Abstract
Malignancy is accompanied by changes in the metabolism of both cells and the organism1,2. Pancreatic ductal adenocarcinoma (PDAC) is associated with wasting of peripheral tissues, a metabolic syndrome that lowers quality of life and has been proposed to decrease survival of patients with cancer3,4. Tissue wasting is a multifactorial disease and targeting specific circulating factors to reverse this syndrome has been mostly ineffective in the clinic5,6. Here we show that loss of both adipose and muscle tissue occurs early in the development of pancreatic cancer. Using mouse models of PDAC, we show that tumour growth in the pancreas but not in other sites leads to adipose tissue wasting, suggesting that tumour growth within the pancreatic environment contributes to this wasting phenotype. We find that decreased exocrine pancreatic function is a driver of adipose tissue loss and that replacement of pancreatic enzymes attenuates PDAC-associated wasting of peripheral tissues. Paradoxically, reversal of adipose tissue loss impairs survival in mice with PDAC. When analysing patients with PDAC, we find that depletion of adipose and skeletal muscle tissues at the time of diagnosis is common, but is not associated with worse survival. Taken together, these results provide an explanation for wasting of adipose tissue in early PDAC and suggest that early loss of peripheral tissue associated with pancreatic cancer may not impair survival.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Koppenol, W. H., Bounds, P. L. & Dang, C. V. Otto Warburg’s contributions to current concepts of cancer metabolism. Nat. Rev. Cancer 11, 325–337 (2011).
Petruzzelli, M. & Wagner, E. F. Mechanisms of metabolic dysfunction in cancer-associated cachexia. Genes Dev. 30, 489–501 (2016).
Dewys, W. D. et al. Prognostic effect of weight loss prior to chemotherapy in cancer patients. Am. J. Med. 69, 491–497 (1980).
Mueller, T. C., Bachmann, J., Prokopchuk, O., Friess, H. & Martignoni, M. E. Molecular pathways leading to loss of skeletal muscle mass in cancer cachexia—can findings from animal models be translated to humans? BMC Cancer 16, 75 (2016).
Fearon, K., Arends, J. & Baracos, V. Understanding the mechanisms and treatment options in cancer cachexia. Nat. Rev. Clin. Oncol. 10, 90–99 (2013).
Penna, F. et al. Anti-cytokine strategies for the treatment of cancer-related anorexia and cachexia. Expert Opin. Biol. Ther. 10, 1241–1250 (2010).
Flint, T. R. et al. Tumor-induced IL-6 reprograms host metabolism to suppress anti-tumor immunity. Cell Metab. 24, 672–684 (2016).
Hingorani, S. R. et al. Preinvasive and invasive ductal pancreatic cancer and its early detection in the mouse. Cancer Cell 4, 437–450 (2003).
Mayers, J. R. et al. Elevation of circulating branched-chain amino acids is an early event in human pancreatic adenocarcinoma development. Nat. Med. 20, 1193–1198 (2014).
Agustsson, T. et al. Mechanism of increased lipolysis in cancer cachexia. Cancer Res. 67, 5531–5537 (2007).
Michaelis, K. A. et al. Establishment and characterization of a novel murine model of pancreatic cancer cachexia. J. Cachexia Sarcopenia Muscle 8, 824–838 (2017).
Rydén, M. et al. Lipolysis—not inflammation, cell death, or lipogenesis—is involved in adipose tissue loss in cancer cachexia. Cancer 113, 1695–1704 (2008).
Shaw, J. H. & Wolfe, R. R. Fatty acid and glycerol kinetics in septic patients and in patients with gastrointestinal cancer. The response to glucose infusion and parenteral feeding. Ann. Surg. 205, 368–376 (1987).
Fearon, K. et al. Definition and classification of cancer cachexia: an international consensus. Lancet Oncol. 12, 489–495 (2011).
Hwang, R. F. et al. Cancer-associated stromal fibroblasts promote pancreatic tumor progression. Cancer Res. 68, 918–926 (2008).
Herrington, M. K., Arnelo, U. & Permert, J. On the role of islet amyloid polypeptide in glucose intolerance and anorexia of pancreatic cancer. Pancreatology 1, 267–274 (2001).
Martin, L. et al. Cancer cachexia in the age of obesity: skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J. Clin. Oncol. 31, 1539–1547 (2013).
Mourtzakis, M. et al. A practical and precise approach to quantification of body composition in cancer patients using computed tomography images acquired during routine care. Appl. Physiol. Nutr. Metab. 33, 997–1006 (2008).
Choi, Y. et al. Skeletal muscle depletion predicts the prognosis of patients with advanced pancreatic cancer undergoing palliative chemotherapy, independent of body mass index. PLoS ONE 10, e0139749 (2015).
Prado, C. M. et al. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: a population-based study. Lancet Oncol. 9, 629–635 (2008).
Rollins, K. E. et al. The impact of sarcopenia and myosteatosis on outcomes of unresectable pancreatic cancer or distal cholangiocarcinoma. Clin. Nutr. 35, 1103–1109 (2016).
Tan, B. H., Birdsell, L. A., Martin, L., Baracos, V. E. & Fearon, K. C. H. Sarcopenia in an overweight or obese patient is an adverse prognostic factor in pancreatic cancer. Clin. Cancer Res. 15, 6973–6979 (2009).
Vujasinovic, M., Valente, R., Del Chiaro, M., Permert, J. & Löhr, J. M. Pancreatic exocrine insufficiency in pancreatic cancer. Nutrients 9, 183 (2017).
Lv, M., Zhu, X., Wang, H., Wang, F. & Guan, W. Roles of caloric restriction, ketogenic diet and intermittent fasting during initiation, progression and metastasis of cancer in animal models: a systematic review and meta-analysis. PLoS ONE 9, e115147 (2014).
Laquente, B. et al. Supportive care in pancreatic ductal adenocarcinoma. Clin. Transl. Oncol. 19, 1293–1302 (2017).
Apte, M. Isolation of quiescent pancreatic stellate cells from rat and human pancreas. Pancreapedia: Exocrine Pancreas Knowledge Base https://doi.org/10.3998/panc.2011.10 (2011).
Loftus, S. K. et al. Acinar cell apoptosis in Serpini2-deficient mice models pancreatic insufficiency. PLoS Genet. 1, e38 (2005).
DiStefano, M. T. et al. The lipid droplet protein hypoxia-inducible gene 2 promotes hepatic triglyceride deposition by inhibiting lipolysis. J. Biol. Chem. 290, 15175–15184 (2015).
Gabbi, C. et al. Pancreatic exocrine insufficiency in LXRβ−/− mice is associated with a reduction in aquaporin-1 expression. Proc. Natl Acad. Sci. USA 105, 15052–15057 (2008).
Kraus, D., Yang, Q. & Kahn, B. B. Lipid extraction from mouse feces. Bio Protoc. 5, e1375 (2015).
Yuan, C. et al. Survival among patients with pancreatic cancer and long-standing or recent-onset diabetes mellitus. J. Clin. Oncol. 33, 29–35 (2015).
Yuan, C. et al. Prediagnostic body mass index and pancreatic cancer survival. J. Clin. Oncol. 31, 4229–4234 (2013).
DerSimonian, R. & Laird, N. Meta-analysis in clinical trials. Control. Clin. Trials 7, 177–188 (1986).
Acknowledgements
We thank members of the Vander Heiden and Wolpin laboratories for discussions and the Koch Institute Swanson Biotechnology Center, particularly the Animal Imaging and Preclinical Testing Facility, for technical assistance. Major funding for this work was provided by the Lustgarten Foundation to B.M.W. and M.G.V.H. L.V.D. was supported by NIH Ruth Kirschstein Fellowship (F32CA210421). A.B. was supported by P50CA127003 and the Robert T. and Judith B. Hale Fund for Pancreatic Cancer Research. A.M. was supported by F32CA213810. E.C.L. was supported by the Damon Runyon Cancer Research Foundation (DRG-2299-17). A.N.L. is a Robert Black Fellow of the Damon Runyon Cancer Research Foundation (DRG-2241-15). B.M.W was supported by Robert T. and Judith B. Hale Fund for Pancreatic Cancer Research, NIH/NCI (U01CA210171), Department of Defense (CA130288), Pancreatic Cancer Action Network, Stand Up To Cancer, Noble Effort Fund, Peter R. Leavitt Family Fund, Wexler Family Fund, and Promises for Purple. M.G.V.H. was supported in part by a Faculty Scholar grant from the Howard Hughes Medical Institute, and acknowledges additional funding from Stand Up To Cancer, The Ludwig Center at MIT, the Koch Institute Frontier Awards, the MIT Center for Precision Cancer Medicine, and the NIH (R01CA168653, P30CA14051).
Reviewer information
Nature thanks M. Löhr and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
L.V.D. designed, performed and analysed the animal experiments with input from M.G.V.H.; L.V.D., A.B., B.M.W. and M.G.V.H. wrote the manuscript with assistance from all other authors. E.A.D. and P.J.-S. assisted with animal experimentation. A.M. immortalized and A.N.L. isolated PSCs. E.C.L. performed caloric restriction experiments. J.R.M. performed muscle volume measurements and blood measurements. K.T. performed non-esterified fatty acid and glycerol assays. A.B., M.H.R., C.B.C. and B.M.W. designed the human study. C.M.P., G.M.P., N.T., M.S., J.J.Y., N.L., N.B., C.F.-d.C., A.S.L., A.C.K., J.B., C.Y., M.W.W., L.K.B., M.H.K. and B.M.W. were involved in patient recruitment and patient data collection. C.D. and C.B.C. were involved in metabolite measurements in patients. A.B. and M.H.R. analysed human data. B.M.W. supervised the human study.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests; however, M.G.V.H. discloses serving on the S.A.B. of Agios Pharmaceuticals and Aeglea Biotherapeutics.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
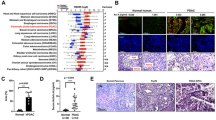
Extended Data Fig. 1 PDAC is associated with adipose and skeletal muscle wasting.
a, Circulating BCAAs (valine, leucine, and isoleucine) in male control (n = 12) and early KP−/−C (n = 10) mice. b, Representative histology of H&E-stained gastrocnemius skeletal muscle of control and early KP−/−C mice. n = 4 per group. c, Relative myofibre area in male control and early KP−/−C mice. n = 3 per group. *P < .0001. d, Representative 3D μCT imaging reconstruction of soleus and gastrocnemius skeletal muscle (highlighted in red). e, Relative soleus and gastrocnemius skeletal muscle as assessed by micro-CT scan of control and early KP−/−C male mice. n = 10 per group. P = 0.04. f, Skeletal muscle tissue mass of the indicated muscle groups in male control (n = 8) and early KP−/−C mice (n = 9). *P = 0.006 (quadriceps), *P = 0.02 (tibialis anterior), *P = 0.004 (soleus). g, Relative mRNA expression of the indicated genes assessed by RT–qPCR. n = 4 per group. P = 0.05 (Mstn), *P = 0.01 (Trim63), *P = 0.07 (Fbxo32), *P = 0.00004 (Map1lc3a), *P = .006 (Gabarapl1). h, Representative histology of H&E-stained epididymal adipose tissue from control and early KP−/−C male mice. n = 4 per group. i, Relative adipocyte area in male control and early KP−/−C mice. n = 3 per group. P < 0.0001. j, Glycerol release in ex vivo adipose tissue explants from control and early KP−/−C male mice. n = 7 per group. P = 0.01. k, Non-esterified fatty acid (NEFA) release in ex vivo adipose tissue explants from control (n = 8) and KP−/−C male (n = 7) mice. P = 0.0002. l, Representative western blot analysis of phosphorylated (p)HSL and HSL expression in adipose tissue of control and early KP−/−C male mice. n = 3 per group. m, Representative H&E histology images of the pancreas of 15-week-old KC male mice. n = 5 per group. Unless otherwise indicated, statistical analysis was performed using unpaired two-sided t-tests, data are mean ± s.e.m. and n represents the number of mice that were analysed.
Extended Data Fig. 2 Systemic circulating factors are not altered in early PDAC.
a–j, Circulating levels of the indicated factors in control and early KP−/−C mice. a, IL-6. n = 16 control and 14 early KP−/−C mice. ns, not significant; P = 0.45. b, TNF-α. n = 12 control and 10 early KP−/−C mice. P = 0.45. c, PTHrP. n = 7 control and 8 early KP−/−C mice. P = 0.94. d, Corticosterone. n = 19 control and 16 early KP−/−C mice. P = 0.40. e, Amylin. n = 7 control and 6 early KP−/−C mice. P = 0.91. f, IL-1β. n = 11 control and 10 early KP−/−C mice. P = 0.39. g, IL-4. n = 12 control and 10 early KP−/−C mice. P = 0.56. h, IFNγ. n = 12 control and 9 early KP−/−C mice. P = 0.08. i, IL-10. n = 12 control and 10 early KP−/−C mice. P = 0.42. j, IL-17. n = 12 and 10 early KP−/−C mice P = 0.29. Unless otherwise indicated, statistical analysis was performed using unpaired two-sided t-tests, data are mean ± s.e.m. and n represents the number of mice that were analysed.
Extended Data Fig. 3 Decreased exocrine pancreatic function in early PDAC disease promotes adipose tissue loss.
a–d, C57BL/6J mice bearing PDAC-derived subcutaneous tumours fed a control diet or the same diet at 40% caloric restriction (CR) for 3 weeks. n = 8 per group. a, Body weight. *P < 0.0001. b, Epididymal adipose tissue (eWAT) and inguinal adipose tissue (iWAT) mass normalized to body weight. *P < 0.0001 for epididymal adipose tissue and *P = 0.0034 for inguinal adipose tissue. c, Skeletal muscle mass of the indicated muscle groups normalized to body weight. d, Tumour volume normalized to body weight. *P = 0.002. n = 15 tumours from 8 control mice (2 tumours per mouse; for one mouse one of the tumours did not grow) and n = 16 tumours from 8 calorie-restricted mice. e, Fed plasma glucose levels of 7-week-old male control and KP−/−C mice that were fed the indicated diets. n = 4 per group. P = 0.18. f, Tissue weights normalized to body weight in KP−/−C mice pair-fed indicated diets. n = 4 per group. *P = 0.01. ns, P = 0.53. g, h, Tumour weight normalized to body weight of KP−/−C mice that were fed the indicated diets for 1 week. g, KP−/−C mice were fed the indicated diets. n = 6 control and 7 enzyme-supplemented. P = 0.9. h, KP−/−C mice were pair-fed the indicated diets. n = 4 per group. P = 0.26. i, Representative histology of H&E-stained autochthonous pancreatic tumours of KP−/−C mice that were fed the indicated diets. n = 4 per group. Unless otherwise indicated, statistical analysis was performed using unpaired two-sided t-tests, data are mean ± s.e.m. and n represents the number of mice that were analysed.
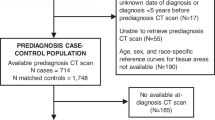
Extended Data Fig. 4 Use of CT imaging to assess patient body composition and relationship between plasma BCAA levels and patient survival by study site.
a, Representative CT image used to analyse body composition. Skeletal muscle is shown in red, intramuscular adipose tissue is shown in green, visceral adipose tissue is shown in yellow and subcutaneous adipose tissue is shown in blue. b. Hazard ratios (HRs) and 95% confidence intervals (CI) for the association between plasma BCAAs and patient survival, comparing the top and bottom quartile, calculated using Cox proportional hazards model adjusted for age at diagnosis (continuous), gender (male or female), race (white, non-white or unknown), year of diagnosis (2000–2005, 2006–2010 or 2011–2015), cancer stage (local, locally advanced, metastatic or unknown), BMI (continuous), diabetes history (none, ≤4 years, >4 years or unknown) and smoking status (never, past, current or unknown). The pooled hazard ratios were calculated using the DerSimonian and Laird random-effects model. The solid squares and horizontal lines correspond to the study site-specific multivariate hazard ratio and 95% confidence interval, respectively. The area of the solid square reflects the study site-specific weight (inverse of the variance). The filled diamond represents the pooled hazard ratio and 95% confidence interval. The solid vertical line indicates a hazard ratio of 1.0. n = 778.
Supplementary information
Supplementary Figure
This file contains the uncropped gel from Extended Data Figure 1.
Rights and permissions
About this article
Cite this article
Danai, L.V., Babic, A., Rosenthal, M.H. et al. Altered exocrine function can drive adipose wasting in early pancreatic cancer. Nature 558, 600–604 (2018). https://doi.org/10.1038/s41586-018-0235-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0235-7
This article is cited by
-
Exploring the causal association between rheumatoid arthritis and the risk of cervical cancer: a two-sample Mendelian randomization study
Arthritis Research & Therapy (2024)
-
Pancreatic cancer is associated with medication changes prior to clinical diagnosis
Nature Communications (2023)
-
Adipose tissue and skeletal muscle wasting precede clinical diagnosis of pancreatic cancer
Nature Communications (2023)
-
Increased glucose availability sensitizes pancreatic cancer to chemotherapy
Nature Communications (2023)
-
Visceral adipose tissue remodeling in pancreatic ductal adenocarcinoma cachexia: the role of activin A signaling
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.