Abstract
In amniotes, the development of the primitive streak and its accompanying ‘organizer’ define the first stages of gastrulation. Although these structures have been characterized in detail in model organisms, the human primitive streak and organizer remain a mystery. When stimulated with BMP4, micropatterned colonies of human embryonic stem cells self-organize to generate early embryonic germ layers1. Here we show that, in the same type of colonies, Wnt signalling is sufficient to induce a primitive streak, and stimulation with Wnt and Activin is sufficient to induce an organizer, as characterized by embryo-like sharp boundary formation, markers of epithelial-to-mesenchymal transition and expression of the organizer-specific transcription factor GSC. Moreover, when grafted into chick embryos, human stem cell colonies treated with Wnt and Activin induce and contribute autonomously to a secondary axis while inducing a neural fate in the host. This fulfils the most stringent functional criteria for an organizer, and its discovery represents a milestone in human embryology.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
12 November 2018
Ref. 7 from Benvenisty and colleagues was inadvertently omitted; this has now been cited in the text and added to the reference list, and subsequent references have been renumbered. The Letter has been corrected online.
References
Warmflash, A., Sorre, B., Etoc, F., Siggia, E. D. & Brivanlou, A. H. A method to recapitulate early embryonic spatial patterning in human embryonic stem cells. Nat. Methods 11, 847–854 (2014).
Spemann, H. & Mangold, H. Induction of embryonic primordia by implantation of organizers from a different species. Int. J. Dev. Biol. 45, 13–38 (2001).
Oppenheimer, J. M. Transplantation experiments on developing teleosts (Fundulus and Perca). J. Exp. Zool. 72, 409–437 (1936).
Waddington, C. H. Experiments on the development of chick and duck embryos, cultivated in vitro. Philos. Trans. R. Soc. London. B 221, 179–230 (1932).
Beddington, R. S. Induction of a second neural axis by the mouse node. Development 120, 613–620 (1994).
Kinder, S. J. et al. The organizer of the mouse gastrula is composed of a dynamic population of progenitor cells for the axial mesoderm. Development 128, 3623–3634 (2001).
Sharon, N., Mor, I., Golan-lev, T., Fainsod, A. & Benvenisty, N. Molecular and functional characterizations of gastrula organizer cells derived from human embryonic stem cells. Stem Cells 29, 600–608 (2011).
Ben-Haim, N. et al. The Nodal precursor acting via Activin receptors induces mesoderm by maintaining a source of its convertases and BMP4. Dev. Cell 11, 313–323 (2006).
Crease, D. J., Dyson, S. & Gurdon, J. B. Cooperation between the Activin and Wnt pathways in the spatial control of organizer gene expression. Proc. Natl Acad. Sci. USA 95, 4398–4403 (1998).
Gritsman, K., Talbot, W. S. & Schier, A. F. Nodal signaling patterns the organizer. Development 127, 921–932 (2000).
Brennan, J. et al. Nodal signalling in the epiblast patterns the early mouse embryo. Nature 411, 965–969 (2001).
Zorn, A. M. & Wells, J. M. Vertebrate endoderm development and organ formation. Annu. Rev. Cell Dev. Biol. 25, 221–251 (2009).
Faial, T. et al. Brachyury and SMAD signalling collaboratively orchestrate distinct mesoderm and endoderm gene regulatory networks in differentiating human embryonic stem cells. Development 142, 2121–2135 (2015).
Williams, M., Burdsal, C., Periasamy, A., Lewandoski, M. & Sutherland, A. Mouse primitive streak forms in situ by initiation of epithelial to mesenchymal transition without migration of a cell population. Dev. Dyn. 241, 270–283 (2012).
Tam, P. P. L. & Loebel, D. A. F. Gene function in mouse embryogenesis: get set for gastrulation. Nat. Rev. Genet. 8, 368–381 (2007).
Robb, L. & Tam, P. P. L. Gastrula organiser and embryonic patterning in the mouse. Semin. Cell Dev. Biol. 15, 543–554 (2004).
Zhu, L., Belo, J. A., De Robertis, E. M. & Stern, C. D. Goosecoid regulates the neural inducing strength of the mouse node. Dev. Biol. 216, 276–281 (1999).
Knoetgen, H., Teichmann, U., Wittler, L., Viebahn, C. & Kessel, M. Anterior neural induction by nodes from rabbits and mice. Dev. Biol. 225, 370–380 (2000).
Chapman, S. C., Collignon, J., Schoenwolf, G. C. & Lumsden, A. Improved method for chick whole-embryo culture using a filter paper carrier. Dev. Dyn. 220, 284–289 (2001).
Etoc, F. et al. A balance between secreted inhibitors and edge sensing controls gastruloid self-organization. Dev. Cell 39, 302–315 (2016).
Vieceli, F. M. et al. The transcription factor chicken Scratch2 is expressed in a subset of early postmitotic neural progenitors. Gene Expr. Patterns 13, 189–196 (2013).
Hamburger, V. & Hamilton, H. L. A series of normal stages in the development of the chick embryo. J. Morphol. 88, 49–92 (1951).
Psychoyos, D. & Finnell, R. Assay for neural induction in the chick embryo. J. Vis. Exp. https://doi.org/10.3791/1027 (2009).
Acknowledgements
The authors are grateful to I. Yan, F. Vieceli and M. Bronner for materials and protocols, to J. Metzger for assistance with 3D image segmentation, and to members of the A.H.B. and E.D.S. laboratories for helpful discussions. This work was supported by grants R01 HD080699, R01 GM101653, the Tri-Institutional Starr Foundation Grant 2016-007, and private funds from the Rockefeller University.
Reviewer information
Nature thanks I. Hyun, O. Pourquié and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
I.M., A.H.B. and E.D.S. conceptualized the work and wrote the paper. I.M. performed stem cell experiments. I.M. devised, and I.M. and T.Y.K. performed chick experiments. A. R. conceived, generated and validated the RUES2-GLR cell line. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Controls for investigating hESC primitive streak initiation hierarchy.
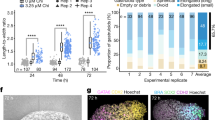
a, Micropatterned hESC colonies were stimulated with IWP2, SB or blank medium. Colonies were fixed after 48 h and stained for germ layer molecular markers. This experiment was repeated at least three times independently with similar results. b, qPCR for BMP4 of unpatterned small colonies stimulated for 4 h as specified. Consistent with the model hierarchy, there was no marked induction of BMP4 by Activin, WNT3A, or BMP4. Data are mean ± s.d. of three biologically independent replicates. c, Quantification of expression data in Fig. 1d. Here, and in all other analyses unless stated otherwise, nuclei were segmented using DAPI and the intensity of immunofluorescence signal for each marker was normalized to the DAPI intensity. Single-cell expression data was binned radially and averaged. The final radial profile represents the mean ± s.d. of n = 25 colonies.
Extended Data Fig. 2 Primitive streak germ layer quantification and EMT timing.
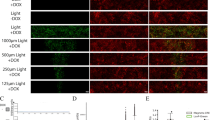
a, Quantification of data presented in Fig. 2a. The radial profile represents the mean ± s.d. of n = 25 colonies. b, Micropatterned hESC colonies were stimulated with BMP4, WNT3A, WNT3A + SB, or WNT3A + Activin, fixed after 12, 24, 36 or 48 h and stained for the primitive streak molecular markers SNAIL, E-cadherin (E-CAD) and N-cadherin (N-CAD). Note that colonies stimulated with WNT3A and WNT3A+Activin turn on expression of EMT markers more rapidly than those stimulated with BMP4 or WNT3A+SB, and have mostly downregulated SNAIL after 48 h. This experiment was repeated at least three times independently with similar results.
Extended Data Fig. 3 Further micropattern fate characterization.
a, b, Micropatterned hESC colonies were stimulated with BMP4, WNT3A, WNT3A + SB, or WNT3A + Activin, fixed at 24 or 48 h after stimulation, and stained for EOMES (a) or PITX2 at 48 h (b). EOMES expression was highest in cells stimulated with WNT3A or WNT3A + Activin and was also dynamic, with the highest expression levels occurring 24 h after stimulation, coinciding with the onset of primitive streak marker expression (Extended Data Fig. 2b). PITX2 is not highly expressed in any of the tested conditions. This experiment was repeated at least three times independently with similar results. c, Quantification of data in Fig. 2j, k and Extended Data Fig. 3a, b. The radial profile represents the mean ± s.d. of n = 25 colonies.
Extended Data Fig. 4 Further characterization of the induced organizer.
a, Micropatterned hESC colonies (1,000-μm and 500-μm diameter) stimulated with WNT3A + Activin, fixed 24 h after stimulation and stained for GSC and BRA. Note that, as previously observed1 for BMP4 induction, shrinking the colony size results in removal of the central micropattern fate region, resulting here in a higher proportion of GSC-expressing cells. This experiment was repeated at least three times independently with similar results. b, Quantification of a. The radial profile represents the mean ± s.d. of n = 25 colonies. c, Scatter plot of single-cell expression of GSC versus BRA. Note that at 24 h most cells co-express BRA and GSC, but by 48 h GSC expression is increased and BRA expression is decreased. We therefore grafted micropatterns at 24 h as well as at 48 h post-stimulation, reasoning that earlier coexpression of BRA and GSC would result in increased graft contribution to axial mesoderm structures. d, qPCR of additional organizer markers. RNA samples were collected from 500-μm-diameter micropatterns stimulated with BMP4, WNT3A, WNT3A + SB, or WNT3A + Activin for 24 or 48 h. With the exception of NOG, the characteristic organizer-secreted inhibitors DKK1, CER1, CHRD, LEFTY1 and LEFTY2, are all most highly expressed in the WNT3A + Activin condition. The high induction of NOG by BMP4 in hESCs has been noted before20, and may represent a species difference between human and mouse. NODAL, which in mouse is restricted to the organizer later in gastrulation, is also most highly expressed in WNT3A + Activin conditions. Data are mean ± s.d. of three biologically independent replicates.
Extended Data Fig. 5 Generation and validation of the RUES2-GLR cell line.
a, Sequencing of the targeted alleles of SOX17, BRA and SOX2 genes. No indels were detected. b, The RUES2-GLR cell line maintains pluripotency normally, as assessed by staining of typical pluripotency markers (OCT4, NANOG and SOX2). Scale bar, 100 μm. This experiment was repeated at least two times independently with similar results. c, The RUES2-GLR cell line was karyotypically normal.
Extended Data Fig. 6 Functional validation of the RUES2-GLR cell line.
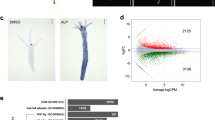
a, Specificity of germ layer reporters. When induced to differentiate to individual germ fates, only the specific reporter was turned on. SOX2–mCitrine was expressed during pluripotency and three days after neural (ectoderm) differentiation, BRA–mCerulean expression commenced after three days of mesodermal differentiation and SOX17-tdTomato reporter was active after three days of endodermal differentiation. Scale bar, 100 μm. b, Snapshots of a time-lapse imaging of the RUES2-GLR cells in micropatterns after treatment with 50 ng ml−1 BMP4, showing how differentiation starts from the edges and extends inwards. Scale bar, 100 μm. c, RUES2-GLR colonies reproducibly generate the typical self-organized concentric rings of germ layers when induced to differentiate with a step presentation of 50 ng ml−1 BMP4 in micropatterns. Scale bar, 200 μm. These experiments were repeated at least three times independently with similar results.
Extended Data Fig. 7 Control chick grafts.
a, Representative grafts for control conditions. With the exception of the BMP4 control condition, grafted hESC colonies were static, with the colonies either growing or dying in place. With BMP4 treatment, the colonies were frequently elongated, possibly owing to hESC migration. There was no induction of SOX2 in the host cells in any of the control conditions. Note that in the case of the WNT3A + SB graft shown, two colonies were grafted into two different locations. Scale bar, 500 μm. Experiments were repeated at least three times independently with similar results. b, Confocal sections showing co-expression of SOX17 (tdTomato) and FOXA2 or OTX2 in human cells that contribute to the secondary axes induced by a 24-h WNT3A + Activin-stimulated micropatterned hESC colony. Scale bar, 20 μm. Experiments were repeated at least three times independently with similar results.
Extended Data Fig. 8 Further characterization of the induced secondary axis.
a, Examples of classifying the notochord-like feature (NLF) based on morphology. At z = +19 μm, the NLF appears as a tighter and brighter rod of cells running north to south that is also distinct and somewhat separated from the surrounding chick epiblast. At z = +46 μm, paired elongated cells stick out ahead of the other cells in a continuation of the originally identified NLF. Other cells belonging to the NLF between z = +46 μm and z = +19 μm are obscured or out of focus in this section, but can be easily identified in individual sections at the other z positions. Scale bar, 100 μm. b, Snapshots from Supplementary Video 1. From top to bottom: yellow shows co-localization of BRA (green) and human (red) cells; purple shows co-localization of Sox17:tdTomato (blue) with human (red) cells; cross-section shows that chick and human cells arrange themselves into germ layers properly, and that they flank the central notochord-like feature indicated by the arrow (cyan); a proportion of human mesoderm cells contribute to part of the notochord-like structure, while the cyan-coloured cells without HNA (red) shows that the remainder of the NLF is composed of host cells. c, Two examples of donor hESC grafts contributing to the induced notochord-like feature, imaged in a live chick embryo 27 h (left) and 23 h (right) after grafting. Scale bars, 200 μm (left), 100 μm (right). Similar notochord-like features were observed in at least ten independent biological replicates.
Supplementary information
Supplementary Tables
This file contains Supplementary Tables 1–4.
Video 1: Fly-through animation of confocal z-stack acquired from the embryo shown in Fig. 3l–r
Stack was first deconvoled with Autoquant software, and then manipulated in Imaris. For classification methodology of the notochord-like feature (NLF) please see Methods and Extended Data Figure 8.
Rights and permissions
About this article
Cite this article
Martyn, I., Kanno, T.Y., Ruzo, A. et al. Self-organization of a human organizer by combined Wnt and Nodal signalling. Nature 558, 132–135 (2018). https://doi.org/10.1038/s41586-018-0150-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0150-y
Keywords
This article is cited by
-
Hypoblast from human pluripotent stem cells regulates epiblast development
Nature (2024)
-
Regulation of long-range BMP gradients and embryonic polarity by propagation of local calcium-firing activity
Nature Communications (2024)
-
Time-integrated BMP signaling determines fate in a stem cell model for early human development
Nature Communications (2024)
-
Middle-out methods for spatiotemporal tissue engineering of organoids
Nature Reviews Bioengineering (2023)
-
Mechanically enhanced biogenesis of gut spheroids with instability-driven morphomechanics
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.