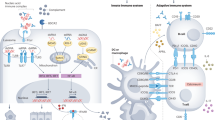
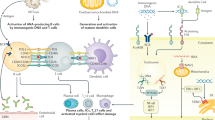
Abstract
Despite the continuing development of immunomodulatory agents and supportive care, the prognosis associated with lupus nephritis (LN) has not improved substantially in the past decade, with end-stage kidney disease still developing in 5–30% of patients within 10 years of LN diagnosis. Moreover, inter-ethnic variation in the tolerance of, clinical response to and level of evidence regarding various therapeutic regimens for LN has led to variation in treatment prioritization in different international recommendations. Modalities that better preserve kidney function and reduce the toxicities of concomitant glucocorticoids are unmet needs in the development of therapeutics for LN. In addition to the conventional recommended therapies for LN, there are newly approved treatments as well as investigational drugs in the pipeline, including the newer generation calcineurin inhibitors and biologic agents. In view of the heterogeneity of LN in terms of clinical presentation and prognosis, the choice of therapies depends on a number of clinical considerations. Molecular profiling, gene-signature fingerprints and urine proteomic panels might enhance the accuracy of patient stratification for treatment personalization in the future.
Key points
-
The development of novel therapeutics to improve the efficacy-to-toxicity balance for lupus nephritis (LN) has unmet needs.
-
Inter-ethnic variation in tolerance to, efficacy of and evidence level for various conventional treatment regimens for LN has led to differences in prioritization in national and international guidelines.
-
Belimumab and voclosporin are the two most recently approved drugs for the treatment of LN in most parts of the world.
-
New-generation anti-CD20 biologic agents, type I interferon antagonists and rituximab–belimumab combination regimens show promise for the treatment of LN.
-
Molecular profiling, gene-signature fingerprints and urine proteomic panels could enhance the accuracy of patient stratification for treatment personalization.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Mok, C. C. Towards new avenues in the management of lupus glomerulonephritis. Nat. Rev. Rheumatol. 12, 221–234 (2016).
Mok, C. C., Kwok, R. C. L. & Yip, P. S. F. Effect of renal disease on the standardized mortality ratio and life expectancy of patients with systemic lupus erythematosus. Arthritis Rheum. 65, 2154–2160 (2013).
Kandane-Rathnayake, R. et al. Longitudinal associations of active renal disease with irreversible organ damage accrual in systemic lupus erythematosus. Lupus 28, 1669–1677 (2019).
Kharawala, S. et al. Health-related quality of life, fatigue and health utilities in lupus nephritis: a systematic literature review. Lupus https://doi.org/10.1177/09612033221100910 (2022).
Jolly, M. et al. Disease-specific quality of life in patients with lupus nephritis. Lupus 27, 257–264 (2018).
Mok, C. C. & Tang, S. S. K. Incidence and predictors of renal disease in Chinese patients with systemic lupus erythematosus. Am. J. Med. 117, 791–795 (2004).
Lewis, M. J. & Jawad, A. S. The effect of ethnicity and genetic ancestry on the epidemiology, clinical features and outcome of systemic lupus erythematosus. Rheumatology 56, i67–i77 (2017).
Parikh, S. V., Almaani, S., Brodsky, S. & Rovin, B. H. Update on lupus nephritis: core curriculum 2020. Am. J. Kidney Dis. 76, 265–281 (2020).
Tanaka, Y., O’Neill, S., Li, M., Tsai, I.-C. & Yang, Y.-W. Systemic lupus erythematosus: targeted literature review of the epidemiology, current treatment, and disease burden in the Asia Pacific Region. Arthritis Care Res. 74, 187–198 (2022).
Jakes, R. W. et al. Systematic review of the epidemiology of systemic lupus erythematosus in the Asia-Pacific region: prevalence, incidence, clinical features, and mortality. Arthritis Care Res. 64, 159–168 (2012).
Sexton, D. J. et al. ESRD from lupus nephritis in the United States, 1995–2010. Clin. J. Am. Soc. Nephrol. 10, 251–259 (2015).
Gómez-Puerta, J. A. et al. Racial and ethnic differences in mortality and cardiovascular events among patients with end-stage renal disease due to lupus nephritis. Arthritis Care Res. 67, 1453–1462 (2015).
Nee, R. et al. Survival disparity of African American versus Non-African American patients with ESRD due to SLE. Am. J. Kidney Dis. 66, 630–637 (2015).
Iwamoto, T. & Niewold, T. B. Genetics of human lupus nephritis. Clin. Immunol. 185, 32–39 (2017).
Freedman, B. I. et al. End-stage renal disease in African Americans with lupus nephritis is associated with APOL1. Arthritis Rheumatol. 66, 390–396 (2014).
Portalatin, G. M., Gebreselassie, S. K. & Bobart, S. A. Lupus nephritis — an update on disparities affecting African Americans. J. Natl Med. Assoc. 114, S34–S42 (2022).
Jordan, J., Thompson, N. J., Dunlop-Thomas, C., Lim, S. S. & Drenkard, C. Relationships among organ damage, social support, and depression in African American women with systemic lupus erythematosus. Lupus 28, 253–260 (2019).
Kumar, K. et al. Beliefs about medicines in patients with rheumatoid arthritis and systemic lupus erythematosus: a comparison between patients of South Asian and White British origin. Rheumatology 47, 690–697 (2008).
Hoi, A. Asian lupus in a multi-ethnic society: what can be learnt? Int. J. Rheum. Dis. 18, 113–116 (2015).
Rodríguez-Almaraz, E. et al. Something new about prognostic factors for lupus nephritis? A systematic review. Lupus 30, 2256–2267 (2021).
Moroni, G. et al. Changing patterns in clinical-histological presentation and renal outcome over the last five decades in a cohort of 499 patients with lupus nephritis. Ann. Rheum. Dis. 77, 1318–1325 (2018).
Jorge, A. et al. All-cause and cause-specific mortality trends of end-stage renal disease due to lupus nephritis from 1995 to 2014. Arthritis Rheumatol. 71, 403–410 (2019).
Tektonidou, M. G., Dasgupta, A. & Ward, M. M. Risk of end-stage renal disease in patients with lupus nephritis, 1971–2015: a systematic review and Bayesian meta-analysis. Arthritis Rheumatol. 68, 1432–1441 (2016).
Anders, H.-J. et al. The management of lupus nephritis as proposed by EULAR/ERA 2019 versus KDIGO 2021. Nephrol. Dial. Transpl. https://doi.org/10.1093/ndt/gfab351 (2021).
Fanouriakis, A. et al. 2019 update of the Joint European League Against Rheumatism and European Renal Association-European Dialysis and Transplant Association (EULAR/ERA-EDTA) recommendations for the management of lupus nephritis. Ann. Rheum. Dis. 79, 713–723 (2020).
Mok, C. C. et al. The Asia-Pacific League of Associations for Rheumatology consensus statements on the management of systemic lupus erythematosus. Lancet Rheumatol. 3, e517–e531 (2021).
Rovin, B. H. et al. Executive summary of the KDIGO 2021 Guideline for the management of glomerular diseases. Kidney Int. 100, 753–779 (2021).
Hahn, B. H. et al. American College of Rheumatology guidelines for screening, treatment, and management of lupus nephritis. Arthritis Care Res. 64, 797–808 (2012).
Houssiau, F. A. et al. The 10-year follow-up data of the Euro-Lupus Nephritis trial comparing low-dose and high-dose intravenous cyclophosphamide. Ann. Rheum. Dis. 69, 61–64 (2010).
Rathi, M. et al. Comparison of low-dose intravenous cyclophosphamide with oral mycophenolate mofetil in the treatment of lupus nephritis. Kidney Int. 89, 235–242 (2016).
Hsu, C.-Y. et al. Cumulative immunosuppressant exposure is associated with diversified cancer risk among 14 832 patients with systemic lupus erythematosus: a nested case-control study. Rheumatology 56, 620–628 (2017).
Mok, C. C., Lau, C. S. & Wong, R. W. Risk factors for ovarian failure in patients with systemic lupus erythematosus receiving cyclophosphamide therapy. Arthritis Rheum. 41, 831–837 (1998).
Mok, C. C. Mycophenolate mofetil for lupus nephritis: an update. Expert. Rev. Clin. Immunol. 11, 1353–1364 (2015).
Mok, C. C. et al. Long-term outcome of a randomised controlled trial comparing tacrolimus with mycophenolate mofetil as induction therapy for active lupus nephritis. Ann. Rheum. Dis. 79, 1070–1076 (2020).
Liu, Z. et al. Multitarget therapy for induction treatment of lupus nephritis: a randomized trial. Ann. Intern. Med. 162, 18–26 (2015).
Mok, C. C. et al. Tacrolimus versus mycophenolate mofetil for induction therapy of lupus nephritis: a randomised controlled trial and long-term follow-up. Ann. Rheum. Dis. 75, 30–36 (2016).
Austin, H. A., Illei, G. G., Braun, M. J. & Balow, J. E. Randomized, controlled trial of prednisone, cyclophosphamide, and cyclosporine in lupus membranous nephropathy. J. Am. Soc. Nephrol. 20, 901–911 (2009).
Merrill, J. T. et al. Efficacy and safety of rituximab in moderately-to-severely active systemic lupus erythematosus: the randomized, double-blind, phase II/III systemic lupus erythematosus evaluation of rituximab trial. Arthritis Rheum. 62, 222–233 (2010).
Rovin, B. H. et al. Efficacy and safety of rituximab in patients with active proliferative lupus nephritis: the Lupus Nephritis Assessment with Rituximab study. Arthritis Rheum. 64, 1215–1226 (2012).
Díaz-Lagares, C. et al. Efficacy of rituximab in 164 patients with biopsy-proven lupus nephritis: pooled data from European cohorts. Autoimmun. Rev. 11, 357–364 (2012).
Terrier, B. et al. Safety and efficacy of rituximab in systemic lupus erythematosus: results from 136 patients from the French Autoimmunity and Rituximab registry. Arthritis Rheum. 62, 2458–2466 (2010).
Rydén-Aulin, M. et al. Off-label use of rituximab for systemic lupus erythematosus in Europe. Lupus Sci. Med. 3, e000163 (2016).
Navarra, S. V. et al. Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: a randomised, placebo-controlled, phase 3 trial. Lancet 377, 721–731 (2011).
Furie, R. et al. A phase III, randomized, placebo-controlled study of belimumab, a monoclonal antibody that inhibits B lymphocyte stimulator, in patients with systemic lupus erythematosus. Arthritis Rheum. 63, 3918–3930 (2011).
Stohl, W. et al. Efficacy and safety of subcutaneous belimumab in systemic lupus erythematosus: a fifty-two-week randomized, double-blind, placebo-controlled study. Arthritis Rheumatol. 69, 1016–1027 (2017).
Zhang, F. et al. A pivotal phase III, randomised, placebo-controlled study of belimumab in patients with systemic lupus erythematosus located in China, Japan and South Korea. Ann. Rheum. Dis. 77, 355–363 (2018).
Brunner, H. I. et al. Safety and efficacy of intravenous belimumab in children with systemic lupus erythematosus: results from a randomised, placebo-controlled trial. Ann. Rheum. Dis. 79, 1340–1348 (2020).
Doria, A. et al. Efficacy and safety of subcutaneous belimumab in anti-double-stranded DNA-positive, hypocomplementemic patients with systemic lupus erythematosus. Arthritis Rheumatol. 70, 1256–1264 (2018).
Maslen, T. et al. Efficacy of belimumab in two serologically distinct high disease activity subgroups of patients with systemic lupus erythematosus: post-hoc analysis of data from the phase III programme. Lupus Sci. Med. 8, e000459 (2021).
Tesar, V. & Hruskova, Z. Belimumab in the management of systemic lupus erythematosus — an update. Expert. Opin. Biol. Ther. 17, 901–908 (2017).
Iaccarino, L. et al. Clinical predictors of response and discontinuation of belimumab in patients with systemic lupus erythematosus in real life setting. Results of a large, multicentric, nationwide study. J. Autoimmun. 86, 1–8 (2018).
Fanouriakis, A. et al. Low disease activity-irrespective of serologic status at baseline-associated with reduction of corticosteroid dose and number of flares in patients with systemic lupus erythematosus treated with belimumab: a real-life observational study. Semin. Arthritis Rheum. 48, 467–474 (2018).
Fanouriakis, A. et al. 2019 update of the EULAR recommendations for the management of systemic lupus erythematosus. Ann. Rheum. Dis. 78, 736–745 (2019).
Ugarte-Gil, M. F. et al. Impact of glucocorticoids on the incidence of lupus-related major organ damage: a systematic literature review and meta-regression analysis of longitudinal observational studies. Lupus Sci. Med. 8, e000590 (2021).
Yurkovich, M., Vostretsova, K., Chen, W. & Aviña-Zubieta, J. A. Overall and cause-specific mortality in patients with systemic lupus erythematosus: a meta-analysis of observational studies. Arthritis Care Res. 66, 608–616 (2014).
Mok, C. C., Ho, L. Y., Chan, K. L., Tse, S. M. & To, C. H. Trend of survival of a cohort of Chinese patients with systemic lupus erythematosus over 25 years. Front. Med. 7, 552 (2020).
van Vollenhoven, R. F. et al. 2021 DORIS definition of remission in SLE: final recommendations from an international task force. Lupus Sci. Med. 8, e000538 (2021).
Rovin, B. H. et al. A randomized, controlled double-blind study comparing the efficacy and safety of dose-ranging voclosporin with placebo in achieving remission in patients with active lupus nephritis. Kidney Int. 95, 219–231 (2019).
Rovin, B. H. et al. Efficacy and safety of voclosporin versus placebo for lupus nephritis (AURORA 1): a double-blind, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet 397, 2070–2080 (2021).
Furie, R. A. et al. B-cell depletion with obinutuzumab for the treatment of proliferative lupus nephritis: a randomised, double-blind, placebo-controlled trial. Ann. Rheum. Dis. 81, 100–107 (2022).
Mok, C. C. et al. Overview of lupus nephritis management guidelines and perspective from Asia. Int. J. Rheum. Dis. 16, 625–636 (2013).
Yap, D. Y. H., Ma, M. K. M., Mok, M. M. Y., Tang, C. S. O. & Chan, T. M. Long-term data on corticosteroids and mycophenolate mofetil treatment in lupus nephritis. Rheumatology 52, 480–486 (2013).
Tse, S. M. & Mok, C. C. Time trend and risk factors of avascular bone necrosis in patients with systemic lupus erythematosus. Lupus 26, 715–722 (2017).
Kunyakham, W., Foocharoen, C., Mahakkanukrauh, A., Suwannaroj, S. & Nanagara, R. Prevalence and risk factor for symptomatic avascular necrosis development in Thai systemic lupus erythematosus patients. Asian Pac. J. Allergy Immunol. 30, 152–157 (2012).
Condon, M. B. et al. Prospective observational single-centre cohort study to evaluate the effectiveness of treating lupus nephritis with rituximab and mycophenolate mofetil but no oral steroids. Ann. Rheum. Dis. 72, 1280–1286 (2013).
Mathian, A. et al. Withdrawal of low-dose prednisone in SLE patients with a clinically quiescent disease for more than 1 year: a randomised clinical trial. Ann. Rheum. Dis. 79, 339–346 (2020).
Kasitanon, N., Fine, D. M., Haas, M., Magder, L. S. & Petri, M. Hydroxychloroquine use predicts complete renal remission within 12 months among patients treated with mycophenolate mofetil therapy for membranous lupus nephritis. Lupus 15, 366–370 (2006).
Pons-Estel, G. J. et al. Protective effect of hydroxychloroquine on renal damage in patients with lupus nephritis: LXV, data from a multiethnic US cohort. Arthritis Rheum. 61, 830–839 (2009).
Dima, A., Jurcut, C., Chasset, F., Felten, R. & Arnaud, L. Hydroxychloroquine in systemic lupus erythematosus: overview of current knowledge. Ther. Adv. Musculoskelet. Dis. 14, 1759720×211073001 (2022).
Lenfant, T. et al. Risk factors for hydroxychloroquine retinopathy in systemic lupus erythematosus: a case-control study with hydroxychloroquine blood-level analysis. Rheumatology 59, 3807–3816 (2020).
Melles, R. B. & Marmor, M. F. The risk of toxic retinopathy in patients on long-term hydroxychloroquine therapy. JAMA Ophthalmol. 132, 1453–1460 (2014).
Dooley, M. A. et al. Mycophenolate versus azathioprine as maintenance therapy for lupus nephritis. N. Engl. J. Med. 365, 1886–1895 (2011).
Tamirou, F. et al. Long-term follow-up of the MAINTAIN Nephritis trial, comparing azathioprine and mycophenolate mofetil as maintenance therapy of lupus nephritis. Ann. Rheum. Dis. 75, 526–531 (2016).
Moroni, G. et al. A randomized pilot trial comparing cyclosporine and azathioprine for maintenance therapy in diffuse lupus nephritis over four years. Clin. J. Am. Soc. Nephrol. 1, 925–932 (2006).
Argolini, L. M. et al. Multicentric study comparing cyclosporine, mycophenolate mofetil and azathioprine in the maintenance therapy of lupus nephritis: 8 years follow up. J. Nephrol. 34, 389–398 (2021).
Takeuchi, T., Wakasugi, N., Uno, S. & Makino, H. Long-term safety and effectiveness of tacrolimus in patients with lupus nephritis: 5-year interim postmarketing surveillance study in Japan (TRUST). J. Rheumatol. 48, 74–81 (2021).
Fu, Q. et al. Leflunomide versus azathioprine for maintenance therapy of lupus nephritis: a prospective, multicentre, randomised trial and long-term follow-up. Ann. Rheum. Dis. https://doi.org/10.1136/ard-2022-222486 (2022).
Jourde-Chiche, N. et al. Weaning of maintenance immunosuppressive therapy in lupus nephritis (WIN-Lupus): results of a multicentre randomised controlled trial. Ann. Rheum. Dis. 81, 1420–1427 (2022).
Wang, G. et al. Association of urinary matrix metalloproteinase 7 levels with incident renal flare in lupus nephritis. Arthritis Rheumatol. 73, 265–275 (2021).
Mejia-Vilet, J. M. et al. Association between urinary epidermal growth factor and renal prognosis in lupus nephritis. Arthritis Rheumatol. 73, 244–254 (2021).
Mejia-Vilet, J. M., Malvar, A., Arazi, A. & Rovin, B. H. The lupus nephritis management renaissance. Kidney Int. 101, 242–255 (2022).
Mok, C. C. & Mohan, C. Urinary biomarkers in lupus nephritis: are we there yet? Arthritis Rheumatol. 73, 194–196 (2021).
Tamirou, F. et al. A proteinuria cut-off level of 0.7 g/day after 12 months of treatment best predicts long-term renal outcome in lupus nephritis: data from the MAINTAIN nephritis trial. Lupus Sci. Med. 2, e000123 (2015).
Dall’Era, M. et al. Predictors of long-term renal outcome in lupus nephritis trials: lessons learned from the Euro-lupus nephritis cohort. Arthritis Rheumatol. 67, 1305–1313 (2015).
Malvar, A. et al. Histologic versus clinical remission in proliferative lupus nephritis. Nephrol. Dial. Transpl. 32, 1338–1344 (2017).
Alvarado, A. S. et al. The value of repeat kidney biopsy in quiescent Argentinian lupus nephritis patients. Lupus 23, 840–847 (2014).
Wakasugi, D. et al. Frequency of class III and IV nephritis in systemic lupus erythematosus without clinical renal involvement: an analysis of predictive measures. J. Rheumatol. 39, 79–85 (2012).
Zabaleta-Lanz, M. E. et al. Further description of early clinically silent lupus nephritis. Lupus 15, 845–851 (2006).
Chedid, A. et al. Low-level proteinuria in systemic lupus erythematosus. Kidney Int. Rep. 5, 2333–2340 (2020).
Zickert, A., Sundelin, B., Svenungsson, E. & Gunnarsson, I. Role of early repeated renal biopsies in lupus nephritis. Lupus Sci. Med. 1, e000018 (2014).
Parodis, I. et al. Per-protocol repeat kidney biopsy portends relapse and long-term outcome in incident cases of proliferative lupus nephritis. Rheumatology 59, 3424–3434 (2020).
Parodis, I., Tamirou, F. & Houssiau, F. A. Treat-to-target in lupus nephritis. what is the role of the repeat kidney biopsy? Arch. Immunol. Ther. Exp. 70, 8 (2022).
Malvar, A. et al. Kidney biopsy-based management of maintenance immunosuppression is safe and may ameliorate flare rate in lupus nephritis. Kidney Int. 97, 156–162 (2020).
Zhang, H. et al. Multitarget therapy for maintenance treatment of lupus nephritis. J. Am. Soc. Nephrol. 28, 3671–3678 (2017).
Saxena, A., Teng, Y. K. O., Collins, C., England, N. & Leher, H. Voclosporin lupus nephritis: results two-year AURORA 2 continuation study. Ann. Rheum. Dis. 81, 325 (2022).
Furie, R. et al. Two-year, randomized, controlled trial of belimumab in lupus nephritis. N. Engl. J. Med. 383, 1117–1128 (2020).
Rovin, B. H. et al. A secondary analysis of the Belimumab International Study in Lupus Nephritis trial examined effects of belimumab on kidney outcomes and preservation of kidney function in patients with lupus nephritis. Kidney Int. 101, 403–413 (2022).
Miyazaki, Y. et al. Efficacy and safety of belimumab during maintenance therapy in patients with systemic lupus erythematosus. Rheumatology 61, 3614–3636 (2022).
Urowitz, M. B. et al. Impact of belimumab on organ damage in systemic lupus erythematosus. Arthritis Care Res. 74, 1822–1828 (2022).
van Vollenhoven, R. F. et al. Long-term safety and limited organ damage in patients with systemic lupus erythematosus treated with belimumab: a phase III study extension. Rheumatology 59, 281–291 (2020).
van Vollenhoven, R. et al. Conceptual framework for defining disease modification in systemic lupus erythematosus: a call for formal criteria. Lupus Sci. Med. 9, e000634 (2022).
Md Yusof, M. Y. et al. Predicting and managing primary and secondary non-response to rituximab using B-cell biomarkers in systemic lupus erythematosus. Ann. Rheum. Dis. 76, 1829–1836 (2017).
Lazarus, M. N., Turner-Stokes, T., Chavele, K.-M., Isenberg, D. A. & Ehrenstein, M. R. B-cell numbers and phenotype at clinical relapse following rituximab therapy differ in SLE patients according to anti-dsDNA antibody levels. Rheumatology 51, 1208–1215 (2012).
Reddy, V. et al. Obinutuzumab induces superior B-cell cytotoxicity to rituximab in rheumatoid arthritis and systemic lupus erythematosus patient samples. Rheumatology 56, 1227–1237 (2017).
Mysler, E. F. et al. Efficacy and safety of ocrelizumab in active proliferative lupus nephritis: results from a randomized, double-blind, phase III study. Arthritis Rheum. 65, 2368–2379 (2013).
Ehrenstein, M. R. & Wing, C. The BAFFling effects of rituximab in lupus: danger ahead? Nat. Rev. Rheumatol. 12, 367–372 (2016).
Kraaij, T. et al. Long-term effects of combined B-cell immunomodulation with rituximab and belimumab in severe, refractory systemic lupus erythematosus: 2-year results. Nephrol. Dial. Transpl. 36, 1474–1483 (2021).
Atisha-Fregoso, Y. et al. Phase II randomized trial of rituximab plus cyclophosphamide followed by belimumab for the treatment of lupus nephritis. Arthritis Rheumatol. 73, 121–131 (2021).
Shipa, M. et al. Effectiveness of belimumab after rituximab in systemic lupus erythematosus: a randomized controlled trial. Ann. Intern. Med. 174, 1647–1657 (2021).
Teng, Y. K. O. et al. Pharmacodynamic effect of sequential belimumab and rituximab therapy in patients with systemic lupus erythematosus: the phase 3, randomized, placebo-controlled BLISS-BELIEVE study [Abstract]. Ann. Rheum. Dis. 81, 186 (2022).
Morand, E. F. et al. Trial of anifrolumab in active systemic lupus erythematosus. N. Engl. J. Med. 382, 211–221 (2020).
Jayne, D. et al. Phase II randomised trial of type I interferon inhibitor anifrolumab in patients with active lupus nephritis. Ann. Rheum. Dis. 81, 496–506 (2022).
Furie, R. A. et al. Type I interferon inhibitor anifrolumab in active systemic lupus erythematosus (TULIP-1): a randomised, controlled, phase 3 trial. Lancet Rheumatol. 1, e208–e219 (2019).
Zhao, Q. Bispecific antibodies for autoimmune and inflammatory diseases: clinical progress to date. BioDrugs 34, 111–119 (2020).
Abuqayyas, L. et al. Safety and biological activity of rozibafusp alfa, a bispecific inhibitor of inducible costimulator ligand and B cell activating factor, in patients with rheumatoid arthritis: results of a phase 1b, randomized, double-blind, placebo-controlled, multiple ascending dose study. ACR Open Rheumatol. 4, 903–911 (2022).
Dörner, T. et al. Treatment of primary Sjögren’s syndrome with ianalumab (VAY736) targeting B cells by BAFF receptor blockade coupled with enhanced, antibody-dependent cellular cytotoxicity. Ann. Rheum. Dis. 78, 641–647 (2019).
Bowman, S. J. et al. Safety and efficacy of subcutaneous ianalumab (VAY736) in patients with primary Sjögren’s syndrome: a randomised, double-blind, placebo-controlled, phase 2b dose-finding trial. Lancet 399, 161–171 (2022).
Mok, C. C. Biological and targeted therapies of systemic lupus erythematosus: evidence and the state of the art. Expert. Rev. Clin. Immunol. 13, 677–692 (2017).
Yan, M.-T., Chao, C.-T. & Lin, S.-H. Chronic kidney disease: strategies to retard progression. Int. J. Mol. Sci. 22, 10084 (2021).
Tse, K. C. et al. Angiotensin inhibition or blockade for the treatment of patients with quiescent lupus nephritis and persistent proteinuria. Lupus 14, 947–952 (2005).
Chang, J. C., Weiss, P. F., Xiao, R., Atkinson, M. A. & Wenderfer, S. E. Use of renin angiotensin aldosterone system inhibitors in children with lupus and time to glucocorticoid discontinuation. Kidney Int. 102, 395–404 (2022).
Del Vecchio, L., Beretta, A., Jovane, C., Peiti, S. & Genovesi, S. A role for SGLT-2 inhibitors in treating non-diabetic chronic kidney disease. Drugs 81, 1491–1511 (2021).
De Nicola, L. et al. Can SGLT2 inhibitors answer unmet therapeutic needs in chronic kidney disease? J. Nephrol. 35, 1605–1618 (2022).
Säemann, M. & Kronbichler, A. Call for action in ANCA-associated vasculitis and lupus nephritis: promises and challenges of SGLT-2 inhibitors. Ann. Rheum. Dis. 81, 614–617 (2022).
Appel, G. B. et al. Mycophenolate mofetil versus cyclophosphamide for induction treatment of lupus nephritis. J. Am. Soc. Nephrol. 20, 1103–1112 (2009).
Isenberg, D. et al. Influence of race/ethnicity on response to lupus nephritis treatment: the ALMS study. Rheumatology 49, 128–140 (2010).
Ginzler, E. et al. Phase III/IV, randomized, fifty-two-week study of the efficacy and safety of belimumab in patients of black African ancestry with systemic lupus erythematosus. Arthritis Rheumatol. 74, 112–123 (2022).
Yasuda, H., Fukusumi, Y., Ivanov, V., Zhang, Y. & Kawachi, H. Tacrolimus ameliorates podocyte injury by restoring FK506 binding protein 12 (FKBP12) at actin cytoskeleton. FASEB J. 35, e21983 (2021).
Vital, E. M. et al. Anifrolumab efficacy and safety by type I interferon gene signature and clinical subgroups in patients with SLE: post hoc analysis of pooled data from two phase III trials. Ann. Rheum. Dis. 81, 951–961 (2022).
Cooles, F. A. H. & Isaacs, J. D. The interferon gene signature as a clinically relevant biomarker in autoimmune rheumatic disease. Lancet Rheumatol. 4, e61–e72 (2022).
Bruce, I. N. et al. Pharmacokinetics, pharmacodynamics, and safety of subcutaneous anifrolumab in patients with systemic lupus erythematosus, active skin disease, and high type I interferon gene signature: a multicentre, randomised, double-blind, placebo-controlled, phase 2 study. Lancet Rheumatol. 3, e101–e110 (2021).
Bolin, K. et al. Association of STAT4 polymorphism with severe renal insufficiency in lupus nephritis. PLoS One 8, e84450 (2013).
Taylor, K. E. et al. Specificity of the STAT4 genetic association for severe disease manifestations of systemic lupus erythematosus. PLoS Genet. 4, e1000084 (2008).
Reid, S. et al. Interaction between the STAT4 rs11889341(T) risk allele and smoking confers increased risk of myocardial infarction and nephritis in patients with systemic lupus erythematosus. Ann. Rheum. Dis. 80, 1183–1189 (2021).
Hagberg, N. et al. The STAT4 SLE risk allele rs7574865[T] is associated with increased IL-12-induced IFN-γ production in T cells from patients with SLE. Ann. Rheum. Dis. 77, 1070–1077 (2018).
Hasni, S. A. et al. Phase 1 double-blind randomized safety trial of the Janus kinase inhibitor tofacitinib in systemic lupus erythematosus. Nat. Commun. 12, 3391 (2021).
Anolik, J. H. et al. The relationship of FcRγIIIa genotype to degree of B cell depletion by rituximab in the treatment of systemic lupus erythematosus. Arthritis Rheum. 48, 455–459 (2003).
Robinson, J. I. et al. Comprehensive genetic and functional analyses of Fc gamma receptors influence on response to rituximab therapy for autoimmunity. EBioMedicine 86, 104343 (2022).
Wahadat, M. J. et al. Gene signature fingerprints stratify SLE patients in groups with similar biological disease profiles: a multicenter longitudinal study. Rheumatology 61, 4344–4354 (2022).
Garantziotis, P. et al. Molecular taxonomy of systemic lupus erythematosus through data-driven patient stratification: molecular endotypes and cluster-tailored drugs. Front. Immunol. 13, 860726 (2022).
Wang, Y.-F. et al. Identification of shared and Asian-specific loci for systemic lupus erythematosus and evidence for roles of type III interferon signaling and lysosomal function in the disease: a multi-ancestral genome-wide association study. Arthritis Rheumatol. 74, 840–848 (2022).
Owen, K. A. et al. Analysis of trans-ancestral SLE risk loci identifies unique biologic networks and drug targets in African and European ancestries. Am. J. Hum. Genet. 107, 864–881 (2020).
Fava, A. et al. Urine proteomics and renal single-cell transcriptomics implicate interleukin-16 in lupus nephritis. Arthritis Rheumatol. 74, 829–839 (2022).
Zhang, T., Duran, V., Vanarsa, K. & Mohan, C. Targeted urine proteomics in lupus nephritis — a meta-analysis. Expert. Rev. Proteom. 17, 767–776 (2020).
Parikh, S. V. et al. Molecular profiling of kidney compartments from serial biopsies differentiate treatment responders from non-responders in lupus nephritis. Kidney Int. 102, 845–865 (2022).
Der, E. et al. Tubular cell and keratinocyte single-cell transcriptomics applied to lupus nephritis reveal type I IFN and fibrosis relevant pathways. Nat. Immunol. 20, 915–927 (2019).
Almaani, S. et al. Rethinking lupus nephritis classification on a molecular level. J. Clin. Med. 8, E1524 (2019).
Author information
Authors and Affiliations
Contributions
C.C.M. and R.S. researched data and wrote the article. All authors contributed substantially to the discussion of content, and to the review and editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
Y.K.O.T. received an unrestricted research grant from GlaxoSmithKline, Aurinia Pharmaceuticals and Vifor Pharma. The Leiden University Medical Center received consulting fees for consultancy provided by Y.K.O.T. from Aurinia Pharmaceuticals, GlaxoSmithKline, KezarBio, Novartis, Otsuka Pharmaceuticals and Vifor Pharma. Y.T. has received speaking fees and/or honoraria from AbbVie, AstraZeneca, Boehringer-Ingelheim, Bristol-Myers Squibb, Chugai, Daiichi-Sankyo, Eli Lilly, Eisai, Gilead, GlaxoSmithKline, Mitsubishi-Tanabe and Pfizer, and has received research grants from AbbVie, Asahi-Kasei, Boehringer-Ingelheim, Chugai, Daiichi-Sankyo, Eisai and Takeda. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Rheumatology thanks Tak Mao Chan, Ioannis Parodis and Agneta Zickert for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mok, C.C., Teng, Y.K.O., Saxena, R. et al. Treatment of lupus nephritis: consensus, evidence and perspectives. Nat Rev Rheumatol 19, 227–238 (2023). https://doi.org/10.1038/s41584-023-00925-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-023-00925-5
This article is cited by
-
LATS2 degradation promoted fibrosis damage and rescued by vitamin K3 in lupus nephritis
Arthritis Research & Therapy (2024)
-
Prevalence and outcomes of end-stage kidney disease in patients with systemic lupus erythematous: a population-based study
Rheumatology International (2023)