Abstract
Mutations in the E3 ubiquitin ligase parkin (PARK2, also known as PRKN) and the protein kinase PINK1 (also known as PARK6) are linked to autosomal-recessive juvenile parkinsonism (AR-JP)1,2; at the cellular level, these mutations cause defects in mitophagy, the process that organizes the destruction of damaged mitochondria3,4. Parkin is autoinhibited, and requires activation by PINK1, which phosphorylates Ser65 in ubiquitin and in the parkin ubiquitin-like (Ubl) domain. Parkin binds phospho-ubiquitin, which enables efficient parkin phosphorylation; however, the enzyme remains autoinhibited with an inaccessible active site5,6. It is unclear how phosphorylation of parkin activates the molecule. Here we follow the activation of full-length human parkin by hydrogen–deuterium exchange mass spectrometry, and reveal large-scale domain rearrangement in the activation process, during which the phospho-Ubl rebinds to the parkin core and releases the catalytic RING2 domain. A 1.8 Å crystal structure of phosphorylated human parkin reveals the binding site of the phospho-Ubl on the unique parkin domain (UPD), involving a phosphate-binding pocket lined by AR-JP mutations. Notably, a conserved linker region between Ubl and the UPD acts as an activating element (ACT) that contributes to RING2 release by mimicking RING2 interactions on the UPD, explaining further AR-JP mutations. Our data show how autoinhibition in parkin is resolved, and suggest a mechanism for how parkin ubiquitinates its substrates via an untethered RING2 domain. These findings open new avenues for the design of parkin activators for clinical use.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Corti, O., Lesage, S. & Brice, A. What genetics tells us about the causes and mechanisms of Parkinson’s disease. Physiol. Rev. 91, 1161–1218 (2011).
Pickrell, A. M. & Youle, R. J. The roles of PINK1, parkin, and mitochondrial fidelity in Parkinson’s disease. Neuron 85, 257–273 (2015).
Harper, J. W., Ordureau, A. & Heo, J.-M. Building and decoding ubiquitin chains for mitophagy. Nat. Rev. Mol. Cell Biol. 19, 93–108 (2018).
Pickles, S., Vigié, P. & Youle, R. J. Mitophagy and quality control mechanisms in mitochondrial maintenance. Curr. Biol. 28, R170–R185 (2018).
Wauer, T., Simicek, M., Schubert, A. & Komander, D. Mechanism of phospho-ubiquitin-induced PARKIN activation. Nature 524, 370–374 (2015).
Kumar, A. et al. Parkin-phosphoubiquitin complex reveals cryptic ubiquitin-binding site required for RBR ligase activity. Nat. Struct. Mol. Biol. 24, 475–483 (2017).
Trempe, J.-F. et al. Structure of parkin reveals mechanisms for ubiquitin ligase activation. Science 340, 1451–1455 (2013).
Wauer, T. & Komander, D. Structure of the human Parkin ligase domain in an autoinhibited state. EMBO J. 32, 2099–2112 (2013).
Riley, B. E. et al. Structure and function of Parkin E3 ubiquitin ligase reveals aspects of RING and HECT ligases. Nat. Commun. 4, 1982 (2013).
Koyano, F. et al. Ubiquitin is phosphorylated by PINK1 to activate parkin. Nature 510, 162–166 (2014).
Kane, L. A. et al. PINK1 phosphorylates ubiquitin to activate Parkin E3 ubiquitin ligase activity. J. Cell Biol. 205, 143–153 (2014).
Kazlauskaite, A. et al. Parkin is activated by PINK1-dependent phosphorylation of ubiquitin at Ser65. Biochem. J. 460, 127–139 (2014).
Ordureau, A. et al. Quantitative proteomics reveal a feedforward mechanism for mitochondrial PARKIN translocation and ubiquitin chain synthesis. Mol. Cell 56, 360–375 (2014).
Wauer, T. et al. Ubiquitin Ser65 phosphorylation affects ubiquitin structure, chain assembly and hydrolysis. EMBO J. 34, 307–325 (2015).
Kondapalli, C. et al. PINK1 is activated by mitochondrial membrane potential depolarization and stimulates Parkin E3 ligase activity by phosphorylating Serine 65. Open Biol. 2, 120080 (2012).
Iguchi, M. et al. Parkin-catalyzed ubiquitin-ester transfer is triggered by PINK1-dependent phosphorylation. J. Biol. Chem. 288, 22019–22032 (2013).
Shiba-Fukushima, K. et al. PINK1-mediated phosphorylation of the Parkin ubiquitin-like domain primes mitochondrial translocation of Parkin and regulates mitophagy. Sci. Rep. 2, 1002 (2012).
Sauvé, V. et al. A Ubl/ubiquitin switch in the activation of Parkin. EMBO J. 34, 2492–2505 (2015).
Kumar, A. et al. Disruption of the autoinhibited state primes the E3 ligase parkin for activation and catalysis. EMBO J. 34, 2506–2521 (2015).
Kazlauskaite, A. et al. Binding to serine 65-phosphorylated ubiquitin primes Parkin for optimal PINK1-dependent phosphorylation and activation. EMBO Rep. 16, 939–954 (2015).
Ordureau, A. et al. Defining roles of PARKIN and ubiquitin phosphorylation by PINK1 in mitochondrial quality control using a ubiquitin replacement strategy. Proc. Natl Acad. Sci. USA 112, 6637–6642 (2015).
Schubert, A. F. et al. Structure of PINK1 in complex with its substrate ubiquitin. Nature 552, 51–56 (2017).
Ordureau, A. et al. Dynamics of PARKIN-dependent mitochondrial ubiquitylation in induced neurons and model systems revealed by digital snapshot proteomics. Mol. Cell 70, 211–227.e8 (2018).
Park, S., Foote, P. K., Krist, D. T., Rice, S. E. & Statsyuk, A. V. UbMES and UbFluor: novel probes for ring-between-ring (RBR) E3 ubiquitin ligase PARKIN. J. Biol. Chem. 292, 16539–16553 (2017).
Pao, K.-C. et al. Probes of ubiquitin E3 ligases enable systematic dissection of parkin activation. Nat. Chem. Biol. 12, 324–331 (2016).
Arkinson, C. & Walden, H. Parkin function in Parkinson’s disease. Science 360, 267–268 (2018).
Harrison, R. A. & Engen, J. R. Conformational insight into multi-protein signaling assemblies by hydrogen–deuterium exchange mass spectrometry. Curr. Opin. Struct. Biol. 41, 187–193 (2016).
Lechtenberg, B. C. et al. Structure of a HOIP/E2~ubiquitin complex reveals RBR E3 ligase mechanism and regulation. Nature 529, 546–550 (2016).
Geisler, S. et al. PINK1/Parkin-mediated mitophagy is dependent on VDAC1 and p62/SQSTM1. Nat. Cell Biol. 12, 119–131 (2010).
Chaudhary, S. et al. Parkin mutations in familial and sporadic Parkinson’s disease among Indians. Parkinsonism Relat. Disord. 12, 239–245 (2006).
Berrow, N. S. et al. A versatile ligation-independent cloning method suitable for high-throughput expression screening applications. Nucleic Acids Res. 35, e45 (2007).
Berndsen, C. E. & Wolberger, C. A spectrophotometric assay for conjugation of ubiquitin and ubiquitin-like proteins. Anal. Biochem. 418, 102–110 (2011).
Gladkova, C. et al. An invisible ubiquitin conformation is required for efficient phosphorylation by PINK1. EMBO J. 36, 3555–3572 (2017).
Wilkinson, K. D., Gan-Erdene, T. & Kolli, N. Derivitization of the C-terminus of ubiquitin and ubiquitin-like proteins using intein chemistry: methods and uses. Methods Enzymol. 399, 37–51 (2005).
Silva, J. C. et al. Quantitative proteomic analysis by accurate mass retention time pairs. Anal. Chem. 77, 2187–2200 (2005).
Waterman, D. G. et al. Diffraction-geometry refinement in the DIALS framework. Acta Crystallogr. D 72, 558–575 (2016).
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Adams, P. D. et al. The Phenix software for automated determination of macromolecular structures. Methods 55, 94–106 (2011).
Yamamoto, A. et al. Parkin phosphorylation and modulation of its E3 ubiquitin ligase activity. J. Biol. Chem. 280, 3390–3399 (2005).
Lu, X. et al. Mitophagy controls beige adipocyte maintenance through a Parkin-dependent and UCP1-independent mechanism. Sci. Signal. 11, eaap8526 (2018).
Avraham, E., Rott, R., Liani, E., Szargel, R. & Engelender, S. Phosphorylation of Parkin by the cyclin-dependent kinase 5 at the linker region modulates its ubiquitin-ligase activity and aggregation. J. Biol. Chem. 282, 12842–12850 (2007).
Durcan, T. M. et al. USP8 regulates mitophagy by removing K6-linked ubiquitin conjugates from parkin. EMBO J. 33, 2473–2491 (2014).
Acknowledgements
We thank beamline scientists at Diamond Light Source (DLS) for support at beamline I24; H. Ovaa and B.-T. Xin for H-Gly-VS hydrochloride; J. Pruneda, P. Elliott and M. Michel for help with crystallography and data collection; and members of the D.K. laboratory for reagents and discussions. Access to DLS was supported in part by the EU FP7 infrastructure grant BIOSTRUCT-X (contract no. 283570). The D.K. laboratory is supported by the Medical Research Council (U105192732), the European Research Council (724804), the Michael J. Fox Foundation and the Lister Institute for Preventive Medicine.
Reviewer information
Nature thanks W. Harper, R. Youle and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
C.G. performed all experiments and analysed the data presented in this manuscript. S.L.M. and J.M.S. performed and analysed HDX-MS experiments. D.K. directed the project, analysed the data, acquired funding and wrote the manuscript with help from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
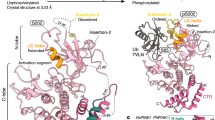
Extended Data Fig. 1 Mechanisms of parkin autoinhibition.
a, Structure of autoinhibited, full-length human parkin (PDB 5C1Z19) shown schematically (top, as in Fig. 1a) and in cartoon representation in the same colours. Two insets show the UPD–RING2 interface (with Cys431 shown in ball-and-stick representation), and the blocked E2 binding site (with the E2 position, modelled according to PDB 5EDV28, shown as grey surface). Zn ions are shown as grey spheres. b, An ‘open-book’ view of the UPD–RING2 interface, with hydrophobic residues coloured white on each surface. c, Structure of phospho-ubiquitin bound to full-length parkin (PDB 5N2W6) as in a. Phospho-ubiquitin binding leads to helix straightening, and IBR domain repositioning, which releases the Ubl domain for phosphorylation5,6. In the shown structures of unphosphorylated parkin, the Ubl and REP (red) inhibit E2 binding, and the RING2–UPD interface is intact, with Cys431 being inaccessible. The Ubl–UPD linker was removed from crystallized constructs in a and c6,19.
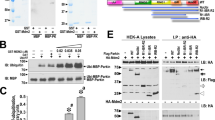
Extended Data Fig. 2 Sample preparation for HDX-MS and selected raw data.
a, Representative LC–MS spectrum of the prepared Ub-VS probe (see Methods). Experiment was performed in duplicate. b, Representative LC–MS spectrum of Ub-VS-reacted phospho-parkin. Experiment was performed in duplicate. c, Samples used in HDX-MS analysis. In HDX-MS, non-covalent complexes with phospho-ubiquitin were used. Covalent complexes are indicated with a dash and non-covalent complexes by a colon. This is representative of at least three independent experiments; for gel source data, see Supplementary Fig. 1. d, Relative deuterium uptake (in Da) is shown for exemplary selected peptides across the parkin molecule, over the timecourse of the experiment. Each point for the technical replicate experiments is shown. Data points were taken at identical time points, but are offset on the x axis for clarity.
Extended Data Fig. 3 Graphical representation of HDX-MS data.
Data from HDX-MS experiments (Fig. 1b–e) were plotted onto a stylized ‘open domain’ model of parkin, with identical colouring (blue, more protected from solvent exchange compared to previous state; red, less protected from solvent exchange compared to previous state). Grey regions correspond to peptides that were not covered or could not be analysed owing to modification. Schematic domain representations indicate an average change of the corresponding interfaces across all time points. White regions indicate no change. a, Parkin compared to parkin–phospho-ubiquitin. b, Parkin–phospho-ubiquitin compared to phospho-parkin–phospho-ubiquitin. c, Phospho-parkin–phospho-ubiquitin compared to phospho-parkin–phospho-ubiquitin in complex with an isopeptide UBE2L3–Ub thioester mimetic (see Methods). This experiment confirmed a previously reported binding site for the E2-conjugated ubiquitin on the RBR6,28 (8). d, Phospho-parkin–phospho-ubiquitin compared to Ub-VS-reacted phospho-parkin–phospho-ubiquitin. Reaction with Ub-VS leads to modification of the catalytic Cys431-containing-peptide, generating non-identical peptides precluding comparison by HDX-MS. Low coverage of the RING2 domain can be explained by ubiquitin resistance to pepsin cleavage, leading to protection of the linked RING2 domain and subsequent peptide loss. To allow comparison, these peptides were also omitted from analysis of the UBE2L3–Ub-bound sample. In c and d, the structure representation is deceiving because REP and RING2 are highly mobile and are no longer bound to the parkin core. Indeed, the high hydrogen–deuterium exchange in the REP sequence in active parkin (Fig. 1d, e; peptide (4) in Extended Data Fig. 2d) indicates an additional loss of secondary structure in this helical element when REP and RING2 are released.
Extended Data Fig. 4 A conserved linker between Ubl and UPD.
Sequence alignment of parkin, with domains coloured corresponding to 5N2W6 as in Extended Data Fig. 1. Phosphate binding pockets are labelled. The linker region between Ubl and UPD (amino acids 76–143) contains two strings of highly conserved residues. Residues upstream and downstream of the conserved region are unconserved both in sequence and linker length. Tsparkin shows the smallest number of residues in the linker (upstream, 25 amino acids in human parkin, 18 amino acids in Tsparkin; downstream, 18 amino acids in human parkin, 11 amino acids in Tsparkin). See also Extended Data Fig. 8d.
Extended Data Fig. 5 Tsparkin and pre-crystallization biochemistry for human parkin.
a, HDX-MS experiment comparing phospho-Tsparkin reacted with phospho-ubiqutin-C3Br and phospho-Tsparkin reacted with phospho-ubiquitin-C3Br and Ub-VS with identical colouring (blue, more protected from solvent exchange; red, less protected from solvent exchange; grey, not covered in all of the compared states, see Fig. 1). The experiment was performed in technical triplicate. The Tsparkin profile is highly similar to the profile of human parkin in an analogous state (Fig. 1e). Higher peptide resolution in this sample reveals protection of the RING2 interface by reacted Ub-VS, but the C terminus of RING2 that binds to the UPD interface is surface exposed. Both phospho-Ubl and the Ubl–UPD linker are protected in activated parkin. b, Limited proteolysis of Tsparkin with elastase, in different stages of activation. In unphosphorylated, autoinhibited Tsparkin, the Ubl is cleaved off in the Ubl–UPD linker. In activated forms of Tsparkin (phospho-Tsparkin, phospho-Tsparkin reacted with phospho-ubiquitin-C3Br, phospho-Tsparkin reacted with phospho-ubiquitin-C3Br and Ub-VS), the RING2 is readily cleaved off, while the Ubl is not efficiently removed. This suggests that the Ubl–UPD linker is not accessible in activated forms of Tsparkin. A representative gel from three independent experiments is shown. For gel source data, see Supplementary Fig. 1. c, A TEV cleavage site was introduced after the IBR domain, so that after activation by phospho-ubiquitin and Ubl-phosphorylation, the released RING2 domain can be removed. Once removed, RING2 is no longer stably associated with the remaining parkin core. Shown is a gel filtration profile illustrating this point. A representative profile from three independent experiments is shown. d, SDS–PAGE analysis of sample preparation process (see Methods). Asterisk denotes ubiquitin probe (Ub-C3Br)-reacted material that modifies the RING2 catalytic Cys, which explains the cleaved, probe-reacted RING2 band (asterisk in step 3). A representative gel from three independent experiments is shown. For gel source data, see Supplementary Fig. 1. e, HDX-MS experiment on Tsparkin, comparing phospho-Tsparkin reacted with phospho-ubiquitin-C3Br with phospho-Tsparkin reacted with phospho-ubiquitin-C3Br and Ub-VS (bottom) or with RING2-TEV-cleaved phospho-Tsparkin reacted with phospho-ubiquitin-C3Br (top), coloured as in a. Identical profiles were obtained, showing that RING2 removal has no effect on the activated core of parkin. This further indicates that RING2 acts independently of the parkin core upon full activation. Notably, in both comparisons, we observed concomitant protection of phospho-Ubl and the Ubl–UPD linker. The experiment was performed in technical triplicate.
Extended Data Fig. 6 Quality control and electron density maps for human phospho-parkin–phospho-ubiquitin.
a, LC–MS spectrum of crystallized human phospho-parkin (amino acids 1–382) bound to phospho-ubiquitin. This is representative of two independent experiments. b, Composite omit map (generated with simulated annealing) shown for the single complex in the asymmetric unit. 2|Fo|−|Fc| electron density is shown at 1σ. c, Electron density as in b for the Ubl–UPD linker. d, Electron density as in b for the Ser65 phospho-Ubl binding site on the UPD linker. e, Electron density as in b for the Ser65 phospho-Ub binding site. As we are missing electron density for disordered regions in the Ubl–ACT and ACT–UPD linkers, we cannot exclude the possibility that phospho-Ubl may interact in trans with a neighbouring parkin molecule. Also see Extended Data Table 1.
Extended Data Fig. 7 The phospho-Ubl binding site on the UPD.
a, Side-by-side view of phospho-parkin–phospho-ubiquitin (left) and parkin–phospho-ubiquitin (PDB 5N2W6, right), and superposition of both (below). The green Ubl domain changes position by >50 Å. b, E2–Ub from the structure of the HOIP RBR domain in complex with UBE2D2–Ub28 was modelled onto phospho-parkin–phospho-ubiquitin, by superposition of the RING1 domains of each complex. The E2-conjugated ubiquitin molecule in the ‘open’ conformation binds to the previously recognized cryptic ubiquitin binding interface on RING1–IBR6. The contact points correlate with HDX-MS data (Fig. 1d, Extended Data Figs. 2, 3c). c, HDX-MS data from Fig. 1e were plotted onto the phospho-parkin–phospho-ubiquitin structure with identical colouring (blue, more protected from solvent exchange; red, less protected from solvent exchange; grey, not covered in all of the compared states, compare with Fig. 1). Protected regions on UPD match the observed phospho-Ubl interface. d, HDX-MS experiments comparing parkin with a mutation in the phospho-acceptor binding site on the UPD (phospho-parkin(K211N)–phospho-ubiquitin) compared with phospho-parkin–phospho-ubiquitin, coloured as in c. The mutant is unable to protect the Ubl, and to release RING2 and REP. Experiments were done as technical triplicate.
Extended Data Fig. 8 A regulatory role of the parkin Ubl–UPD linker.
a, b, E2 discharge assay resolved on a Coomassie stained SDS–PAGE gel (a) and quantified from band intensities (b) for phospho-parkin and phospho-parkin(R104A). This is representative of at least two independent experiments; for gel source data, see Supplementary Fig. 1. The mutation in the ACT element leads to a reduction in discharge activity, suggesting that the residue is required to dislodge RING2 from the parkin core. c, Parkin(R104A) is equally stable as wild-type parkin, in the unphosphorylated or phosphorylated form. Thermal denaturation experiments were performed as technical triplicate. d, Sequence detail of the Ubl–UPD linker, which contains the ACT element described here. In the ACT element as bound to phospho-parkin–phospho-ubiquitin, the positions of two annotated (in PhosphoSitePlus) parkin phosphorylation sites, Ser101 and Ser108, are resolved. Phosphorylation of Ser101 decreases parkin activity40, which is probably explained by phosphorylation preventing phospho-Ubl and/or linker binding to the UPD. It is hence highly likely that phosphorylation of parkin on these residues provides additional layers of parkin regulation that remain to be uncovered in future work. As an example, parkin phosphorylation by PKA was recently reported to be a mechanism of parkin inhibition in beige-to-white adipocyte transition, although phosphorylation sites remained unclear41. Residues before the ACT element (amino acids 73–99) and after the ACT element (amino acids 109–142) are disordered in our structure. The last ordered residue, Ser108, is tantalizingly close to the REP binding site as well as to the phospho-ubiquitin binding pocket, but disorder suggests that clear binding sites for other conserved linker residues, in particular for the parkin GLAVIL motif, are not present. HDX-MS also does not reveal additional protection of the linker, even when the E2–Ub conjugate is bound, suggesting that the GLAVIL motif may not bind the E2 (Fig. 1d, Extended Data Figs. 2, 3c). On the other hand, there are at least three additional annotated phosphorylation sites, Ser116, Ser131 and Ser13615,40,42,43, suggesting that the second part of the linker may also be regulated. Phosphorylation on these residues could change the ability of the disordered parts of the linker to interact with parkin in cis. For example, we would speculate that a phosphorylated Ser116 could for example, reach the phosphate binding pocket occupied by phospho-Ser65 of ubiquitin. Alternatively, the remaining Ubl–UPD linker may be important for substrate recruitment, or involved in other, PINK1-independent mechanisms of parkin activation.
Supplementary information
Supplementary Fig. 1
Uncropped versions of SDS-PAGE gels.
Rights and permissions
About this article
Cite this article
Gladkova, C., Maslen, S.L., Skehel, J.M. et al. Mechanism of parkin activation by PINK1. Nature 559, 410–414 (2018). https://doi.org/10.1038/s41586-018-0224-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0224-x
This article is cited by
-
Optineurin provides a mitophagy contact site for TBK1 activation
The EMBO Journal (2024)
-
A mutational atlas for Parkin proteostasis
Nature Communications (2024)
-
The unifying catalytic mechanism of the RING-between-RING E3 ubiquitin ligase family
Nature Communications (2023)
-
The mitophagy pathway and its implications in human diseases
Signal Transduction and Targeted Therapy (2023)
-
GRAF1 integrates PINK1-Parkin signaling and actin dynamics to mediate cardiac mitochondrial homeostasis
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.