Abstract
The thymus is responsible for generating a diverse yet self-tolerant pool of T cells1. Although the thymic medulla consists mostly of developing and mature AIRE+ epithelial cells, recent evidence has suggested that there is far greater heterogeneity among medullary thymic epithelial cells than was previously thought2. Here we describe in detail an epithelial subset that is remarkably similar to peripheral tuft cells that are found at mucosal barriers3. Similar to the periphery, thymic tuft cells express the canonical taste transduction pathway and IL-25. However, they are unique in their spatial association with cornified aggregates, ability to present antigens and expression of a broad diversity of taste receptors. Some thymic tuft cells pass through an Aire-expressing stage and depend on a known AIRE-binding partner, HIPK2, for their development. Notably, the taste chemosensory protein TRPM5 is required for their thymic function through which they support the development and polarization of thymic invariant natural killer T cells and act to establish a medullary microenvironment that is enriched in the type 2 cytokine, IL-4. These findings indicate that there is a compartmentalized medullary environment in which differentiation of a minor and highly specialized epithelial subset has a non-redundant role in shaping thymic function.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kyewski, B. & Klein, L. A central role for central tolerance. Annu. Rev. Immunol. 24, 571–606 (2006).
Takahama, Y., Ohigashi, I., Baik, S. & Anderson, G. Generation of diversity in thymic epithelial cells. Nat. Rev. Immunol. 17, 295–305 (2017).
Gerbe, F. & Jay, P. Intestinal tuft cells: epithelial sentinels linking luminal cues to the immune system. Mucosal Immunol. 9, 1353–1359 (2016).
Metzger, T. C. et al. Lineage tracing and cell ablation identify a post-Aire-expressing thymic epithelial cell population. Cell Rep. 5, 166–179 (2013).
Eckhart, L., Lippens, S., Tschachler, E. & Declercq, W. Cell death by cornification. Biochim. Biophys. Acta 1833, 3471–3480 (2013).
Derbinski, J. et al. Promiscuous gene expression in thymic epithelial cells is regulated at multiple levels. J. Exp. Med. 202, 33–45 (2005).
White, A. J. et al. Lymphotoxin signals from positively selected thymocytes regulate the terminal differentiation of medullary thymic epithelial cells. J. Immunol. 185, 4769–4776 (2010).
Odaka, C. et al. TGF-β type II receptor expression in thymic epithelial cells inhibits the development of Hassall’s corpuscles in mice. Int. Immunol. 25, 633–642 (2013).
Hassall, A. H. The Microscopic Anatomy of the Human Body, in Health and Disease Vol. 2, Plate LXI, Figure 10 (Samuel Highley, London, 1849).
Farr, A. G., Dooley, J. L. & Erickson, M. Organization of thymic medullary epithelial heterogeneity: implications for mechanisms of epithelial differentiation. Immunol. Rev. 189, 20–27 (2002).
Gerbe, F., Legraverend, C. & Jay, P. The intestinal epithelium tuft cells: specification and function. Cell. Mol. Life Sci. 69, 2907–2917 (2012).
von Moltke, J., Ji, M., Liang, H.-E. & Locksley, R. M. Tuft-cell-derived IL-25 regulates an intestinal ILC2-epithelial response circuit. Nature 529, 221–225 (2016).
Howitt, M. R. et al. Tuft cells, taste-chemosensory cells, orchestrate parasite type 2 immunity in the gut. Science 351, 1329–1333 (2016).
Gerbe, F. et al. Intestinal epithelial tuft cells initiate type 2 mucosal immunity to helminth parasites. Nature 529, 226–230 (2016).
Hoover, B. et al. The intestinal tuft cell nanostructure in 3D. Sci. Rep. 7, 1652 (2017).
Krasteva, G. et al. Cholinergic chemosensory cells in the trachea regulate breathing. Proc. Natl Acad. Sci. USA 108, 9478–9483 (2011).
Deckmann, K. et al. Bitter triggers acetylcholine release from polymodal urethral chemosensory cells and bladder reflexes. Proc. Natl Acad. Sci. USA 111, 8287–8292 (2014).
Panneck, A. R. et al. Cholinergic epithelial cell with chemosensory traits in murine thymic medulla. Cell Tissue Res. 358, 737–748 (2014).
Soultanova, A. et al. Cholinergic chemosensory cells of the thymic medulla express the bitter receptor Tas2r131. Int. Immunopharmacol. 29, 143–147 (2015).
Bezençon, C. et al. Murine intestinal cells expressing Trpm5 are mostly brush cells and express markers of neuronal and inflammatory cells. J. Comp. Neurol. 509, 514–525 (2008).
Wong, G. T., Gannon, K. S. & Margolskee, R. F. Transduction of bitter and sweet taste by gustducin. Nature 381, 796–800 (1996).
Zhang, Y. et al. Coding of sweet, bitter, and umami tastes: different receptor cells sharing similar signaling pathways. Cell 112, 293–301 (2003).
Yano, M. et al. Aire controls the differentiation program of thymic epithelial cells in the medulla for the establishment of self-tolerance. J. Exp. Med. 205, 2827–2838 (2008).
Rattay, K. et al. Homeodomain-interacting protein kinase 2, a novel autoimmune regulator interaction partner, modulates promiscuous gene expression in medullary thymic epithelial cells. J. Immunol. 194, 921–928 (2015).
Lee, Y. J. et al. Tissue-specific distribution of iNKT cells impacts their cytokine response. Immunity 43, 566–578 (2015).
Lee, Y. J., Holzapfel, K. L., Zhu, J., Jameson, S. C. & Hogquist, K. A. Steady-state production of IL-4 modulates immunity in mouse strains and is determined by lineage diversity of iNKT cells. Nat. Immunol. 14, 1146–1154 (2013).
Matsumoto, I., Ohmoto, M., Narukawa, M., Yoshihara, Y. & Abe, K. Skn-1a (Pou2f3) specifies taste receptor cell lineage. Nat. Neurosci. 14, 685–687 (2011).
Anderson, M. S. et al. Projection of an immunological self shadow within the thymus by the Aire protein. Science 298, 1395–1401 (2002).
Damak, S. et al. Trpm5 null mice respond to bitter, sweet, and umami compounds. Chem. Senses 31, 253–264 (2006).
Gordon, J. et al. Specific expression of lacZ and cre recombinase in fetal thymic epithelial cells by multiplex gene targeting at the Foxn1 locus. BMC Dev. Biol. 7, 69 (2007).
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 13, 133–140 (2010).
Picelli, S. et al. Full-length RNA-seq from single cells using Smart-seq2. Nat. Protoc. 9, 171–181 (2014).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Velten, L. et al. Human haematopoietic stem cell lineage commitment is a continuous process. Nat. Cell Biol. 19, 271–281 (2017).
Kharchenko, P. V., Silberstein, L. & Scadden, D. T. Bayesian approach to single-cell differential expression analysis. Nat. Methods 11, 740–742 (2014).
Acknowledgements
We thank A. Chan, M. Waterfield, L. Velten and the Anderson, Locksley and Steinmetz laboratories for helpful discussions; K. Wu, Y. Wang and E. Li for experimental support. Tetramers were from the NIH Tetramer Core Facility. Pou2f3−/− mice were from the DTCC-KOMP2 Consortium from The Canadian Mouse Mutant Repository. Biostatistics support was provided by the UCSF Functional Genomics Core. This work was supported by NIH grant R01 AI097457 (C.N.M., I.P., I.S.K., T.C.M. and M.S.A.); Larry Hillblom Foundation 2017-D-012-FEL (I.P.); NIH Medical Scientist Training Program grant T32 GM007618 to UCSF (I.S.K.); NSF GRFP DGE 1656518 (K.L.W.); NSF GRFP DGE 1656518 (A.R.R.); NIH grant R37 AI039560 (H.W. and K.A.H.); Damon Runyon Cancer Research Foundation DRG-2162-13 (J.v.M.); Howard Hughes Medical Institute (J.v.M. and R.M.L.); Sandler Asthma Basic Research Center (A.C.F., J.v.M. and R.M.L.); German Cancer Research Center (K.R.); European Research Council grant ERC-2012-AdG (B.K.); NIH grant U01 DK107383 (A.V.P. and M.S.A.); NIH grant P01 HG00020527 (L.M.S.); NIH grant R01 AI026918 (R.M.L.); NIH Diabetes Research Center grant P30 DK063720 (A.C.F., M.S.A. and Single Cell Analysis Center); NIH Shared Instrument Grant 1S10OD021822-01 (Single Cell Analysis Center).
Reviewer information
Nature thanks H.-R. Rodewald and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
C.N.M. conceived the study, designed and performed experiments and analysed data. I.P. conceived and designed experiments with C.N.M., performed thymocyte flow cytometry and analysed data. J.v.M. designed and performed bulk tuft RNA-seq, provided Flare25 and Trpm5−/− mice and analysed data with R.M.L. K.L.W. and A.R.R. designed and performed single-cell RNA-seq and analysed data with L.M.S. H.W. designed and performed F1 experiments and analysed data with K.A.H. K.R. designed and performed flow cytometry on thymic Hipk2-knockout mTECs and analysed data with B.K. I.S.K. and T.C.M. designed and performed mTEC iALT RNA-seq. J.L.P. designed and performed biostatistical analysis of RNA-seq data and analysed data with D.J.E. A.C.F. designed and performed quantitative immunofluorescence image analysis. A.V.P. designed and performed human mTEC experiments. E.J.W. designed and performed immunofluorescence experiments and analysed data. W.W.L. performed RLBA experiments. M.S.A. directed the study and wrote the manuscript with C.N.M. and I.P.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 RNA-seq of FACS-sorted iALT mTEC subsets.
a, Heat map of Aire and representative Aire-dependent TSAs in pre-, early-, late- and post-Aire populations as defined in Fig. 1a. b, Heat map of differentially expressed genes with a FDR < 0.01 and |fold change| > 8. Columns are organized by unsupervised hierarchical clustering (union of all comparisons) with a dendrogram representing similarity between clustered columns. Note that the early- and late-Aire-expressing populations are grouped, as expected.
Extended Data Fig. 2 Medullary localization of DCLK1bright cells and KRT10 bodies.
Representative confocal maximum projections (25 μm) of immunofluorescence staining of thymic slices at low magnification. a, KRT5 (red) and KRT10 (green) (left) and KRT5 (red) and DCLK1 (green) (right) showing medullary localization of KRT10 and DCLK1. b, Left, KRT5 (red) and DCKL1 (green). Right, DCLK1 alone. Region was selected for presence of multicellular DCLK1bright cell clumps as indicated by white arrows. Scale bars, 100 μm. n = 3 mice; images are representative of three independent experiments.
Extended Data Fig. 3 Non-random distribution of medullary DCLK1bright cells.
a, Stitched confocal maximum projection (77 μm) of immunofluorescence staining of semi-thick (200 μm) thymic slice from a C57BL/6 mouse at low magnification. KRT10 (red) and DCLK1 (green). Field, 3,670 × 3,670 µm. Small rectangles indicate volumes selected for quantitative image analysis from this slice. b, Expanded area of image from a indicated in lower left corner with KRT10 signal converted into surfaces and DLCK1 signal converted to centre of intensity coordinates. Scale bar, 100 μm. c, Pair correlation function (PCF) analysis of 12 identically sized regions of interest (586 × 272 × 77 µm) from three different stitched thymic slices (n = 3 thymic slices). Results are presented as a histogram for a range of distances from KRT10 surfaces and are interpreted as follows: g = 1 indicates a spatial distribution that follows a random Poisson distribution, illustrated by a dashed line and grey envelope; g < 1 indicates regularity in the distribution; g > 1 indicates clustering. d, Schematic representation of the PCF analysis. The PCF counts the number of objects from a surface at a radial distance (r) and compares this number to the expected number of events for a random Poisson-distributed population at this distance.
Extended Data Fig. 4 Human thymus contains medullary DCLK1bright cells.
a, Representative immunohistochemistry of neonatal (21-day-old) human thymus stained for DCLK1 or isotype control. Arrows point to individual DCLK1bright cells or clusters. HC, Hassall’s corpuscle. Scale bars, 25 μm. b, Flow cytometry plot from enzymatically digested prenatal human thymus (22.3 gestational weeks) gated on CD45−EPCAM+ TECs showing intracellular DCLK1 or isotype control. Note that human DCLK1+ events are approximately 3.5% of prenatal TECs. Data are representative of two independent experiments.
Extended Data Fig. 5 Characterization of thymic tuft cells.
a, DCLK1 and RFP (IL-25) in the thymus of C57BL/6 control and Flare25 mice. Confocal maximal projection. Scale bar, 5 μm. n = 3 mice, two independent experiments. b, RFP (IL-25) in (CD11c−CD45−EPCAM+) mTECs from C57BL/6 control and Flare25 mice. n = 5 mice; three independent experiments. c, Gating strategy for FACS sorting of CD11c−CD45−EpCAM+Ly51− mTECs from Flare25-reporter mice for qPCR analysis of mTEChigh (RFP−MHC-IIhigh), mTEClow (RFP+MHC-IIlow), and thymic tuft (RFP+) populations. d, qPCR analysis of expression of indicated genes of interest on populations sorted in c normalized to mTEChigh. Data are mean ± s.d. n = 3 mice. Two-way non-parametric ANOVA with multiple comparisons correction. e, Representative confocal maximum projection (10 μm) stained for KRT8/KRT18 (red) and DCLK1 (green). Scale bar, 50 μm. n = 3 mice, two independent experiments. f–i, Expression levels (normalized reads from Fig. 2b) from bulk RNA sequencing of small-intestinal (SI; n = 3 mice) and thymic tuft (n = 4 mice) cells. f, Expression of major MHC-I genes and B2m. FDR > 0.1. g, Expression of major MHC-II genes and Cd74 in small-intestinal tuft cells. h, Expression of minor MHC-II genes in thymic tuft cells. i, Expression of Tas2r family members in thymic tuft cells. g–i, Data are mean ± s.d. g, i, The red line corresponds to a cut-off of 5 reads per million; data for which the mean ± s.d. fall above this cut-off are indicated with an asterisk.
Extended Data Fig. 6 Tas2r expression in single thymic tuft cells is not stochastic.
a, Correlation plot of Tas2r genes in single Flare25 tuft cells as analysed in Fig. 2f. Colour and circle size represent pairwise correlation value. b, Empirical cumulative distribution function (ECDF) plot of pairwise gene expression correlation of single Flare25 tuft cells. Red points represent Tas2r genes that were significantly highly correlated compared to the background gene set (P < 0.05). c, List of correlated Tas2r gene pairs from b and their corresponding empirical P values.
Extended Data Fig. 7 Analysis of Tas2r expression in single Flare25 and Flare25;Aire−/− thymic tuft cells.
a, Intracellular DCLK1 in C57BL/6 control and Aire−/− mTECs (CD11c−CD45−EPCAM+). n = 5 mice; three independent experiments. b, Histogram showing the number of Tas2r family members expressed in each individual thymic tuft cell. Note that the distribution is similar between wild-type and Aire−/− cells. c, Correlation plot of Tas2r genes in single Flare25;Aire−/− tuft cells as analysed in Fig. 3c. Colour and circle size represent pairwise correlation values. d, Empirical cumulative distribution function plot of pairwise gene expression correlation of single Flare25;Aire−/− tuft cells. Red points represent Tas2r genes that were significantly highly correlated compared to the background gene set (P < 0.05). e, List of correlated Tas2r gene pairs from d and their corresponding empirical P values. f, Representative confocal maximum projections (25 μm) of immunofluorescence analysis of C57BL/6 control and Aire−/− thymi stained for KRT5 (red) and KRT10 (green). Scale bars, 100 μm. n = 3 mice; two independent experiments. f, Analysis of DCLK1+ cells in Hipk2fl/fl controls or Foxn1creHipk2fl/fl (mTEC Hipk2−/−) thymus gated on CD45−CDR1−EPCAM+ mTECs as quantified in Fig. 3h. n = 6 mice; three independent experiments.
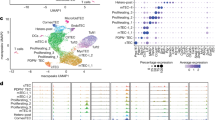
Extended Data Fig. 8 Single-cell RNA-seq data quality and inclusion criteria.
a, Histograms of total read counts (left) or features (right) for each single cell. Red represents Flare25 thymic tuft cells and blue represents Flare25;Aire−/− thymic tuft cells. Cells with fewer than 100,000 reads or 750 detected features were discarded from further analysis. b, Scatter plot of total features versus mitochondrial read percentage. Red represents Flare25 thymic tuft cells and blue represents Flare25;Aire−/− thymic tuft cells. Triangles and circles denote cells from two separate sorts. Cells with more than 10% of their reads mapping to mitochondrial genes were discarded from further analysis as indicated by the black line.
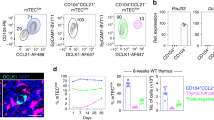
Extended Data Fig. 9 Diphtheria toxin ablation of thymic tuft cells in Kn2 BALB/cByJ × AireDTR C57BL/6 F1 mice disrupts the medullary NKT2–IL-4 axis.
a–d, Analysis of thymus from Kn2 BALB/cByJ × AireDTR C57BL/6 F1 mice or AireDTR transgene-negative F1 controls treated for 9 days with diphtheria toxin. a, Immunofluorescence staining of thin thymic sections for KRT10 (red) and DCLK1 (green). n = 3 mice; two independent experiments. b, Counts of CD11c−CD45−EPCAM+ mTECs and DCLK1+ tuft cells in non-transgenic control thymus (n = 9 mice) or F1 thymus (n = 12 mice). c, Flow cytometry plots gated on TCRβintCD1d+ iNKT cells showing intracellular PLZF and RORγt staining for iNKT subset analysis. Right, counts of NKT1 (PLZF−RORγt−), NKT2 (PLZF+RORγt−) and NKT17 (PLZF−RORγt+) in non-transgenic control thymus (n = 35 mice) or F1 thymus (n = 23 mice). d, Flow cytometry plots gated on TCRβ+CD8+ single-positive thymocytes showing intracellular EOMES staining. Right, counts of EOMES+ cells in non-transgenic control thymus (n = 20 mice) or F1 thymus (n = 18 mice). b–d, Data are mean ± s.d. Unpaired, parametric, two-tailed Student’s t-test. Pooled from three independent experiments.
Extended Data Fig. 10 Characterization of Pou2f3−/− mice.
a, Representative on-edge, full-face mid-thymic sections (5 μm) stained with haematoxylin and eosin showing grossly normal thymic architecture in Pou2f3−/− mice. n = 3 mice; two independent experiments. b, Flow cytometry plots (left) and frequencies (right) of mTEC subsets from C57BL/6 control and Pou2f3−/− mice. n = 5 mice per genotype; three independent experiments. c, Flow cytometry plots (left) and counts (right) of thymocyte subsets from C57BL/6 control and Pou2f3−/− mice. n = 5 mice per genotype; three independent experiments. d, Flow cytometry plots (left) of TCRβintCD1d+ thymic iNKT cells in C57BL/6 control (n = 26 mice), Pou2f3−/− (n = 19 mice) or Trpm5−/− (n = 10 mice) mice. e, Gating strategy of iNKT subset analysis (left) and counts (right) of NKT1 (PLZF−RORγt−) and NKT17 (PLZF−RORγt+) in C57BL/6 control (n = 15 mice), Pou2f3−/− (n = 19 mice) or Trpm5−/− (n = 10 mice) mice. f, Flow cytometry plots gated on TCRβ+CD8+ single-positive thymocytes showing intracellular EOMES staining. Quantified in Fig. 4d. g, Flow cytometry plots gated on splenic TCRβintCD1d+ iNKT cells showing intracellular PLZF and RORγt staining for iNKT subset analysis, NKT1 (PLZF−RORγt−), NKT2 (PLZF+RORγt−) and NKT17 (PLZF−RORγt+). Right, counts of NKT1 and NKT17 in C57BL/6 control (n = 5 mice) or Pou2f3−/− (n = 4 mice) mice. Two independent experiments. b–e, g, Data are mean ± s.d. b, c, g, Unpaired, parametric, two-tailed Student’s t-test. d, e, One-way, non-parametic ANOVA (Kruskal–Wallis test). Pooled from three independent experiments.
Supplementary information
Rights and permissions
About this article
Cite this article
Miller, C.N., Proekt, I., von Moltke, J. et al. Thymic tuft cells promote an IL-4-enriched medulla and shape thymocyte development. Nature 559, 627–631 (2018). https://doi.org/10.1038/s41586-018-0345-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0345-2
This article is cited by
-
Expression of FOXI1 and POU2F3 varies among different salivary gland neoplasms and is higher in Warthin tumor
Discover Oncology (2024)
-
Primary explants of the postnatal thymus allow the expansion of clonogenic thymic epithelial cells that constitute thymospheres
Stem Cell Research & Therapy (2023)
-
An exploratory study for tuft cells in the breast and their relevance in triple-negative breast cancer: the possible relationship of SOX9
BMC Cancer (2023)
-
New insights into the function of Interleukin-25 in disease pathogenesis
Biomarker Research (2023)
-
Bitter taste cells in the ventricular walls of the murine brain regulate glucose homeostasis
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.