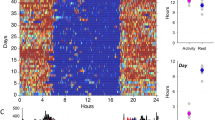
Abstract
During sleep, animals do not eat, reproduce or forage. Sleeping animals are vulnerable to predation. Yet, the persistence of sleep despite evolutionary pressures, and the deleterious effects of sleep deprivation, indicate that sleep serves a function or functions that cannot easily be bypassed. Recent research demonstrates sleep to be phylogenetically far more pervasive than previously appreciated; it is possible that the very first animals slept. Here, we give an overview of sleep across various species, with the aim of determining its original purpose. Sleep exists in animals without cephalized nervous systems and can be influenced by non-neuronal signals, including those associated with metabolic rhythms. Together, these observations support the notion that sleep serves metabolic functions in neural and non-neural tissues.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Zhdanova, I. V., Wang, S. Y., Leclair, O. U. & Danilova, N. P. Melatonin promotes sleep-like state in zebrafish. Brain Res. 903, 263–268 (2001).
Tobler, I. Effect of forced locomotion on the rest–activity cycle of the cockroach. Behav. Brain Res. 8, 351–360 (1983).
Raizen, D. M. et al. Lethargus is a Caenorhabditis elegans sleep-like state. Nature 451, 569–572 (2008).
Vorster, A. P., Krishnan, H. C., Cirelli, C. & Lyons, L. C. Characterization of sleep in Aplysia californica. Sleep 37, 1453–1463 (2014).
Omond, S. et al. Inactivity is nycthemeral, endogenously generated, homeostatically regulated, and melatonin modulated in a free-living platyhelminth flatworm. Sleep 40, zsx124 (2017).
Nath, R. D. et al. The jellyfish Cassiopea exhibits a sleep-like state. Curr. Biol. 27, 2984–2990 (2017).
Walker, J. M. & Berger, R. J. Sleep as an adaptation for energy conservation functionally related to hibernation and shallow torpor. Prog. Brain Res. 53, 255–278 (1980).
Tu, B. P. & McKnight, S. L. Metabolic cycles as an underlying basis of biological oscillations. Nat. Rev. Mol. Cell Biol. 7, 696–701 (2006).
Tu, B. P. & McKnight, S. L. The yeast metabolic cycle: insights into the life of a eukaryotic cell. Cold Spring Harb. Symp. Quant. Biol. 72, 339–343 (2007).
Schmidt, M. H. The energy allocation function of sleep: a unifying theory of sleep, torpor, and continuous wakefulness. Neurosci. Biobehav. Rev. 47, 122–153 (2014).
Buxton, O. M. et al. Adverse metabolic consequences in humans of prolonged sleep restriction combined with circadian disruption. Sci. Transl Med. 4, 129ra43 (2012).
Van Cauter, E., Spiegel, K., Tasali, E. & Leproult, R. Metabolic consequences of sleep and sleep loss. Sleep Med. 9, S23–S28 (2008).
Nedeltcheva, A. V. & Scheer, F. A. Metabolic effects of sleep disruption, links to obesity and diabetes. Curr. Opin. Endocrinol. Diabetes Obes. 21, 293–298 (2014).
Campbell, S. S. & Tobler, I. Animal sleep: a review of sleep duration across phylogeny. Neurosci. Biobehav. Rev. 8, 269–300 (1984).
Franken, P., Chollet, D. & Tafti, M. The homeostatic regulation of sleep need is under genetic control. J. Neurosci. 21, 2610–2621 (2001).
Shaw, P. J., Cirelli, C., Greenspan, R. J. & Tononi, G. Correlates of sleep and waking in Drosophila melanogaster. Science 287, 1834–1837 (2000).
Hendricks, J. C. et al. Rest in Drosophila is a sleep-like state. Neuron 25, 129–138 (2000).
Tobler, I. I. & Neuner-Jehle, M. 24-h variation of vigilance in the cockroach Blaberus giganteus. J. Sleep Res. 1, 231–239 (1992).
Kaiser, W. & Steiner-Kaiser, J. Neuronal correlates of sleep, wakefulness and arousal in a diurnal insect. Nature 301, 707–709 (1983).
Singh, R. N. & Sulsston, J. E. Some observations on moulting in Caenorhabditis elegans. Nematologica 24, 63–71 (1978).
Cassada, R. C. & Russell, R. L. The dauerlarva, a post-embryonic developmental variant of the nematode Caenorhabditis elegans. Dev. Biol. 46, 326–342 (1975).
Singh, K., Ju, J. Y., Walsh, M. B., DiIorio, M. A. & Hart, A. C. Deep conservation of genes required for both Drosphila melanogaster and Caenorhabditis elegans sleep includes a role for dopaminergic signaling. Sleep 37, 1439–1451 (2014).
Schwarz, J., Lewandrowski, I. & Bringmann, H. Reduced activity of a sensory neuron during a sleep-like state in Caenorhabditis elegans. Curr. Biol. 21, R983–R984 (2011).
Nagy, S. et al. Homeostasis in C. elegans sleep is characterized by two behaviorally and genetically distinct mechanisms. eLife 3, e04380 (2014).
Driver, R. J., Lamb, A. L., Wyner, A. J. & Raizen, D. M. DAF-16/FOXO regulates homeostasis of essential sleep-like behavior during larval transitions in C. elegans. Curr. Biol. 23, 501–506 (2013).
Kayser, M. S. & Biron, D. Sleep and development in genetically tractable model organisms. Genetics 203, 21–33 (2016).
Trojanowski, N. F. & Raizen, D. M. Call it worm sleep. Trends Neurosci. 39, 54–62 (2016).
Satterlie, R. A. Do jellyfish have central nervous systems? J. Exp. Biol. 214, 1215–1223 (2011).
Dzirasa, K. et al. Dopaminergic control of sleep-wake states. J. Neurosci. 26, 10577–10589 (2006).
Kume, K., Kume, S., Park, S. K., Hirsh, J. & Jackson, F. R. Dopamine is a regulator of arousal in the fruit fly. J. Neurosci. 25, 7377–7384 (2005).
Andretic, R., van Swinderen, B. & Greenspan, R. J. Dopaminergic modulation of arousal in Drosophila. Curr. Biol. 15, 1165–1175 (2005).
Turek, M., Besseling, J., Spies, J. P., Konig, S. & Bringmann, H. Sleep-active neuron specification and sleep induction require FLP-11 neuropeptides to systemically induce sleep. eLife 5, e12499 (2016).
Nelson, M. D. et al. FMRFamide-like FLP-13 neuropeptides promote quiescence following heat stress in Caenorhabditis elegans. Curr. Biol. 24, 2406–2410 (2014).
Nath, R. D., Chow, E. S., Wang, H., Schwarz, E. M. & Sternberg, P. W. C. elegans stress-induced sleep emerges from the collective action of multiple neuropeptides. Curr. Biol. 26, 2446–2455 (2016).
Shang, Y. et al. Short neuropeptide F is a sleep-promoting inhibitory modulator. Neuron 80, 171–183 (2013).
Lenz, O., Xiong, J., Nelson, M. D., Raizen, D. M. & Williams, J. A. FMRFamide signaling promotes stress-induced sleep in Drosophila. Brain Behav. Immun. 47, 141–148 (2015).
Lee, D. A. et al. Genetic and neuronal regulation of sleep by neuropeptide VF. eLife 6, e25727 (2017).
Deregnaucourt, S., Mitra, P. P., Feher, O., Pytte, C. & Tchernichovski, O. How sleep affects the developmental learning of bird song. Nature 433, 710–716 (2005).
Hendricks, J. C., Kirk, D., Panckeri, K., Miller, M. S. & Pack, A. I. Modafinil maintains waking in the fruit fly Drosophila melanogaster. Sleep 26, 139–146 (2003).
Panckeri, K. A., Schotland, H. M., Pack, A. I. & Hendricks, J. C. Modafinil decreases hypersomnolence in the English bulldog, a natural animal model of sleep-disordered breathing. Sleep 19, 626–631 (1996).
Rihel, J. et al. Zebrafish behavioral profiling links drugs to biological targets and rest/wake regulation. Science 327, 348–351 (2010).
Foltenyi, K., Greenspan, R. J. & Newport, J. W. Activation of EGFR and ERK by rhomboid signaling regulates the consolidation and maintenance of sleep in Drosophila. Nat. Neurosci. 10, 1160–1167 (2007).
Kushikata, T., Fang, J., Chen, Z., Wang, Y. & Krueger, J. M. Epidermal growth factor enhances spontaneous sleep in rabbits. Am. J. Physiol. 275, R509–R514 (1998).
Kramer, A. et al. Regulation of daily locomotor activity and sleep by hypothalamic EGF receptor signaling. Science 294, 2511–2515 (2001).
Van Buskirk, C. & Sternberg, P. W. Epidermal growth factor signaling induces behavioral quiescence in Caenorhabditis elegans. Nat. Neurosci. 10, 1300–1307 (2007).
Cirelli, C. & Tononi, G. Differences in brain gene expression between sleep and waking as revealed by mRNA differential display and cDNA microarray technology. J. Sleep Res. 8, S44–S52 (1999).
Naidoo, N., Giang, W., Galante, R. J. & Pack, A. I. Sleep deprivation induces the unfolded protein response in mouse cerebral cortex. J. Neurochem. 92, 1150–1157 (2005).
Jones, S., Pfister-Genskow, M., Benca, R. M. & Cirelli, C. Molecular correlates of sleep and wakefulness in the brain of the white-crowned sparrow. J. Neurochem. 105, 46–62 (2008).
Sanders, J., Scholz, M., Merutka, I. & Biron, D. Distinct unfolded protein responses mitigate or mediate effects of nonlethal deprivation of C. elegans sleep in different tissues. BMC Biol. 15, 67 (2017).
Yurgel, M. E., Masek, P., DiAngelo, J. & Keene, A. C. Genetic dissection of sleep–metabolism interactions in the fruit fly. J. Comp. Physiol. A 201, 869–877 (2015).
Seugnet, L., Galvin, J. E., Suzuki, Y., Gottschalk, L. & Shaw, P. J. Persistent short-term memory defects following sleep deprivation in a Drosophila model of Parkinson disease. Sleep 32, 984–992 (2009).
Siegel, J. M. Clues to the functions of mammalian sleep. Nature 437, 1264–1271 (2005).
Lyamin, O., Pryaslova, J., Lance, V. & Siegel, J. Animal behaviour: continuous activity in cetaceans after birth. Nature 435, 1177 (2005).
Lesku, J. A. et al. Adaptive sleep loss in polygynous pectoral sandpipers. Science 337, 1654–1658 (2012).
Rattenborg, N. C. et al. Migratory sleeplessness in the white-crowned sparrow (Zonotrichia leucophrys gambelii). PLOS Biol. 2, E212 (2004).
Rattenborg, N. C. et al. Evidence that birds sleep in mid-flight. Nat. Commun. 7, 12468 (2016).
Mukhametov, L. M. Unihemispheric slow-wave sleep in the Amazonian dolphin Inia geoffrensis. Neurosci. Lett. 79, 128–132 (1987).
Siegel, J. M. Do all animals sleep? Trends Neurosci. 31, 208–213 (2008).
Fontvieille, A. M., Rising, R., Spraul, M., Larson, D. E. & Ravussin, E. Relationship between sleep stages and metabolic rate in humans. Am. J. Physiol. 267, E732–E737 (1994).
Brebbia, D. R. & Altshuler, K. Z. Oxygen consumption rate and electroencephalographic stage of sleep. Science 150, 1621–1623 (1965).
Dement, W. & Kleitman, N. Cyclic variations in EEG during sleep and their relation to eye movements, body motility, and dreaming. Electroencephalogr. Clin. Neurophysiol. 9, 673–690 (1957).
Rechtschaffen, A. & Kales, A. (eds) A manual of standardized terminology, techniques and scoring system for sleep stages of human subjects (US National Institute of Neurological Diseases and Blindness, 1968).
Jouvet, M. & Michel, F. Electromyographic correlations of sleep in the chronic decorticate & mesencephalic cat [French]. C. R. Seances Soc. Biol. Fil. 153, 422–425 (1959).
Shein-Idelson, M., Ondracek, J. M., Liaw, H. P., Reiter, S. & Laurent, G. Slow waves, sharp waves, ripples, and REM in sleeping dragons. Science 352, 590–595 (2016).
Nichols, A. L. A., Eichler, T., Latham, R. & Zimmer, M. A global brain state underlies C. elegans sleep behavior. Science 356, eaam6851 (2017).
Yap, M. H. W. et al. Oscillatory brain activity in spontaneous and induced sleep stages in flies. Nat. Commun. 8, 1815 (2017).
Nitz, D. A., van Swinderen, B., Tononi, G. & Greenspan, R. J. Electrophysiological correlates of rest and activity in Drosophila melanogaster. Curr. Biol. 12, 1934–1940 (2002).
Ramon, F., Hernandez-Falcon, J., Nguyen, B. & Bullock, T. H. Slow wave sleep in crayfish. Proc. Natl Acad. Sci. USA 101, 11857–11861 (2004).
Trojanowski, N. F., Nelson, M. D., Flavell, S. W., Fang-Yen, C. & Raizen, D. M. Distinct mechanisms underlie quiescence during two Caenorhabditis elegans sleep-like states. J. Neurosci. 35, 14571–14584 (2015).
van Alphen, B., Yap, M. H., Kirszenblat, L., Kottler, B. & van Swinderen, B. A dynamic deep sleep stage in Drosophila. J. Neurosci. 33, 6917–6927 (2013).
Blumberg, M. S., Coleman, C. M., Gerth, A. I. & McMurray, B. Spatiotemporal structure of REM sleep twitching reveals developmental origins of motor synergies. Curr. Biol. 23, 2100–2109 (2013).
Dilley, L. C., Vigderman, A., Williams, C. E. & Kayser, M. S. Behavioral and genetic features of sleep ontogeny in Drosophila. Sleep 41, zsy086 (2018).
Hobson, J. A. Sleep is of the brain, by the brain and for the brain. Nature 437, 1254–1256 (2005).
Saper, C. B., Scammell, T. E. & Lu, J. Hypothalamic regulation of sleep and circadian rhythms. Nature 437, 1257–1263 (2005).
Joiner, W. J., Crocker, A., White, B. H. & Sehgal, A. Sleep in Drosophila is regulated by adult mushroom bodies. Nature 441, 757–760 (2006).
Bringmann, H. Sleep-active neurons: conserved motors of sleep. Genetics 208, 1279–1289 (2018).
Lim, J. & Dinges, D. F. Sleep deprivation and vigilant attention. Ann. NY Acad. Sci. 1129, 305–322 (2008).
Kirszenblat, L. & van Swinderen, B. The yin and yang of sleep and attention. Trends Neurosci. 38, 776–786 (2015).
Cirelli, C. & Tononi, G. Sleep and synaptic homeostasis. Sleep 38, 161–162 (2015).
Krueger, J. M. & Tononi, G. Local use-dependent sleep; synthesis of the new paradigm. Curr. Top. Med. Chem. 11, 2490–2492 (2011).
Vyazovskiy, V. V. & Harris, K. D. Sleep and the single neuron: the role of global slow oscillations in individual cell rest. Nat. Rev. Neurosci. 14, 443–451 (2013).
Krueger, J. M., Huang, Y. H., Rector, D. M. & Buysse, D. J. Sleep: a synchrony of cell activity-driven small network states. Eur. J. Neurosci. 38, 2199–2209 (2013).
Saper, C. B., Fuller, P. M., Pedersen, N. P., Lu, J. & Scammell, T. E. Sleep state switching. Neuron 68, 1023–1042 (2010).
Hinard, V. et al. Key electrophysiological, molecular, and metabolic signatures of sleep and wakefulness revealed in primary cortical cultures. J. Neurosci. 32, 12506–12517 (2012).
Jewett, K. A. et al. Tumor necrosis factor enhances the sleep-like state and electrical stimulation induces a wake-like state in co-cultures of neurons and glia. Eur. J. Neurosci. 42, 2078–2090 (2015).
Laposky, A. et al. Deletion of the mammalian circadian clock gene BMAL1/Mop3 alters baseline sleep architecture and the response to sleep deprivation. Sleep 28, 395–409 (2005).
Ehlen, J. C. et al. Bmal1 function in skeletal muscle regulates sleep. eLife 6, e26557 (2017).
Williams, J. A., Sathyanarayanan, S., Hendricks, J. C. & Sehgal, A. Interaction between sleep and the immune response in Drosophila: a role for the NFκB Relish. Sleep 30, 389–400 (2007).
Bennett, H. L. et al. Normal sleep bouts are not essential for C. elegans survival and FoxO is important for compensatory changes in sleep. BMC Neurosci. 19, 10 (2018).
Iannacone, M. J. et al. The RFamide receptor DMSR-1 regulates stress-induced sleep in C. elegans. eLife 6, e19837 (2017).
Smith, C. L. et al. Novel cell types, neurosecretory cells, and body plan of the early-diverging metazoan Trichoplax adhaerens. Curr. Biol. 24, 1565–1572 (2014).
Senatore, A., Reese, T. S. & Smith, C. L. Neuropeptidergic integration of behavior in Trichoplax adhaerens, an animal without synapses. J. Exp. Biol. 220, 3381–3390 (2017).
Smith, C. L., Pivovarova, N. & Reese, T. S. Coordinated feeding behavior in Trichoplax, an animal without synapses. PLOS ONE 10, e0136098 (2015).
Varoqueaux, F. et al. High cell diversity and complex peptidergic signaling underlie placozoan behavior. Curr. Biol. 28, 3495–3501 (2018).
Sakarya, O. et al. A post-synaptic scaffold at the origin of the animal kingdom. PLOS ONE 2, e506 (2007).
Nickel, M. Kinetics and rhythm of body contractions in the sponge Tethya wilhelma (Porifera: Demospongiae). J. Exp. Biol. 207, 4515–4524 (2004).
Ludeman, D. A., Farrar, N., Riesgo, A., Paps, J. & Leys, S. P. Evolutionary origins of sensation in metazoans: functional evidence for a new sensory organ in sponges. BMC Evol. Biol. 14, 3 (2014).
de Mairan, J. J. D. Histoire de l’Académie Royale des Sciences (Année 1729) 35–36 (Imprimerie Royale, 1731).
Hattori, A. et al. Identification of melatonin in plants and its effects on plasma melatonin levels and binding to melatonin receptors in vertebrates. Biochem. Mol. Biol. Int. 35, 627–634 (1995).
Arnao, M. B. & Hernandez-Ruiz, J. Functions of melatonin in plants: a review. J. Pineal Res. 59, 133–150 (2015).
Poroyko, V. A. et al. Chronic sleep disruption alters gut microbiota, induces systemic and adipose tissue inflammation and insulin resistance in mice. Sci. Rep. 6, 35405 (2016).
Szuperak, M. et al. A sleep state in Drosophila larvae required for neural stem cell proliferation. eLife 7, e33220 (2018).
Kayser, M. S., Yue, Z. & Sehgal, A. A critical period of sleep for development of courtship circuitry and behavior in Drosophila. Science 344, 269–274 (2014).
Roffwarg, H. P., Muzio, J. N. & Dement, W. C. Ontogenetic development of the human sleep–dream cycle. Science 152, 604–619 (1966).
Davis, K. C. & Raizen, D. M. A mechanism for sickness sleep: lessons from invertebrates. J. Physiol. 595, 5415–5424 (2016).
Prather, A. A., Janicki-Deverts, D., Hall, M. H. & Cohen, S. Behaviorally assessed sleep and susceptibility to the common cold. Sleep 38, 1353–1359 (2015).
Kuo, T. H. & Williams, J. A. Increased sleep promotes survival during a bacterial infection in Drosophila. Sleep 37, 1077–1086 (2014).
Hill, A. J., Mansfield, R., Lopez, J. M., Raizen, D. M. & Van Buskirk, C. Cellular stress induces a protective sleep-like state in C. elegans. Curr. Biol. 24, 2399–2405 (2014).
Huang, T. C., Tu, J., Chow, T. J. & Chen, T. H. Circadian rhythm of the prokaryote Synechococcus sp. RF-1. Plant Physiol. 92, 531–533 (1990).
Postgate, J. Nitrogen Fixation 3rd edn (Cambridge Univ. Press, 1998).
Liu, Y. et al. Circadian orchestration of gene expression in cyanobacteria. Genes Dev. 9, 1469–1478 (1995).
Tu, B. P., Kudlicki, A., Rowicka, M. & McKnight, S. L. Logic of the yeast metabolic cycle: temporal compartmentalization of cellular processes. Science 310, 1152–1158 (2005).
Chen, Z., Odstrcil, E. A., Tu, B. P. & McKnight, S. L. Restriction of DNA replication to the reductive phase of the metabolic cycle protects genome integrity. Science 316, 1916–1919 (2007).
Xie, L. et al. Sleep drives metabolite clearance from the adult brain. Science 342, 373–377 (2013).
Maret, S. et al. Homer1a is a core brain molecular correlate of sleep loss. Proc. Natl Acad. Sci. USA 104, 20090–20095 (2007).
Cirelli, C., Gutierrez, C. M. & Tononi, G. Extensive and divergent effects of sleep and wakefulness on brain gene expression. Neuron 41, 35–43 (2004).
Anafi, R. C. et al. Sleep is not just for the brain: transcriptional responses to sleep in peripheral tissues. BMG Genomics 14, 362 (2013).
Mackiewicz, M. et al. Macromolecule biosynthesis: a key function of sleep. Physiol. Genom. 31, 441–457 (2007).
Thompson, C. L. et al. Molecular and anatomical signatures of sleep deprivation in the mouse brain. Front. Neurosci. 4, 165 (2010).
Archer, S. N. et al. Mistimed sleep disrupts circadian regulation of the human transcriptome. Proc. Natl Acad. Sci. USA 111, E682–E691 (2014).
Balsalobre, A. et al. Resetting of circadian time in peripheral tissues by glucocorticoid signaling. Science 289, 2344–2347 (2000).
Kajimoto, J., Matsumura, R., Node, K. & Akashi, M. Potential role of the pancreatic hormone insulin in resetting human peripheral clocks. Genes Cells 23, 393–399 (2018).
Hardman, J. A., Haslam, I. S., Farjo, N., Farjo, B. & Paus, R. Thyroxine differentially modulates the peripheral clock: lessons from the human hair follicle. PLOS ONE 10, e0121878 (2015).
Brown, S. A., Zumbrunn, G., Fleury-Olela, F., Preitner, N. & Schibler, U. Rhythms of mammalian body temperature can sustain peripheral circadian clocks. Curr. Biol. 12, 1574–1583 (2002).
Franken, P. & Dijk, D. J. Circadian clock genes and sleep homeostasis. Eur. J. Neurosci. 29, 1820–1829 (2009).
Durkin, J. & Aton, S. J. Sleep-dependent potentiation in the visual system is at odds with the synaptic homeostasis hypothesis. Sleep 39, 155–159 (2016).
Liu, Z. W., Faraguna, U., Cirelli, C., Tononi, G. & Gao, X. B. Direct evidence for wake-related increases and sleep-related decreases in synaptic strength in rodent cortex. J. Neurosci. 30, 8671–8675 (2010).
Hengen, K. B., Torrado Pacheco, A., McGregor, J. N., Van Hooser, S. D. & Turrigiano, G. G. Neuronal firing rate homeostasis is inhibited by sleep and promoted by wake. Cell 165, 180–191 (2016).
Tononi, G. & Cirelli, C. Sleep and the price of plasticity: from synaptic and cellular homeostasis to memory consolidation and integration. Neuron 81, 12–34 (2014).
Raichle, M. E. & Mintun, M. A. Brain work and brain imaging. Annu. Rev. Neurosci. 29, 449–476 (2006).
Lesku, J. A. & Ly, L. M. T. Sleep origins: restful jellyfish are sleeping jellyfish. Curr. Biol. 27, R1060–R1062 (2017).
Weljie, A. M. et al. Oxalic acid and diacylglycerol 36:3 are cross-species markers of sleep debt. Proc. Natl Acad. Sci. USA 112, 2569–2574 (2015).
Davies, S. K. et al. Effect of sleep deprivation on the human metabolome. Proc. Natl Acad. Sci. USA 111, 10761–10766 (2014).
Tu, B. P. et al. Cyclic changes in metabolic state during the life of a yeast cell. Proc. Natl Acad. Sci. USA 104, 16886–16891 (2007).
Yurgel, M. E. et al. Ade2 functions in the Drosophila fat body to promote sleep. G3 8, 3385–3395 (2018).
Thimgan, M. S., Suzuki, Y., Seugnet, L., Gottschalk, L. & Shaw, P. J. The perilipin homologue, lipid storage droplet 2, regulates sleep homeostasis and prevents learning impairments following sleep loss. PLOS Biol. 8, e1000466 (2010).
Skora, S., Mende, F. & Zimmer, M. Energy scarcity promotes a brain-wide sleep state modulated by insulin signaling in C. elegans. Cell Rep. 22, 953–966 (2018).
Rechtschaffen, A., Bergmann, B. M., Everson, C. A., Kushida, C. A. & Gilliland, M. A. Sleep deprivation in the rat: X. Integration and discussion of the findings. Sleep 12, 68–87 (1989).
Shaw, P. J., Tononi, G., Greenspan, R. J. & Robinson, D. F. Stress response genes protect against lethal effects of sleep deprivation in Drosophila. Nature 417, 287–291 (2002).
Rechtschaffen, A. Current perspectives on the function of sleep. Perspect. Biol. Med. 41, 359–390 (1998).
Everson, C. A. Clinical assessment of blood leukocytes, serum cytokines, and serum immunoglobulins as responses to sleep deprivation in laboratory rats. Am. J. Physiol. Regul. Integr.Comp. Physiol. 289, R1054–R1063 (2005).
McHill, A. W. & Wright, K. P. Jr. Role of sleep and circadian disruption on energy expenditure and in metabolic predisposition to human obesity and metabolic disease. Obes. Rev. 18, S15–S24 (2017).
Naidoo, N. et al. Aging and sleep deprivation induce the unfolded protein response in the pancreas: implications for metabolism. Aging Cell 13, 131–141 (2014).
Rechtschaffen, A., Gilliland, M. A., Bergmann, B. M. & Winter, J. B. Physiological correlates of prolonged sleep deprivation in rats. Science 221, 182–184 (1983).
Walker, M. P. & Stickgold, R. Sleep, memory, and plasticity. Annu. Rev. Psychol. 57, 139–166 (2006).
Benington, J. H. & Frank, M. G. Cellular and molecular connections between sleep and synaptic plasticity. Prog. Neurobiol. 69, 71–101 (2003).
Zhang, J. et al. Extended wakefulness: compromised metabolics in and degeneration of locus ceruleus neurons. J. Neurosci. 34, 4418–4431 (2014).
Spiegel, K., Leproult, R. & Van Cauter, E. Impact of sleep debt on metabolic and endocrine function. Lancet 354, 1435–1439 (1999).
Imeri, L. & Opp, M. R. How (and why) the immune system makes us sleep. Nat. Rev. Neurosci. 10, 199–210 (2009).
Dubowy, C. et al. Genetic dissociation of daily sleep and sleep following thermogenetic sleep deprivation in Drosophila. Sleep 39, 1083–1095 (2016).
Seidner, G. et al. Identification of neurons with a privileged role in sleep homeostasis in Drosophila melanogaster. Curr. Biol. 25, 2928–2938 (2015).
Alvarenga, T. A., Andersen, M. L., Papale, L. A., Antunes, I. B. & Tufik, S. Influence of long-term food restriction on sleep pattern in male rats. Brain Res. 1057, 49–56 (2005).
Slocumb, M. E. et al. Enhanced sleep is an evolutionarily adaptive response to starvation stress in Drosophila. PLOS ONE 10, e0131275 (2015).
Duboue, E. R., Keene, A. C. & Borowsky, R. L. Evolutionary convergence on sleep loss in cavefish populations. Curr. Biol. 21, 671–676 (2011).
Keene, A. C. et al. Clock and cycle limit starvation-induced sleep loss in Drosophila. Curr. Biol. 20, 1209–1215 (2010).
Goetting, D. L., Soto, R. & Van Buskirk, C. Food-dependent plasticity in Caenorhabditis elegans stress-induced sleep is mediated by TOR-FOXA and TGF-β signaling. Genetics 209, 1183–1195 (2018).
Ramm, P. & Smith, C. T. Rates of cerebral protein synthesis are linked to slow wave sleep in the rat. Physiol. Behav. 48, 749–753 (1990).
Simor, A. et al. The short- and long-term proteomic effects of sleep deprivation on the cortical and thalamic synapses. Mol. Cell. Neurosci. 79, 64–80 (2017).
Siegel, J. M. Sleep viewed as a state of adaptive inactivity. Nat. Rev. Neurosci. 10, 747–753 (2009).
Acknowledgements
The authors thank A. Rohacek and S. Belfer for comments. R.C.A. is supported by US Defense Advanced Research Projects Agency grant D17AP00003; M.S.K. is supported by K08NS090461 (US National Institutes of Health), a Burroughs Wellcome Career Award for Medical Scientists, a March of Dimes Basil O’Connor Scholar Award and a Sloan Research Fellowship; and D.M.R. is supported by R01NS088432 (US National Institutes of Health).
Reviewer information
Nature Reviews Neuroscience thanks D. Prober, M. Zimmer and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
All authors wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Anafi, R.C., Kayser, M.S. & Raizen, D.M. Exploring phylogeny to find the function of sleep. Nat Rev Neurosci 20, 109–116 (2019). https://doi.org/10.1038/s41583-018-0098-9
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41583-018-0098-9
This article is cited by
-
A midbrain GABAergic circuit constrains wakefulness in a mouse model of stress
Nature Communications (2024)
-
A perfect storm: sleep loss causes systemic inflammation and death
Cell Research (2024)
-
Waking Up the Sleep Field: An Overview on the Implications of Genetics and Bioinformatics of Sleep
Molecular Biotechnology (2024)
-
Sleep-mediated regulation of reward circuits: implications in substance use disorders
Neuropsychopharmacology (2023)
-
Development and validation of a sleep questionnaire, SNoRE 3.0, to evaluate sleep in companion dogs
Scientific Reports (2023)