). Tactile stimulation after SNI in the presence of an intact CST directly activates neurons in laminae III–V and indirectly increases activation of lamina I projection neurons. The latter effect is likely to result from disinhibition of polysynaptic circuits that connect laminae III–V with lamina I4,20,23. CST ablation blocked the mechanical allodynia caused by spinal disinhibition produced pharmacologically by inhibiting GABA (γ-aminobutyric acid) and glycine activity21,23 (Extended Data Fig. 6). These data indicate that inputs from the corticospinal pathway to their direct targets in laminae III–V are required for low-threshold afferent inputs to activate ascending nociceptive pathways after SNI. Consistent with this conclusion, the activation of multiple brain areas, including CSNs in S1/S2, produced by tactile stimulation after SNI is blocked by CST ablation (Extended Data Fig. 7).
We next investigated which neurons in the dorsal horn are targeted by the S1/S2 CST. Excitatory cholecystokinin (CCK)iresCre-labelled cells in the deep dorsal horn are co-innervated by the CST and Aβ primary afferent fibres, and selective silencing of CCK+ neurons results in tactile defects11, making it likely that CCK neurons are CST targets. Indeed, selective ablation of CCK+ interneurons in the lumbar spinal cord (T13–L6) phenocopied CST lesions (Figs. 1, 2), producing selective defects in tactile sensitivity in intact mice and a reduction in mechanical allodynia after SNI (Fig. 4a–d, Extended Data Fig. 8a–f). Ablation of CCK+ interneurons also significantly reduced the number of c-Fos+ cells after brush stimulation in all dorsal horn laminae in mice with SNI (two-sided t-test, P = 0.0045 and 0.0028 for laminae I–II and laminae III–V, respectively, Extended Data Fig. 8g, i), but to a lesser extent than pyramidotomy (Fig. 3d, f), suggesting that other neurons co-innervated by CST and Aβ fibres also contribute to this process11. Nevertheless, these results show that CCK+ interneurons in deep laminae of the dorsal horn are important contributors to the facilitatory effects of the CST on tactile sensitivity and mechanical allodynia.
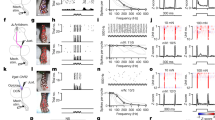
a, b, Representative images (a) of cervical and lumbar spinal cord dorsal horn in control mice (AAV-PLAP, n = 5) or mice in which CCK–tdTomato neurons are ablated (AAV-FLEX-DTR, n = 7) with quantification (b). P = 0.35 and P < 0.0001 for cervical and lumbar, respectively; two-sided t-test. c, d, Measurement of punctate (c) and dynamic (d) mechanical allodynia after SNI in control mice (n = 8) or mice in which CCK–tdTomato interneurons have been ablated (n = 7). Two-way repeated measures ANOVA followed by Bonferroni correction. P = 0.002, 0.01, 0.03, 0.03, 0.04 and P < 0.0001, P = 0.0003, 0.0003, 0.0009, 0.0014 for 1, 3, 7, 14, 21 d in c and d, respectively. e, f, Averaged traces of EPSCs evoked by Aβ, CST, or co-stimulation (−70 mV, voltage clamp, left) and representative trace of EPSP or action potential (AP; current clamp, right) in untreated CCK–tdTomato neurons (e), or neurons treated with bicuculline and strychnine (f). Bar graphs under EPSC curves: ratio of EPSC amplitude evoked by Aβ and CST co-stimulation over the sum of amplitudes evoked by individual stimulation (e, n = 8, P = 0.02; f, n = 6, P = 0.04), one-sided paired t-test. Data shown as mean ± s.e.m.
To test whether CCK+ interneurons are directly activated by the CST and Aβ fibres, we performed whole-cell patch recordings in ex vivo spinal cord slices21,24,25 with attached dorsal roots prepared from CCK–tdTomato mice26 in which S1/S2-derived CST axons were labelled with channelrhodopsin2 and yellow fluorescent protein (ChR2–YFP) (Extended Data Fig. 9a). Among all recorded CCK–tdTomato cells, 50% exhibited excitatory monosynaptic responses to both Aβ and CST stimulation (Extended Data Fig. 9b, c), but in only a few was single stimulation of either input sufficient to evoke action potentials (Extended Data Fig. 9b, c). However, in those neurons with direct connections from Aβ fibres and the CST (Extended Data Fig. 9d), co-stimulation of the afferents and descending pathway led to super-additive synaptic responses, with a substantially increased action potential output (Fig. 4e). In the presence of bicuculline (a GABAA receptor inhibitor) and strychnine (a glycine receptor inhibitor), which mimics the spinal disinhibition that occurs in SNI21,23, such additive responses were further enhanced, with action potential trains occurring in all recorded CCK–tdTomato neurons (Fig. 4f). In mice with SNI, half of c-Fos+ cells found in the dorsal horn after repetitive touch stimulation expressed CCK (Extended Data Fig. 8g), and c-Fos immunoreactivity in these cells was abolished by pyramidotomy (Extended Data Fig. 8g, h). Together, these results suggest that CST inputs onto CCK+ interneurons have a crucial sensitizing role in the processing of tactile input in naive mice, and that this role is increased after nerve injury.
Finally, we found that optical stimulation of CSNs in S1 in mice expressing ChR2 targeted to CSNs (Extended Data Fig. 10a) significantly extended the duration of paw flinching or licking after a light brush stimulus (two-sided t-test, P = 0.02, Extended Data Fig. 10b, c), and increased brush-induced pERK in lamina I NK1R neurons (Extended Data Fig. 10d). We conclude that the direct output from the somatosensory cortex to the spinal dorsal horn via the CST mediates powerful facilitation of tactile sensory processing in the spinal cord, which amplifies transmission of tactile-related signalling to the brain. This represents a spino-cortico-spinal feed-forward sensitization loop that is crucial for controlling tactile sensation in normal conditions and allodynia in neuropathic pain states. Normalizing the excitability of somatosensory CSNs could, therefore, be a potential target for treating neuropathic pain by pharmacological or electromagnetic manipulation. Furthermore, by identifying direct cortical control of tactile processing within spinal circuits, these results provide a plausible explanation for how mental states could directly increase or decrease normal and pathological tactile sensations in different contexts or mood states, by controlling activity in transmission pathways from the spinal cord to the brain.
Methods
Mouse strains
All experimental procedures were performed in compliance with animal protocols approved by the Institutional Animal Care and Use Committee at Boston Children’s Hospital or National Institute of Mental Health. C57Bl/6 wild-type, Rosa-LSL-ChR2EYFP (Jax#12569), Rosa-LSL-tdTomato27 and CCKCre (Jax#012706) mouse strains were maintained on a C57Bl/6 genetic background. For each set of behavioural measurements, animals used in experimental and control groups were littermates. The body weight and sexes were randomized and assigned to different treatment groups, and no other specific randomization was used for the animal studies. Behavioural tests were run blinded to the experimental condition. No statistical methods were used to predetermine sample size.
Virus production and intraspinal injection
Lentivirus vectors of HiRet-GFP and HiRet-Cre (all lenti-virus titres were adjusted to 1.6–2 × 1012 copies/ml for injection) were constructed based on the HiRet backbone13. To retrogradely target supraspinal neurons, HiRet lentiviruses were injected into the mouse spinal cord guided by a Vevo 770 ultrasound system (VisualSonics) as described28.
Regional ablation or silencing of corticospinal neurons or CCK+ interneurons
For regional CSN ablation, 2 μl HiRet-Cre viruses (2 × 1012 copies/ml) were intraspinally injected into wild-type mice at P4 (pilot experiment, data not shown) or P14. 1 μl AAV2/8-FLEX-DTR viruses (5 × 1012 copies/ml) or AAV2/8-FLEX-PLAP viruses (5 × 1012 copies/ml) as control were injected to the cortical areas covering ACC/PFC (AP: 0.8–2.5 mm, ML: 0.2–0.8 mm to bregma) or the somatosensory cortex (S1: −0.3 to −1.6 mm, ML: 1.5–2.4 mm to bregma; S2: AP: −1.8 to −2.1 mm, ML: 3.3–4.0 mm to bregma), respectively. To monitor injection position and targeting efficiency, all mice were co-injected with AAV2/8-GFP into the cortex (5 × 1012 copies/ml). Diphtheria toxin (DT, Sigma, D0564, 100 μg/kg) was administered intraperitoneally into adult animals (age 8 weeks). After behavioural and histological assessments were performed blindly, mice with incomplete CST ablation, as defined by more than 25% spared GFP+ axons in the lumbar spinal cord, were excluded from further analysis. In our pilot experiment (P4 injection), we found that DT-mediated CSN ablation in S1/S2, but not PFC/ACC, significantly impaired responses to light touch, whereas the responses to noxious heat or mechanical stimuli were not affected by these manipulations.
To transiently silence CSN activity, 2 μl HiRet-Cre or PLAP viruses (2 × 1012 copies/ml) were injected to the lumbar spinal cord at P14 or 1 μl AAV-2/8-FLEX-PSAML141F-Y115F-GlyR-IRES-GFP viruses (5 × 1012 copies/ml) were injected into the cortical areas covering hindlimb S1/S2 cortical areas at P28. After von Frey and brush measurements (see ‘Behavioural assessment’) after spared nerve ligation (SNI), PSEM308 (Apex Scientific, 10 mg/kg) was administered intraperitoneally. von Frey and brush measurements were performed at 20, 40 and 120 min post PSEM administration. We verified the expression (GFP+) of GlyR in the somatosensory cortex as post hoc histological analysis.
To ablate lumbar spinal CCK+ interneurons, we injected 3 μl AAV2/8-FLEX-DTR viruses (5 × 1012 copies/ml) or AAV2/8-FLEX-PLAP viruses (5 × 1012 copies/ml) as control at P21 into the lumbar spinal cord (T13–L6) of CCK–tdTomato mice. DT (Sigma, D0564, 100 μg/kg) was administered intraperitoneally at P35. Animals were subjected to gross motor and sensory tests 2 weeks after DT injection. For post hoc examination, CCK–tdTomato+ cells were quantified in the cervical and lumbar spinal cord in mice injected with AAV-FLEX-DTR or AAV-FLEX-PLAP, respectively.
Immunohistochemistry and imaging
Mice were perfused transcardially with 4% paraformaldehyde (PFA) in phosphate-buffered saline (PBS) followed by further 4% PFA in PBS post-fixation overnight. The tissue was cryo-protected with 30% sucrose and processed using a cryostat (section thickness 30 μm for spinal cord and 60 μm for brain). Sections were intensively washed with PBS and then treated with a blocking solution containing 10% goat normal serum (GNS), 0.5% Triton-100 for 2 h at room temperature before staining. The primary antibodies used were chicken anti-GFP [Abcam (ab13970), 1:1,000], rabbit anti-RFP [Abcam (ab34771), 1:1,000], rabbit anti-PKCγ [Santa Cruz (sc211), 1:100], rabbit anti-c-fos [Cell Signaling Technology (2250), 1:200], mouse anti-pERK [Cell Signaling Technology (9106), 1:100], and rabbit-anti-NK1R [Sigma-Aldrich (S8305), 1:1,000]. Secondary antibodies including Alexa Fluor 488-conjugated goat anti-chicken, Alexa Fluor 488, 594-conjugated goat anti-rabbit, and the fluorescent IB4 (Alex Fluor 488 conjugated) were purchased from Invitrogen. Floating brain tissue sections (60 μm) were incubated with primary antibodies overnight at 4 °C and washed three times for 10 min with PBS before being incubated with secondary antibodies, or with IB4 (1:400 dilution) for 2 h. After thorough washing with PBS, sections were mounted with ProLong antifade mounting medium with or without DAPI for imaging and analysis. Sections were imaged with a confocal laser-scanning microscope (Zeiss 700), or a wide field Nikon 80i Microscope (air).
Surgery
For pyramidotomy, the procedure is similar to that described previously8,9. In brief, mice were anaesthetized with ketamine and xylazine. An incision was made at the left side of the trachea, followed by blunt dissection to expose the skull base. A craniotomy in the occipital bone was then performed to allow access to the medullary pyramids. The left and right pyramids were cut with a fine scalpel medially up to the basilar artery. The wound was closed in layers with 6.0 sutures. After pyramidotomy, mice were tested for gross motor functions. Most animals showed normal gross motor functions, consistent with previous findings9,12,29,30. At the end point, lumbar spinal cord sections from all mice were subjected to immunostaining with antibodies against PKCγ and the remaining PKCγ immunofluorescence in the dorsal column was quantified. As shown in Extended Data Fig. 1, a few mice with inefficient CST ablation showed neither nociceptive nor tactile sensation deficits. With these criteria, we excluded animals with inefficient CST ablation and/or gross motor defects from all other independent experiments (CFA, c-fos, pERK quantification).
For anterograde labelling of CST axons (Fig. 2a, b), AAV2/1-ChR2-mCherry (1 × 1013 copies/ml for injection, produced by Boston Children’s Hospital, viral core) was injected into mouse sensorimotor cortex at multiple sites as described previously12.
Spared nerve ligation (SNI) surgery was performed under isoflurane (3% for induction /2% for maintenance) on adult mice (8–12 weeks old). In brief, the tibial and common peroneal branches of the sciatic nerve were tightly ligated with a 5.0 silk suture and transected distally, while the sural nerve was left intact15. After injury, the incision was sutured and mice were allowed to recover on heated pads before being returned to their home cages. Punctate and dynamic mechanical allodynia, cold allodynia and mechanical hyperalgesia were measured 1–21 days after SNI. To induce spinal disinhibition, we intrathecally injected 10 μl saline containing bicuculline (0.02 μg, Sigma-Aldrich, 14340) and strychnine (0.05 μg Sigma-Aldrich, S0532) into the spinal cord21. We started to measure mechanical allodynia 10 min after injection. For CFA-induced inflammation, mice were anaesthetized with isoflurane (2%). 20 µl of CFA (Sigma) was then injected into the plantar surface of the left hindpaw. Both punctate and dynamic mechanical allodynia were measured 1–7 days after CFA treatment.
Behavioural experiments
Contact heat pain (hot plate test)
Mice were placed on a metallic plate heated to a set temperature (30, 50 or 54 °C) within an acrylic container (Bioseb), and videotaped. The latency to flinching and licking one of the hindpaws was blindly measured by video analysis. One temperature was tested per day.
Laser heat pain
Individual mice were habituated for 20 min in a small (7.5 × 7.5 × 15 cm) plastic cage. A laser heat source (500 mW, 445 nm; Shanghai Laser & Optics Century) was targeted at the paw and the latency to withdraw was measured.
Acetone test (cold allodynia)
Mice were habituated for 20 min in a small (7.5 × 7.5 × 15 cm) plastic cage and a small volume of acetone (5 µl) was applied onto the plantar surface of the hindpaw. The time spent flinching or licking the paw was recorded for one minute.
Pinprick
Mice were habituated for 20 min in a small (7.5 × 7.5 × 15 cm) plastic cage. The tester used an Austerlitz pin (000; Fine Scientific Tools) to touch the plantar surface of the hindpaw without skin penetration. Numbers of withdrawal responses per 10 trials (within 1 min) were counted.
Tape removal
Mice were habituated in a transparent cylinder. A 9-mm diameter circular adhesive Microtube Tough-Spots label (Diversified Biotech) was attached to the plantar surface of the hindpaw. The latency to biting or licking to remove the tape (sense time) was measured. Three measurements were performed per mouse.
Brush (dynamic mechanical stimuli)
Each mouse was habituated in a small (7.5 × 7.5 × 15 cm) plastic cage for at least 20 min before testing. The plantar hindpaw was stimulated by light stroking from heel to toe with a paintbrush. For baseline dynamic mechanical sensitivity, the test was repeated ten times. The number of occurrences of walking away or occasionally brief paw lifting was measured. For dynamic mechanical allodynia, the paw flinching/licking time was recorded as an indicator and averaged over three trials for individual animals.
von Frey filaments (punctate mechanical stimuli)
Each mouse was habituated in a small (7.5 × 7.5 × 15 cm) plastic cage for at least 20 min before testing. Mechanical sensitivity was determined with a series of von Frey filaments (bending forces: 0.02, 0.04, 0.07, 0.16, 0.4, 0.6, 1 and 2 g) applied within the sciatic nerve territory (lateral part of the hindpaw). Each filament was tested ten times in increasing order from the lowest force. Between individual measurements, von Frey filaments were applied at least 3 s after the mice had returned to their initial resting state. For baseline static mechanical sensitivity, all filaments were applied and the number of withdrawals was recorded. For tactile allodynia, the minimal force filament for which animals presented either a brisk paw withdrawal and/or an escape attempt in response to at least five of the ten stimulations was determined as the mechanical response threshold.
Open field
To test overall locomotor activity, mice were individually placed on the side of a circular open arena that consisted of an unmarked floor enclosed by a 45 cm diameter wall and allowed to roam freely for 20 min. The arena was divided into the outermost periphery (wall), the centre of the arena (15 cm diameter), and the neutral zone between the centre and the wall (25 cm diameter). Video tracking software (Ethovision XT v11.5, Noldus BV) was used to quantify the distance travelled in each zone by the animal. The arena was housed in a dimly lit room (30 lx) with no noise, and examiners were not present in the room during testing, to ensure limited distractions to the mice.
Ground walking
For ground walking, mice were placed in the MotoRater (TSE Systems)12 and kinematic analysis was performed based on data collected by the MotoRater.
Rotarod
Mice were trained five times on a single speed (15 r.p.m.) rotarod (IITC Life Science). After that, the latency to fall was recorded on the next day. The experiment was repeated twice at 20-min intervals and the average was calculated as rotarod latency.
Calcium imaging in freely behaving mice
Mice used for calcium imaging experiments were injected with HiRet-Cre (5 × 1012 copies/ml) into the spinal cord on P14. In adult mice, a cranial window was installed over the injected area as described12,31,32,33. During the procedures, mice were anaesthetized with Avertin (1.5% solution given at 0.01 ml/g, i.p.) and treated with dexamethasone (0.2 mg/kg, s.c.) and carprofen (5 mg/kg, s.c.) to prevent brain swelling and inflammation. A piece of skull (3.5 mm in diameter) above the somatosensory cortex was removed after high-speed dental drilling. AAV2/9-FLEX-syn-GCaMP6 s (5 × 1013 copies/ml, 0.45 μl per animal) was injected into primary sensory (hindlimb S1) cortex using a micro-syringe pump. A 3-mm coverslip was used to seal the cranial window and the exposed scalp was sutured. After 7 days, the baseplate of a miniaturized integrated fluorescent microscope (Inscopix)34 was fixed on top of the coverslip17. Animals were then habituated to the attachment of the microscope for 2 days. Calcium imaging was then performed in freely moving mice without sensory stimulation, or in intact mice with light brush, von Frey fibre and laser heat applied to the hindpaw. SNI was then performed and calcium imaging was conducted on the same animals at 3 days post-SNI. Sham controls were conducted in separate animals by cutting the skin on the hindlimb without damaging the nerves involved in SNI, and imaging sessions were performed before and after sham procedures. Each imaging session include 5 blocks: 20 min habituation, 4 min free moving followed by 13–17 trials (trial interval: 10 s) of brush, von Frey and laser heat stimuli.
Calcium imaging was performed in freely moving mice using the head-attached microscope (Inscopix; LED power: 0.6–1.0 mW; camera resolution: 1,440 × 1,080 pixels). Images were acquired at 30 Hz using nVista HD software (Inscopix). At the beginning of each imaging session, the protective cap of the previously implanted baseplate was removed and the microscope was attached. The imaging field of view was approximately 900 × 600 μm2 at 0.65 μm/pixel resolution and the imaging depth was selected by adjusting the focus of the microscope until clear dendritic trunk signals were observed, which appeared as bright spots in the images. The focal plane was 150–250 μm below the lens. Mouse behaviour was recorded with a video camera (Canon), which was synchronized with calcium imaging using the trigger-out signal from nVista HD12.
Calcium imaging videos were analysed using Mosaic software (Inscopix) and custom-written scripts in MATLAB following published algorithms35,36. Raw videos were first down-sampled by fourfold along spatial dimensions to reduce file size and noise. The mean fluorescence intensity of each pixel during a recording session (4–8 min) was calculated as F0 and changes in pixel intensity at time t were expressed as (Ft − F0)/F0 or ΔF/F0. To extract active dendritic signals, principal component and independent component analysis (PCA-ICA) was applied to the spatial-temporal data matrices of ΔF/F0 using CellSort and fastICA toolboxes (these toolboxes are freely downloadable from MATLAB Central35,36). This analysis decomposes a spatiotemporal data matrix into independent components based on the skewness of data distribution. Each component has a characteristic spatial filter over the imaged area and a corresponding temporal signal during the imaging period. The spatial filter and the temporal signal of each component were graphically displayed and inspected by human observers who were blind to the experimental conditions of each video. If the spatial filter for a component overlapped with the dark shadows casted by blood vessels in the F0 image, this component was likely to be contributed by blood flow and was therefore rejected. In addition, because calcium signals show characteristic fast-rising and slow-decaying time course37, the temporal skewness of calcium signals is expected to be positive and those components with skewness less than 1 were rejected35. For each selected component, the location of the dendritic trunk was identified as the brightest spot (3 × 3 pixel) of the spatial filter. The corresponding temporal signal of the dendritic trunk was calculated from the ΔF/F0 video by subtracting the median value of the background area (outside the dendritic tree) from the average value of the dendritic trunk area. Calcium activity in dendritic trunks tightly correlated with that in the soma in freely behaving mice12, similar to what has been reported before by two-photon microscopy38,39.
To identify periods of increased neuronal activity, we searched for the rising phase of each calcium event (peak ΔF/F0 > 3 s.d. of baseline fluctuation), which has been shown to be closely associated with neuronal spiking activity37,40,41. The start of this rising phase is detected when the first derivative of ΔF/F0 (calculated in a 200-ms moving window) rises above 0 and continues to increase above 5 s.d. of baseline fluctuation41, and the end of this rising phase is detected when the first derivative of ΔF/F0 falls below 0. For each dendrite, the amplitude of each detected calcium event was normalized by the s.d. of the whole calcium trace. To visualize the activity patterns of detected dendrites during stimulation, the active event traces of each CSN were aligned to the time when the brush, von Frey filament or laser heat was applied to the left plantar surface of the hindlimb. The resulting traces from all the CSNs were sorted by their peak activation time during the window and displayed in temporal raster plots.
To quantify the difference in activity between pre- and post-SNI conditions, we first calculated the trial-averaged activities of CSN dendrites in response to different sensory stimuli, then compared the responses between pre- and post-SNI conditions within 1 s of stimulation (rank sum tests). A significant increase in CSN activity in response to von Frey filaments was observed after SNI (Fig. 3c), but not in sham controls (data not shown).
To quantify the proportion of activated neurons responding to brush only, von Frey only, or both stimuli in pre- and post-SNI conditions, we concatenated the calcium videos of the brush and von Frey sessions. Responsive neurons were classified based on whether they showed maximal trial average activity within 1 s after either or both stimuli.
Spinal cord slice preparation and electrophysiology
To label CST axons, AAV-2/1-ChR2-YFP was injected unilaterally into the somatosensory cortex in neonatal CCK–tdTomato mice (P1). At P28–P31, mice were deeply anaesthetized with isoflurane, decapitated and the lumbar spinal cord was quickly removed to ice-cold modified artificial cerebrospinal fluid (ACSF): NaCl, 80; KCl, 2.5; NaH2PO4, 1.25; CaCl2, 0.5; MgCl2, 3.5; NaHCO3, 25; sucrose, 75; sodium ascorbate, 1.3; sodium pyruvate, 3.0 (all in mM), with pH at 7.4 and osmolarity at 310–320 mOsm, and oxygenated with 95% O2 and 5% CO2. Sagittal spinal cord slices (350–500 μm) with dorsal roots (3–5 mm) attached were cut using a vibratome (VT1000S, Leica). Slices were then incubated for about 1 h at 35 °C in a solution that contains (in mM): NaCl, 125; KCl, 2.5; CaCl2, 2; MgCl2, 1, NaH2PO4, 1.25; NaHCO3, 26; d-glucose, 25; sodium ascorbate, 1.3; sodium pyruvate, 3.0 with pH at 7.2 and measured osmolality at 310–320 mOsm, and oxygenated with 95% O2 and 5% CO2. The slice was then transferred into a recording chamber and perfused with oxygenated recording solution at a rate of 5 ml/min before electrophysiological recordings at room temperature (23 °C).
For whole cell patch-clamp recording, CCK–tdTomato neurons were viewed using an upright microscope (Olympus, BX51WI) with a 63× water-immersion objective, infrared differential interference contrast (IR-DIC) and fluorescence. Patch pipettes (5–10 MΩ) were made from borosilicate glass (Sutter) using a P-1000 horizontal micropipette puller (Sutter). Internal solution contained (in mM): potassium gluconate 130, KCl 5, MgATP 4, NaGTP 0.5, HEPES 10, EGTA 0.5, pH 7.28 with KOH, and measured osmolality at 280–300 mOsm. This internal solution allows separation of AMPAR and GABAA currents at the reversal potential of the other receptor. Data were acquired with pClamp 10.0 software (Molecular Devices) using a MultiClamp 700B patch-clamp amplifier and Digidata 1440A (Molecular Devices). Responses were low-pass filtered online at 4 kHz and digitized at 10 kHz. Junction potential was around 6–8 mV. No correction for junction potential was applied. Membrane capacitance (24.3 ± 1.1 pF), membrane resistance (493.4 ± 29.4 mΩ) were monitored continuously and cells were excluded from analysis if these values changed by more than 10% during an experiment. Following established procedure21, recordings with series resistance greater than 30 mΩ or resting membrane potential more depolarized than −50 mV were excluded from analysis. All recordings were performed at room temperature (23 °C).
Voltage clamp recordings were performed by holding membrane potential at −70 mV. Under this condition, evoked inhibitory postsynaptic currents (eIPSCs) were minimized, and evoked excitatory postsynaptic currents (eEPSCs) were detected. This was used to study whether a neuron received inputs directly (monosynaptic) or indirectly (polysynaptic) from dorsal root or CST axons. Current clamp was also carried out at resting membrane potential in order to record evoked action potential output. Dorsal root stimulation was generated with ISO-Flex stimulus isolator (A.M.P.I.) and delivered with suction electrode (A-M Systems). The stimulation pulse width was 0.1 ms and intensity was 25 µA for Aβ fibres. High-frequency stimulation at 20 Hz was used to determine monosynaptic input for Aβ and CST fibres. Laser stimulation (473 nm, 5 ms, 5 mW) for activating CST axon terminals was delivered using an optical fibre (200 µm core, 0.22NA; Thorlabs) directly illuminating the recording area. Monosynaptic transmission for opto-stimulation was verified by applying tetrodotoxin (TTX; 0.5 µM, Abcam) in the recording bath to block sodium channels, followed by 4-aminopyridine (4-AP; 2 mM, Sigma) to augment light-induced, direct depolarization of ChR2 axon terminals42. NBQX (5 µM) and CPP (20 µM) were used to block AMPAR and NMDAR currents, respectively. To mimic the spinal disinhibition condition, both voltage and current clamps were performed in the presence of both bicuculline (10 μM, Sigma) and strychnine (2 μM, Sigma).
Innocuous mechanical stimulation for c-Fos and/or pERK induction
To induce c-Fos expression by innocuous mechanical (dynamic brush) stimulation, animals were lightly restrained using a rodent restraint bag (traditional triangle shape, AIMS). We used a paintbrush to stroke the hindpaw. Each touch, lasting 2 s and moving from the middle of the foot to the distal foot pad along the peripheral side that belongs to the sural nerve territory, was applied once every 4 s for 10 min. This touch stimulus does not elicit a flexion reflex in normal mice. Animals were perfused two hours later and spinal cord transverse sections were prepared for c-Fos immunostaining. For pERK induction, the same procedures were applied, except that mice were killed immediately after repetitive mechanical brushing or 5 min after thermal stimulation.
Light-based CSN stimulation
The HiRet-Cre virus (2 × 1012 copies/ml) was injected intraspinally into the lumbar spinal cord of LSL-ChR2-YFP mice at P14 to retrogradely label hindlimb corticospinal neurons. Four weeks later, the animals were subjected to optical stimulation experiments. For optic fibre implantation, animals were anaesthetized with ketamine/xylazine (100 mg/kg ketamine, 10 mg/kg xylazine) and mounted on a stereotaxic frame. A 1-mm hole was drilled on the right side of the skull, centred at −1.2 mm posterior to Bregma and 1.65 mm lateral to midline, with the dura left intact. All SNI surgeries were performed on the left hindpaw. A customized fibre optic cannula (Ø1.25 mm Ceramic Ferrule, Ø200 µm Core, 0.50 NA, 2 mm projection; Thorlabs) was positioned on the cortical surface through the hole. Silicon elastomer and dental cement were applied to protect the skull window and secure the cannula. The incision was sutured and animals were allowed to recover for 7 days before behavioural testing. Before optogenetic stimulation, an optogenetic patch cable was connected to the cannula via a mating sleeve. The patch cable was connected to the laser source with a rotary joint (FRJ 1 × 1, Doric) to allow free movement of the animal. Laser stimulation (473 nm, 5mW; 100 pulses of 5 ms, 20 Hz) was delivered during sensory tests. We verified the expression of ChR2-YFP in the hindlimb S1 by post hoc histological analysis.
For pERK induction, animals were lightly restrained using a rodent restraint bag (traditional triangle shape, AIMS). Laser stimulation (473 nm, 5 mW; 100 pulses of 5 ms, 20 Hz) was delivered at 2-s intervals for 10 min, and repeated three times with a 1-min rest between each session in mice with SNI (contralateral cortex). Repetitive light brush (see ‘Innocuous mechanical stimulation for c-Fos and/or pERK induction’) was applied during optogenetic stimulation. Animals were perfused immediately after stimulation.
Quantification and statistical analysis
The normality and variance similarity were measured by STATA (version 12) before we applied any parametric tests. Two-sided or one-sided t-tests were used for single comparisons between two groups. The rest of the data were analysed using one-way or two-way ANOVAs, depending on the appropriate design. Post hoc comparisons were carried out only when a main effect showed statistical significance. P values of multiple comparisons were adjusted using Bonferroni’s correction. Error bars in all figures represent mean ± s.e.m. ***P < 0.001, **P < 0.01, *P < 0.05, NS, no statistical significance (P ≥ 0. 05).
Reporting summary
Further information on experimental design is available in the Nature Research Reporting Summary linked to this paper.
Code availability
All custom codes used in this study are available from the corresponding author on reasonable request.
Data availability.
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Owens, D. M. & Lumpkin, E. A. Diversification and specialization of touch receptors in skin. Cold Spring Harb. Perspect. Med. 4, a013656 (2014).
Abraira, V. E. & Ginty, D. D. The sensory neurons of touch. Neuron 79, 618–639 (2013).
Basbaum, A. I., Bautista, D. M., Scherrer, G. & Julius, D. Cellular and molecular mechanisms of pain. Cell 139, 267–284 (2009).
Costigan, M., Scholz, J. & Woolf, C. J. Neuropathic pain: a maladaptive response of the nervous system to damage. Annu. Rev. Neurosci. 32, 1–32 (2009).
Lemon, R. N. Descending pathways in motor control. Annu. Rev. Neurosci. 31, 195–218 (2008).
Liu, K. et al. PTEN deletion enhances the regenerative ability of adult corticospinal neurons. Nat. Neurosci. 13, 1075–1081 (2010).
Jin, D. et al. Restoration of skilled locomotion by sprouting corticospinal axons induced by co-deletion of PTEN and SOCS3. Nat. Commun. 6, 8074 (2015).
Boada, M. D. & Woodbury, C. J. Myelinated skin sensory neurons project extensively throughout adult mouse substantia gelatinosa. J. Neurosci. 28, 2006–2014 (2008).
Boada, M. D. & Woodbury, C. J. Physiological properties of mouse skin sensory neurons recorded intracellularly in vivo: temperature effects on somal membrane properties. J. Neurophysiol. 98, 668–680 (2007).
Lemon, R. N. & Griffiths, J. Comparing the function of the corticospinal system in different species: organizational differences for motor specialization? Muscle Nerve 32, 261–279 (2005).
Abraira, V. E. et al. The cellular and synaptic architecture of the mechanosensory dorsal horn. Cell 168, 295–310.e19 (2017).
Wang, X. et al. Deconstruction of corticospinal circuits for goal-directed motor skills. Cell 171, 440–455.e14 (2017).
Kinoshita, M. et al. Genetic dissection of the circuit for hand dexterity in primates. Nature 487, 235–238 (2012).
Liu, Y. et al. A sensitized IGF1 treatment restores corticospinal axon-dependent functions. Neuron 95, 817–833.e4 (2017).
O’Leary, D. D. Development of connectional diversity and specificity in the mammalian brain by the pruning of collateral projections. Curr. Opin. Neurobiol. 2, 70–77 (1992).
O’Leary, D. D. & Koester, S. E. Development of projection neuron types, axon pathways, and patterned connections of the mammalian cortex. Neuron 10, 991–1006 (1993).
Decosterd, I. & Woolf, C. J. Spared nerve injury: an animal model of persistent peripheral neuropathic pain. Pain 87, 149–158 (2000).
Atasoy, D., Betley, J. N., Su, H. H. & Sternson, S. M. Deconstruction of a neural circuit for hunger. Nature 488, 172–177 (2012).
Li, X. et al. Skin suturing and cortical surface viral infusion improves imaging of neuronal ensemble activity with head-mounted miniature microscopes. J. Neurosci. Methods 291, 238–248 (2017).
Todd, A. J. Neuronal circuitry for pain processing in the dorsal horn. Nat. Rev. Neurosci. 11, 823–836 (2010).
Cheng, L. et al. Identification of spinal circuits involved in touch-evoked dynamic mechanical pain. Nat. Neurosci. 20, 804–814 (2017).
Ji, R. R., Baba, H., Brenner, G. J. & Woolf, C. J. Nociceptive-specific activation of ERK in spinal neurons contributes to pain hypersensitivity. Nat. Neurosci. 2, 1114–1119 (1999).
Torsney, C. & MacDermott, A. B. Disinhibition opens the gate to pathological pain signaling in superficial neurokinin 1 receptor-expressing neurons in rat spinal cord. J. Neurosci. 26, 1833–1843 (2006).
Duan, B. et al. Identification of spinal circuits transmitting and gating mechanical pain. Cell 159, 1417–1432 (2014).
Bourane, S. et al. Identification of a spinal circuit for light touch and fine motor control. Cell 160, 503–515 (2015).
Taniguchi, H. et al. A resource of Cre driver lines for genetic targeting of GABAergic neurons in cerebral cortex. Neuron 71, 995–1013 (2011).
Sun, F. et al. Sustained axon regeneration induced by co-deletion of PTEN and SOCS3. Nature 480, 372–375 (2011).
Arlotta, P. et al. Neuronal subtype-specific genes that control corticospinal motor neuron development in vivo. Neuron 45, 207–221 (2005).
Metz, G. A., Dietz, V., Schwab, M. E. & van de Meent, H. The effects of unilateral pyramidal tract section on hindlimb motor performance in the rat. Behav. Brain Res. 96, 37–46 (1998).
Muir, G. D. & Whishaw, I. Q. Complete locomotor recovery following corticospinal tract lesions: measurement of ground reaction forces during overground locomotion in rats. Behav. Brain Res. 103, 45–53 (1999).
Mastwal, S. et al. Phasic dopamine neuron activity elicits unique mesofrontal plasticity in adolescence. J. Neurosci. 34, 9484–9496 (2014).
Cao, V. Y. et al. In vivo two-photon imaging of experience-dependent molecular changes in cortical neurons. J. Vis. Exp. 71, 50148 (2013).
Cao, V. Y. et al. Motor learning consolidates Arc-expressing neuronal ensembles in secondary motor cortex. Neuron 86, 1385–1392 (2015).
Ziv, Y. et al. Long-term dynamics of CA1 hippocampal place codes. Nat. Neurosci. 16, 264–266 (2013).
Mukamel, E. A., Nimmerjahn, A. & Schnitzer, M. J. Automated analysis of cellular signals from large-scale calcium imaging data. Neuron 63, 747–760 (2009).
Hyvärinen, A. & Oja, E. Independent component analysis: algorithms and applications. Neural Netw. 13, 411–430 (2000).
Chen, T. W. et al. Ultrasensitive fluorescent proteins for imaging neuronal activity. Nature 499, 295–300 (2013).
Hill, D. N., Varga, Z., Jia, H., Sakmann, B. & Konnerth, A. Multibranch activity in basal and tuft dendrites during firing of layer 5 cortical neurons in vivo. Proc. Natl Acad. Sci. USA 110, 13618–13623 (2013).
Peters, A. J., Lee, J., Hedrick, N. G., O’Neil, K. & Komiyama, T. Reorganization of corticospinal output during motor learning. Nat. Neurosci. 20, 1133–1141 (2017).
Cichon, J., Blanck, T. J. J., Gan, W. B. & Yang, G. Activation of cortical somatostatin interneurons prevents the development of neuropathic pain. Nat. Neurosci. 20, 1122–1132 (2017).
Peters, A. J., Chen, S. X. & Komiyama, T. Emergence of reproducible spatiotemporal activity during motor learning. Nature 510, 263–267 (2014).
Holloway, B. B. et al. Monosynaptic glutamatergic activation of locus coeruleus and other lower brainstem noradrenergic neurons by the C1 cells in mice. J. Neurosci. 33, 18792–18805 (2013).
Acknowledgements
We thank T. Huang, Y. Zhang and Q. Ma for advice and D. Ginty, S. Hegarty, Q. Ma, F. Wang and P. Williams for critical reading. This study was supported by grants from the Craig Neilsen Foundation (Y.L. and X.W.), Paralyzed Veterans of America Foundation (Y.L.), Dr. Miriam and Sheldon G. Adelson Medical Research Foundation and NINDS (C.J.W. and Z.H.) and NIMH intramural research program ZIA MH002897 (K.H.W. and X.L.). IDDRC and viral cores supported by the grants NIH P30 HD018655 and P30EY012196 were used for this study.
Reviewer information
Nature thanks R. Ji and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
Y.L., A.L., X.L., Z.Z., K.H.W., C.J.W. and Z.H. conceived the experiments. Y.L., A.L., X.L., Z.Z., M.C., X.W., C.F., C.A., J.Z., Z.G., B.C., X.D., J.-Y.Z. and Y.Z. performed the experiments. Y.L., A.L., X.L., C.C., K.H.W., C.J.W. and Z.H. prepared the manuscript with input from all authors. K.H.W., C.J.W. and Z.H. co-supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Effects of pyramidotomy on tactile behaviour and gross locomotion in mice.
a, Correlation between CST ablation and tactile behaviours in mice with pyramidotomy. For individual animals that received pyramidotomy, tag number, image and quantification of L3 spinal cord sections stained with anti-PKCγ antibodies showing remaining CST axons, percentage of withdrawal response to low-threshold von Frey filament (0.16g) and light brush, and sense time to the tape are shown. b, c, Performance on ground walking (b, P = 0.14, hindlimb weight support; P = 0.81, hindlimb retraction; P = 0.41, hindlimb protraction; P = 0.81, inter-limb coordination; P = 0.20, fore–hindlimb coordination), and rotarod test (c, P = 0.87) in mice with sham surgery (n = 8) or pyramidotomy (n = 12). n.s., no statistical significance. Two-sided student’s t-test. Data shown as mean ± s.e.m.
Extended Data Fig. 2 CST axon termination in the lumbar spinal cord.
a, Representative transverse spinal section (L3) from an Emx1-tdTomato (red) reporter line. b, Sections were co-stained with IB4 (green), a lamina IIi marker, and anti-PKCγ, a laminae IIi/III marker, in the spinal dorsal horn. Scale bar, 500 μm. For a and b, three and four mice, respectively, showed similar results.
Extended Data Fig. 3 Efficiency of somatosensory CSN ablation by P14 intraspinal injection.
a, Left, schematic of regional CSN ablation by P14 lumbar (T13–L6) intraspinal injection. Right, representative image (n = 8 animals with similar results) of the cortex with GFP+ areas covering hindlimb S1/S2. b, Representative images (n = 6 animals with similar results) of cortical sections showing hindlimb CSNs retrogradely labelled by lumbar (T13–L6) intraspinal injection of HiRet-GFP at P14. Scale bar, 100 μm. c, To assess ablation efficiency, at the end point, retrograde-targeting rAAV-mCherry was injected into the lumbar spinal cord in some animals. Representative images of cortical sections showing retrogradely labelled mCherry+ CSNs (left) within the GFP+ cortical areas (right) (S1/S2) in control or AAV-FLEX-DTR injected animals with quantification (normalized to those in controls as 100). **P < 0.01 (P < 0.0001), two-sided Student’s t-test. n = 5 and 5 for control and AAV-FLEX-DTR injected mice, respectively. Scale bar, 100 μm. d, Representative images of transverse lumbar spinal cord sections showing residual CST axons labelled by GFP (from mice co-injected with AAV-GFP to S1/S2 and AAV-FLEX-DTR) in control or S1/S2 CSN ablated animals with quantification. **P < 0.01 (P < 0.0001), two-sided Student’s t-test. n = 7 or 8 for control or AAV-FLEX-DTR-injected mice, respectively. Scale bar, 500 μm. Data shown as mean ± s.e.m.
Extended Data Fig. 4 Mechanical allodynia induced by SNI or CFA injection is compromised in mice with pyramidotomy, but cold allodynia and mechanical hyperalgesia induced by SNI are not.
a, Schematic drawing of experimental paradigm. b–e, Measurement of punctate (b) and dynamic (c) mechanical allodynia, cold allodynia (d) and mechanical hyperalgesia (e) after SNI in mice that underwent sham surgery (n = 8) or pyramidotomy (n = 9) 1–21 days after SNI. b, P < 0.0001, P < 0.0001, P = 0.0012, P = 0.0004 and P = 0.041; c, P < 0.0001, P < 0.0001, P = 0.0004, P = 0.001 and P = 0.0045; d, P = 0.26, P = 0.33, P = 0.29, P > 0.99 and P > 0.99; e, P > 0.99, P = 0.15, P = 0.56, P > 0.99 and P = 0.74 for 1, 3, 7, 14 and 21 d post SNI, respectively. f, g, Measurement of punctate (f) and dynamic (g) mechanical allodynia in mice that underwent sham surgery (n = 6) or pyramidotomy (n = 6) 1–7 days after hindpaw CFA injection. f, P = 0.01, P = 0.01, P = 0.01, P < 0.0001 and P < 0.0001; g, P < 0.0001 for 1, 2, 3, 5 and 7d post CFA injection, respectively. Two-way repeated measures ANOVA followed by Bonferroni correction. Data shown as mean ± s.e.m.
Extended Data Fig. 5 Calcium imaging of CSN activity in intact and SNI mice.
a, Schematic of experimental procedures. b, Confocal fluorescence images of coronal brain sections showing specific expression of GCaMP6s in CSNs. Left, a 10× image showing labelled CSN soma and dendrites; right, a 25× image showing the magnified view of apical dendritic trunks. Dotted line, expected focal plane of head-mounted microscope. Scale bars, 100 μm. c, Procedures for identifying the active events of CSN dendrites. Left, example of dendrites identified from a calcium movie by ICA analysis. The brightest spot in a dendritic tree, corresponding to the trunk (red circle), is used as a region of interest for temporal signal analysis. Top trace, temporal signal of the dendrite. Bottom trace, magnified calcium events. Horizontal bars indicate rising phases of calcium events, which are associated with neuronal activation and used in subsequent analysis. Scale bar, 100 µm. d, Example calcium movie frames showing dendritic activities of hindlimb S1 CSNs upon different sensory stimuli in intact mice. In these examples, brush stimuli activated CSNs, whereas von Frey (0.04 g) and laser heat stimuli did not. Calcium signals are expressed as ∆F/F0 (F0 is the time-average fluorescence of the whole movie). Scale bar, 200 µm. For b, d, the experiments were repeated independently four times with similar results. e, Pie charts showing the proportions of neurons that responded to brushes, von Frey filaments or both brush and von Frey stimulation before and after SNI. Few neurons responded to both stimuli before SNI, but this overlapping proportion increased after SNI.
Extended Data Fig. 6 Mechanical allodynia induced by spinal disinhibition (treatment with bicuculline and strychnine) is compromised in mice with pyramidotomy.
a, Drawing of experimental paradigm. b, c, Measurement of punctate (b) and dynamic (c) mechanical allodynia after intrathecal injection of bicuculline and strychnine in mice that had undergone sham surgery (n = 6) or pyramidotomy (n = 7). **P < 0.01 (P < 0.0001 for b and c at 10, 30, and 90 min post drug), two-way repeated measures ANOVA followed by Bonferroni correction. Data shown as mean ± s.e.m.
Extended Data Fig. 7 Neuronal activity in cortical and subcortical areas upon light touch after SNI.
a–c, Drawings of c-Fos immunostaining in intact mice (a), mice with SNI only (b) and mice with SNI after light brush stimulation (c) from control mice and mice with pyramidotomy. mPFC, medial prefrontal cortex; ACC, anterior cingulate cortex; S1HL, hindlimb primary somatosensory cortex; S2, secondary somatosensory cortex; Pir, piriform cortex; PV, periventricular nucleus of the thalamus; VM, ventromedial nucleus of the hypothalamus; Amyg, amygdala. d, Quantification of c-Fos+ cells in multiple cortical areas in intact mice (with CSN, n = 3; with pyramidotomy, n = 3), mice with SNI only (with CSN, n = 3; with pyramidotomy, n = 3), and mice with SNI after light brush stimulation (with CSN, n = 4; with pyramidotomy, n = 3). **P < 0.01; n.s., no statistical significance. PFC SNI only with or without Py: P > 0.99; PFC SNI + brush with or without Py: P < 0.0001; ACC SNI only with or without Py: P > 0.99; ACC SNI + brush with or without Py: P = 0.0002; M1 SNI only with or without Py: P > 0.99; M1 SNI + brush with or without Py: P > 0.99; S1 SNI only with or without Py: P > 0.99; S1 SNI + brush with or without Py: P < 0.0001; insula SNI only with or without Py: P > 0.99; insula SNI + brush with or without Py: P < 0.0001; S2 SNI + brush with or without Py: P = 0.0075; Pir all conditions (ANOVA): P = 0.82. One-way ANOVA followed by Bonferroni correction. e, Representative images of multiple cortical areas stained with c-Fos (red) and GFP (green, HiRet-GFP injection) in mice with SNI after light brush stimulation. Arrowheads mark the co-localization of c-Fos+ and GFP+ CSNs. Scale bars, 20 μm. f, Quantification of c-Fos+ and GFP+ CSN co-localization in multiple cortical areas in animals with SNI after light brush stimulation without (n = 4) or with pyramidotomy (Py, n = 3). *P < 0.05; **P < 0.01, P = 0.03, 0.04, 0.0009, and 0.02 for mPFC, ACC, S1, and S2, respectively. Two-sided Student’s t-test. Data shown as mean ± s.e.m.
Extended Data Fig. 8 Ablation of lumbar CCK+ interneurons reduces tactile sensitivity and dorsal horn neuronal activation after SNI, but not nociceptive response or gross locomotion.
a–d, Measurements of sensitivity to laser heat (a, P = 0.76), acetone (b, P = 0.86), von Frey filaments (c, P = 0.03 for 0.016 g) and brush (d, P = 0.002) stimuli in control mice (n = 8) or mice in which CCK–tdTomato interneurons had been ablated (n = 7). a, b, d, Two-sided Student’s t-test; c, two-way repeated measures ANOVA followed by Bonferroni correction. e, f, Performance on open field (e, P = 0.54) and ground walking (f, P = 0.68, 0.72, and 0.50 for hindlimb weight support, protraction, and retraction, respectively) in control mice (n = 8) or mice in which CCK–tdTomato interneurons had been ablated (n = 7). n.s., no statistical significance, two-sided Student’s t-test. g–i, Representative images of c-Fos (green) activity (g) and quantification of CCK+/c-Fos+ cells (h) and c-Fos+ neurons in different laminae of the dorsal horn of the spinal cord (L3–4) of CCK–tdTomato (red) mice after SNI and brush stimulation in mice that had undergone sham surgery (n = 4), pyramidotomy (n = 5) or lumbar CCK–tdTomato ablation (n = 3) (i). Scale bars, 500 μm (g, right, middle, and left columns) and 50 μm (g, zoom in panels). **P < 0.01, two-sided Student’s t-test. h, P < 0.0001; i, P = 0.0045 and 0.0028 for laminae I–II and laminae III–V, respectively. Data shown as mean ± s.e.m.
Extended Data Fig. 9 Characterization of Aβ fibre and CST inputs onto CCK–tdTomato neurons.
a, Schematic of the stimulation and whole-cell patch recording set-up for tdTomato-labelled CCK+ interneurons. CST axons labelled by AAV-ChR2-YFP were stimulated with a 473-nm laser. A single dorsal root (L4–L6) was stimulated with a glass suction electrode. b, c, Representative consecutive traces (n = 3) of Aβ fibre (left) and opto-CST (right) stimulation-evoked responses (b) and summarization (c) of whole cell patch-clamp recordings on CCK–tdTomato interneurons. Three recording conditions were used: first, to detect evoked EPSCs (eEPSCs), we held the membrane potential at −70 mV, which is the equilibrium potential of Cl− and therefore minimizes the flow of eIPSCs. Second, by holding the membrane potential at 0 mV, we examined the polysynaptic, inhibitory inputs (eIPSCs) onto CCK–tdTomato interneurons. Third, we used current clamp mode to examine whether the stimulation drove action potential firing at the resting membrane potential. Type 1: CCK–tdTomato neurons receive only excitatory inputs, and few of them generated AP outputs when Aβ or CST inputs were stimulated. Type 2: CCK–tdTomato neurons receive both excitatory inputs and feed-forward inhibitory inputs, with no AP output. Type 3: CCK–tdTomato neurons receive predominant feed-forward inhibition, with no AP output. Type 4: CCK–tdTomato neurons show no response at either voltage or current clamp recording. d, Left, representative recording of an Aβ (25 μA) dorsal root evoked EPSC at −70 mV. Latency and jitter properties (magnified in inset) with quantifications (n = 8 neurons) are consistent with monosynaptic sensory connectivity. Right, opto-stimulation evoked EPSCs (averaged traces) at −70 mV in the same cell as shown on the left. The evoked EPSC was blocked by AMPA and NMDA antagonists (NBQX (5 μM)/CPP (20 μM)). In addition, opto-stimulation evoked EPSCs were eliminated by TTX (0.5 μM), and reinstated by 4-AP (2 mM), indicating a monosynaptic connection between the CST and CCK–tdTomato interneurons. Bar graph, quantification of eEPSC amplitudes after administration of drugs as shown. **P < 0.0001; one-way ANOVA followed by Bonferroni correction. n = 8 neurons. Data shown as mean ± s.e.m.
Extended Data Fig. 10 Reinforcement of tactile allodynia in mice with SNI by optogenetic stimulation of somatosensory CSNs.
a, Drawing of experimental paradigm. b, c, Measurement of punctate (b, von Frey filaments) and dynamic (c, brush) mechanical allodynia upon opto-stimulation in control mice (n = 6) and mice expressing ChR2-YFP in CSNs (n = 6) after SNI. n.s., no statistical significance; *P < 0.05. b, c, P = 0.18, 0.08 and 0.41, 0.02 for PLAP and Cre, without or with laser, respectively; two-sided Student’s t-test. d, Representative images and quantification of pERK (red) and NK1R (green) immunostaining in the superficial dorsal horn (laminae I–II) of the spinal cord (L3–4) in control mice (n = 3) or mice expressing ChR2-YFP in hindlimb CSNs (n = 3) with SNI and brush stimulation coupled with opto-stimulation. Scale bar, 100 μm. *P < 0.05; P = 0.02 and 0.01 for pERK and pERK/NK1R ratio, respectively; two-sided Student’s t-test. Six sections crossing the lumbar spinal cord (L3–4) were quantified for individual animals. Data shown as mean ± s.e.m.
Supplementary information
Source data
Rights and permissions
About this article
Cite this article
Liu, Y., Latremoliere, A., Li, X. et al. Touch and tactile neuropathic pain sensitivity are set by corticospinal projections. Nature 561, 547–550 (2018). https://doi.org/10.1038/s41586-018-0515-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0515-2
Keywords
This article is cited by
-
The developmental timing of spinal touch processing alterations predicts behavioral changes in genetic mouse models of autism spectrum disorders
Nature Neuroscience (2024)
-
Somatosensory cortex and central amygdala regulate neuropathic pain-mediated peripheral immune response via vagal projections to the spleen
Nature Neuroscience (2024)
-
Distinct local and global functions of mouse Aβ low-threshold mechanoreceptors in mechanical nociception
Nature Communications (2024)
-
Microglia enhance post-anesthesia neuronal activity by shielding inhibitory synapses
Nature Neuroscience (2024)
-
The secondary somatosensory cortex gates mechanical and heat sensitivity
Nature Communications (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.