Abstract
A placebo is an inert substance normally used in clinical trials for comparison with an active substance. However, a placebo has been shown to have an effect on its own; commonly known as the placebo effect. A placebo is an essential component in the design of conclusive clinical trials but has itself become the focus of intense research. The placebo effect is partly the result of positive expectations of the recipient on the state of health. Conversely, a nocebo effect is when negative expectations from a substance lead to poor treatment outcomes and/or adverse events. Randomized controlled trials in functional urology have demonstrated the importance of the placebo and nocebo effects across different diseases such as overactive bladder, urinary incontinence, lower urinary tract symptoms and interstitial cystitis/painful bladder syndrome, as well as male and female sexual dysfunction. Understanding the true nature of the placebo–nocebo complex and the scope of its effect in functional urology could help urologists to maximize the positive effects of this phenomenon while minimizing its potentially negative effects.
Key points
-
Placebo and nocebo effects have major roles in functional urological ailments.
-
The mechanisms by which the placebo and nocebo effects function are not fully understood.
-
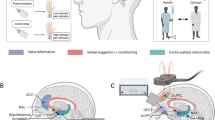
The pontine micturition centre might be affected by positive and negative expectations through a cascade of events resulting in improvement or worsening of functional urological symptoms.
-
Clinicians need to consider the placebo and nocebo phenomena when managing a patient with a functional urological ailment.
-
Clinicians should be trained regarding the placebo and nocebo effects with the aim of maximizing the benefits of the placebo effect and minimizing the harms of the nocebo effect.
-
An individualized approach and shared decision-making should be performed when dealing with placebo and nocebo effects, as each individual has different perceptions with regard to placebo and nocebo phenomena.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Stewart-Williams, S. & Podd, J. The placebo effect: dissolving the expectancy versus conditioning debate. Psychol. Bull. 130, 324 (2004).
McDonald, C. J., Mazzuca, S. A. & McCabe, G. P. Jr How much of the placebo ‘effect’ is really statistical regression? Stat. Med. 2, 417–427 (1983).
Colloca, L. The placebo effect in pain therapies. Annu. Rev. Pharmacol. Toxicol. 59, 191–211 (2019).
Aronson, J. Please, please me. BMJ 318, 716 (1999).
Benedetti, F., Mayberg, H. S., Wager, T. D., Stohler, C. S. & Zubieta, J.-K. Neurobiological mechanisms of the placebo effect. J. Neurosci. 25, 10390–10402 (2005).
Gaab, J. The placebo and its effects: a psychoneuroendocrinological perspective. Psychoneuroendocrinology 105, 3–8 (2019).
Evers, A. W. et al. Implications of placebo and nocebo effects for clinical practice: expert consensus. Psychother. Psychosom. 87, 204–210 (2018).
Ernst, E. & Resch, K. L. Concept of true and perceived placebo effects. BMJ 311, 551–553 (1995).
Colloca, L. & Barsky, A. J. Placebo and nocebo effects. N. Engl. J. Med. 382, 554–561 (2020).
Gupta, U. & Verma, M. Placebo in clinical trials. Perspect. Clin. Res. 4, 49 (2013).
Lipman, J. J. et al. Peak B endorphin concentration in cerebrospinal fluid: reduced in chronic pain patients and increased during the placebo response. Psychopharmacology 102, 112–116 (1990).
Pollo, A. et al. Expectation modulates the response to subthalamic nucleus stimulation in Parkinsonian patients. Neuroreport 13, 1383–1386 (2002).
Howick, J. et al. Are treatments more effective than placebos? A systematic review and meta-analysis. PLoS ONE 8, e62599 (2013).
Howick, J. H. The Philosophy of Evidence-Based Medicine (John Wiley & Sons, 2011).
Mangera, A., Chapple, C. R., Kopp, Z. S. & Plested, M. The placebo effect in overactive bladder syndrome. Nat. Rev. Urol. 8, 495 (2011).
Chvetzoff, G. & Tannock, I. F. Placebo effects in oncology. J. Natl Cancer Inst. 95, 19–29 (2003).
Kaptchuk, T. J. & Miller, F. G. Placebo effects in medicine. N. Engl. J. Med. 373, 8–9 (2015).
Bishop, F. L. et al. What techniques might be used to harness placebo effects in non-malignant pain? A literature review and survey to develop a taxonomy. BMJ Open 7, e015516 (2017).
Beecher, H. K. The powerful placebo. J. Am. Med. Assoc. 159, 1602–1606 (1955).
Häuser, W., Hansen, E. & Enck, P. Nocebo phenomena in medicine: their relevance in everyday clinical practice. Dtsch. Ärztebl. Int. 109, 459 (2012).
Kravvariti, E., Kitas, G. D., Mitsikostas, D. D. & Sfikakis, P. P. Nocebos in rheumatology: emerging concepts and their implications for clinical practice. Nat. Rev. Rheumatol. 14, 727–740 (2018).
Colagiuri, B., Schenk, L. A., Kessler, M. D., Dorsey, S. G. & Colloca, L. The placebo effect: from concepts to genes. Neuroscience 307, 171–190 (2015).
Fields, H. L. & Levine, J. D. Placebo analgesia — a role for endorphins? Trends Neurosci. 7, 271–273 (1984).
Peciña, M., Heffernan, J., Wilson, J., Zubieta, J. & Dombrovski, A. Prefrontal expectancy and reinforcement-driven antidepressant placebo effects. Transl. Psychiatry 8, 1–11 (2018).
Mostafaei, H. et al. Nocebo response in the pharmacological management of overactive bladder: a systematic review and meta-analysis. Eur. Urol. Focus 7, 1143–1153 (2020).
Colloca, L. & Miller, F. G. Harnessing the placebo effect: the need for translational research. Philos. Trans. R. Soc. B Biol. Sci. 366, 1922–1930 (2011).
Hróbjartsson, A. & Gøtzsche, P. C. Placebo interventions for all clinical conditions (2010 update). Cochrane Database Syst. Rev. 2010, CD003974 (2010).
Hróbjartsson, A. & Gøtzsche, P. C. Is the placebo powerless? Update of a systematic review with 52 new randomized trials comparing placebo with no treatment. J. Intern. Med. 256, 91–100 (2004).
Barnett, A. G., Van Der Pols, J. C. & Dobson, A. J. Regression to the mean: what it is and how to deal with it. Int. J. Epidemiol. 34, 215–220 (2005).
Kienle, G. S. & Kiene, H. The powerful placebo effect: fact or fiction? J. Clin. Epidemiol. 50, 1311–1318 (1997).
Hróbjartsson, A. & Gøtzsche, P. C. Placebo interventions for all clinical conditions. Cochrane Database Syst. Rev. 2004, CD003974 (2004).
Benedetti, F., Lanotte, M., Lopiano, L. & Colloca, L. When words are painful: unraveling the mechanisms of the nocebo effect. Neuroscience 147, 260–271 (2007).
Kong, J. et al. A functional magnetic resonance imaging study on the neural mechanisms of hyperalgesic nocebo effect. J. Neurosci. 28, 13354–13362 (2008).
Nolan, T. The placebo effect in general practice. InnovAiT 12, 404–407 (2019).
Wells, R. E. & Kaptchuk, T. J. To tell the truth, the whole truth, may do patients harm: the problem of the nocebo effect for informed consent. Am. J. Bioeth. 12, 22–29 (2012).
Tyrer, P., Eilenberg, T., Fink, P., Hedman, E. & Tyrer, H. Health anxiety: the silent, disabling epidemic. BMJ 353, i2250 (2016).
Planès, S., Villier, C. & Mallaret, M. The nocebo effect of drugs. Pharm. Res. Perspect. 4, e00208 (2016).
Cohen, S. The nocebo effect of informed consent. Bioethics 28, 147–154 (2014).
Ockene, J. K. et al. Symptom experience after discontinuing use of estrogen plus progestin. JAMA 294, 183–193 (2005).
Vase, L., Robinson, M. E., Verne, G. N. & Price, D. D. The contributions of suggestion, desire, and expectation to placebo effects in irritable bowel syndrome patients: an empirical investigation. Pain 105, 17–25 (2003).
Price, D. D. et al. An analysis of factors that contribute to the magnitude of placebo analgesia in an experimental paradigm. Pain 83, 147–156 (1999).
Benedetti, F., Arduino, C. & Amanzio, M. Somatotopic activation of opioid systems by target-directed expectations of analgesia. J. Neurosci. 19, 3639–3648 (1999).
Goebel, M. U. et al. Behavioral conditioning of immunosuppression is possible in humans. FASEB J. 16, 1869–1873 (2002).
Benedetti, F. et al. Conscious expectation and unconscious conditioning in analgesic, motor, and hormonal placebo/nocebo responses. J. Neurosci. 23, 4315–4323 (2003).
Petrie, K. J. & Rief, W. Psychobiological mechanisms of placebo and nocebo effects: pathways to improve treatments and reduce side effects. Annu. Rev. Psychol. 70, 599–625 (2019).
Bąbel, P. Classical conditioning as a distinct mechanism of placebo effects. Front. Psychiatry 10, 449 (2019).
Schedlowski, M., Enck, P., Rief, W. & Bingel, U. Neuro-bio-behavioral mechanisms of placebo and nocebo responses: implications for clinical trials and clinical practice. Pharmacol. Rev. 67, 697–730 (2015).
Colloca, L. & Finniss, D. Nocebo effects, patient-clinician communication, and therapeutic outcomes. JAMA 307, 567–568 (2012).
Nestoriuc, Y., Orav, E. J., Liang, M. H., Horne, R. & Barsky, A. J. Prediction of nonspecific side effects in rheumatoid arthritis patients by beliefs about medicines. Arthritis Care Res. 62, 791–799 (2010).
Papadopoulos, D. & Mitsikostas, D. Nocebo effects in multiple sclerosis trials: a meta-analysis. Mult. Scler. J. 16, 816–828 (2010).
He, J., Morales, D. R. & Guthrie, B. Exclusion rates in randomized controlled trials of treatments for physical conditions: a systematic review. Trials 21, 1–11 (2020).
Jensen, J. S., Bielefeldt, A. Ø. & Hróbjartsson, A. Active placebo control groups of pharmacological interventions were rarely used but merited serious consideration: a methodological overview. J. Clin. Epidemiol. 87, 35–46 (2017).
Faria, V. et al. Do you believe it? Verbal suggestions influence the clinical and neural effects of escitalopram in social anxiety disorder: a randomized trial. EBioMedicine 24, 179–188 (2017).
Colloca, L., Lopiano, L., Lanotte, M. & Benedetti, F. Overt versus covert treatment for pain, anxiety, and Parkinson’s disease. Lancet Neurol. 3, 679–684 (2013).
Santana, L. & Fontenelle, L. F. A review of studies concerning treatment adherence of patients with anxiety disorders. Patient Prefer. Adherence 5, 427–439 (2011).
Chaliha, C. & Stanton, S. The ethnic cultural and social aspects of incontinence — a pilot study. Int. Urogynecol. J. 10, 166–170 (1999).
Greville-Harris, M. & Dieppe, P. Bad is more powerful than good: the nocebo response in medical consultations. Am. J. Med. 128, 126–129 (2015).
van Leeuwen, J. H. S., Castro, R., Busse, M. & Bemelmans, B. L. The placebo effect in the pharmacologic treatment of patients with lower urinary tract symptoms. Eur. Urol. 50, 440–453 (2006).
Enck, P., Bingel, U., Schedlowski, M. & Rief, W. The placebo response in medicine: minimize, maximize or personalize? Nat. Rev. Drug Discov. 12, 191 (2013).
Colloca, L., Sigaudo, M. & Benedetti, F. The role of learning in nocebo and placebo effects. Pain 136, 211–218 (2008).
Mondaini, N. et al. Finasteride 5 mg and sexual side effects: how many of these are related to a nocebo phenomenon? J. Sex. Med. 4, 1708–1712 (2007).
Many sexual adverse effects of finasteride are attributable to a nocebo effect. Nat. Clin. Pract. Urol. 5, 66–66 (2008).
Walsh, J. J. Old-Time Makers of Medicine: The Story of The Students And Teachers of the Sciences Related to Medicine During the Middle Ages (Good Press, 2019).
Gliedman, L. H., Gantt, W. H. & Teitelbaum, H. A. Some implications of conditional reflex studies for placebo research. Am. J. Psychiatry 113, 1103–1107 (1957).
Kurland, A. A. The drug placebo — its psychodynamic and conditional reflex action. Behav. Sci. 2, 101–110 (1957).
Pavlov, I. P. & Anrep, G. V. Conditioned Reflexes: an Investigation of the Physiological Activity of the Cerebral Cortex (Oxford University Press, 1927).
Lang, W. & Rand, M. A placebo response as a conditional reflex to glyceryl trinitrate. Med. J. Aust. 1, 12–14 (1969).
Kirsch, I. & Heap, M. Hypnosis: Theory, Research and Application (Routledge, 2017).
Kirsch, I. Response expectancy as a determinant of experience and behavior. Am. Psychol. 40, 1189 (1985).
Kirsch, I. & Weixel, L. J. Double-blind versus deceptive administration of a placebo. Behav. Neurosci. 102, 319 (1988).
Bąbel, P. et al. How classical conditioning shapes placebo analgesia: hidden versus open conditioning. Pain. Med. 19, 1156–1169 (2018).
Bąbel, P. et al. Classical conditioning without verbal suggestions elicits placebo analgesia and nocebo hyperalgesia. PLoS ONE 12, e0181856 (2017).
Manchikanti, L., Giordano, J., Fellows, B. & Hirsch, J. A. Placebo and nocebo in interventional pain management: a friend or a foe-or simply foes. Pain. Phys. 14, E157–E175 (2011).
Wager, T. D. et al. Placebo-induced changes in FMRI in the anticipation and experience of pain. Science 303, 1162–1167 (2004).
Amanzio, M., Benedetti, F., Porro, C. A., Palermo, S. & Cauda, F. Activation likelihood estimation meta-analysis of brain correlates of placebo analgesia in human experimental pain. Hum. Brain Mapp. 34, 738–752 (2013).
Benedetti, F. Placebo effects: from the neurobiological paradigm to translational implications. Neuron 84, 623–637 (2014).
Blok, B. F. Central pathways controlling micturition and urinary continence. Urology 59, 13–17 (2002).
Benedetti, F. Placebo Effects (Oxford University Press, USA, 2014).
Colloca, L., Flaten, M. A. & Meissner, K. Placebo and Pain: from Bench to Bedside (Academic Press, 2013).
Holstege, G. The emotional motor system and micturition control. Neurourol. Urodyn. 29, 42–48 (2010).
Blok, B. F. & Holstege, G. Direct projections from the periaqueductal gray to the pontine micturition center (M-region). An anterograde and retrograde tracing study in the cat. Neurosci. Lett. 166, 93–96 (1994).
Yoshimura, N. et al. Therapeutic receptor targets for lower urinary tract dysfunction. Naunyn Schmiedebergs Arch. Pharmacol. 377, 437–448 (2008).
Andersson, K.-E. & Gratzke, C. in Textbook of the Neurogenic Bladder 95–114 (CRC Press, 2008).
De la Fuente-Fernández, R. et al. Expectation and dopamine release: mechanism of the placebo effect in Parkinson’s disease. Science 293, 1164–1166 (2001).
de la Fuente-Fernández, R., Schulzer, M. & Stoessl, A. J. Placebo mechanisms and reward circuitry: clues from Parkinson’s disease. Biol. Psychiatry 56, 67–71 (2004).
Van der Aa, F., Ost, D. & De Ridder, D. J. M. K. Interstitial cells of the bladder: the missing link? BJOG 111, 57–60 (2004).
Amanzio, M. & Benedetti, F. Neuropharmacological dissection of placebo analgesia: expectation-activated opioid systems versus conditioning-activated specific subsystems. J. Neurosci. 19, 484–494 (1999).
Levine, J., Gordon, N. & Fields, H. The mechanism of placebo analgesia. Lancet 312, 654–657 (1978).
Benedetti, F. The opposite effects of the opiate antagonist naloxone and the cholecystokinin antagonist proglumide on placebo analgesia. Pain 64, 535–543 (1996).
Pertwee, R. G. & Fernando, S. R. Evidence for the presence of cannabinoid CB1 receptors in mouse urinary bladder. Br. J. Pharmacol. 118, 2053–2058 (1996).
Freeman, R. et al. The effect of cannabis on urge incontinence in patients with multiple sclerosis: a multicentre, randomised placebo-controlled trial (CAMS-LUTS). Int. Urogynecol. J. 17, 636–641 (2006).
Vase, L. & Wartolowska, K. Pain, placebo, and test of treatment efficacy: a narrative review. Br. J. Anaesth. 123, e254–e262 (2019).
Benedetti, F. & Amanzio, M. Mechanisms of the placebo response. Pulm. Pharmacol. Ther. 26, 520–523 (2013).
Benedetti, F., Amanzio, M., Casadio, C., Oliaro, A. & Maggi, G. Blockade of nocebo hyperalgesia by the cholecystokinin antagonist proglumide. Pain 71, 135–140 (1997).
Galton, F. Regression towards mediocrity in hereditary stature. J. Anthropol. Inst. Gt. Br. Irel. 15, 246–263 (1886).
Sech, S. M. et al. The so-called “placebo effect” in benign prostatic hyperplasia treatment trials represents partially a conditional regression to the mean induced by censoring. Urology 51, 242–250 (1998).
Chapple, C. What is new in functional urology? Eur. Urol. Focus. 5, 307–309 (2019).
Haylen, B. T. et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic floor dysfunction. Neurourol. Urodynam. 29, 4–20 (2010).
Coyne, K. S. et al. National community prevalence of overactive bladder in the United States stratified by sex and age. Urology 77, 1081–1087 (2011).
Irwin, D. E. et al. Understanding the elements of overactive bladder: questions raised by the EPIC study. BJU Int. 101, 1381–1387 (2008).
Stewart, W. et al. Prevalence and burden of overactive bladder in the United States. World J. Urol. 20, 327–336 (2003).
Milsom, I. et al. How widespread are the symptoms of an overactive bladder and how are they managed? A population-based prevalence study. BJU Int. 87, 760–766 (2001).
Eapen, R. S. & Radomski, S. B. Review of the epidemiology of overactive bladder. Res. Rep. Urol. 8, 71 (2016).
Andersson, K.-E. The overactive bladder: pharmacologic basis of drug treatment. Urology 50, 74–84 (1997).
Robinson, D. & Cardozo, L. Managing overactive bladder. Climacteric 22, 250–256 (2019).
Yamaguchi, O. et al. Randomized, double-blind, placebo- and propiverine-controlled trial of the once-daily antimuscarinic agent solifenacin in Japanese patients with overactive bladder. BJU Int. 100, 579–587 (2007).
Shin, D. G. et al. Mirabegron as a treatment for overactive bladder symptoms in men (MIRACLE study): efficacy and safety results from a multicenter, randomized, double-blind, placebo-controlled, parallel comparison phase IV study. Neurourol. Urodyn. 38, 295–304 (2019).
Cruz, F. et al. Efficacy and safety of onabotulinumtoxinA in patients with urinary incontinence due to neurogenic detrusor overactivity: a randomised, double-blind, placebo-controlled trial. Eur. Urol. 60, 742–750 (2011).
Mostafaei, H. et al. Placebo response in patients with oral therapy for overactive bladder: a systematic review and meta-analysis. Eur. Urol. Focus https://doi.org/10.1016/j.euf.2021.02.005 (2021).
Sand, P. K., Dmochowski, R. R., Reddy, J. & van der Meulen, E. A. Efficacy and safety of low dose desmopressin orally disintegrating tablet in women with nocturia: results of a multicenter, randomized, double-blind, placebo controlled, parallel group study. J. Urol. 190, 958–964 (2013).
Johnson, T. M. et al. Changes in nocturia from medical treatment of benign prostatic hyperplasia: secondary analysis of the Department of Veterans Affairs Cooperative Study Trial. J. Urol. 170, 145–148 (2003).
Blaivas, J. G. et al. Overactive bladder phenotypes: development and preliminary data. Can. J. Urol. 28, 10699–10704 (2021).
Herschorn, S., Chapple, C. R., Snijder, R., Siddiqui, E. & Cardozo, L. Could reduced fluid intake cause the placebo effect seen in overactive bladder clinical trials? Analysis of a large solifenacin integrated database. Urology 106, 55–59 (2017).
Baines, G., Araklitis, G., Flint, R., Robinson, D. & Cardozo, L. What affects the placebo effect? Eur. J. Obstet. Gynecol. Reprod. Biol. 246, 134–137 (2020).
Avery, J. C., Braunack-Mayer, A. J., Stocks, N. P., Taylor, A. & Duggan, P. Psychological Perspectives in Urinary Incontinence: a Metasynthesis (OA Women’s Health, 2013).
Irwin, D. E., Kopp, Z. S., Agatep, B., Milsom, I. & Abrams, P. Worldwide prevalence estimates of lower urinary tract symptoms, overactive bladder, urinary incontinence and bladder outlet obstruction. BJU Int. 108, 1132–1138 (2011).
Milsom, I. & Gyhagen, M. The prevalence of urinary incontinence. Climacteric 22, 217–222 (2019).
Mostafaei, H. et al. Prevalence of female urinary incontinence in the developing world: a systematic review and meta-analysis — a report from the Developing World Committee of the International Continence Society and Iranian Research Center for Evidence Based Medicine. Neurourol. Urodyn. 39, 1063–1086 (2020).
Wilson, L., Brown, J. S., Shin, G. P., Luc, K.-O. & Subak, L. L. Annual direct cost of urinary incontinence. Obstet. Gynecol. 98, 398–406 (2001).
Ganz, M. L. et al. Economic costs of overactive bladder in the United States. Urology 75, 526–532 (2010).
D’Ancona, C. et al. The International Continence Society (ICS) report on the terminology for adult male lower urinary tract and pelvic floor symptoms and dysfunction. Neurourol. Urodyn. 38, 433–477 (2019).
Abrams, P. et al. 6th International Consultation on Incontinence. Recommendations of the International Scientific Committee: evaluation and treatment of urinary incontinence, pelvic organ prolapse and faecal incontinence. Neurourol. Urodyn. 37, 2271–2272 (2018).
Yalcin, I. & Bump, R. C. The effect of previous treatment experience and incontinence severity on the placebo response of stress urinary incontinence. Am. J. Obstet. Gynecol. 191, 194–197 (2004).
Klarskov, N., Darekar, A., Scholfield, D., Whelan, L. & Lose, G. Effect of fesoterodine on urethral closure function in women with stress urinary incontinence assessed by urethral pressure reflectometry. Int. Urogynecol. J. 25, 755–760 (2014).
Blaganje, M. et al. Non-ablative Er: YAG laser therapy effect on stress urinary incontinence related to quality of life and sexual function: a randomized controlled trial. Eur. J. Obstet. Gynecol. Reprod. Biol. 224, 153–158 (2018).
Li, J. et al. The role of duloxetine in stress urinary incontinence: a systematic review and meta-analysis. Int. Urol. Nephrol. 45, 679–686 (2013).
Parish, S. J. et al. Toward a more evidence-based nosology and nomenclature for female sexual dysfunctions — part II. J. Sex. Med. 13, 1888–1906 (2016).
Laumann, E. O., Paik, A. & Rosen, R. C. Sexual dysfunction in the United States: prevalence and predictors. JAMA 281, 537–544 (1999).
Basson, R. et al. Report of the international consensus development conference on female sexual dysfunction: definitions and classifications. J. Urol. 163, 888–893 (2000).
Bradford, A. & Meston, C. Correlates of placebo response in the treatment of sexual dysfunction in women: a preliminary report. J. Sex. Med. 4, 1345–1351 (2007).
Modelska, K. & Cummings, S. Female sexual dysfunction in postmenopausal women: systematic review of placebo-controlled trials. Am. J. Obstet. Gynecol. 188, 286–293 (2003).
Weinberger, J. M. et al. Female sexual dysfunction and the placebo effect: a meta-analysis. Obstet. Gynecol. 132, 453–458 (2018).
Wiegel, M., Meston, C. & Rosen, R. The female sexual function index (FSFI): cross-validation and development of clinical cutoff scores. J. Sex. Marital. Ther. 31, 1–20 (2005).
Bradford, A. & Meston, C. M. Behavior and symptom change among women treated with placebo for sexual dysfunction. J. Sex. Med. 8, 191–201 (2011).
Bradford, A. Listening to placebo in clinical trials for female sexual dysfunction. J. Sex. Med. 10, 451–459 (2013).
DeRogatis, L. R. & Burnett, A. L. The epidemiology of sexual dysfunctions. J. Sex. Med. 5, 289–300 (2008).
Gur, S. et al. Update on drug interactions with phosphodiesterase-5 inhibitors prescribed as first-line therapy for patients with erectile dysfunction or pulmonary hypertension. Curr. Drug Metab. 14, 265–269 (2013).
Swearingen, D., Nehra, A., Morelos, S. & Peterson, C. A. Hemodynamic effect of avanafil and glyceryl trinitrate coadministration. Drugs Context 2013, 212248 (2013).
De Araujo, A. C. et al. The management of erectile dysfunction with placebo only: does it work? J. Sex. Med. 6, 3440–3448 (2009).
Seidman, S. N., Roose, S. P., Menza, M. A., Shabsigh, R. & Rosen, R. C. Treatment of erectile dysfunction in men with depressive symptoms: results of a placebo-controlled trial with sildenafil citrate. Am. J. Psychiatry 158, 1623–1630 (2001).
Mulhall, J. P., Carlsson, M., Stecher, V. & Tseng, L.-J. Predictors of erectile function normalization in men with erectile dysfunction treated with placebo. J. Sex. Med. 15, 866–872 (2018).
Stridh, A. et al. Placebo responses among men with erectile dysfunction enrolled in phosphodiesterase 5 inhibitor trials: a systematic review and meta-analysis. JAMA Netw. Open 3, e201423–e201423 (2020).
Melnik, T. & Abdo, C. H. Psychogenic erectile dysfunction: comparative study of three therapeutic approaches. J. Sex. Marital. Ther. 31, 243–255 (2005).
Faasse, K., Martin, L. R., Grey, A., Gamble, G. & Petrie, K. J. Impact of brand or generic labeling on medication effectiveness and side effects. Health Psychol. 35, 187 (2016).
Konkle, K. S. et al. Comparison of an interstitial cystitis/bladder pain syndrome clinical cohort with symptomatic community women from the RAND Interstitial Cystitis Epidemiology study. J. Urol. 187, 508–512 (2012).
Nigro, D. A. et al. Associations among cystoscopic and urodynamic findings for women enrolled in the Interstitial Cystitis Data Base (ICDB) Study. Urology 49, 86–92 (1997).
Nickel, J. C. Interstitial cystitis: characterization and management of an enigmatic urologic syndrome. Rev. Urol. 4, 112 (2002).
Nickel, J. C. & Moldwin, R. FDA BRUDAC 2018 Criteria for Interstitial Cystitis/Bladder Pain Syndrome Clinical Trials: future direction for research. J. Urol. 200, 39–42 (2018).
Hunner, G. L. A rare type of bladder ulcer in women; report of cases. Boston Med. Surgical J. 172, 660–664 (1915).
Hanno, P. International Consultation on IC-Rome, September 2004/Forging an International Consensus: progress in painful bladder syndrome/interstitial cystitis. Int. Urogynecol. J. 16, S2 (2005).
Doggweiler, R. et al. A standard for terminology in chronic pelvic pain syndromes: a report from the chronic pelvic pain working group of the International Continence Society. Neurourol. Urodyn. 36, 984–1008 (2017).
Berry, S. H. et al. Prevalence of symptoms of bladder pain syndrome/interstitial cystitis among adult females in the United States. J. Urol. 186, 540–544 (2011).
Parsons, C. L. et al. The prevalence of interstitial cystitis in gynecologic patients with pelvic pain, as detected by intravesical potassium sensitivity. Am. J. Obstet. Gynecol. 187, 1395–1400 (2002).
Sant, G. R. Etiology, pathogenesis, and diagnosis of interstitial cystitis. Rev. Urol. 4, S9 (2002).
Foster, H. E. et al. Effect of amitriptyline on symptoms in treatment naive patients with interstitial cystitis/painful bladder syndrome. J. Urol. 183, 1853–1858 (2010).
van Ophoven, A., Vonde, K., Koch, W., Auerbach, G. & Maag, K. P. Efficacy of pentosan polysulfate for the treatment of interstitial cystitis/bladder pain syndrome: results of a systematic review of randomized controlled trials. Curr. Med. Res. Opin. 35, 1495–1503 (2019).
Nickel, J. C. et al. Pentosan polysulfate sodium for treatment of interstitial cystitis/bladder pain syndrome: insights from a randomized, double-blind, placebo controlled study. J. Urol. 193, 857–862 (2015).
Bosch, P. C. Examination of the significant placebo effect in the treatment of interstitial cystitis/bladder pain syndrome. Urology 84, 321–326 (2014).
Kuo, H. C., Jiang, Y. H., Tsai, Y. C. & Kuo, Y. C. Intravesical botulinum toxin‐A injections reduce bladder pain of interstitial cystitis/bladder pain syndrome refractory to conventional treatment–a prospective, multicenter, randomized, double-blind, placebo-controlled clinical trial. Neurourol. Urodyn. 35, 609–614 (2016).
Pinto, R. A. et al. Intratrigonal onabotulinumtoxinA improves bladder symptoms and quality of life in patients with bladder pain syndrome/interstitial cystitis: a pilot, single center, randomized, double-blind, placebo controlled trial. J. Urol. 199, 998–1003 (2018).
Lai, H. H. Clinical trials: placebo effects in interstitial cystitis/bladder pain syndrome. Nat. Rev. Urol. 11, 494 (2014).
Coyne, K. S. et al. The prevalence of lower urinary tract symptoms (LUTS) in the USA, the UK and Sweden: results from the Epidemiology of LUTS (EpiLUTS) study. BJU Int. 104, 352–360 (2009).
Kupelian, V. et al. Prevalence of lower urinary tract symptoms and effect on quality of life in a racially and ethnically diverse random sample: the Boston Area Community Health (BACH) Survey. Arch. Intern. Med. 166, 2381–2387 (2006).
Martin, S. A., Haren, M. T., Marshall, V. R., Lange, K. & Wittert, G. A. Prevalence and factors associated with uncomplicated storage and voiding lower urinary tract symptoms in community-dwelling Australian men. World J. Urol. 29, 179–184 (2011).
Abrams, P. et al. The standardisation of terminology in lower urinary tract function: report from the standardisation sub-committee of the International Continence Society. Urology 61, 37–49 (2003).
Gratzke, C. et al. EAU guidelines on the assessment of non-neurogenic male lower urinary tract symptoms including benign prostatic obstruction. Eur. Urol. 67, 1099–1109 (2015).
Brasure, M. et al. Newer Medications for Lower Urinary Tract Symptoms Attributed to Benign Prostatic Hyperplasia: A Review (Agency for Healthcare Research and Quality 2016).
Meng, J. et al. Age, height, BMI and FBG predict prostate volume in ageing benign prostatic hyperplasia: evidence from 5285 patients. Int. J. Clin. Pract. 73, e13438 (2019).
Zhang, W. et al. Prevalence of lower urinary tract symptoms suggestive of benign prostatic hyperplasia (LUTS/BPH) in China: results from the China Health and Retirement Longitudinal Study. BMJ Open 9, e022792 (2019).
Vuichoud, C. & Loughlin, K. R. Benign prostatic hyperplasia: epidemiology, economics and evaluation. Can. J. Urol. 22, 1–6 (2015).
Sorokin, I., Schatz, A. & Welliver, C. Placebo medication and sham surgery responses in benign prostatic hyperplasia treatments: implications for clinical trials. Curr. Urol. Rep. 16, 73 (2015).
Barry, M. J. et al. Benign prostatic hyperplasia specific health status measures in clinical research: how much change in the American Urological Association symptom index and the benign prostatic hyperplasia impact index is perceptible to patients? J. Urol. 154, 1770–1774 (1995).
McConnell, J. D. et al. The long-term effect of doxazosin, finasteride, and combination therapy on the clinical progression of benign prostatic hyperplasia. N. Engl. J. Med. 349, 2387–2398 (2003).
Kirby, R. S. et al. Efficacy and tolerability of doxazosin and finasteride, alone or in combination, in treatment of symptomatic benign prostatic hyperplasia: the Prospective European Doxazosin and Combination Therapy (PREDICT) trial. Urology 61, 119–126 (2003).
Lepor, H. et al. The efficacy of terazosin, finasteride, or both in benign prostatic hyperplasia. N. Engl. J. Med. 335, 533–540 (1996).
Byrnes, C. A. et al. Efficacy, tolerability, and effect on health-related quality of life of finasteride versus placebo in men with symptomatic benign prostatic hyperplasia: a community-based study. Clin. Ther. 17, 956–969 (1995).
Nickel, J. C. et al. Efficacy and safety of finasteride therapy for benign prostatic hyperplasia: results of a 2-year randomized controlled trial (the PROSPECT study). PROscar Safety Plus Efficacy Canadian Two year Study. CMAJ 155, 1251 (1996).
Nickel, J. Placebo therapy of benign prostatic hyperplasia: a 25-month study. Canadian PROSPECT Study Group. Br. J. Urol. 81, 383–387 (1998).
Roehrborn, C. G. et al. The prostatic urethral lift for the treatment of lower urinary tract symptoms associated with prostate enlargement due to benign prostatic hyperplasia: the LIFT Study. J. Urol. 190, 2161–2167 (2013).
Porst, H. et al. Efficacy and safety of tadalafil 5 mg once daily for lower urinary tract symptoms suggestive of benign prostatic hyperplasia: subgroup analyses of pooled data from 4 multinational, randomized, placebo-controlled clinical studies. Urology 82, 667–673 (2013).
Regadas, R. P. et al. Urodynamic effects of the combination of tamsulosin and daily tadalafil in men with lower urinary tract symptoms secondary to benign prostatic hyperplasia: a randomized, placebo-controlled clinical trial. Int. Urol. Nephrol. 45, 39–43 (2013).
Eredics, K., Madersbacher, S. & Schauer, I. A relevant midterm (12 months) placebo effect on lower urinary tract symptoms and maximum flow rate in male lower urinary tract symptom and benign prostatic hyperplasia — a meta-analysis. Urology 106, 160–166 (2017).
Zimmern, P. Medical treatment modalities for lower urinary tract symptoms: what are the relevant differences in randomised controlled trials? Eur. Urol. 38, 18–24 (2000).
Enck, P. & Klosterhalfen, S. Does sex/gender play a role in placebo and nocebo effects? Conflicting evidence from clinical trials and experimental studies. Front. Neurosci. 13, 160 (2019).
Vambheim, S. M. & Flaten, M. A. A systematic review of sex differences in the placebo and the nocebo effect. J. Pain. Res. 10, 1831 (2017).
Liccardi, G. et al. Evaluation of the nocebo effect during oral challenge in patients with adverse drug reactions. J. Investig. Allergol. Clin. Immunol. 14, 104–107 (2004).
Colloca, L., Pine, D. S., Ernst, M., Miller, F. G. & Grillon, C. Vasopressin boosts placebo analgesic effects in women: a randomized trial. Biol. Psychiatry 79, 794–802 (2016).
Siegel, S. Drug tolerance, drug addiction, and drug anticipation. Curr. Direct Psychol. Sci. 14, 296–300 (2005).
Weimer, K., Colloca, L. & Enck, P. Age and sex as moderators of the placebo response — an evaluation of systematic reviews and meta-analyses across medicine. Gerontology 61, 97–108 (2015).
Cumming, G. Understanding the New Statistics: Effect Sizes, Confidence Intervals, and Meta-Analysis (Routledge, 2013).
Tufanaru, C. What’s in a name: ‘significant’ results? JBI Evid. Synth. 13, 1–3 (2015).
Rai, S. K., Yazdany, J., Fortin, P. R. & Aviña-Zubieta, J. A. Approaches for estimating minimal clinically important differences in systemic lupus erythematosus. Arthritis Res. Ther. 17, 143 (2015).
Copay, A. G., Subach, B. R., Glassman, S. D., Polly, D. W. Jr & Schuler, T. C. Understanding the minimum clinically important difference: a review of concepts and methods. Spine J. 7, 541–546 (2007).
Wyrwich, K. W., Tierney, W. M. & Wolinsky, F. D. Further evidence supporting an SEM-based criterion for identifying meaningful intra-individual changes in health-related quality of life. J. Clin. Epidemiol. 52, 861–873 (1999).
Jacobson, N. S., Follette, W. C. & Revenstorf, D. Psychotherapy outcome research: methods for reporting variability and evaluating clinical significance. Behav. Ther. 15, 336–352 (1984).
Jacobson, N. S., Follette, W. C. & Revenstorf, D. Toward a standard definition of clinically significant change. Behav. Ther. 17, 308–311 (1986).
Black, N. et al. Consensus development methods: a review of best practice in creating clinical guidelines. J. Health Serv. Res. Policy 4, 236–248 (1999).
McKenna, H. P. The Delphi technique: a worthwhile research approach for nursing? J. Adv. Nurs. 19, 1221–1225 (1994).
Lim, R., Liong, M. L., Lim, K. K., Leong, W. S. & Yuen, K. H. The minimum clinically important difference of the International Consultation on Incontinence Questionnaires (ICIQ-UI SF and ICIQ-LUTSqol). Urology 133, 91–95 (2019).
Fuller, T. W., Ristau, B. T., Tepe, S. M. & Benoit, R. M. Characterizing clinically meaningful changes in lower urinary tract symptoms using the American Urological Association symptom index. Urology 115, 139–143 (2018).
Barsky, A. J., Saintfort, R., Rogers, M. P. & Borus, J. F. Nonspecific medication side effects and the nocebo phenomenon. JAMA 287, 622–627 (2002).
Petrie, K. J., Cameron, L. D., Ellis, C. J., Buick, D. & Weinman, J. Changing illness perceptions after myocardial infarction: an early intervention randomized controlled trial. Psychosom. Med. 64, 580–586 (2002).
Kaptchuk, T. J. et al. Components of placebo effect: randomised controlled trial in patients with irritable bowel syndrome. BMJ 336, 999–1003 (2008).
Petrie, K. J. et al. Effect of providing information about normal test results on patients’ reassurance: randomised controlled trial. BMJ 334, 352 (2007).
Eikelboom, R. & Stewart, J. Conditioning of drug-induced physiological responses. Psychol. Rev. 89, 507 (1982).
Brody, H. The lie that heals: the ethics of giving placebos. Ann. Intern. Med. 97, 112–118 (1982).
Schaefer, M., Sahin, T. & Berstecher, B. Why do open-label placebos work? A randomized controlled trial of an open-label placebo induction with and without extended information about the placebo effect in allergic rhinitis. PLoS ONE 13, e0192758 (2018).
Blease, C., Colloca, L. & Kaptchuk, T. J. Are open-label placebos ethical? Informed consent and ethical equivocations. Bioethics 30, 407–414 (2016).
Charlesworth, J. E. et al. Effects of placebos without deception compared with no treatment: a systematic review and meta‐analysis. J. Evid. Based Med. 10, 97–107 (2017).
Author information
Authors and Affiliations
Contributions
H.M., S.J. and E.L. researched data for the article. H.M., K.M., F.Q., V.M.S., A.A., C.G.R., S.F.S. and S.H. made substantial contributions to discussions of content. H.M., G.L.C. and R.S.M. wrote the article. H.M., B.P., C.G.R., S.F.S. and S.H. reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
S.F.S. is an advisory board member and/or speaker for Astellas, AstraZeneca, Bayer, BMS, Cepheid, Ferring, Ipsen, Jansen, Lissy, MSD, Olympus, Pfizer, Pierre Fabre, Roche, Sanochemia, Sanofi and Wolff. C.G.R. is a consultant for GSK, Lilly, Procept, NxThera, Neotract and Sophiris and has previously received grants or research support from NxThera, Neotract, Procept and Astellas. The other authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Urology thanks Linda Cardozo, Francisco Cruz and Gommert van Koeveringe for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Regression to the mean
-
Regression to the mean is a statistical term that indicates that if a variable from a population is extreme in the first measure it is likely that it will move to the mean (become less extreme) in the next measure.
- Natural history
-
The natural history of a disease is the course of a disease from the beginning to resolution.
- Symptom fluctuations
-
Symptom fluctuations are defined as a constantly changing symptom presentation between one level or another.
- Classical conditioning
-
Classical conditioning is a behavioural process in which an unconditioned stimulus (such as food) is paired with a conditioned stimulus (such as a bell).
- Hidden conditioning
-
Hidden conditioning is when the process of classical conditioning proceeds without the target individual noticing.
- Open conditioning
-
Open conditioning is when the target of conditioning is aware of the process of classical conditioning.
- Anchor-based method
-
The anchor-based method compares changes in scores with an ‘anchor’ as a reference. An anchor establishes whether the patient is better after treatment than baseline according to the patient’s own experience.
- Distribution-based method
-
The distribution-based approach is a method of determining minimal clinically important changes. It relies on the variability of data and the statistical characteristics of estimates of magnitudes of change.
- Delphi technique
-
The Delphi method is a process used to reach an agreement or decision by surveying a panel of experts.
Rights and permissions
About this article
Cite this article
Mostafaei, H., Jilch, S., Carlin, G.L. et al. The placebo and nocebo effects in functional urology. Nat Rev Urol 19, 171–189 (2022). https://doi.org/10.1038/s41585-021-00545-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41585-021-00545-2