Abstract
Despite significant scientific advances in the modern three-piece inflatable penile prosthesis implant surgery, it is not without surgical risks and can carry additional cosmetic and psychosocial consequences in poorly selected and consented individuals. To address this problem, an international group of key opinion leaders and high-volume prosthetic surgeons reviewed the current guidelines and clinical evidence, discussed their experiences, and formed a consensus regarding inflatable penile prosthesis surgery. The findings of this consensus panel were presented at the 17th biennial Asia Pacific Society of Sexual Medicine scientific meeting. The experts concluded that proper patient selection, informed consent and strict adherence to safe surgical principles are important to optimize clinical outcomes. Furthermore, most intraoperative complications, if recognized, can be addressed intraoperatively to enable placement of the device at the time of initial surgery. Men with significant corporal fibrosis due to Peyronie’s disease, prior prosthesis explantation and priapism, and men who have undergone construction of a neophallus, as well as men who receive concurrent continence surgery, are complex cases requiring additional care and advanced techniques to obtain optimal surgical outcomes. Variability in patient care — in terms of postoperative antibiotic use, pain management, scrotal care, and cycling of the penile prosthesis implant — must be reduced to enable optimization and assessment of outcomes across patient groups.
Similar content being viewed by others
Introduction
The introduction of the modern inflatable penile prosthesis (IPP) in 1974 completely revolutionized the treatment for erectile dysfunction (ED)1. For nearly 50 years, considerable advances have been made in IPP technology and, coupled with improved scientific knowledge in terms of perioperative management and innovative surgical techniques, IPP surgery has been accepted as an effective and safe treatment for ED in many men2.
Categorization of the IPP can largely be divided into single-piece, two-piece, and three-piece prostheses, based on whether the devices have a small reservoir at the end of each cylinder (single-piece) or attached to the pump (two-piece), or a larger (separate) reservoir that is connected to the pump (three-piece). Several key factors, including patient preference, surgeon experience, underlying reasons for surgery, manual dexterity, cost, penile size, and prior penile surgery, will contribute to decision making regarding the type of IPP surgery2. Non-inflatable or malleable penile prostheses are an alternative to IPPs, and these simpler devices are less costly, easier to perform and are associated with fewer mechanical complications than the IPP2. However, the three-piece IPP is often considered a physiologically superior device as it produces penile rigidity and flaccidity that closely resembles a more natural penile erection2. Although a two-piece IPP does not require placement of a separate reservoir and is, therefore, easier to place than the three-piece IPP, it has limited cylinder inflation owing to the small volume of fluid in the compartmentalized reservoir and is associated with a lower patient satisfaction rate2,3,4.
Four main IPP products are available commercially: the Boston Scientific AMS 700 series (American Medical Systems (AMS), now Boston Scientific (BSci), Marlborough, MA, USA), the Coloplast Titan series (Coloplast Corp., Minneapolis, MN, USA), the Zephyr ZSI 475 (Zephyr Surgical Implants SRAL, Geneva, Switzerland), and the Infla10® series (Rigicon Inc., Ronkonkoma, NY, USA). These IPPs have undergone stringent review and clinical testing from relevant national regulatory bodies to ensure that they are safe and mechanically reliable5,6,7,8. Boston Scientific has manufactured three variations of the AMS 700 series since 1983: the AMS 700 LGX (previously Ultrex), AMS 700 CX and AMS 700 CXR2,5. The Coloplast Titan series, formerly the Mentor prosthesis, was introduced in 2002 and is available in Titan (standard), Narrow Base (NB) and extra-large (XL) cylinders6. The Zephyr ZSI 475 was launched in the early 2010s and the company manufactures the ZSI 475FtM in 2016, specifically for female-to-male gender assignment surgery2,7. The most recent addition to the IPP market, in 2019, is the Infla10® series (Rigicon Inc., Ronkonkoma, NY, USA), which consists of three different IPP products, namely the Infla10®X, Infla10®AX (anatomical expansion) and Infla10® NarrowBody8.
In 2020, clinical recommendations were released by the European Society of Sexual Medicine3; these recommendations focus predominantly on sexual parameters following penile prosthesis implantation, particularly regarding partner expectations, satisfaction in male and phalloplasty cohorts, the effect of penile length, girth and implant type, reservoir placement, and the influence of comorbidities and social circumstances. By contrast, the present consensus statement builds on and expands the surgical aspects of the 4th International Consultation on Sexual Medicine guideline on penile prosthesis implant, which was endorsed by the International Society of Sexual Medicine and published in 2016 (ref.4). This consensus was reached by the collective agreement of a group of experts in penile prosthesis surgery and is not endorsed by any society.
This International Penile Prosthesis Implant consensus statement provides a clinical framework around several key domains in patient selection, preoperative evaluation and management of common complications and postoperative care after implantation of a three-piece IPP. A detailed analysis of all relevant studies and a full description of the surgical techniques is not the goal of this consensus statement; instead, it provides an overview of the current evidence-based management strategies for commonly encountered dilemmas when managing patients using IPP. Given the lack of high-quality randomized controlled trials in IPP surgery, specific emphasis is placed on information from narrative review articles and published guidelines (Box 1).
Methods
Databases including MEDLINE, EMBASE and Cochrane CENTRAL trials register were searched for the following terms “inflatable penile prosthesis implant”, “clinical outcome”, “complication”, “urinary incontinence”, “transgender”, “priapism”, “augmentation” and “penile reconstruction”. As only a few prospective and randomized controlled trials involving IPP surgery have been published, a full Preferred Reporting Items for Systematic Reviews and Meta-Analyses protocol was not adopted for this article; instead, a narrative approach was taken. Available literature was reviewed by the faculty and outcomes were presented at the International Penile Prosthesis Implant Consensus Forum on Thursday 11 April 2019 at the 17th biennial Asia Pacific Society of Sexual Medicine scientific meeting in Brisbane, Australia. This session was not sponsored by any IPP device company, and the clinical recommendations were made independent of the IPP device manufacturers.
The panel of nine experts who took part in the process are key opinion leaders and high-volume IPP surgeons from North America, Europe, Latin America, and the Asia Pacific region. This Asia Pacific Society of Sexual Medicine session was not sponsored by any IPP device company, and the clinical recommendations were made independently based on the professional experience and expertise of the panellists. The panel was asked to evaluate and comment on four clinically relevant subheadings concerning IPP surgery: clinical assessment and patient selection, surgical principles and precautionary measures, specific populations, and postoperative care. Clinical findings from published studies were discussed by the panel and the quality of evidence was graded based on the Oxford Centre for Evidence-Based Medicine recommendations and the AGREE II (Appraisal of Guidelines for Research and Evaluation) instrument9. Any disagreements were resolved with subsequent group discussion and consensus development was developed based on the strength of data and a modified Delphi method that included multiple rounds of feedback. A consensus agreement was received, and all authors agreed on the list of recommendations.
Clinical assessment and patient selection
Penile prosthesis implantation is often considered the last option in men who are refractory to or unwilling to use phosphodiesterase-5 inhibitors, intraurethral or intracavernosal injections, and a vacuum erection device10. In men with Peyronie’s disease (PD) and concomitant ED, a concurrent penile prosthesis implant is often advocated in men who require complex penile reconstructive surgery, or in those who have a high likelihood of postoperative ED11.
Before the introduction of phosphodiesterase-5 inhibitors, vasoactive penile injection testing and colour penile Duplex ultrasonography were routinely obtained as part of the diagnostic algorithm12. Penile colour Duplex ultrasonography is not mandatory before IPP surgery, as most cases of ED are likely to be due to diabetes mellitus, vascular disease, or post-prostate cancer treatments3,4. Furthermore, most third-party insurance payers, such as private health funds, have accepted that these diagnostic tests do not alter the decision regarding whether a patient undergoes IPP surgery3,4.
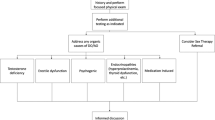
Known patient factors that increase the risk of prosthetic complications and lower satisfaction rates are those with diabetes mellitus, the presence of pathological nasal and skin flora (for example, Staphylococcus spp.), long-term steroid use, poor personal hygiene, suboptimal antimicrobial practice, poor cardiovascular status, history of radiation therapy, spinal cord injury, urinary catheterization, immunosuppression and concurrent genitalia reconstructive surgery13. Optimization of these risk factors and adequate patient counselling are essential before surgery. The informed consent should clearly define the advantages and disadvantages of IPP surgery, explain alternative treatment options, provide cost, and describe potential complications, such as infection and its consequences, bleeding, pain, mechanical failure, failure to regain preoperative penile size, glans softness, injury to surrounding structures (urethra, bladder, bowel, or vessels), and device migration or erosion3,4,14. Information regarding the mechanics of IPP, such as how IPP functions and how the patient operates the device, should be included in the preoperative counselling. Patients must be fully informed that any pre-existing natural erection will be lost and that the procedure is irreversible — subsequent removal of the penile prosthesis will not restore normal erectile capability. The informed consent process provides legal documentation for the IPP surgery and is unique to each institution, although several organizations such as the British Association of Urological Surgeons15 or the Sexual Medicine Society of North America16 have published a standardized template that can be adapted to suit the individual surgeon.
IPP surgery is an invasive surgery and potential complications in the perioperative and postoperative settings can add to the overall economic burden, patient dissatisfaction and future medicolegal risk17. Strong evidence suggests that high-risk patient populations, such as men with uncontrolled diabetes mellitus, corporal fibrosis (for example, PD or priapism) and those who are undergoing salvage penile implants are at a greater risk of prosthetic complications, especially device infection and erosion18. Other reasons for patient dissatisfaction following penile implants include loss of perceived penile length, poor penile glans engorgement and unnaturalness of penile cylinders as perceived by the partner19.
Diabetic men
Men with diabetes mellitus are likely to suffer from an earlier onset of ED and have more severe ED and higher prosthetic complication rates, especially in patients with uncontrolled diabetes9. The increased frequency of infections in patients with diabetes is caused by an underlying hyperglycaemic environment that favours immune dysfunction. Such factors include damage to neutrophil function, depression of the antioxidant system, and humoral immunity, microangiopathies and macroangiopathies, neuropathy, and lower antibacterial activity of urine as well as the greater likelihood of coexisting medical comorbidities and the need for medical interventions20,21.
Several studies have increased interest in the use of cut-off points for glycosylated haemoglobin (HbA1c) to identify diabetic men at a high risk of penile periprosthetic infections22,23,24,25,26. Some studies including systematic reviews and meta-analyses from 2021 reported that an HbA1c level >8.5% might be associated with a higher risk of penile prosthesis infection20,22,23,24,25,26,27, although this observation is limited by the quality of available evidence such as heterogeneous study population, selection bias and studies derived from the manufacturer’s database. However, the debate around whether a meaningful difference in an actual HbA1c threshold can result in a statistically significant increased risk of periprosthetic infection is ongoing, as most studies in this field are heterogeneous, underpowered, or do not control for confounders such as other medical comorbidities and perioperative antibiotics use20,23,24. Furthermore, evidence from a 2016 study suggests that tight intraoperative glycaemic control can minimize infection risk and postoperative hypoglycaemia21.
Avoiding preoperative hyperglycaemia and ensuring stringent peri-operative glycaemic control are paramount to avoiding postoperative diabetic-related complications28. Elective IPP surgery must often be delayed in men with poorly controlled diabetes20,23. Early consultation with an endocrinologist is valuable to ensure optimal glycaemic control during the perioperative period24. Hypoglycaemic agents such as sodium-glucose co-transporter-2 inhibitors should be stopped before surgery to minimize the risk of diabetic ketoacidosis postoperatively28.
Antibiotic and/or antifungal use
In 2016, the International Consultation on Sexual Medicine recommended that patients being prepared for penile prosthesis surgery should be screened to exclude symptoms or signs of cellulitis around the genitalia and lower abdomen, which increases the risk of prosthetic infection and wound-related complications4. A formal urine microscopy test should be obtained to ensure that there is no bacterial growth in the urine before surgery. Appropriate precautions should be undertaken to minimize contamination of the surgical field according to the WHO Guidelines for Safe Surgery, such as the use of a preoperative shower with antibacterial agents, intraoperative hair removal, use of alcoholic formulations for skin prep, appropriate surgical attire with full protective, positive pressure airflow system and minimal traffic within the operating room28. The meticulous care with surgical wound sterility, including multiple changes in surgical draping, the use of antibiotics and/or diluted betadine irrigation and minimizing device–skin contact or “no-touch” techniques, are aimed at minimizing intraoperative skin flora and atypical bacterial contamination4,29.
Coated implants and bacterial contamination
Perioperative antibiotics prophylaxis and antibiotic-coated devices can be coupled with meticulous surgical care to reduce infection risk in IPP implantation13,30,31. The InhibiZone®-coated AMS 700 series is not available in many non-Western countries owing to regulatory issues5. The current Coloplast Titan series, Zephyr ZSI 475 series and Infla10® series have a hydrophilic coating that absorbs water-based substances, and can, therefore, facilitate the use of the surgeon’s antibiotic of choice for antimicrobial elution and protection6,7,8. The 2008 American Urological Association Best Policy Statement advocates the administration of two different intravenous antibiotics, specifically an aminoglycoside (or aztreonam) plus either a first-generation or second-generation cephalosporin or vancomycin at least 1 h before skin incision32. However, the choice of antibiotics is likely dependent on hospital prescribing guidelines, surgeon’s preference, patient’s drug allergy and/or sensitivity, antibiotic availability and local anti-microbiograms.
Biofilm formation is thought to contribute to prosthetic infection33. Once bacterial seeding occurs, the resultant biofilm can promote the growth of other bacteria species and increase antimicrobial resistance and tissue damage33,34,35. Furthermore, biofilm can allow dormant bacteria to be released later, resulting in delayed device infection35. In addition to surgical sterility, perioperative checklists, appropriate perioperative antibiotics coverage and the use of an anti-infective biomaterial-coated IPP can minimize infection risk34,35. Although the use of postoperative antibiotics lacks strong scientific evidence, many surgeons will prescribe postoperative antibiotics to cover for common skin organisms (for example, Staphylococcus spp.)3,4. However, most experts agree that postoperative antibiotics should be prescribed to patients undergoing revision and salvage procedures3,4,36.
Fungal infection
Evidence is increasing regarding the role of fungal species, particularly Candida spp., in medical device infections35. Fungal infections are especially common in certain patient demographics (for example, immunosuppressed) and geographical locations (for example, in developing countries)13,28. The use of antifungals in IPP surgery should be reserved for unusual cases, such as in patients with poorly controlled diabetes, or in the setting of salvaging an infected IPP that is not responsive to intravenous antibiotics4,35. Fungal species such as Candida albicans can form biofilms by themselves or by participating in polymicrobial biofilms alongside bacterial species37. The prescription of antifungals should be done in consultation with an infectious disease physician and the clinical decision should be made to promptly explant the infected IPP if the patient fails to improve after 48 h or in the presence of pus or urosepsis.
Antiplatelet and anticoagulant use
The established risk factors between ED and cardiovascular disease38 and the high rates of comorbidity between the two mean that many patients, especially the ageing population and those with pre-existing cardiovascular history, will be using various oral antiplatelet and anticoagulant medications. Surgeons are increasingly likely to be faced with the dilemma of either continuing these agents and accepting potential serious bleeding or withholding therapy and risking fatal thromboembolic complications4,39. The IPP surgery can be performed with a low risk of clinically significant haemorrhage and thromboembolic risk for a patient on aspirin alone39 whereas antiplatelets (for example, clopidogrel) and anticoagulants (for example, warfarin or ticagrelor) should be withheld4. Generally, patients at a high risk of thromboembolism should be considered for a more aggressive perioperative management strategy with appropriate bridging therapy. Postoperative haematoma can often be successfully managed conservatively with simple analgesia, antibiotics and proper scrotal support40.
Current recommendations from the International Consultation on Urological Disease and the American Urological Association regarding anticoagulant and/or antiplatelet drugs in prosthetic urological surgery are largely based on evidence derived from other surgical specialities; these recommendations vary substantially owing to individual complexity, types of surgery and local hospital’s practice guidelines on venous thromboembolism prevention41. Early and continuous effective communication among patients, general practitioners, and specialists (including the surgeons, cardiologists, and haematologists) is pivotal in managing such high-risk patients who are taking blood thinners during the perioperative period. Guidelines regarding the timing of the resumption of anticoagulant and/or antiplatelet prophylaxis are not available, other than stating that it be resumed as soon as the surgeon believes the risk of bleeding has decreased and assuming no clinically significant surgical haematoma has formed41.
Summary of assessment and selection recommendations
Penile prosthesis implantation is considered an appropriate treatment option for a man who is not achieving a satisfactory erection and desires a permanent solution for ED (Grade C). Proper patient selection, counselling and informed consent are essential, coupled with optimization of existing medical conditions especially diabetes (Grade B). Adequate prophylactic measures should be taken to minimize prosthetic infection and bleeding or thromboembolic risk to ensure good clinical outcomes (Grade B) (Box 1).
Surgical principles and precautionary measures
Meticulous preoperative preparation, adherence to key surgical principles and safe surgical techniques are crucial to optimize clinical outcomes. Prevention remains the best approach to troubleshoot prosthetic-related complications such as corporal perforation, urethral injury and suboptimal placement of IPP components.
Learning curve and centre of excellence
IPP surgery is complex and should be performed by clinicians who have adequate experience in prosthetic surgery. No published study has identified the actual minimum number of IPP cases a surgeon requires to be proficient, given that each patient is different in terms of their underlying cause of ED, body habitus, associated medical comorbidities and the state of their corporal tissue. However, the relationship between patient outcomes and surgical volume is well documented for a variety of other surgeries and is also relevant for IPP surgery42. Awareness of the various intraoperative problems that can arise and preparedness for surgical troubleshooting is of critical importance, as many urologists who place penile prostheses might not have the benefit of high surgical volume or experience in this procedure43. A high-volume or frequent implanter has been defined as a surgeon who routinely implants more than 25 prostheses per year44; evidence suggests that patients treated by high-volume implanters are less likely to require reoperation owing to a lower incidence of prosthetic complications in patients of experienced surgeons45,46.
The concept of a prosthetic centre of excellence — whereby patients are seen by a high-volume specialist surgeon — has been suggested to improve clinical outcomes, streamline surgical care and lower complication rates46,47. In the era of patient choice and good clinical governance for safe clinical practice, the patient should choose a surgeon with whom he is comfortable and who can provide the best quality care in IPP surgery. Although formal fellowship training in IPP surgery is ideal, surgical workshops or mentorship programs can often be useful to improve a surgeon’s understanding of IPP care and surgical techniques if a formal fellowship is not available4. Thus, an appropriately trained and safe surgeon with high-volume expertise in penile prostheses will deliver high-quality outcomes, and potentially, increased economic value47.
Surgical approaches
IPP surgery can be performed through penoscrotal (or trans-scrotal), infra-pubic and sub-coronal approaches (Fig. 1 and Table 1). Surgeons should have adequate knowledge of these surgical approaches; the preferred method is often based on surgeon experience, the patient’s specific anatomy and whether a concurrent penile reconstructive surgery is undertaken48,49,50. Each surgical approach has its benefits and weaknesses, with no clear advantages favouring one surgical approach over another in terms of patient satisfaction4.
a | The trans-scrotal approach incision involves a vertical or horizontal incision over the upper part of the scrotum. b | A penoscrotal approach uses an incision over the penoscrotal junction or extending into the penile shaft, which can provide better corporal exposure. c | An infrapubic incision is made one fingerbreadth above the penile pubic junction while gently pulling down on the penis and can provide direct visualization when inserting the reservoir. d | The subcoronal approach involves a subcoronal incision below the glans penis with degloving of the penis, enabling concurrent corporal reconstructive surgery at the time of inflatable penile prosthesis surgery as nearly the entire corpora can be accessed through the same incision.
Penoscrotal or trans-scrotal approach
The surgical incision involves a vertical or horizontal incision over the upper part of the scrotum (trans-scrotal) (Fig. 1a) or extending into the penile shaft (penoscrotal junction) (Fig. 1b), which can provide better corporal exposure (by extending the incision further along the penile shaft). Most surgeons are comfortable with the anatomy50. Furthermore, the pump can be directly anchored in the scrotum. However, the insertion of the reservoir into the retropubic space is performed blindly in this approach4.
Infra-pubic approach
In contrast with the penoscrotal or trans-scrotal approaches, the infra-pubic incision (Fig. 1c) is made one fingerbreadth above the penile pubic junction while gently pulling down on the penis. This approach can provide a direct visualization when inserting the reservoir and is thought to be associated with a lower rate of scrotal bruising as the scrotal tissue is not surgically dissected. Thus, the infra-pubic approach enables potentially earlier pump cycling. However, this approach can result in damage to the sensory nerves of the penis, and more difficult corporal dilation in obese patients with significant truncal fat pad and scrotal pump misplacement or migration4.
Subcoronal approach
The subcoronal approach involves a subcoronal incision below the glans penis with degloving of the penis (Fig. 1d), enabling concurrent corporal reconstructive surgery at the time of IPP surgery, as nearly the entire corpora can be accessed through the same incision. However, this technique is used much less commonly than the penoscrotal and infra-pubic approaches owing to longer operative time and offering no additional benefit in patients who do not require multiple penile reconstructive surgeries4.
Device characteristics and placement
Cylinder considerations
The appropriate cylinder size is selected based on the maximum corporal length measurement with the aim of maximum cylinder size with minimum rear tip extenders (Table 2). The proximal corporal measurement often determines the selection of cylinder size and selection of rear tip extender length to ensure that the proximal cylinder tip can be placed accurately4. The recommended cavernosal dilation is 12 mm for standard IPP cylinder insertion and 10 mm if a smaller diameter or narrow-base device is utilized4. A smaller diameter cylinder can be useful in cases of significant corporal scarring, smaller penile size or decreased tunical elasticity seen in PD4,11.
For the same cylinder length, the Coloplast Titan provides a larger cylinder girth diameter (up to 21 mm each cylinder) than other IPPs and the Bioflex material is thought to be stronger and more fatigue resistant than silicone material in ex vivo and in vitro studies51. The Titan cylinder is suggested to provide a bigger penile prosthesis size and stronger axial rigidity than the AMS 700 cylinder for the same device length2,6. Data are still accruing for the ZSI 475 and Rigicon IPPs5,6,7,8.
Corporal cylinder complications — including crossover, proximal or distal corporal perforations — can occur in any patient owing to poor surgical technique and in men with corporal fibrosis4,11. These complications can be rectified at the time of surgery (Table 3). A discrepancy (usually >1.5 cm) between each corporal measurement probably signifies corporal perforation4. If a urethral injury is suspected, it is standard of care to abort the IPP surgery rather than repairing urethral defects and proceeding with placing penile implant cylinders, as proceeding with the implantation can increase the risk of device infection and the placement of a prosthetic cylinder can delay urethral healing4. In the case of a unilateral perforation, a single-cylinder placement on the normal corporal chamber (contralateral side of urethral perforation) can be undertaken, although reported patient satisfaction is considerably lower in a single inflatable cylinder than with two-cylinder placement4.
Reservoir considerations
The AMS 700 series has two different reservoirs — the Conceal Low Profile reservoir, which has a flat pancake-like shape when filled with saline, and a traditional round sphere reservoir. AMS 700 comes in several reservoir sizes: 65 ml and 100 ml spherical and 100 ml Conceal reservoirs5. By contrast, the Coloplast Cloverleaf reservoir is available in 75-ml and 125-ml sizes and has been approved for ectopic submuscular reservoir placement in men in whom retropubic access could be difficult owing to scarring or the presence of bowel in the retropubic space6. The Titan Cloverleaf reservoir has a residing lock-out valve mechanism to minimize auto-inflation and prevent a sudden change in pressure. Both the ZSI 475 and Rigicon IPP series use a more traditional (round) reservoir shape. The ZSI 475 reservoir can be filled to 80 ml or 100 ml7 whereas the Rigicon standard reservoir accommodates 65 ml and the Rigicon Adaptive Reservoir can be expanded from 70 ml to 110 ml8. As a general guide, a 100-ml reservoir should be inserted if the IPP cylinder is larger than 18 cm and the reservoir should be filled to the manufacturer’s specified capacity to ensure adequate expansion before adjusting the final reservoir volume according to the size of the IPP cylinders, for example, 40 ml saline is sufficient to inflate a 16-cm cylinder4.
The IPP reservoir is usually placed into the retroperitoneal Retzius space to avoid complications such as poor cosmesis (from the visibility of the reservoir outline), reservoir herniation or device auto-inflation (from direct compression on the reservoir)4. Retropubic reservoir placement is achieved by blind puncture through the transversalis fascia in a penoscrotal approach, with a potential risk (1–2%) of inadvertent organ injury (vascular, bladder or bowel) especially following robotic surgery. Other unusual reservoir complications include erosion into the bladder or bowel, intraperitoneal reservoir placement and iliac vein compression52.
Ectopic reservoir placement, either between the transversus abdominis anteriorly and the transversalis fascia posteriorly or more medially between the rectus abdominis muscle anteriorly and the transversalis fascia posteriorly, has gained popularity owing to the difficulty in the placement of a reservoir in an obliterated retropubic space following radical pelvic surgery53. However, ectopic reservoir placement might not be ideal in thin patients in whom the reservoir outline might be visible, and potential complications such as inadvertent vascular injury and reservoir migration or herniation54,55.
If the retropubic or ectopic placement of the reservoir is not feasible or might result in a suboptimal outcome, a separate transverse hypogastric incision can be made to enable direct placement of the reservoir into the retropubic space. In this scenario, the reservoir tubing should be tunnelled through the subcutaneous abdominal fat into the penoscrotal wound for connection to the pump tubing.
Pump considerations
The AMS 700 series initially used a Tactile pump but has since switched to the current Momentary Squeeze pump, which is modified to be smaller and easier to handle, with better concealment and quicker pump deflation2,5. A lock-out valve was incorporated into the Momentary Squeeze pump to prevent auto-inflation of the penile cylinders against extensive force or sudden elevated pressure within the reservoir. The Momentary Squeeze pump includes a deactivation button on the midline ventral aspect.
Similarly, the Titan pumps have also been improved in terms of their mechanical reliability and durability across different generations such as the Genesis pump, One-Touch Release pump and the Titan touch pump2,6. The ZSI 475 pump has two valves: a simple open-close valve to reduce the risk of mechanical failure and a reinforced valve to decrease the risk of incidental auto-inflation7. The Rigicon Rapid-Pump is quite similar to the AMS 700 Momentary Squeeze pump in appearance, apart from a deactivation button on the side of the pump rather than the ventral midline8.
Ideally, the pump should be placed in the midline scrotal–spongiosis septum in the subdartos space in between the external and internal spermatic fascia4. Stay sutures can be placed at the edges of the scrotal fascia opening to prevent upward pump migration. The pump should be easily accessible by the patient and not positioned too superficially, which can cause poor cosmesis or discomfort during sexual intercourse and an increased risk of pump erosion or extrusion; too high near the shaft of the penis, which can cause poor cosmesis or discomfort during sexual penetration; or too low towards the perineum, which can cause accidental device inflation. The tubing should be buried properly within the scrotal dartos layers for improved concealment and to avoid the tubes kinking and the pump migrating.
Summary recommendations
Strict adherence to meticulous surgical principles can minimize postoperative complications and prosthesis-related complications can usually be addressed intraoperatively to enable placement of the device at the time of surgery (Grade B). The IPP implantation should generally be aborted in the presence of urethral injury (Grade B) (Box 1).
Specific populations
Specific patient populations have different needs when considering prosthesis placement.
Patients with corporal fibrosis
Corporal fibrosis is often encountered in men with priapism, a prior history of explanted implant and advanced PD, and these disorders can present considerable surgical challenges as well as being associated with higher complication rates11,18. Management strategies for implanting prostheses in men with corporal fibrosis include sharp dissection with Metzenbaum scissors, serial corporal dilation with Hegar or Brooks dilators or the use of other specialized tools such as cavernotomes (Carrion-Rossello, Minneapolis, MN, USA) and Uramix (Lansdowne, PA, USA)4,18. In some instances, extended corporotomies with incision or excision of corporal fibrosis followed by the use of graft material might be necessary18,56. A narrow cylinder implant, such as the AMS 700 CXR or Coloplast Titan NB cylinder can be used, with the option of exchanging the device for a regular-sized prosthesis in the future57. A malleable implant can also be used to provide a cheaper and easier alternative and patients can then elect to have an IPP implanted at a later date58,59,60,61.
A residual penile curvature of less than 15° after PPI is not uncommon in men with PD with extensive corporal fibrosis and tunical plaque11. Strategies to correct residual penile curvature include manual penile modelling (with inflated cylinders), intracorporal plaque incision, extracorporal plaque-releasing incision and/or graft reconstruction11,62. One paper reported no statistically significant difference between the AMS 700 CX and Coloplast Titan device in terms of device mechanical survival and patient satisfaction rate63.
Salvaging an infected penile prosthesis
A prosthesis infection can occur in any patient, despite meticulous surgical sterility and strict adherence to antimicrobial prophylaxis; however, the use of infection-retardant coated devices has significantly reduced this devastating complication to <1%4,64. The decision to perform a salvage implant procedure should always involve the surgeon and patient and the patient’s partner, with due consideration of the various options and scenarios4. For a suspected subclinical penile implant infection (with persistent pain beyond 4–6 weeks or the presence of haemoserous drainage after 2 weeks), a course of intravenous antibiotics, preferably piperacillin/tazobactam (4.5 g three times a day) or meropenem (1 g three times a day) can be given provided that the patient is observed closely as an inpatient and surgical intervention should be undertaken if the patient fails to respond to the antibiotics or if infectious symptoms and signs worsen4,18. Prolonged oral antibiotics such as quinolones (for example, ciprofloxacin 500 mg twice a day) are often necessary following discussion with a local infectious clinician and in certain cases with a suspected fungal infection, the use of antifungals such as amphotericin B (intravenous 0.5–0.7 mg/kg/day) or fluconazole (oral 400 mg daily) might be appropriate. Explantation of the infected device without salvage remains the safest option, although this approach affects the possibility of future device implantation, which will be difficult owing to dense corporal fibrosis with ensuing penile size loss18.
Mulcahy et al. popularized the concept of salvaging an infected penile prosthesis in the 1990s65. Immediate salvage surgery can potentially preserve implant component(s), prevent loss of penile size, and minimize subsequent surgical challenges associated with corporal fibrosis. The presence of tissue necrosis and purulence in the corporal bodies, or exposed device component(s) are contraindications for salvage surgery18. In the salvage procedures, all components of the device should be removed, followed by vigorous mechanical lavage with solutions comprising a combination of diluted betadine, half-strength hydrogen peroxide and antibiotic agents, such as kanamycin and/or bacitracin antibiotic solution18. The use of a malleable device or biomaterial case insert such as calcium sulphate at the time of salvage surgery could help to minimize corporal fibrosis and loss in penile size, as well as facilitate revision surgery at a later date4. Penile traction devices can be used as an adjunct to preserve penile size in this instance66,67.
Concurrent penile lengthening and augmentation surgery
Most men with long-standing ED will develop loss of penile length owing to corporal hypoxia and fibrosis11,18. Complex penile reconstruction with concurrent placement is a demanding surgery and should only be performed by surgeons with extensive prosthetic and reconstructive experience. Proper informed consent should be obtained, as the risk of sensory loss, glans ischaemia and/or necrosis, prosthesis-related complications, and failure to gain any meaningful length are serious concerns and cannot be underestimated66.
Several advanced surgical techniques and adjuvant manoeuvres to increase penile length during IPP insertion, including circumferential incision68, the sliding technique with a double dorsal-ventral approach69, the modified sliding technique70 and the multiple slice technique71. These techniques require mobilization of neurovascular bundles and urethra, incision and/or excision of coexisting penile plaque and complete transection of corpora to stretch the penis to the maximum corrected length. The circumferential technique has been largely abandoned, owing to the risk of prosthesis fracture from poor axial rigidity, whereas sliding techniques can be performed with or without grafting72. The maximum length gain is likely limited by the elasticity of the neurovascular bundles66,72. Other surgical techniques that can enhance the perception of increased length include ventral phalloplasty, suprapubic lipectomy, and suspensory ligament release, all of which have been used to provide a subjective penile length gain66.
Girth enhancement can be obtained using augmentation corporoplasty and glans augmentation with hyaluronic acid and other fillers. Augmentation corporoplasty is not commonly done as an adjunct to PPI as it is not cost-effective and provides a marginal improvement (<10%) in the final penile size72. The use of synthetic materials (such as Gore-Tex, silicone), human grafts (venous or dermal) and treated biological materials (including InteXen, Tutoplast, AlloDerm) have been described for girth enhancement68,73. In general, synthetic materials are less preferred as their use is associated with intense fibrosis66. Human autologous grafts are associated with longer operative time and donor site complications66. The Penuma silicone sleeve implant, an FDA-cleared reversible penile implant for girth enhancement, is not intended to augment erectile function and can be associated with complications including seroma (4.8%), scar formation (4.5%), and infection (3.3%)74. At present, no published data are available regarding the Penuma cast in the setting of penile implants and it is not recognized as a standard of care.
Concurrent male continence surgery
In patients who have both ED and stress urinary incontinence, synchronous or sequential (delayed) IPP with male sling or artificial urinary sphincter (AUS) surgery can be performed effectively and safely18. Patients should be counselled regarding the benefits and drawbacks of single versus staged surgery. Technical considerations for the device placement will vary depending on the sequence of prosthetic surgery and the surgeon’s preferences.
Candidates for synchronous surgery are usually patients in whom conservative management for both conditions has been unsuccessful and who wish to undergo a single surgical procedure. In a carefully selected group of patients who understand the implications of combined procedures, a single-incision approach such as trans-scrotal insertion of IPP and AUS75 or a single perineal incision for combined IPP and MS76 is feasible and potentially more cost-effective than having a staged procedure77.
Pertinent points to discuss with prospective patients interested in synchronous dual implants include counselling on the complexity of the surgery, the potential higher complication rates, and the risk of losing both devices in the event of prosthetic infection18. Continence surgery should generally be performed before IPP surgery, any inadvertent urethral injury would require abandoning and would preclude preparation and implantation of both prostheses. The corporal dilation and placement of the IPP cylinder in the proximal corporal bodies can be completed without fear of dislodging both arms of the male sling. In AUS surgery, corporal dilatation during IPP surgery must be performed with caution in the proximal part of the corporal tissue near the AUS cuff, as both corporal bodies have already diverged; a single-step dilatation can sometimes be a better option than sequential dilatation to avoid injuring the urethra18.
A two-stage procedure requires more attentive surgery to avoid damaging various components of the existing implant and adds the complexity of possibly operating in less well-defined tissue planes from previous surgery78. Careful preoperative planning is required, including a review of the patient’s previous surgical records; and having pre-operative imaging with CT or MRI might be necessary at times to better delineate the anatomy. Great care should be taken to avoid damaging the existing device and a cutting current should be used wherever possible. In patients with a pre-existing AUS, the placement of IPP should be planned so that the pump and reservoir are on the contralateral side78.
Transgender men
Penile prosthesis surgery in the neophallus after gender-affirming surgery is often complex and should be reserved for when the neourethra has healed and reasonable genital sensitivity has developed. The neophallus and native phallus have pertinent anatomical differences, which can present surgical challenges, as the neophallus lacks proper corporal bodies and surrounding tunica albuginea79. Thus, IPP placement in the neophallus risks subsequent prosthesis malposition and distal erosion in up to one-third of cases79. Furthermore, anchoring the proximal end of the penile implant can be challenging owing to the lack of proximal crura and attachment to ischial rami. Extra care should be undertaken to avoid compromising the vascular supply of the neophallus; the use of intraoperative Doppler ultrasonography can be useful to identify the vascular pedicle to the neophallus79,80. The dorsal clitoral nerve should be preserved to provide sensation to the neophallus as decreased tactile sensation in the neophallus can be associated with a higher distal prosthesis erosion rate in transgender patients79,80.
An IPP is thought to be better and safer than malleable implants in the neophallus because it enables a more physiologically normal erection process, without constant pressure against the distal neophallus flap. The decision on the implantation of one or two cylinders is often made based on the size and girth of the neophallus to ensure that the cylinder is aesthetically acceptable, symmetrical, and not too superficial to the overlying skin. The proximal cylinder end can be anchored to the pubic bone to prevent proximal cylinder migration, either with direct placement of permanent sutures onto the proximal cylinder base or rear tip extender, or within a graft–cylinder complex (for example, the Dacron polyester vascular graft)80. The placement of the IPP reservoir and pump are in a standard manner although a smaller reservoir is sufficient in a single-cylinder implant and the pump is often placed on one side of the hemi-scrotum so that a separate testicular prosthesis can be inserted into the contralateral side. A specialized IPP designed to meet the specific needs of transgender patients after phalloplasty is a great unmet need, as transgender patients experience higher revision rates than cisgender men owing to complications related to prosthetic infection (8 to 50%), erosion (4 to 10%), malposition (3 to 30%) and mechanical failure (9 to 20%)79,80. The ZSI 475FtM is a specifically designed prosthesis for use in a neophallus, which consists of a single-cylinder (12, 15 or 17 cm in length) with a large, more glans-like distal tip, large stainless steel and silicone proximal base for pubic bone fixation, and a pump that resembles a testicle81.
Summary recommendations
Men with significant corporal fibrosis; those who receive concurrent continence surgery; and transgender men with a neophallus who are keen to undergo IPP surgery are complex cases that require expert care and advanced prosthetic techniques to obtain optimal surgical outcomes (Grade C). Men with significant corporal fibrosis due to priapism, PD, and prior explant of an infected IPP will require careful corporal dissection and dilation using specialized tools and, in some cases, a narrow cylinder implant or malleable implant can be used (Grade C). The decision to perform a salvage implant procedure in suspected or confirmed infected IPP should always involve both the surgeon and patient and the patient’s partner, with due consideration of the various options and scenarios (Grade C). Several advanced surgical techniques and adjuvant manoeuvres can be used to increase penile length during IPP insertion although these manoeuvres should be performed by an expert surgeon. The use of synthetic materials in girth enhancement surgery should be discouraged (Grade C). In patients who have both ED and stress urinary incontinence, synchronous or sequential (delayed) IPP with a male sling or artificial urinary sphincter surgery can be performed, and patients should be counselled regarding the benefits and drawbacks of single versus staged surgery (Grade C). Penile prosthesis surgery in the neophallus after gender-affirming surgery is often complex and should be performed by an expert surgeon owing to the anatomy of the neophallus and higher revision rates than for conventional IPP surgery (Grade C) (Box 1).
Postoperative care
The use of prostheses with antibiotic coatings has reduced the rates of infection or revision surgery related to infection82,83,84. Although the antibiotic-impregnated AMS 700 penile prosthesis series is widely available, non-antibiotic coated implants are used in some parts of the world owing to local device registration and governmental regulatory approval5,85. Thus, postoperative antimicrobial prophylaxis should be recommended only when the potential benefits outweigh the risks and anticipated costs — including the expense of the agent and its administration, the risk of allergic reactions or other adverse effects, and the potential for induction of bacterial resistance. However, many surgeons will prescribe postoperative antibiotics to fit and healthy patients based on their personal preference or intuition, which can be contrary to recommendations from local institution infectious disease guidelines86,87. Published best practice guidelines on surgery antimicrobial prophylaxis from the Urologic Surgery Antimicrobial Prophylaxis Best Practice Policy Panel in 2008 and the National Surgical Infection Prevention Project in 2005 (refs31,88) recommended that antibiotics should be discontinued within 24 h unless justified for specific antimicrobial prophylactic regimens in specific groups of patients, such as those who are immunosuppressed. Consultation with a local institution infectious disease physician regarding the types and duration of antimicrobial prophylaxis is valuable and can help with the selection of the best prophylactic agent, in terms of cost, convenience and safety profile.
Postoperative pain is a critical factor in the overall patient experience in penile prosthetic surgery and can substantially influence patient decision-making, particularly given the elective nature of IPP surgery and the growing concerns with long-term opioid use. Current practice has shifted towards improved perioperative local anaesthesia and postoperative analgesia cover89,90. Refinements in surgical techniques with less aggressive dissection and better surgical tools4,56 coupled with innovative intraoperative analgesia strategies such as dorsal penile nerve block and crural block91,92,93,94 and the use of drug elution hydrophilic IPPs95 have been shown to minimize postoperative pain scores and improve patient satisfaction rates. Furthermore, multimodal analgesia protocols adopted from other surgical fields and subspecialties have been implemented in IPP surgery with good outcomes96,97.
Several strategies to minimize the risk of scrotal bleeding and haematoma formation have been described, including watertight closure for corporotomies, partial cylinder inflation, compressive dressing and closed drainage98. Patients should be advised to avoid strenuous physical activity in the early postoperative period for the first 4 weeks. In most instances, the postoperative scrotal haematoma can be managed expectantly without a need for further surgical intervention40,99,100, using a combination of bed rest, ice application, scrotal support, compression dressings and prophylactic antibiotic use40. Surgical intervention with clot evacuation is mandated if patients present with an enlarging scrotal haematoma, prolonged pain and impending skin infection40,98,100. However, surgical exploration can be challenging during the early postoperative period owing to tissue oedema and can be associated with an increased risk of prosthesis infection40,100.
Early cycling of the IPP is thought to minimize corporal scarring and improve penile prosthesis appearance4,11,56; however, to do this, patients need to recover sufficiently to be able to distinguish the configuration of and squeeze the pump in the scrotum. Furthermore, the timing of the initial penile prosthesis inflation is dependent on many factors, such as postoperative scrotal pain, residual swelling, the extent of wound healing and the timing of the patient’s postoperative review appointment100. Most patients should be able to cycle their IPP between 4 to 6 weeks postoperatively, with minimal discomfort4,98,99,100 and patients who underwent an infrapubic or subcoronal approach in penile prosthesis implantation might be able to cycle their pump earlier than those who had penoscrotal surgery4,98. However, no randomized direct comparative study or properly designed and powered clinical study has shown significant benefits of earlier penile prosthesis implant cycling.
Summary recommendations
Immediate postoperative management centres around four domains: antibiotics use, pain management, scrotal care and cycling of penile prosthesis implant. No universal guidelines are currently available, meaning that clinical practice varies considerably. Postoperative antibiotics use can be discontinued within 24 h unless justified for specific antimicrobial prophylactic regimens in specific groups of patients and postoperative antimicrobial prescription is often based on the surgeon’s own personal preference in consultation with a local institution’s antimicrobial guidelines (Grade C). A multimodal analgesia protocol including intraoperative analgesia strategies can minimize postoperative pain and improve patient satisfaction rates (Grade C). Several strategies are aimed at minimizing the risk of scrotal bleeding and haematoma formation, and in most instances, the postoperative scrotal haematoma can be managed expectantly without a need for further surgical intervention (Grade B). Early cycling of the IPP is thought to minimize corporal scarring and improve the appearance of the penile prosthesis, and patients should be able to cycle their IPP between 4 to 6 weeks postoperatively, with minimal discomfort (Grade C) (Box 1).
Conclusions
The increased understanding of IPP surgery and advances in prosthetic surgical techniques have resulted in higher quality products and better surgical outcomes. Continued investment in research and development by device companies has resulted in the creation of a newer generation of penile prosthesis implants, whereas clinical governance by regulatory bodies ensures that these devices remain safe and reliable. Conducting true randomized, placebo-controlled trials in IPP surgery is difficult owing to ethical issues and patient recruitment; thus, more multi-institutional collaborative studies with longer-term follow-up should be undertaken with strict methodology and meaningful objective outcome measures.
Overall, IPP surgery is an effective, safe and durable treatment option for ED. Strict patient selection and counselling are essential, coupled with the use of evidence-based surgical principles and safe surgical techniques, as well as vigilant postoperative care, are paramount to ensuring the best clinical outcomes and patient satisfaction rates.
References
Scott, F. B., Bradley, W. E. & Timm, G. W. Management of erectile impotence: use of implantable inflatable prosthesis. Urology 2, 80–82 (1973).
Chung, E. Penile prosthesis implant: scientific advances and technological innovations over the last four decades. Transl. Androl. Urol. 6, 37–45 (2017).
Osmonov, D., Christopher, A. N. & Blecher, G. A. Clinical recommendations from the European Society of Sexual Medicine exploring partner expectations, satisfaction in male and phalloplasty cohorts, the impact of penile length, girth and implant type, reservoir placement, and influence of comorbidities and social circumstances. J. Sex. Med 17, 210–237 (2020).
Levine, L. A. et al. Penile prosthesis surgery: current recommendations from the International Consultation on Sexual Medicine. J. Sex. Med. 13, 489–518 (2016).
The Boston Scientific AMS 700 series. https://www.bostonscientific.com/en-US/products/penile-prosthesis/ams-700-inflatable-penile-prosthesis.html (2021).
The Coloplast Titan series. https://www.coloplastmenshealth.com/erectile-dysfunction/penile-implants/titan-inflatable-penile-implant/ (2021).
The Zephyr ZSI475 series. https://www.zsimplants.ch/en/products-en/erectile-dysfunction (2021).
The Rigicon Infla10 series. https://www.rigicon.com/inflatable-penile-prosthesis (2021).
Guyatt, G. H., GRADE Working Group et al. Going from evidence to recommendations. BMJ 336, 1049 (2008).
Porst, H. et al. SOP conservative (medical and mechanical) treatment of erectile dysfunction. J. Sex. Med 10, 130–171 (2013).
Chung, E. et al. Evidence-based management guidelines on Peyronie’s disease. J. Sex. Med 13, 905–923 (2016).
Meuleman, E. J. et al. Diagnostic tests for male erectile dysfunction revisited. Committee consensus report of the international consultation in sexual medicine. J. Sex. Med 7, 2375–2381 (2010).
Hebert, K. J. & Kohler, T. S. Penile prosthesis infection: myths and realities. World J. Mens Health 37, 276–287 (2019).
Scherzer, N. D., Dick, B., Gabrielson, A. T., Alzweri, L. M. & Hellstrom, W. J. G. Penile prosthesis complications: planning, prevention, and decision making. Sex. Med. Rev. 7, 349–359 (2019).
BAUS. https://www.baus.org.uk/_userfiles/pages/files/Patients/Leaflets/Penile%20prostheses.pdf (2020).
SMSNA. https://www.smsna.org/V1/images/SMSNAIPP_policy.pdf (2021).
Sunaryo, P. L., Colaco, M. & Terlecki, R. Penile prostheses and the litigious patient: a legal database review. J. Sex. Med. 11, 2589–2594 (2014).
Chung, E. Penile prosthesis implant in the special populations: diabetics, neurogenic conditions, fibrotic cases, concurrent urinary incontinence, and salvage implants. Asian J. Androl. 22, 39–44 (2020).
Trost, L. W., Baum, N. & Hellstrom, W. J. G. Managing the difficult penile prosthesis patient. J. Sex. Med 10, 893–907 (2013).
Gon, L. M. et al. A systematic review of penile prosthesis infection and meta-analysis of diabetes mellitus role. BMC Urol. 21, 35 (2021).
Pearson-Stuttard, J., Blundell, S., Harris, T., Cook, D. G. & Critchley, J. Diabetes and infection: assessing the association with glycemic control in population-based studies. Lancet Diabetes Endocrinol. 4, 148–158 (2016).
Habous, M. et al. Defining a glycated haemoglobin (HbA1c) level that predicts increased risk of penile implant infection. BJU Int 121, 293–300 (2018).
Dick, B. P., Yousif, A., Raheem, O. & Hellstrom, W. J. G. Does lowering the haemoglobin A1c reduce penile prosthesis infection: a systematic review. Sex. Med Rev. S2050-0521, 30068–30068 (2020).
Huynh, L. M., Huang, E., El-Khatib, F. M., Gross, M. S. & Yafi, F. A. A systematic review of literature regarding whether immediate preoperative haemoglobin A1c or serum glucose are risk factors for infection following penile prosthesis implantation. Urology 152, 12–54 (2021).
Talib, R. et al. HbA1c over 8.5% is not predictive of increased infection rate following penile prosthesis implant in diabetic patients with erectile dysfunction. Andrologia 53, e14132 (2021).
Osman, M. M. et al. Immediate preoperative blood glucose and hemoglobin a1c levels are not predictive of postoperative infections in diabetic men undergoing penile prosthesis implant. Int. J. Impot. Res 33, 296–302 (2021).
Carvajal, A., Benavides, J., Garcia-Perdomo, H. A. & Henry, G. D. Risk factors association with penile prosthesis infection: systematic review and meta-analysis. Int. J. Impot. Res 32, 587–597 (2020).
Aniskevich, S., Renew, J. R., Chadha, R. M. & Irizarry-Alvarado, J. M. Pharmacology and perioperative considerations for diabetes mellitus medications. Curr. Clin. Pharm. 12, 157–163 (2017).
WHO guidelines for safe surgery 2009: Safe surgery saves lives. Geneva: World Health Organization; 2006. Objective 6, the team will consistently use methods known to minimize the minimize the risk for surgical site infection. https://www.ncbi.nlm.nih.gov/books/NBK143246 (2021).
Pineda, M. & Burnett, A. L. Penile prosthesis infections — a review of risk factors, prevention, and treatment. Sex. Med. Rev. 4, 389–398 (2016).
Mandava, S. H., Serefoglu, E. C., Freier, M. T., Wilson, S. K. & Hellstrom, W. J. Infection retardant coated inflatable penile prostheses decrease the incidence of infection: a systematic review and meta-analysis. J. Urol. 188, 1855–1860 (2012).
Wolf, J. S. Jr et al. Urologic surgery antimicrobial prophylaxis best practice policy panel. Best practice policy statement on urologic surgery antimicrobial prophylaxis. J. Urol. 179, 1379–1390 (2008).
Herati, A. S. & Lo, E. M. Penile prosthesis biofilm formation and emerging therapies against them. Trans. Androl. Urol. 7, 960–967 (2018).
Katz, B. F. et al. Use of a preoperative checklist reduces risk of penile prosthesis infection. J. Urol. 192, 130–135 (2014).
Gross, M. S. et al. Multicenter investigation of the micro-organisms involved in penile prosthesis infection: an analysis of the efficacy of the AUA and EAU guidelines for penile prosthesis prophylaxis. J. Sex. Med 14, 455–463 (2017).
Darouiche, R. O. et al. North American consensus document on infection of penile prostheses. Urology 82, 937–942 (2013).
Giles, C., Lamont-Friedrich, S. J., Michl, T. D., Griesser, H. J. & Coad, B. R. The importance of fungal pathogens and antifungal coatings in medical device infections. Biotechnol. Adv. 36, 264–280 (2018).
Nehra, A. et al. The Princeton III consensus recommendations for the management of erectile dysfunction and cardiovascular disease. Mayo Clin. Proc. 87, 766–778 (2012).
Scherzer, N. D., Dick, B., Gabrielson, A. T., Alzweri, L. M. & Hellstrom, W. J. G. Penile prosthesis complications: planning, prevention, and decision making. Sex. Med. Rev. 7, 349–359 (2019).
Garber, B. B. & Bickell, M. Delayed postoperative hematoma formation after inflatable penile prosthesis implantation. J. Sex. Med 12, 265–269 (2015).
Culkin, D. J. et al. Anticoagulation and antiplatelet therapy in urological practice: ICUD/AUA review paper. J. Urol. 192, 1026–1034 (2014).
Lotan, Y. et al. Factors influencing the outcomes of penile prosthesis surgery at a teaching institution. Urology 62, 918–921 (2003).
Sharma, D. & Smith, R. P. Troubleshooting intraoperative complications of penile prosthesis placement. Trans. Androl. Urol. 6, S892–S897 (2017).
Oberlin, D. T. et al. National practice patterns of treatment of erectile dysfunction with penile prosthesis implantation. J. Urol. 193, 2040–2044 (2015).
Onyeji, I. C. et al. Impact of surgeon case volume on reoperation rates after inflatable penile prosthesis surgery. J. Urol. 197, 223–229 (2017).
Henry, G. D. et al. Centers of excellence concept and penile prostheses: an outcome analysis. J. Urol. 181, 1264–1268 (2009).
Kashanian, J. A. et al. Trends in penile prosthetics: influence of patient demographics, surgeon volume, and hospital volume on type of penile prosthesis inserted in New York State. J. Sex. Med 15, 245–250 (2018).
Palmisano, F. et al. Comparison of infrapubic vs penoscrotal approaches for 3-piece inflatable penile prosthesis placement: do we have a winner? Sex. Med. Rev. 6, 631–639 (2018).
Trost, L. W., Boonjindasup, A. G. & Hellstrom, W. J. Comparison of infrapubic versus transscrotal approaches for inflatable penile prosthesis placement: a multi-institution report. Int. J. Impot. Res. 27, 86–89 (2015).
Henry, G. D. et al. Surgical techniques in penoscrotal implantation of an inflatable penile prosthesis: a guide to increasing patient satisfaction and surgeon ease. Sex. Med. Rev. 3, 36–47 (2015).
Wallen, J. J. et al. Biochemical comparison of inflatable penile implants: a cadaveric pilot study. J. Sex. Med 15, 1034–1040 (2018).
Levine, L. A. & Hoeh, M. P. Review of penile prosthetic reservoir: complications and presentation of a modified reservoir placement technique. J. Sex. Med 9, 2759–2769 (2012).
Perito, P. E. & Wilson, S. K. Traditional (retroperitoneal) and abdominal wall (ectopic) reservoir placement. J. Sex. Med. 8, 656–659 (2011).
Karpman, E. et al. Reservoir alternate surgical implantation technique: preliminary outcomes of initial PROPPER study of low profile or spherical reservoir implantation in submuscular location or traditional prevesical space. J. Urol. 193, 239–244 (2015).
Karpman, E., Sadeghi-Nejad, H., Henry, G., Khera, M. & Morey, A. F. Current opinions on alternative reservoir placement for inflatable penile prosthesis among members of the Sexual Medicine Society of North America. J. Sex. Med. 10, 2115–2120 (2013).
Trost, L., Patil, M. & Kramer, A. Critical appraisal and review of management strategies for severe fibrosis during penile implant surgery. J. Sex. Med. 12, 439–447 (2015).
Wilson, S. K., Delk, J. R., Mulcahy, J. J., Cleves, M. & Salem, E. A. Upsizing of inflatable penile implant cylinders in patients with corporal fibrosis. J. Sex. Med. 3, 736–742 (2006).
Zacharakis, E. et al. Penile prosthesis insertion in patients with refractory ischaemic priapism: early vs delayed implantation. BJU Int. 114, 576–581 (2014).
Ralph, D. J. et al. The immediate insertion of a penile prosthesis for acute ischaemic priapism. Eur. Urol. 56, 1033–1038 (2009).
Durazi, M. H. & Jalal, A. A. Penile prosthesis implantation for treatment of postpriapism erectile dysfunction. Urol. J. 5, 115–119 (2008).
Sedigh, O. et al. Early insertion of inflatable prosthesis for intractable ischemic priapism: our experience and review of the literature. Int. J. Impot. Res 23, 158–164 (2011).
Chung, E., Wang, R., Ralph, D., Levine, L. & Brock, G. A worldwide survey on Peyronie’s disease surgical practice patterns among surgeons. J. Sex. Med 15, 568–575 (2018).
Chung, E., Solomon, M., De Young, L. & Brock, G. B. Comparison between AMS 700 CX and Coloplast Titan inflatable penile prosthesis for Peyronie’s disease treatment and remodelling: clinical outcomes and patient satisfaction. J. Sex. Med. 10, 2855–2860 (2013).
Lao M., Graydon R. J., Bieniek J. M. Salvage penile prosthetic surgery utilizing temporary malleable implants. Trans. Androl. Urol. 6, S806–S812 (2017).
Brant, M. D., Ludlow, J. K. & Mulcahy, J. J. Prosthesis salvage operation: immediate replacement of infected penile prostheses. J. Urol. 155, 155–157 (1996).
Chung, E. Penile reconstructive surgery in Peyronie’s disease: challenges in restoring normal penis size, shape and function. World J. Mens. Health 36, e10–e17 (2018).
Sultana, A., Grice, P., Vukina, J., Pearce, I. & Modgil, V. Indications and characteristics of penile traction and vacuum erection devices. Nat. Rev. Urol. 19, 84–100 (2022).
Sansalone, S. et al. Simultaneous penile lengthening and penile prosthesis implantation in patients with Peyronie’s disease, refractory erectile dysfunction, and severe penile shortening. J. Sex. Med. 9, 316–321 (2012).
Rolle, L. et al. A new, innovative, lengthening surgical procedure for Peyronie’s disease by penile prosthesis implantation with double dorsal-ventral patch graft: the “sliding technique. J. Sex. Med. 9, 2389–2395 (2012).
Egydio, P. H., Kuehhas, F. E. & Valenzuela, R. J. Modified sliding technique (MoST) for penile lengthening with insertion of inflatable penile prosthesis. J. Sex. Med. 12, 1100–1104 (2015).
Egydio, P. H. & Kuehhas, F. E. The multiple-slit technique (MUST) for penile length and girth restoration. J. Sex. Med. 15, 261–269 (2018).
Tran, H., Goldfarb, R., Ackerman, A. & Valenzuela, R. J. Penile lengthening, girth, and size preservation at the time of penile prosthesis insertion. Sex. Med. Rev. 5, 403–412 (2017).
Austoni, E., Guarneri, A. & Cazzaniga, A. A new technique for augmentation phalloplasty: albugineal surgery with bilateral saphenous grafts — three years of experience. Eur. Urol. 42, 245–253 (2002). discussion 252–253.
Elist, J. J. et al. A single-surgeon, retrospective, and preliminary evaluation of the safety and effectiveness of the penuma silicone sleeve implant for elective cosmetic correction of the flaccid penis. J. Sex. Med. 15, 1216–1223 (2018).
Rolle, L. et al. Surgical implantation of artificial urinary device and penile prosthesis through trans-scrotal incision for postprostatectomy urinary incontinence and erectile dysfunction: synchronous or delayed procedure? Urology 80, 1046–1050 (2012).
Gorbatiy, V., Westney, O. L., Romero, C. & Wang, R. Outcomes of simultaneous placement of an inflatable penile prosthesis and a male urethral sling through a single perineal incision. J. Sex. Med. 7, 832–838 (2010).
Patel, N. et al. A contemporary analysis of dual inflatable penile prosthesis and artificial urinary sphincter outcomes. J. Urol. 201, 141–146 (2019).
Chung, E. Artificial urinary sphincter surgery in the special populations: neurological, revision, concurrent penile prosthesis and female stress urinary incontinence groups. Asian J. Androl. 22, 45–50 (2020).
Rooker, S. A. et al. The rise of the neophallus: a systematic review of penile prosthetic outcomes and complications in gender-affirming surgery. J. Sex. Med 16, 661–672 (2019).
Kang, A., Aizen, J. M., Cohen, A. J., Bales, G. T. & Pariser, J. J. Techniques and considerations of prosthetic surgery after phalloplasty in the transgender male. Transl. Androl. Urol. 8, 273–282 (2019).
Neuville, P. et al. First outcomes of the ZSI 475 FtM, a specific prosthesis designed for phalloplasty. J. Sex. Med. 16, 316–322 (2019).
Dhabuwala, C., Sheth, S. & Zamzow, B. Infection rates of rifampin/gentamicin-coated Titan Coloplast penile implants. Comparison with Inhibizone-impregnated AMS penile implants. J. Sex. Med. 8, 315–320 (2011).
Wosnitzer, M. S. & Greenfield, J. M. Antibiotic patterns with inflatable penile prosthesis insertion. J. Sex. Med. 8, 1521–1528 (2011).
Abouassaly, R., Angermeier, K. W. & Montague, D. K. Risk of infection with an antibiotic coated penile prosthesis at device replacement for mechanical failure. J. Urol. 176, 2471–2473 (2006).
Song, W. D. et al. Penile prosthesis implant in Chinese patients with severe erectile dysfunction: 10-year experience. Asian J. Androl. 15, 658–661 (2013).
Dropkin, B. M. et al. Penile prosthesis insertion in the era of antibiotic stewardship are postoperative antibiotics necessary? J. Urol. 203, 611–614 (2020).
Hinds, P. R., Wilson, S. K. & Sadeghi-Nejad, H. Dilemmas of inflatable penile prosthesis revision surgery: what practices achieve the best outcomes and the lower infection rates? (CME). J. Sex. Med. 9, 2483–2491 (2012).
Bratzler, D. W. & Houck, P. M., Surgical Infection Prevention Guideline Writers Workgroup. Antimicrobial prophylaxis for surgery: an advisory statement from the national surgical infection prevention project. Am. J. Surg. 189, 395–404 (2005).
Reinstatler, L., Shee, K. & Gross, M. S. Pain management in penile prosthetic surgery: a review of the literature. Sex. Med. Rev. 6, 162–169 (2018).
Ellis, J. L., Higgins, A. M. & Simhan, J. Pain management strategies in penile implantation. Asian J. Androl. 22, 34–38 (2020).
Raynor, M. C., Smith, A., Vyas, S. N., Selph, J. P. & Carson, C. C. Dorsal penile nerve block prior to inflatable penile prosthesis. J. Sex. Med. 9, 2975–2979 (2012).
Weinberg, A., Pagano, M. & Valenzuela, R. Preoperative local anesthetic block reduces the need for narcotic medications following penile prosthesis surgery. J. Sex. Med. 13, S66–S67 (2016).
Hsu, G. et al. Outpatient penile implantation with the patient under a novel method of crural block. Int. J. Androl. 151, 147–151 (2004).
Xie, D. et al. Effect of operative local anesthesia on postoperative pain outcomes of inflatable penile prosthesis: prospective comparison of two medications. Int. J. Impot. Res 30, 93–96 (2018).
Chung, E. Local anaesthesia eluting property of Coloplast titan penile prosthesis hydrophilic coating: an in-vitro drug elution profile and a randomized double-blind clinical outcome study. J. Sex. Med. 15, S149 (2018).
Ellis, J. L. et al. Pain management strategies in contemporary penile implant recipients. Curr. Urol. Rep. 22, 17 (2021).
Tong, C. M. C. et al. Novel multi-modal analgesia protocol significantly decreases opioid requirements in inflatable penile prosthesis patients. J. Sex. Med. 15, 1187–1194 (2018).
O’Rourke, T. K., Erbella, A., Zang, Y. & Wosnitzer, M. S. Prevention, identification, and management of post-operative penile implant complications of infection, hematoma, and device malfunction. Transl. Androl. Urol. 6, S832–S848 (2017).
Chung, E. et al. Penile prosthesis implantation for the treatment for male erectile dysfunction: clinical outcomes and lessons learnt after 955 procedures. World J. Urol. 31, 591–595 (2013).
Sadeghi-Nejad, H. et al. Multi-institutional outcome study on the efficacy of closed-suction drainage of the scrotum in three-piece inflatable penile prosthesis surgery. Int. J. Impot. Res. 17, 535–538 (2005).
Author information
Authors and Affiliations
Contributions
All authors researched data for the article, contributed substantially to discussion of the content, and reviewed and/or edited the manuscript before submission. E.C. and G.B. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
E.C., C.B., P.E., C.L., D.O., S.P., D.R. and G.B. declare honoraria and/or speaker’s fees from Coloplast. E.C., C.B., D.O., S.P., D.R., Z.C.X. and G.B. declare honoraria and/or speaker’s fees from Boston Scientific.
Peer review
Peer review information
Nature Reviews Urology thanks Steven Wilson, John Mulcahy and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chung, E., Bettocchi, C., Egydio, P. et al. The International Penile Prosthesis Implant Consensus Forum: clinical recommendations and surgical principles on the inflatable 3-piece penile prosthesis implant. Nat Rev Urol 19, 534–546 (2022). https://doi.org/10.1038/s41585-022-00607-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41585-022-00607-z
This article is cited by
-
Penile implants in low flow priapism
International Journal of Impotence Research (2023)
-
History of penile implants: from implants made of bone to modern inflatable penile implants
International Journal of Impotence Research (2023)
-
Comment on “Comparison of patient-reported outcomes after penile prosthesis placement in men with and without Peyronie’s disease”
International Journal of Impotence Research (2023)
-
A narrative review on malleable and inflatable penile implants: choosing the right implant for the right patient
International Journal of Impotence Research (2023)
-
Perioperative outcomes of penile prosthesis implantation in Germany: results from the GRAND study
International Journal of Impotence Research (2023)