Abstract
Extracellular ATP (eATP) is an ancient ‘danger signal’ used by eukaryotes to detect cellular damage1. In mice and humans, the release of eATP during inflammation or injury stimulates both innate immune activation and chronic pain through the purinergic receptor P2RX72,3,4. It is unclear, however, whether this pathway influences the generation of immunological memory, a hallmark of the adaptive immune system that constitutes the basis of vaccines and protective immunity against re-infection5,6. Here we show that P2RX7 is required for the establishment, maintenance and functionality of long-lived central and tissue-resident memory CD8+ T cell populations in mice. By contrast, P2RX7 is not required for the generation of short-lived effector CD8+ T cells. Mechanistically, P2RX7 promotes mitochondrial homeostasis and metabolic function in differentiating memory CD8+ T cells, at least in part by inducing AMP-activated protein kinase. Pharmacological inhibitors of P2RX7 provoked dysregulated metabolism and differentiation of activated mouse and human CD8+ T cells in vitro, and transient P2RX7 blockade in vivo ameliorated neuropathic pain but also compromised production of CD8+ memory T cells. These findings show that activation of P2RX7 by eATP provides a common currency that both alerts the nervous and immune system to tissue damage, and promotes the metabolic fitness and survival of the most durable and functionally relevant memory CD8+ T cell populations.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Heil, M. & Land, W. G. Danger signals — damaged-self recognition across the tree of life. Front. Plant Sci. 5, 578 (2014).
Surprenant, A., Rassendren, F., Kawashima, E., North, R. A. & Buell, G. The cytolytic P2Z receptor for extracellular ATP identified as a P2X receptor (P2X7). Science 272, 735–738 (1996).
Chessell, I. P. et al. Disruption of the P2X7 purinoceptor gene abolishes chronic inflammatory and neuropathic pain. Pain 114, 386–396 (2005).
Mariathasan, S. et al. Cryopyrin activates the inflammasome in response to toxins and ATP. Nature 440, 228–232 (2006).
Williams, M. A. & Bevan, M. J. Effector and memory CTL differentiation. Annu. Rev. Immunol. 25, 171–192 (2007).
Schenkel, J. M. & Masopust, D. Tissue-resident memory T cells. Immunity 41, 886–897 (2014).
Di Virgilio, F., Dal Ben, D., Sarti, A. C., Giuliani, A. L. & Falzoni, S. The P2X7 receptor in infection and inflammation. Immunity 47, 15–31 (2017).
Rissiek, B., Haag, F., Boyer, O., Koch-Nolte, F. & Adriouch, S. P2X7 on mouse T cells: one channel, many functions. Front. Immunol. 6, 204 (2015).
Proietti, M. et al. ATP-gated ionotropic P2X7 receptor controls follicular T helper cell numbers in Peyer’s patches to promote host-microbiota mutualism. Immunity 41, 789–801 (2014).
Trautmann, A. Extracellular ATP in the immune system: more than just a “danger signal”. Sci. Signal. 2, pe6 (2009).
Utzschneider, D. T. et al. T cell factor 1-expressing memory-like CD8+ T cells sustain the immune response to chronic viral infections. Immunity 45, 415–427 (2016).
Im, S. J. et al. Defining CD8+ T cells that provide the proliferative burst after PD-1 therapy. Nature 537, 417–421 (2016).
He, R. et al. Follicular CXCR5- expressing CD8+ T cells curtail chronic viral infection. Nature 537, 412–428 (2016).
Pearce, E. L., Poffenberger, M. C., Chang, C. H. & Jones, R. G. Fueling immunity: insights into metabolism and lymphocyte function. Science 342, 1242454 (2013).
van der Windt, G. J. et al. Mitochondrial respiratory capacity is a critical regulator of CD8+ T cell memory development. Immunity 36, 68–78 (2012).
Buck, M. D., O’Sullivan, D. & Pearce, E. L. T cell metabolism drives immunity. J. Exp. Med. 212, 1345–1360 (2015).
Ledderose, C. et al. Mitochondrial dysfunction, depleted purinergic signaling, and defective T cell vigilance and immune defense. J. Infect. Dis. 213, 456–464 (2016).
Schenk, U. et al. Purinergic control of T cell activation by ATP released through pannexin-1 hemichannels. Sci. Signal. 1, ra6 (2008).
Chang, J. T., Wherry, E. J. & Goldrath, A. W. Molecular regulation of effector and memory T cell differentiation. Nat. Immunol. 15, 1104–1115 (2014).
Sprent, J. & Surh, C. D. Normal T cell homeostasis: the conversion of naive cells into memory-phenotype cells. Nat. Immunol. 12, 478–484 (2011).
Carrio, R., Bathe, O. F. & Malek, T. R. Initial antigen encounter programs CD8+ T cells competent to develop into memory cells that are activated in an antigen-free, IL-7- and IL-15-rich environment. J. Immunol. 172, 7315–7323 (2004).
Buck, M. D. et al. Mitochondrial dynamics controls T cell fate through metabolic programming. Cell 166, 63–76 (2016).
Donnelly-Roberts, D. L. & Jarvis, M. F. Discovery of P2X7 receptor-selective antagonists offers new insights into P2X7 receptor function and indicates a role in chronic pain states. Br. J. Pharmacol. 151, 571–579 (2007).
Saez, P. J. et al. ATP promotes the fast migration of dendritic cells through the activity of pannexin 1 channels and P2X7 receptors. Sci. Signal. 10, eaah7107 (2017)
Blagih, J. et al. The energy sensor AMPK regulates T cell metabolic adaptation and effector responses in vivo. Immunity 42, 41–54 (2015).
Pearce, E. L. et al. Enhancing CD8 T-cell memory by modulating fatty acid metabolism. Nature 460, 103–107 (2009).
Schenkel, J. M. et al. T cell memory. Resident memory CD8 T cells trigger protective innate and adaptive immune responses. Science 346, 98–101 (2014).
Bartlett, R., Stokes, L. & Sluyter, R. The P2X7 receptor channel: recent developments and the use of P2X7 antagonists in models of disease. Pharmacol. Rev. 66, 638–675 (2014).
Matzinger, P. Tolerance, danger, and the extended family. Annu. Rev. Immunol. 12, 991–1045 (1994).
Janeway, C. A., Jr & Medzhitov, R. Innate immune recognition. Annu. Rev. Immunol. 20, 197–216 (2002).
Thompson, E. A., Beura, L. K., Nelson, C. E., Anderson, K. G. & Vezys, V. Shortened intervals during heterologous boosting preserve memory CD8 T cell function but compromise longevity. J. Immunol. 196, 3054–3063 (2016).
Schenkel, J. M., Fraser, K. A., Vezys, V. & Masopust, D. Sensing and alarm function of resident memory CD8+ T cells. Nat. Immunol. 14, 509–513 (2013).
McGaraughty, S. et al. P2X7-related modulation of pathological nocicpetion in rats. Neuroscience. 146, 1817–1828 (2007).
Araki, K. et al. mTOR regulates memory CD8 T-cell differentiation. Nature 460, 108–112 (2009).
Steinert, E. M. et al. Quantifying memory CD8 T cells reveals regionalization of immunosurveillance. Cell 161, 737–749 (2015).
Skon, C. N. et al. Transcriptional downregulation of S1pr1 is required for the establishment of resident memory CD8+ T cells. Nat. Immunol. 14, 1285–1293 (2013).
Renkema, K. R. et al. IL-4 sensitivity shapes the peripheral CD8+ T cell pool and response to infection. J. Exp. Med. 213, 1319–1329 (2016).
Daniels, M. A. & Jameson, S. C. Critical role for CD8 in T cell receptor binding and activation by peptide/major histocompatibility complex multimers. J. Exp. Med. 191, 335–346 (2000).
Anderson, K. G. et al. Intravascular staining for discrimination of vascular and tissue leukocytes. Nat. Protocols 9, 209–222 (2014).
Schenkel, J. M. et al. IL-15-independent maintenance of tissue-resident and boosted effector memory CD8 T cells. J. Immunol. 196, 3920–3926 (2016).
Kieper, W. C. & Jameson, S. C. Homeostatic expansion and phenotypic conversion of naïve T cells in response to self peptide/MHC ligands. Proc. Natl Acad. Sci. USA 96, 13306–13311 (1999).
Praetorius, H. A. & Leipziger, J. ATP release from non-excitable cells. Purinergic Signal. 5, 433–446 (2009).
Decosterd, I. & Woolf, C. J. Spared nerve injury: an animal model of persistent peripheral neuropathic pain. Pain 87, 149–158 (2000).
Bourquin, A. F. et al. Assessment and analysis of mechanical allodynia-like behavior induced by spared nerve injury (SNI) in the mouse. Pain 122, 14.e11-14 (2006).
Chaplan, S. R., Bach, F. W., Pogrel, J. W., Chung, J. M. & Yaksh, T. L. Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods 53, 55–63 (1994).
Acknowledgements
We thank the UMN Flow Cytometry Resource for cell sorting, C. Henzler (UMN Supercomputing Institute) for bioinformatics analysis, F. Zhou (UMN Characterization Facility) for transmission electron microscopy, M. Pierson for viral plaque assays, the NIH Tetramer Core for peptide/MHC tetramers, and A. Goldrath, S. Kaech, G. Shadel, R. Jones, E. Pearce, M. Jenkins, V. Vezys and members of the Jamequist laboratory and UMN Center for Immunology for discussions. The UMN Characterization Facility is a member of the NSF-funded Materials Research Facilities Network (https://www.mrfn.org) via the MRSEC program. This work was supported by NIH grants AI38903 and AI75168 (S.C.J.), CA157971 (A.K.), and MN Partnership Infrastructure Award MNPIF#16.09 (A.K.). H.B.d.S. was supported by a CNPq research fellowship from the Ministry of Science, Technology and Innovation of Brazil.
Reviewer information
Nature thanks F. Grassi, J. Linden and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
S.C.J. and H.B.d.S. designed, analysed and interpreted the experiments. H.B.d.S., L.K.B., H.W., E.A.H., R.G., M.C.S., D.A.W., K.E.B., R.F. and Y.Y. performed experiments. R.G. performed spared nerve injury surgical procedures. E.A.H. and A.K. provided assistance with extracellular flux analysis. K.L.H., B.R.B., D.M., A.K., L.V. and K.A.H. contributed critical reagents and biological samples. S.C.J. and H.B.d.S. wrote the manuscript, with all authors contributing to editing the final text.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
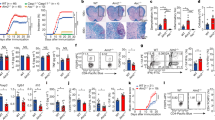
Extended Data Fig. 1 P2RX7 is required for CD103high TRM and TCM cell generation upon acute viral infection, and for establishment of Ag-specific CD8+ T cells upon chronic viral infection.
a–e, Wild-type and P2rx7−/− P14 CD8+ T cells were mixed 1:1 and co-adoptively transferred into B6.SJL mice that were subsequently infected with LCMV, and donor cells were identified as in Fig. 1. Data from 3–4 independent experiments, n = 4 FRT and n = 5–7 other organs from all experiments. a, Ratios of P2rx7−/− to wild-type P14 cells in different non-lymphoid organs over time. b, Flow cytometric plots showing CD69 and CD103 co-expression by wild-type and P2rx7−/− P14 cells from SI-IELs and SI LP (small intestine lamina propria) 4 weeks post-infection (representative of three experiments). c, Ratios of P2rx7−/− to wild-type P14 cells for CD103high, CD103int and CD103low subsets among SI LP over time. d, e, We also evaluated the role of P2RX7 in the generation and maintenance of memory CD8+ T cell subsets based on CX3CR1 expression. d, Representative plot depicting the subpopulations studied (left) and P2RX7 median in these subsets (right). e, Ratio of P2rx7−/− to wild-type P14 CD8+ T cells in spleen, gated on indicated subsets. Data from 2–3 independent experiments, n = 4–7 mice total. f, CD8+ T cells from wild-type and P2rx7−/− OT-I TCR transgenic (OT-I) mice were mixed 1:1 and co-adoptively transferred into B6 mice subsequently infected with VSV-OVA (two independent experiments, n = 7 from all experiments). Ratio of wild-type to P2rx7−/− OT-I cells in indicated tissues was determined 4 weeks after VSV-OVA infection. g, The indicated radiation bone marrow chimaeras were generated and infected with LCMV. Percentages of splenic Db/gp33-tetramer binding (gp33+) CD8+ TCM cells were determined 8 weeks post-infection with LCMV (data from two independent experiments, n = 5 from all experiments). h, Wild-type or P2rx7−/− mice were infected with LCMV-Arm or LCMV-Cl13, and the numbers (left) and percentages of CXCR5+PD1low (right) gp33+ CD8+ T cells were evaluated 4 weeks after infection (n = 6–13 from all experiments). a, c–h, Mean ± s.e.m.; g, h, two-tailed Student’s t-test, *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001.
Extended Data Fig. 2 Long-lived memory CD8+ T cells express higher levels of functional P2RX7.
a, Flow cytometry histograms for P2RX7 staining on naive wild-type or P2rx7−/− P14 cells (representative of three experiments). b, Median values for P2RX7 staining on naive wild-type and P2rx7−/− P14 cells, and on wild-type P14 cells 7 d post-LCMV infection (data from three independent experiments, n = 10 total). c–h, Indicated subsets of adoptively transferred wild-type P14 CD8+ T cells from listed tissues were assayed for P2RX7 expression and functional response to the P2RX7 agonist BzATP following priming with LCMV for the indicated time (data from three independent experiments, n = 6–21 from all experiments). c, d, Percentage of P2RX7high wild-type P14 cells in MPEC and SLEC (c) and TCM and TEM subsets (d) four weeks after infection. e, P2RX7 median fold increase (relative to expression in naive CD8+ T cells, showed as a dashed line) of P14 cells in blood, lymphoid and non-lymphoid organs (four weeks after infection). f, P2RX7 median fold increase (relative to naive CD8+ T cells) of CD103+ versus CD103− P14 cells in SI-IELs and SI LP. g, h, Adoptively transferred and LCMV primed wild-type and P2rx7−/− P14 cells were incubated with DAPI then stimulated during flow cytometry with 300 μM BzATP (which mediates P2RX7 pore opening and DAPI uptake). g, Flow cytometry plots of DAPI uptake by SI-IELs and SI LP wild-type and P2rx7−/− P14 CD8+ T cells over 30 min (representative of six samples). h, Compiled data for percentage DAPI+ P14 cells (defined as the percentage above P2rx7−/− DAPI levels 5 min after BzATP stimulation) in spleen, SI IEL and SI LP P14 cells (n = 6 from all experiments). b–f, h, Mean ± s.e.m.; b, one-way ANOVA with Tukey’s post-test; c, d, f, h, two-tailed Student’s t-test; *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001.
Extended Data Fig. 3 P2RX7 is required for optimal metabolism of MPECs and TCM cells.
Wild-type and P2rx7−/− P14 CD8+ T cells were mixed 1:1 and co-adoptively transferred into B6.SJL mice that were subsequently infected with LCMV. Donor cells were identified as in Fig. 1. a, Numbers of wild-type and P2rx7−/− P14 MPECs and SLECs in spleens (three independent experiments, n = 5–6 from all experiments). b, Gene expression profile from wild-type versus P2rx7−/− P14 MPECs (sorted two weeks post-infection with LCMV). Gene expression in SLEC populations, sorted and analysed at the same time, also showed minimal differences between wild-type and P2rx7−/− groups (data not shown). c–g, At day 8 (c, e, f) or 14 (d, g), splenic wild-type and P2rx7−/− MPEC and SLEC subpopulations were stained for MTG (c), Cox-IV (d), TMRE (e) or 2-NBDG uptake (f, g); two independent experiments, n = 4–5 from all experiments. In h, the ECAR levels in MPECs and SLECs (from the experiments described in Fig. 2d–f) are shown (three independent experiments, n = 8–12). i–l, Four weeks after priming with LCMV, wild-type and P2rx7−/− P14 CD8+ TCM and TEM subsets were assessed for expression of Cox-IV (i), Glut1 (j) or CPT1a (l), or uptake of Bodipy (k). j–l, Left, representative flow cytometric plots for TCM populations (relative to naive wild-type host CD8+ T cells); right, median difference in staining relative to naive wild-type host CD8+ T cells. i–l, Three independent experiments, n = 5–10 from all experiments. a, c–l, Mean ± s.e.m.; a, c, e–l, two-tailed Student’s t-test; d, one-way ANOVA with Tukey’s post-test; *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001.
Extended Data Fig. 4 P2RX7 signalling is required for survival rather than homeostatic proliferation of long-lived memory CD8+ T cells.
a–g, Wild-type (black) and P2rx7−/− (red) P14 CD8+ T cells were mixed 1:1 and co-adoptively transferred into B6.SJL mice that were subsequently infected with LCMV, and donor cells were identified as in Fig. 1 (data from three independent experiments, n = 4–10 from all experiments). a–d, Percentages of Ki-67+ cells (ex vivo staining; a), BrdU+ cells (b) and annexin V+ Live–Dead+ cells (c), and BCL2 median fold increase (relative to median values of naive CD8+ T cells) for bulk, TCM and TEM subsets (d) was determined for wild-type and P2rx7−/− P14 CD8+ T cells eight weeks after infection. e, Median fold increase (relative to naive CD8+ T cells) in expression of TCF1 (left), BCL2 (centre) and EOMES (right) in wild-type and P2rx7−/− P14 MPEC and SLEC CD8+ T cells at the indicated times. f, Expression of TCF1 (left) and EOMES (right) (shown as median expression relative to naive CD8+ T cells) in splenic TEM and TCM subsets of wild-type and P2rx7−/− P14 CD8+ T cells, four weeks after infection. g, CD127 (left) and CD122 (right) median expression (relative to naive CD8+ T cells) for splenic bulk cells and TCM and TEM subsets four weeks after infection. h, i, Wild-type and P2rx7−/− P14 CD8+ T cells were mixed 1:1 and co-adoptively transferred into B6.SJL or IL-15−/− mice, which were subsequently infected with LCMV (data from two independent experiments, n = 4–5 from all experiments). h, Numbers of wild-type and P2rx7−/− P14 CD8+ T cells in each host (four weeks after infection). i, Ratio of total and TCM P2rx7−/− to wild-type P14 CD8+ T cells in spleens from wild-type and IL-15−/− hosts at indicated times post-infection. j, k, Congenically distinct wild-type and P2rx7−/− P14 CD8+ T cells were stained with CFSE, mixed 1:1 and co-adoptively transferred into Rag2−/− mice. Data from two independent experiments, n = 6 total. j, Ratio of P2rx7−/− to wild-type P14 CD8+ T cells in the blood of Rag2−/− hosts at indicated times post-transfer. k, Percentages of P2rx7−/− and wild-type P14 CD8+ T cells proliferating over four cycles in spleens of Rag2−/− hosts three weeks after transfer. All data shown as mean ± s.e.m. a–d, f–h, k, Two-tailed Student’s t-test; e, two-way ANOVA with Bonferroni’s post-test; *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001.
Extended Data Fig. 5 Defective metabolism of IL-15-polarized CD8+ T cells in the absence of P2RX7.
a, Viability of wild-type and P2rx7−/− P14 cells maintained under culture conditions used for extracellular flux assays for 4 h (data from two independent experiments, n = 11 from all experiments). b–d, Wild-type or P2rx7−/− P14 cells were activated in vitro and subsequently polarized in IL-2 or IL-15 for 72 h and assayed for ECAR to measure aerobic glycolysis (b); uptake of TMRE to measure mitochondrial membrane potential (data normalized to MTG staining; c); and staining with MTG to determine total mitochondrial mass (d). Data from three independent experiments, n = 5–6 from all experiments. e, Wild-type and P2rx7−/− P14 cells were activated in vitro for 72 h, then stimulated with IL-15 (or not) for 30 min and immediately assayed for pSTAT5 expression. Data are representative of two independent experiments (n = 6 total). f, Wild-type and P2rx7−/− P14 cells were activated in vitro for 72 h, then stimulated with IL-15 or IL-2 for 72 h and assayed for expression of CD62L. Data are representative of three independent experiments (n = 6 total). g, Numbers of viable wild-type and P2rx7−/− P14 cells following activation and subsequent culture in IL-15 or IL-2 for the indicated number of days. Values for wild-type and P2rx7−/− cells were not significantly different (P > 0.05). Data from three independent experiments (n = 3–6 total). a–d, g, Mean ± s.e.m.; a, c, d, g, two-tailed Student’s t-test; **P ≤ 0.01.
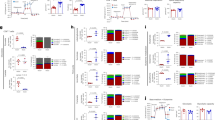
Extended Data Fig. 6 P2RX7 controls mitochondrial integrity in CD8+ T cells during immune responses.
a, Left, representative electron micrographs showing mitochondrial structures; right, mitochondrial area measurements of in vitro-activated wild-type and P2rx7−/− P14 cells following culture for 72 h in IL-2 or IL-15. Representative of 3 independent experiments (n = 8–9 in total). Black bars indicate 500 nm. b, c, Wild-type and P2rx7−/− P14 cells were activated in vitro for 72 h and assayed at that time (b) or after a further 72 h of culture in IL-2 or IL-15 (as in Fig. 3b) (c) for OCR. Graphs show values for proton leak (the difference in OCR values after oligomycin and after antimycin A/rotenone addition; Fig. 3b). Data are from three independent experiments (n = 11–18 total). d, Calculated proton leak derived from OCR measurements on in vivo activated wild-type and P2rx7−/− P14 CD8+ MPECs described in Fig. 2d–f (data from three independent experiments, data pooled from five mice per experiment; n = 4–6 wells in total). e–h, Wild-type and P2rx7−/− P14 cells were activated in vitro and polarized with either IL-2 or IL-15 (as in Fig. 3b); total cell (e–g) and mitochondrial (g, h) protein extracts were collected for protein quantification experiments. Data from three independent experiments, samples pooled from n = 6 mice total (2 mice per experiment). e, Representative blot showing Opa1 expression in polarized wild-type or P2rx7−/− P14 cells, in comparison with β-actin (for gel source data, see Supplementary Fig. 1). f, Opa1 protein levels in polarized wild-type or P2rx7−/− P14 cells, normalized to β-actin. g, Mitochondrial concentration (normalized by total protein concentration) in polarized wild-type or P2rx7−/− P14 cells. h, Opa1 protein levels in polarized wild-type or P2rx7−/− P14 cells, normalized to total mitochondrial concentration. a–d, f–h, Mean ± s.e.m.; a, b, d, two-tailed Student’s t-test; c, f–h, two-sided Mann–Whitney’s test; *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001.
Extended Data Fig. 7 Normal metabolic function of naive P2rx7−/− P14 CD8+ T cells, while pharmacological inhibition of P2RX7 compromises aerobic glycolysis of in vitro-activated CD8+ T cells.
a–c, Naive wild-type and P2rx7−/− P14 cells were isolated and evaluated for different metabolism parameters (data from three independent experiments, n = 6 total). d, e, Human CD8+ T cells were activated in vitro, with the P2RX7 inhibitor A-438079 or vehicle control added 20 h after initiation of the culture, and assessed at 72 h for ECAR (d) or for viability of cells cultured in parallel under the same conditions as those used for extracellular flux assays (e). d, e, Data are from three independent experiments, n = 4–6 from all experiments. All data shown as mean ± s.e.m.; b, c, e, Two-tailed Student’s t-test.
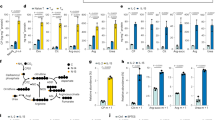
Extended Data Fig. 8 P2RX7-mediated eATP sensing is crucial for optimal CD8+ T cell immunometabolism by regulation of the AMPK–mTOR pathway.
a–g, Wild-type and P2rx7−/− P14 cells were activated in vitro and polarized with IL-15 (as in Fig. 3b). Data from three independent experiments, samples pooled from n = 6 mice per experiment; n = 3–12 total samples. a–c, Numbers (left) and viability (right) of P14 cells in cultures supplemented with apyrase (a), oATP (b) or BzATP (c) during cell culture. d, e, g, IL-15-polarized wild-type or P2rx7−/− P14 cells were assayed for OCR 1 h after addition of apyrase (d) or oATP (e), or 6 h after addition of A-438079 (g). f, IL-15-polarized cells or ex vivo wild-type P14 TCM cells (isolated 4 weeks after LCMV infection) were incubated with DAPI (left) or Indo-1 (right) and stimulated with the indicated concentrations of BzATP during kinetic flow cytometric analysis. The percentage of cells showing DAPI uptake (left) or Ca2+ influx (right) over 30 min are shown. f, Data from two independent experiments, samples pooled from n = 5 mice total; n = 2–5 samples. h, In vitro-activated (72 h) wild-type and P2rx7−/− P14 cells were assayed for intracellular ATP concentrations. Data from three independent experiments, n = 9 total. i, In vitro-activated, IL-15 polarized (24 h post-polarization) wild-type or P2rx7−/− P14 cells were assayed for extracellular ATP concentration, following culture without or with the Panx1 inhibitor 10Panx. Data from two independent experiments, n = 3–4 total samples (pooled from six mice). j, Wild-type and P2rx7−/− P14 cells were co-adoptively transferred and assayed 4 weeks after LCMV infection (as in Fig. 1a) and the ex vivo frequency of pS6-expressing cells was determined by flow cytometry. Data are from two independent experiments (n = 6 total). k, Expression of pACC in IL-15-polarized wild-type (black) and P2rx7−/− (red) P14 cells (relative to Fig. 3l; representative of three independent experiments, n = 6 total). l, In vitro-activated and IL-15-polarized wild-type and P2rx7−/− P14 cells were cultured for 6 h with the indicated concentrations of BzATP, then stained for pACC (left) and pS6 (centre), and the pACC/pS6 ratio was determined (right). Data from three independent experiments, n = 6–8 total. m, In vitro-activated wild-type and P2rx7−/− P14 cells were IL-15-polarized in the presence or absence of AICAR as in Fig. 3l. The percentage of viable cells at the indicated times following initiation of IL-15 culture with or without AICAR is indicated. Data are from three independent experiments (n = 3–6 total; samples pooled from n = 6 mice total). n–p, Wild-type and P2rx7−/− P14 CD8+ T cells were mixed 1:1 and co-adoptively transferred into B6.SJL mice that were subsequently infected with LCMV, and donor cells identified as in Fig. 1. The mice were treated with metformin or PBS control during the first week of LCMV infection, and the cells were analysed at day 30. Data are compiled from three independent experiments (n = 11–12 total, n = 4 for FRT samples). n, Relates to Fig. 3m; ratio of P2rx7−/− to wild-type P14 cells in the indicated non-lymphoid tissues (n = 9 except FRT, n = 4). o, p, Measurements of mitochondrial mass (measured using MTG) (o) and mitochondrial membrane potential (measured by TMRE staining, normalized to MTG staining) (p) for the indicated splenocyte subsets (n = 3–6 total samples). q, Wild-type and P2rx7−/− P14 CD8+ T cells were mixed 1:1 and co-adoptively transferred into B6.SJL mice that were subsequently infected with LCMV, and donor cells were identified as in Fig. 1. The mice were treated with rapamycin or PBS control between days 4 and 8 post-LCMV infection, and the cells were analysed at day 30. The numbers of wild-type and P2rx7−/− P14 cells are shown (log-transformed). Data are compiled from three independent experiments (n = 15 total). a–j, l–q, Mean ± s.e.m.; a–c, h–j, m–p, two-tailed Student’s t-test; l, q, one-way ANOVA with Tukey’s post-test; *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001.
Extended Data Fig. 9 P2RX7 deficiency compromises memory CD8+ T cell function, and P2RX7 pharmacological blockade impairs generation of CD8+ T cell memory cells in vivo.
a, Wild-type or P2rx7−/− mice were infected with LCMV-Cl13, and kidney PFU levels were quantified 4 weeks later. Data from 3 independent experiments, n = 7 total. b–f, Wild-type and P2rx7−/− P14 CD8+ T cells were mixed 1:1 and co-adoptively transferred into B6.SJL mice that were subsequently infected with LCMV and then challenged (or not) with Lm-gp33 6–8 weeks later (b). Data are from 2 independent experiments (n = 5–7 total). c, Ratio of P2rx7−/− to wild-type splenic P14 CD8+ T cells before (0 d) and after challenge (5 d). d, Fold increase in numbers of wild-type or P2rx7−/− P14 cells in indicated tissues, relative to mice that did not receive Lm-gp33 challenge. e, Percentage of cells in cell cycle, determined by Ki-67 staining. f, Frequency of dying cells indicated by the percentage of annexin V+Live–Dead+ cells in mice 5 d after Lm-gp33 challenge. g, h, Wild-type and P2rx7−/− P14 CD8+ T cells were individually transferred into B6.SJL mice that were subsequently infected with LCMV. After 6–8 weeks, the mice were transcervically challenged with gp33 or PBS as in Fig. 4d. g, Flow cytometry plots for IFN-γ production by wild-type or P2rx7−/− P14 cells in mice treated with PBS or gp33. h, Percentage of IFN-γ+ bystander (non-P14) CD8+ T cells (left) and percentage of CCR7+CD86+ dendritic cells (right) in the FRT 12 h later. g, h, Data are from three independent experiments, n = 4–11 total. i, Schematic of experimental scheme combining spared nerve injury (SNI), LCMV infection and A-438079 treatment. For surgery, two of the three branches of the sciatic nerve in one hind limb were exposed and cut (Fig. 4) or left uncut (sham). After 2 weeks mice were assayed for pain sensitivity, then infected with LCMV with or without A-438079 treatment for the first week after infection. Mice were assayed again for pain sensitivity (day 7) and subsequent development of central (CD62L+) and effector (CD62L−) memory cells specific for the LCMV epitope gp33 (after day 30). Data are compiled from two independent experiments, n = 6 from all experiments. j, Pain sensitivity of sham-surgery mice (pre- and post-treatment). k, Percentages of gp33-specific TCM cells (left) and numbers of gp33-specific TCM and TEM cells (right) in sham surgery animals. l–p, In other studies, B6 or Balb/C mice were adoptively transferred or not with wild-type P14 cells, infected with LCMV, and treated with A-438079 in the time frames indicated, relative to infection. Data from 2–3 independent experiments, n = 5–10 total. m, Percentages of CD62L+CD44+ (TCM) P14 cells per spleen (left) and spleen P14 TCM and TEM cell numbers (right) from the different treatment groups at 4 weeks post-infection. n, P14 recipient mice were treated with PBS or A-438079 for the first week following LCMV infection, then assayed at 3 weeks for numbers of MPECs and SLECs. o, Balb/C mice were infected with LCMV, treated with A-438079 throughout the first week post-infection, and the numbers of LCMV-specific (NPtet+) CD8+ T cells were quantified in the spleen, lymph nodes and SI-IEL at 4 weeks post-infection. p, B6 mice infected with LCMV were treated with A-438079 between days 40 and 50 post-infection and the numbers of gp33+ CD8+ T cells (TCM and TEM) per spleen were quantified at 8 weeks post-infection. Data shown as mean ± s.e.m.; a, c–f, k, n–p, two-tailed Student’s t-test; j, two-sided Mann–Whitney’s test; h, m, one-way ANOVA with Tukey’s post-test; *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001.
Extended Data Fig. 10 Model of P2RX7 function in regulating long-lived CD8+ T cell memory.
a, After activation of CD8+ T cells, P2RX7 is stimulated by eATP (derived from damaged cells or exported from live activated cells). This induces calcium influx, increased mitochondrial metabolic activity and activation of the ATP export channel PANX1 (bold arrows). Generation of eATP through PANX1 sustains P2RX7 activation, further promoting mitochondrial function and T cell homeostasis. b, The effect of P2RX7 function is magnified as the CD8+ T cell response progresses into memory phase, with P2RX7 deficiency having little effect on effector T cells, while severely compromising survival and metabolic function in memory T cells, impairing the generation of long-lived central memory (TCM) and CD103highCD69high resident memory (TRM) populations. P2RX7 activation is proposed to stimulate AMPK activation, which may arise from both calcium influx and increased AMP/ATP ratios as a result of P2RX7 and PANX1 activity. A requirement for eATP stimulation of P2RX7 persists throughout memory CD8+ T cell maintenance.
Supplementary information
Supplementary Figures 1-2
This file contains the original western blot and the flow cytometry gating strategy
Supplementary Data
This file contains a list of P values for all figures
Rights and permissions
About this article
Cite this article
Borges da Silva, H., Beura, L.K., Wang, H. et al. The purinergic receptor P2RX7 directs metabolic fitness of long-lived memory CD8+ T cells. Nature 559, 264–268 (2018). https://doi.org/10.1038/s41586-018-0282-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0282-0
This article is cited by
-
Cancer immunometabolism: advent, challenges, and perspective
Molecular Cancer (2024)
-
Long-term activation of anti-tumor immunity in pancreatic cancer by a p53-expressing telomerase-specific oncolytic adenovirus
British Journal of Cancer (2024)
-
Bone marrow plasma cells require P2RX4 to sense extracellular ATP
Nature (2024)
-
The P2X7 receptor in mucosal adaptive immunity
Purinergic Signalling (2024)
-
Immunogenic cell death-based prognostic model for predicting the response to immunotherapy and common therapy in lung adenocarcinoma
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.