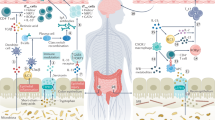
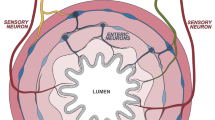
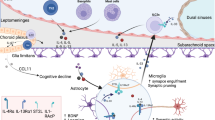
Abstract
Interactions between the immune and nervous systems are of central importance in neuropathic pain, a common and debilitating form of chronic pain caused by a lesion or disease affecting the somatosensory system. Our understanding of neuroimmune interactions in pain research has advanced considerably. Initially considered as passive bystanders, then as culprits in the pathogenesis of neuropathic pain, immune responses in the nervous system are now established to underpin not only the initiation and progression of pain but also its resolution. Indeed, immune cells and their mediators are well-established promoters of neuroinflammation at each level of the neural pain pathway that contributes to pain hypersensitivity. However, emerging evidence indicates that specific subtypes of immune cells (including antinociceptive macrophages, pain-resolving microglia and T regulatory cells) as well as immunoresolvent molecules and modulators of the gut microbiota–immune system axis can reduce the pain experience and contribute to the resolution of neuropathic pain. This Review provides an overview of the immune mechanisms responsible for the resolution of neuropathic pain, including those involved in innate, adaptive and meningeal immunity as well as interactions with the gut microbiome. Specialized pro-resolving mediators and therapeutic approaches that target these neuroimmune mechanisms are also discussed.
Key points
-
Accumulating evidence indicates that subpopulations of immune cells and mediators help to suppress nociception and promote the resolution of neuropathic pain.
-
Pain-resolving immune cells infiltrate the peripheral nervous system following nerve damage, inhibit pronociceptive immune or glial cells, and dampen primary afferent excitability via the release of anti-inflammatory cytokines and opioids.
-
Pain-resolving meningeal immune cells and resident spinal cord microglia suppress central sensitization by inhibiting pronociceptive reactive glial cells and neuronal hyperexcitability.
-
Gut microbiota production of bile acids and butyrate, and restoration of a dysbiotic gut microbiome with probiotics, antibiotics or faecal microbiota transplantation, promote pain resolution by modulating neuroimmune signalling pathways.
-
Specialized pro-resolving mediators regulate glial reactivity and inflammatory signalling and modulate primary nociceptive neuronal activity by acting on transient receptor potential channels and G protein-coupled receptors.
-
Current treatments for neuropathic pain have limited efficacy; novel therapeutic approaches that target pain-resolving neuroimmune mechanisms provide exciting new avenues to treat neuropathic pain.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Austin, P. J. & Moalem-Taylor, G. The neuro-immune balance in neuropathic pain: involvement of inflammatory immune cells, immune-like glial cells and cytokines. J. Neuroimmunol. 229, 26–50 (2010).
Grace, P. M., Hutchinson, M. R., Maier, S. F. & Watkins, L. R. Pathological pain and the neuroimmune interface. Nat. Rev. Immunol. 14, 217–231 (2014).
Moalem, G. & Tracey, D. J. Immune and inflammatory mechanisms in neuropathic pain. Brain Res. Rev. 51, 240–264 (2006).
Watkins, L. R. & Maier, S. F. Beyond neurons: evidence that immune and glial cells contribute to pathological pain states. Physiol. Rev. 82, 981–1011 (2002).
Kuner, R. Central mechanisms of pathological pain. Nat. Med. 16, 1258–1266 (2010).
Mogil, J. S. Qualitative sex differences in pain processing: emerging evidence of a biased literature. Nat. Rev. Neurosci. 21, 353–365 (2020).
Cohen, S. P., Vase, L. & Hooten, W. M. Chronic pain: an update on burden, best practices, and new advances. Lancet 397, 2082–2097 (2021).
Raja, S. N. et al. The revised International Association for the Study of Pain definition of pain: concepts, challenges, and compromises. Pain 161, 1976–1982 (2020).
Attal, N. et al. Neuropathic pain: are there distinct subtypes depending on the aetiology or anatomical lesion? Pain 138, 343–353 (2008).
Finnerup, N. B., Kuner, R. & Jensen, T. S. Neuropathic pain: from mechanisms to treatment. Physiol. Rev. 101, 259–301 (2021).
van Hecke, O., Austin, S. K., Khan, R. A., Smith, B. H. & Torrance, N. Neuropathic pain in the general population: a systematic review of epidemiological studies. Pain 155, 654–662 (2014).
Colloca, L. et al. Neuropathic pain. Nat. Rev. Dis. Prim. 3, 17002 (2017).
Nicholson, B. & Verma, S. Comorbidities in chronic neuropathic pain. Pain Med. 5 (Suppl. 1), S9–S27 (2004).
Doth, A. H., Hansson, P. T., Jensen, M. P. & Taylor, R. S. The burden of neuropathic pain: a systematic review and meta-analysis of health utilities. Pain 149, 338–344 (2010).
Dyer, O. US opioid epidemic: FDA demands studies of whether opioids do control chronic pain. BMJ 364, l959 (2019).
Martel, M. O. et al. Self-reports of medication side effects and pain-related activity interference in patients with chronic pain: a longitudinal cohort study. Pain 156, 1092–1100 (2015).
Louveau, A., Harris, T. H. & Kipnis, J. Revisiting the mechanisms of CNS immune privilege. Trends Immunol. 36, 569–577 (2015).
Croese, T., Castellani, G. & Schwartz, M. Immune cell compartmentalization for brain surveillance and protection. Nat. Immunol. 22, 1083–1092 (2021).
Ellis, A. & Bennett, D. L. Neuroinflammation and the generation of neuropathic pain. Br. J. Anaesth. 111, 26–37 (2013).
Austin, P. J., Kim, C. F., Perera, C. J. & Moalem-Taylor, G. Regulatory T cells attenuate neuropathic pain following peripheral nerve injury and experimental autoimmune neuritis. Pain 153, 1916–1931 (2012).
Bonomo, R. R. et al. Fecal transplantation and butyrate improve neuropathic pain, modify immune cell profile, and gene expression in the PNS of obese mice. Proc. Natl Acad. Sci. USA 117, 26482–26493 (2020).
Davoli-Ferreira, M. et al. Regulatory T cells counteract neuropathic pain through inhibition of the Th1 response at the site of peripheral nerve injury. Pain 161, 1730–1743 (2020).
Duffy, S. S. et al. Regulatory T cells and their derived cytokine, interleukin-35, reduce pain in experimental autoimmune encephalomyelitis. J. Neurosci. 39, 2326–2346 (2019).
Fischer, R. et al. TNFR2 promotes Treg-mediated recovery from neuropathic pain across sexes. Proc. Natl Acad. Sci. USA 116, 17045–17050 (2019).
Kuhn, J. A. et al. Regulatory T-cells inhibit microglia-induced pain hypersensitivity in female mice. eLife 10, 69056 (2021).
Niehaus, J. K., Taylor-Blake, B., Loo, L., Simon, J. M. & Zylka, M. J. Spinal macrophages resolve nociceptive hypersensitivity after peripheral injury. Neuron 109, 1274–1282.e6 (2021).
Singh, S. K. et al. CD8+ T cell-derived IL-13 increases macrophage IL-10 to resolve neuropathic pain. JCI Insight 7, 154194 (2022).
Kohno, K. et al. A spinal microglia population involved in remitting and relapsing neuropathic pain. Science 376, 86–90 (2022).
Moore, R. A., Chi, C. C., Wiffen, P. J., Derry, S. & Rice, A. S. Oral nonsteroidal anti-inflammatory drugs for neuropathic pain. Cochrane Database Syst. Rev. https://doi.org/10.1002/14651858.CD010902.pub2 (2015).
Parisien, M. et al. Acute inflammatory response via neutrophil activation protects against the development of chronic pain. Sci. Transl. Med. 14, eabj9954 (2022).
Brack, A. et al. Control of inflammatory pain by chemokine-mediated recruitment of opioid-containing polymorphonuclear cells. Pain 112, 229–238 (2004).
Rittner, H. L. et al. Mycobacteria attenuate nociceptive responses by formyl peptide receptor triggered opioid peptide release from neutrophils. PLoS Pathog. 5, e1000362 (2009).
Davies, A. J. et al. Natural killer cells degenerate intact sensory afferents following nerve injury. Cell 176, 716–728.e18 (2019).
Xie, W., Strong, J. A. & Zhang, J. M. Active nerve regeneration with failed target reinnervation drives persistent neuropathic pain. eNeuro https://doi.org/10.1523/ENEURO.0008-17.2017 (2017).
Gao, Y. H. et al. NK cells mediate the cumulative analgesic effect of electroacupuncture in a rat model of neuropathic pain. BMC Complement. Altern. Med. 14, 316 (2014).
Lassen, J. et al. Protective role of natural killer cells in neuropathic pain conditions. Pain 162, 2366–2375 (2021).
Davidson, S. et al. Fibroblasts as immune regulators in infection, inflammation and cancer. Nat. Rev. Immunol. 21, 704–717 (2021).
Zhang, S. et al. Fibroblastic SMOC2 suppresses mechanical nociception by inhibiting coupled activation of primary sensory neurons. J. Neurosci. 42, 4069–4086 (2022).
Soderblom, C. et al. Perivascular fibroblasts form the fibrotic scar after contusive spinal cord injury. J. Neurosci. 33, 13882–13887 (2013).
Kobayashi, D., Kiguchi, N., Saika, F., Kishioka, S. & Matsuzaki, S. Insufficient efferocytosis by M2-like macrophages as a possible mechanism of neuropathic pain induced by nerve injury. Biochem. Biophys. Res. Commun. 525, 216–223 (2020).
Takahashi, Y., Hasegawa-Moriyama, M., Sakurai, T. & Inada, E. The macrophage-mediated effects of the peroxisome proliferator-activated receptor-γ agonist rosiglitazone attenuate tactile allodynia in the early phase of neuropathic pain development. Anesth. Analg. 113, 398–404 (2011).
Celik, M. O., Labuz, D., Keye, J., Glauben, R. & Machelska, H. IL-4 induces M2 macrophages to produce sustained analgesia via opioids. JCI Insight 5, 133093 (2020).
Kiguchi, N. et al. Peripheral interleukin-4 ameliorates inflammatory macrophage-dependent neuropathic pain. Pain 156, 684–693 (2015).
Labuz, D., Celik, M. O., Seitz, V. & Machelska, H. Interleukin-4 induces the release of opioid peptides from M1 macrophages in pathological pain. J. Neurosci. 41, 2870–2882 (2021).
Pannell, M. et al. Adoptive transfer of M2 macrophages reduces neuropathic pain via opioid peptides. J. Neuroinflammation 13, 262 (2016).
Iwai, H. et al. Tissue-resident M2 macrophages directly contact primary sensory neurons in the sensory ganglia after nerve injury. J. Neuroinflammation 18, 227 (2021).
Tonello, R. et al. Local sympathectomy promotes anti-inflammatory responses and relief of paclitaxel-induced mechanical and cold allodynia in mice. Anesthesiology 132, 1540–1553 (2020).
Kong, F. et al. PD-L1 improves motor function and alleviates neuropathic pain in male mice after spinal cord injury by inhibiting MAPK pathway. Front. Immunol. 12, 670646 (2021).
van der Vlist, M. et al. Macrophages transfer mitochondria to sensory neurons to resolve inflammatory pain. Neuron 110, 613–626.e9 (2022).
Lim, T. K., Rone, M. B., Lee, S., Antel, J. P. & Zhang, J. Mitochondrial and bioenergetic dysfunction in trauma-induced painful peripheral neuropathy. Mol. Pain 11, 58 (2015).
Luo, X. et al. Macrophage Toll-like receptor 9 contributes to chemotherapy-induced neuropathic pain in male mice. J. Neurosci. 39, 6848–6864 (2019).
Luo, X. et al. IL-23/IL-17A/TRPV1 axis produces mechanical pain via macrophage-sensory neuron crosstalk in female mice. Neuron 109, 2691–2706.e5 (2021).
Saika, F. et al. Chemogenetic regulation of CX3CR1-expressing microglia using Gi-DREADD exerts sex-dependent anti-allodynic effects in mouse models of neuropathic pain. Front. Pharmacol. 11, 925 (2020).
Sorge, R. E. et al. Different immune cells mediate mechanical pain hypersensitivity in male and female mice. Nat. Neurosci. 18, 1081–1083 (2015).
Sun, J. S. et al. Minocycline attenuates pain by inhibiting spinal microglia activation in diabetic rats. Mol. Med. Rep. 12, 2677–2682 (2015).
Fiore, N. T. et al. Sex-specific transcriptome of spinal microglia in neuropathic pain due to peripheral nerve injury. Glia 70, 675–696 (2022).
Tansley, S. et al. Single-cell RNA sequencing reveals time- and sex-specific responses of spinal cord microglia to peripheral nerve injury and links ApoE to neuropathic pain. Nat. Commun. 13, 843 (2022).
Vacca, V. et al. Higher pain perception and lack of recovery from neuropathic pain in females: a behavioural, immunohistochemical, and proteomic investigation on sex-related differences in mice. Pain 155, 388–402 (2014).
Romero-Sandoval, A., Nutile-McMenemy, N. & DeLeo, J. A. Spinal microglial and perivascular cell cannabinoid receptor type 2 activation reduces behavioral hypersensitivity without tolerance after peripheral nerve injury. Anesthesiology 108, 722–734 (2008).
Huo, W. et al. Dehydrocorydaline attenuates bone cancer pain by shifting microglial M1/M2 polarization toward the M2 phenotype. Mol. Pain 14, 1744806918781733 (2018).
Burke, N. N., Kerr, D. M., Moriarty, O., Finn, D. P. & Roche, M. Minocycline modulates neuropathic pain behaviour and cortical M1-M2 microglial gene expression in a rat model of depression. Brain Behav. Immun. 42, 147–156 (2014).
Jin, J. et al. M2-Like microglia polarization attenuates neuropathic pain associated with Alzheimer’s disease. J. Alzheimers Dis. 76, 1255–1265 (2020).
Duffy, S. S., Hayes, J. P., Fiore, N. T. & Moalem-Taylor, G. The cannabinoid system and microglia in health and disease. Neuropharmacology 190, 108555 (2021).
Wu, J., Hocevar, M., Bie, B., Foss, J. F. & Naguib, M. Cannabinoid type 2 receptor system modulates paclitaxel-induced microglial dysregulation and central sensitization in rats. J. Pain 20, 501–514 (2019).
Dos Santos, R. S. et al. Involvement of spinal cannabinoid CB2 receptors in exercise-induced antinociception. Neuroscience 418, 177–188 (2019).
Wu, H. Y., Mao, X. F., Fan, H. & Wang, Y. X. p38β mitogen-activated protein kinase signaling mediates exenatide-stimulated microglial β-endorphin expression. Mol. Pharmacol. 91, 451–463 (2017).
Li, T. F., Wu, H. Y., Wang, Y. R., Li, X. Y. & Wang, Y. X. Molecular signaling underlying bulleyaconitine A (BAA)-induced microglial expression of prodynorphin. Sci. Rep. 7, 45056 (2017).
Wu, H. Y. et al. Spinal interleukin-10 produces antinociception in neuropathy through microglial β-endorphin expression, separated from antineuroinflammation. Brain Behav. Immun. 73, 504–519 (2018).
Wu, H. Y., Tang, X. Q., Mao, X. F. & Wang, Y. X. Autocrine interleukin-10 mediates glucagon-like peptide-1 receptor-induced spinal microglial β-endorphin expression. J. Neurosci. 37, 11701–11714 (2017).
Tang, X., Wu, H., Mao, X., Li, X. & Wang, Y. The GLP-1 receptor herbal agonist morroniside attenuates neuropathic pain via spinal microglial expression of IL-10 and β-endorphin. Biochem. Biophys. Res. Commun. 530, 494–499 (2020).
Apryani, E. et al. The spinal microglial IL-10/β-endorphin pathway accounts for cinobufagin-induced mechanical antiallodynia in bone cancer pain following activation of α7-nicotinic acetylcholine receptors. J. Neuroinflammation 17, 75 (2020).
Ali, U. et al. Low frequency electroacupuncture alleviates neuropathic pain by activation of spinal microglial IL-10/β-endorphin pathway. Biomed. Pharmacother. 125, 109898 (2020).
Mao, X. F. et al. Activation of GPR40 produces mechanical antiallodynia via the spinal glial interleukin-10/β-endorphin pathway. J. Neuroinflammation 16, 84 (2019).
Ma, L. et al. Spinal microglial β-endorphin signaling mediates IL-10 and exenatide-induced inhibition of synaptic plasticity in neuropathic pain. CNS Neurosci. Ther. 27, 1157–1172 (2021).
Shoaib, R. M., Ahmad, K. A. & Wang, Y. X. Protopanaxadiol alleviates neuropathic pain by spinal microglial dynorphin A expression following glucocorticoid receptor activation. Br. J. Pharmacol. 178, 2976–2997 (2021).
Okutani, H., Yamanaka, H., Kobayashi, K., Okubo, M. & Noguchi, K. Recombinant interleukin-4 alleviates mechanical allodynia via injury-induced interleukin-4 receptor α in spinal microglia in a rat model of neuropathic pain. Glia 66, 1775–1787 (2018).
Ji, R. R., Chamessian, A. & Zhang, Y. Q. Pain regulation by non-neuronal cells and inflammation. Science 354, 572–577 (2016).
Fiore, N. T. & Austin, P. J. Are the emergence of affective disturbances in neuropathic pain states contingent on supraspinal neuroinflammation? Brain Behav. Immun. 56, 397–411 (2016).
Moalem, G., Xu, K. & Yu, L. T lymphocytes play a role in neuropathic pain following peripheral nerve injury in rats. Neuroscience 129, 767–777 (2004).
Leger, T., Grist, J., D’Acquisto, F., Clark, A. K. & Malcangio, M. Glatiramer acetate attenuates neuropathic allodynia through modulation of adaptive immune cells. J. Neuroimmunol. 234, 19–26 (2011).
Labuz, D., Schreiter, A., Schmidt, Y., Brack, A. & Machelska, H. T lymphocytes containing β-endorphin ameliorate mechanical hypersensitivity following nerve injury. Brain Behav. Immun. 24, 1045–1053 (2010).
Xu, Z. et al. Pain relief dependent on IL-17-CD4+ T cell-β-endorphin axis in rat model of brachial plexus root avulsion after electroacupuncture therapy. Front. Neurosci. 14, 596780 (2020).
Lees, J. G., Duffy, S. S., Perera, C. J. & Moalem-Taylor, G. Depletion of Foxp3+ regulatory T cells increases severity of mechanical allodynia and significantly alters systemic cytokine levels following peripheral nerve injury. Cytokine 71, 207–214 (2015).
Chen, H. et al. Exploring the correlation between the regulation of macrophages by regulatory t cells and peripheral neuropathic pain. Front. Neurosci. 16, 813751 (2022).
Liu, X. J. et al. Nociceptive neurons regulate innate and adaptive immunity and neuropathic pain through MyD88 adapter. Cell Res. 24, 1374–1377 (2014).
Hu, R., Zhang, J., Liu, X., Huang, D. & Cao, Y. Q. Low-dose interleukin-2 and regulatory T cell treatments attenuate punctate and dynamic mechanical allodynia in a mouse model of sciatic nerve injury. J. Pain Res. 14, 893–906 (2021).
Heyn, J. et al. miR-124a and miR-155 enhance differentiation of regulatory T cells in patients with neuropathic pain. J. Neuroinflammation 13, 248 (2016).
Luchting, B. et al. Anti-inflammatory T-cell shift in neuropathic pain. J. Neuroinflammation 12, 12 (2015).
Xing, Q., Hu, D., Shi, F. & Chen, F. Role of regulatory T cells in patients with acute herpes zoster and relationship to postherpetic neuralgia. Arch. Dermatol. Res. 305, 715–722 (2013).
Moalem-Taylor, G. et al. Immune dysregulation in patients with carpal tunnel syndrome. Sci. Rep. 7, 8218 (2017).
Saligrama, N. et al. Opposing T cell responses in experimental autoimmune encephalomyelitis. Nature 572, 481–487 (2019).
Krukowski, K. et al. CD8+ T cells and endogenous IL-10 are required for resolution of chemotherapy-induced neuropathic pain. J. Neurosci. 36, 11074–11083 (2016).
Laumet, G., Edralin, J. D., Dantzer, R., Heijnen, C. J. & Kavelaars, A. Cisplatin educates CD8+ T cells to prevent and resolve chemotherapy-induced peripheral neuropathy in mice. Pain 160, 1459–1468 (2019).
Kaufmann, I. et al. Lymphocyte subsets and the role of TH1/TH2 balance in stressed chronic pain patients. Neuroimmunomodulation 14, 272–280 (2007).
Lopes, D. M. et al. Sex differences in peripheral not central immune responses to pain-inducing injury. Sci. Rep. 7, 16460 (2017).
Rosen, S. F. et al. T-cell mediation of pregnancy analgesia affecting chronic pain in mice. J. Neurosci. 37, 9819–9827 (2017).
Cao, L. & DeLeo, J. A. CNS-infiltrating CD4+ T lymphocytes contribute to murine spinal nerve transection-induced neuropathic pain. Eur. J. Immunol. 38, 448–458 (2008).
Costigan, M. et al. T-cell infiltration and signaling in the adult dorsal spinal cord is a major contributor to neuropathic pain-like hypersensitivity. J. Neurosci. 29, 14415–14422 (2009).
Liang, Z. et al. A transcriptional toolbox for exploring peripheral neuroimmune interactions. Pain 161, 2089–2106 (2020).
Chernov, A. V. et al. A myelin basic protein fragment induces sexually dimorphic transcriptome signatures of neuropathic pain in mice. J. Biol. Chem. 295, 10807–10821 (2020).
Lee, H. J. et al. Sex-specific B cell and anti-myelin autoantibody response after peripheral nerve injury. Front. Cell Neurosci. 16, 835800 (2022).
Guo, T. Z. et al. Pronociceptive autoantibodies in the spinal cord mediate nociceptive sensitization, loss of function, and spontaneous pain in the lumbar disc puncture model of chronic back pain. Pain 164, 421–434 (2022).
Li, W. W. et al. Autoimmunity contributes to nociceptive sensitization in a mouse model of complex regional pain syndrome. Pain 155, 2377–2389 (2014).
Helyes, Z. et al. Transfer of complex regional pain syndrome to mice via human autoantibodies is mediated by interleukin-1-induced mechanisms. Proc. Natl Acad. Sci. USA 116, 13067–13076 (2019).
Guo, T. Z. et al. Passive transfer autoimmunity in a mouse model of complex regional pain syndrome. Pain 158, 2410–2421 (2017).
Cuhadar, U. et al. Autoantibodies produce pain in complex regional pain syndrome by sensitizing nociceptors. Pain 160, 2855–2865 (2019).
de Mos, M. et al. The incidence of complex regional pain syndrome: a population-based study. Pain 129, 12–20 (2007).
Schneider, S., Randoll, D. & Buchner, M. Why do women have back pain more than men? A representative prevalence study in the federal republic of Germany. Clin. J. Pain 22, 738–747 (2006).
Dalakas, M. C. et al. Placebo-controlled trial of rituximab in IgM anti-myelin-associated glycoprotein antibody demyelinating neuropathy. Ann. Neurol. 65, 286–293 (2009).
Rosser, E. C. & Mauri, C. Regulatory B cells: origin, phenotype, and function. Immunity 42, 607–612 (2015).
Hua, D. et al. Gut microbiome and plasma metabolome signatures in middle-aged mice with cognitive dysfunction induced by chronic neuropathic pain. Front. Mol. Neurosci. 14, 806700 (2021).
Mollgard, K. et al. A mesothelium divides the subarachnoid space into functional compartments. Science 379, 84–88 (2023).
Alves de Lima, K., Rustenhoven, J. & Kipnis, J. Meningeal immunity and its function in maintenance of the central nervous system in health and disease. Annu. Rev. Immunol. 38, 597–620 (2020).
Louveau, A. et al. Structural and functional features of central nervous system lymphatic vessels. Nature 523, 337–341 (2015).
Cugurra, A. et al. Skull and vertebral bone marrow are myeloid cell reservoirs for the meninges and CNS parenchyma. Science https://doi.org/10.1126/science.abf7844 (2021).
Du, B. et al. CD4+ αβ T cell infiltration into the leptomeninges of lumbar dorsal roots contributes to the transition from acute to chronic mechanical allodynia after adult rat tibial nerve injuries. J. Neuroinflammation 15, 81 (2018).
Maganin, A. G. et al. Meningeal dendritic cells drive neuropathic pain through elevation of the kynurenine metabolic pathway in mice. J. Clin. Invest. 132, e153805 (2022).
Echeverry, S., Shi, X. Q., Rivest, S. & Zhang, J. Peripheral nerve injury alters blood-spinal cord barrier functional and molecular integrity through a selective inflammatory pathway. J. Neurosci. 31, 10819–10828 (2011).
Alexander, J. F. et al. Targeting the meningeal compartment to resolve chemobrain and neuropathy via nasal delivery of functionalized mitochondria. Adv. Healthc. Mater. 11, e2102153 (2022).
Alexander, J. F. et al. Nasal administration of mitochondria reverses chemotherapy-induced cognitive deficits. Theranostics 11, 3109–3130 (2021).
Guan, Z. et al. Injured sensory neuron-derived CSF1 induces microglial proliferation and DAP12-dependent pain. Nat. Neurosci. 19, 94–101 (2016).
Belkaid, Y. & Harrison, O. J. Homeostatic immunity and the microbiota. Immunity 46, 562–576 (2017).
Fan, Y. & Pedersen, O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol. 19, 55–71 (2021).
Guo, R., Chen, L. H., Xing, C. & Liu, T. Pain regulation by gut microbiota: molecular mechanisms and therapeutic potential. Br. J. Anaesth. 123, 637–654 (2019).
Minerbi, A. et al. Altered microbiome composition in individuals with fibromyalgia. Pain 160, 2589–2602 (2019).
Pane, K. et al. Role of gut microbiota in neuropathy and neuropathic pain states: a systematic preclinical review. Neurobiol. Dis. 170, 105773 (2022).
Ellis, R. J., Heaton, R. K., Gianella, S., Rahman, G. & Knight, R. Reduced gut microbiome diversity in people with HIV who have distal neuropathic pain. J. Pain 23, 318–325 (2022).
Morais, L. H., Schreiber, H. L. T. & Mazmanian, S. K. The gut microbiota–brain axis in behaviour and brain disorders. Nat. Rev. Microbiol. 19, 241–255 (2021).
Chen, P. et al. Alterations in the gut microbiota and metabolite profiles in the context of neuropathic pain. Mol. Brain 14, 50 (2021).
Lian, N. et al. Drinking hydrogen-rich water alleviates chemotherapy-induced neuropathic pain through the regulation of gut microbiota. J. Pain Res. 14, 681–691 (2021).
Ma, P. et al. Gut microbiota depletion by antibiotics ameliorates somatic neuropathic pain induced by nerve injury, chemotherapy, and diabetes in mice. J. Neuroinflammation 19, 169 (2022).
Shen, S. et al. Gut microbiota is critical for the induction of chemotherapy-induced pain. Nat. Neurosci. 20, 1213–1216 (2017).
Wardill, H. R. et al. Irinotecan-induced gastrointestinal dysfunction and pain are mediated by common TLR4-dependent mechanisms. Mol. Cancer Ther. 15, 1376–1386 (2016).
Alemi, F. et al. The TGR5 receptor mediates bile acid-induced itch and analgesia. J. Clin. Invest. 123, 1513–1530 (2013).
Minerbi, A. et al. Altered serum bile acid profile in fibromyalgia is associated with specific gut microbiome changes and symptom severity. Pain 164, e66–e76 (2022).
Ramakrishna, C. et al. Dominant role of the gut microbiota in chemotherapy induced neuropathic pain. Sci. Rep. 9, 20324 (2019).
Zhou, F. et al. Short-chain fatty acids contribute to neuropathic pain via regulating microglia activation and polarization. Mol. Pain 17, 1744806921996520 (2021).
Jing, Y. et al. Effect of fecal microbiota transplantation on neurological restoration in a spinal cord injury mouse model: involvement of brain-gut axis. Microbiome 9, 59 (2021).
Sideris-Lampretsas, G. & Malcangio, M. Pain-resolving microglia. Science 376, 33–34 (2022).
Erny, D. et al. Host microbiota constantly control maturation and function of microglia in the CNS. Nat. Neurosci. 18, 965–977 (2015).
Ding, W. et al. Gut microbiota influences neuropathic pain through modulating proinflammatory and anti-inflammatory T cells. Anesth. Analg. 132, 1146–1155 (2021).
Arpaia, N. et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 504, 451–455 (2013).
Wang, Z. et al. Lactobacillus paracasei S16 alleviates lumbar disc herniation by modulating inflammation response and gut microbiota. Front. Nutr. 8, 701644 (2021).
Singh, V. et al. Microbiota dysbiosis controls the neuroinflammatory response after stroke. J. Neurosci. 36, 7428–7440 (2016).
Markle, J. G. et al. Sex differences in the gut microbiome drive hormone-dependent regulation of autoimmunity. Science 339, 1084–1088 (2013).
Serhan, C. N. Pro-resolving lipid mediators are leads for resolution physiology. Nature 510, 92–101 (2014).
Serhan, C. N. Discovery of specialized pro-resolving mediators marks the dawn of resolution physiology and pharmacology. Mol. Asp. Med. 58, 1–11 (2017).
Serhan, C. N. A search for endogenous mechanisms of anti-inflammation uncovers novel chemical mediators: missing links to resolution. Histochem. Cell Biol. 122, 305–321 (2004).
Serhan, C. N. et al. Resolvins: a family of bioactive products of omega-3 fatty acid transformation circuits initiated by aspirin treatment that counter proinflammation signals. J. Exp. Med. 196, 1025–1037 (2002).
Dalli, J. & Serhan, C. N. Pro-resolving mediators in regulating and conferring macrophage function. Front. Immunol. 8, 1400 (2017).
Chiang, N., Bermudez, E. A., Ridker, P. M., Hurwitz, S. & Serhan, C. N. Aspirin triggers antiinflammatory 15-epi-lipoxin A4 and inhibits thromboxane in a randomized human trial. Proc. Natl Acad. Sci. USA 101, 15178–15183 (2004).
Basil, M. C. & Levy, B. D. Specialized pro-resolving mediators: endogenous regulators of infection and inflammation. Nat. Rev. Immunol. 16, 51–67 (2016).
Serhan, C. N., Dalli, J., Colas, R. A., Winkler, J. W. & Chiang, N. Protectins and maresins: new pro-resolving families of mediators in acute inflammation and resolution bioactive metabolome. Biochim. Biophys. Acta 1851, 397–413 (2015).
Chiang, N. & Serhan, C. N. Structural elucidation and physiologic functions of specialized pro-resolving mediators and their receptors. Mol. Asp. Med. 58, 114–129 (2017).
Chiang, N. & Serhan, C. N. Specialized pro-resolving mediator network: an update on production and actions. Essays Biochem. 64, 443–462 (2020).
Serhan, C. N. Treating inflammation and infection in the 21st century: new hints from decoding resolution mediators and mechanisms. FASEB J. 31, 1273–1288 (2017).
Kasuga, K. et al. Rapid appearance of resolvin precursors in inflammatory exudates: novel mechanisms in resolution. J. Immunol. 181, 8677–8687 (2008).
Serhan, C. N. & Petasis, N. A. Resolvins and protectins in inflammation resolution. Chem. Rev. 111, 5922–5943 (2011).
Dalli, J. & Serhan, C. N. Identification and structure elucidation of the pro-resolving mediators provides novel leads for resolution pharmacology. Br. J. Pharmacol. 176, 1024–1037 (2019).
Chavez-Castillo, M. et al. Specialized pro-resolving lipid mediators: the future of chronic pain therapy? Int. J. Mol. Sci. 22, 10370 (2021).
Leuti, A., Fava, M., Pellegrini, N. & Maccarrone, M. Role of specialized pro-resolving mediators in neuropathic pain. Front. Pharmacol. 12, 717993 (2021).
Fattori, V., Zaninelli, T. H., Rasquel-Oliveira, F. S., Casagrande, R. & Verri, W. A. Jr Specialized pro-resolving lipid mediators: a new class of non-immunosuppressive and non-opioid analgesic drugs. Pharmacol. Res. 151, 104549 (2020).
Zhang, L. Y., Jia, M. R. & Sun, T. The roles of special proresolving mediators in pain relief. Rev. Neurosci. 29, 645–660 (2018).
Zhang, J., Li, Z., Fan, M. & Jin, W. Lipoxins in the nervous system: brighter prospects for neuroprotection. Front. Pharmacol. 13, 781889 (2022).
Ji, R. R., Xu, Z. Z., Strichartz, G. & Serhan, C. N. Emerging roles of resolvins in the resolution of inflammation and pain. Trends Neurosci. 34, 599–609 (2011).
Luo, X., Gu, Y., Tao, X., Serhan, C. N. & Ji, R. R. Resolvin D5 inhibits neuropathic and inflammatory pain in male but not female mice: distinct actions of D-series resolvins in chemotherapy-induced peripheral neuropathy. Front. Pharmacol. 10, 745 (2019).
Baggio, D. F. et al. Sex dimorphism in resolvin D5-induced analgesia in rat models of trigeminal pain. J. Pain https://doi.org/10.1016/j.jpain.2022.12.013 (2022).
Liu, Z. H. et al. Resolvin D1 inhibits mechanical hypersensitivity in sciatica by modulating the expression of nuclear factor-κB, phospho-extracellular signal-regulated kinase, and pro- and antiinflammatory cytokines in the spinal cord and dorsal root ganglion. Anesthesiology 124, 934–944 (2016).
Zhang, L. et al. Distinct analgesic actions of DHA and DHA-derived specialized pro-resolving mediators on post-operative pain after bone fracture in mice. Front. Pharmacol. 9, 412 (2018).
Wang, J. C. & Strichartz, G. R. Prevention of chronic post-thoracotomy pain in rats by intrathecal resolvin D1 and D2: effectiveness of perioperative and delayed drug delivery. J. Pain 18, 535–545 (2017).
Huang, L., Wang, C. F., Serhan, C. N. & Strichartz, G. Enduring prevention and transient reduction of postoperative pain by intrathecal resolvin D1. Pain 152, 557–565 (2011).
Klein, C. P. et al. Effects of D-series resolvins on behavioral and neurochemical changes in a fibromyalgia-like model in mice. Neuropharmacology 86, 57–66 (2014).
Bang, S. et al. Resolvin D1 attenuates activation of sensory transient receptor potential channels leading to multiple anti-nociception. Br. J. Pharmacol. 161, 707–720 (2010).
Park, C. K. et al. Resolvin D2 is a potent endogenous inhibitor for transient receptor potential subtype V1/A1, inflammatory pain, and spinal cord synaptic plasticity in mice: distinct roles of resolvin D1, D2, and E1. J. Neurosci. 31, 18433–18438 (2011).
Tao, X., Lee, M. S., Donnelly, C. R. & Ji, R. R. Neuromodulation, specialized proresolving mediators, and resolution of pain. Neurotherapeutics 17, 886–899 (2020).
Serhan, C. N., de la Rosa, X. & Jouvene, C. Novel mediators and mechanisms in the resolution of infectious inflammation: evidence for vagus regulation. J. Intern. Med. 286, 240–258 (2019).
Tao, X. et al. Spinal Cord stimulation attenuates mechanical allodynia and increases central resolvin D1 levels in rats with spared nerve injury. Front. Physiol. 12, 687046 (2021).
Bang, S. et al. Activation of GPR37 in macrophages confers protection against infection-induced sepsis and pain-like behaviour in mice. Nat. Commun. 12, 1704 (2021).
Xu, Z. Z. et al. Neuroprotectin/protectin D1 protects against neuropathic pain in mice after nerve trauma. Ann. Neurol. 74, 490–495 (2013).
Park, C. K. et al. Resolving TRPV1- and TNF-α-mediated spinal cord synaptic plasticity and inflammatory pain with neuroprotectin D1. J. Neurosci. 31, 15072–15085 (2011).
Nesman, J. I. et al. A new synthetic protectin D1 analog 3-oxa-PD1n-3 DPA reduces neuropathic pain and chronic itch in mice. Org. Biomol. Chem. 19, 2744–2752 (2021).
Pham, T. L. & Bazan, H. E. P. Docosanoid signaling modulates corneal nerve regeneration: effect on tear secretion, wound healing, and neuropathic pain. J. Lipid Res. 62, 100033 (2021).
Pham, T. L. et al. Defining a mechanistic link between pigment epithelium-derived factor, docosahexaenoic acid, and corneal nerve regeneration. J. Biol. Chem. 292, 18486–18499 (2017).
Chiang, N., Libreros, S., Norris, P. C., de la Rosa, X. & Serhan, C. N. Maresin 1 activates LGR6 receptor promoting phagocyte immunoresolvent functions. J. Clin. Invest. 129, 5294–5311 (2019).
Serhan, C. N. et al. Macrophage proresolving mediator maresin 1 stimulates tissue regeneration and controls pain. FASEB J. 26, 1755–1765 (2012).
Fattori, V. et al. The specialised pro-resolving lipid mediator maresin 1 reduces inflammatory pain with a long-lasting analgesic effect. Br. J. Pharmacol. 176, 1728–1744 (2019).
Gao, J. et al. Pro-resolving mediator maresin 1 ameliorates pain hypersensitivity in a rat spinal nerve ligation model of neuropathic pain. J. Pain Res. 11, 1511–1519 (2018).
Wang, Y. H. et al. Maresin 1 attenuates radicular pain through the inhibition of NLRP3 inflammasome-induced pyroptosis via NF-κB signaling. Front. Neurosci. 14, 831 (2020).
Francos-Quijorna, I. et al. Maresin 1 promotes inflammatory resolution, neuroprotection, and functional neurological recovery after spinal cord injury. J. Neurosci. 37, 11731–11743 (2017).
Herová, M., Schmid, M., Gemperle, C. & Hersberger, M. ChemR23, the receptor for chemerin and resolvin E1, is expressed and functional on M1 but not on M2 macrophages. J. Immunol. 194, 2330–2337 (2015).
Tiberi, M. & Chiurchiu, V. Specialized pro-resolving lipid mediators and glial cells: emerging candidates for brain homeostasis and repair. Front. Cell. Neurosci. 15, 673549 (2021).
Arita, M. et al. Resolvin E1 selectively interacts with leukotriene B4 receptor BLT1 and ChemR23 to regulate inflammation. J. Immunol. 178, 3912–3917 (2007).
Xu, Z. Z., Berta, T. & Ji, R. R. Resolvin E1 inhibits neuropathic pain and spinal cord microglial activation following peripheral nerve injury. J. Neuroimmune Pharmacol. 8, 37–41 (2013).
Xu, Z. Z. et al. Resolvins RvE1 and RvD1 attenuate inflammatory pain via central and peripheral actions. Nat. Med. 16, 592–597 (2010).
Jo, Y. Y., Lee, J. Y. & Park, C. K. Resolvin E1 inhibits substance P-induced potentiation of TRPV1 in primary sensory neurons. Mediators Inflamm. 2016, 5259321 (2016).
Bang, S., Yoo, S., Oh, U. & Hwang, S. W. Endogenous lipid-derived ligands for sensory TRP ion channels and their pain modulation. Arch. Pharm. Res. 33, 1509–1520 (2010).
Roh, J., Go, E. J., Park, J. W., Kim, Y. H. & Park, C. K. Resolvins: potent pain inhibiting lipid mediators via transient receptor potential regulation. Front. Cell. Dev. Biol. 8, 584206 (2020).
Nolan, K. & Godson, C. in Encyclopedia of Signaling Molecules (ed. Choi, S.) 1854–1862 (Springer, 2018).
Liu, G. J. et al. Functions of resolvin D1-ALX/FPR2 receptor interaction in the hemoglobin-induced microglial inflammatory response and neuronal injury. J. Neuroinflammation 17, 239 (2020).
Cooray, S. N. et al. Ligand-specific conformational change of the G-protein–coupled receptor ALX/FPR2 determines proresolving functional responses. Proc. Natl Acad. Sci. USA 110, 18232–18237 (2013).
Maciuszek, M., Cacace, A., Brennan, E., Godson, C. & Chapman, T. M. Recent advances in the design and development of formyl peptide receptor 2 (FPR2/ALX) agonists as pro-resolving agents with diverse therapeutic potential. Eur. J. Med. Chem. 213, 113167 (2021).
Sun, T. et al. Lipoxin A4 induced antinociception and decreased expression of NF-κB and pro-inflammatory cytokines after chronic dorsal root ganglia compression in rats. Eur. J. Pain 16, 18–27 (2012).
Martini, A. C. et al. Lipoxin A4 inhibits microglial activation and reduces neuroinflammation and neuropathic pain after spinal cord hemisection. J. Neuroinflammation 13, 75 (2016).
Svensson, C. I., Zattoni, M. & Serhan, C. N. Lipoxins and aspirin-triggered lipoxin inhibit inflammatory pain processing. J. Exp. Med. 204, 245–252 (2007).
Miao, G. S. et al. Lipoxin A4 attenuates radicular pain possibly by inhibiting spinal ERK, JNK and NF-κB/p65 and cytokine signals, but not p38, in a rat model of non-compressive lumbar disc herniation. Neuroscience 300, 10–18 (2015).
Wang, Z. F. et al. Aspirin-triggered lipoxin A4 attenuates mechanical allodynia in association with inhibiting spinal JAK2/STAT3 signaling in neuropathic pain in rats. Neuroscience 273, 65–78 (2014).
Li, Q. et al. Involvement of the spinal NALP1 inflammasome in neuropathic pain and aspirin-triggered-15-epi-lipoxin A4 induced analgesia. Neuroscience 254, 230–240 (2013).
Liu, Z. Q., Zhang, H. B., Wang, J., Xia, L. J. & Zhang, W. Lipoxin A4 ameliorates ischemia/reperfusion induced spinal cord injury in rabbit model. Int. J. Clin. Exp. Med. 8, 12826–12833 (2015).
Finnerup, N. B. et al. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol. 14, 162–173 (2015).
Veluchamy, A., Hébert, H. L., Meng, W., Palmer, C. N. A. & Smith, B. H. Systematic review and meta-analysis of genetic risk factors for neuropathic pain. Pain 159, 825–848 (2018).
Albrecht, D. S. et al. Brain glial activation in fibromyalgia — a multi-site positron emission tomography investigation. Brain Behav. Immun. 75, 72–83 (2019).
Loggia, M. L. et al. Evidence for brain glial activation in chronic pain patients. Brain 138, 604–615 (2015).
Gravius, N. et al. Selective L4 dorsal root ganglion stimulation evokes pain relief and changes of inflammatory markers: part I, profiling of saliva and serum molecular patterns. Neuromodulation 22, 44–52 (2019).
Kinfe, T. M. et al. Unilateral L4-dorsal root ganglion stimulation evokes pain relief in chronic neuropathic postsurgical knee pain and changes of inflammatory markers: part II, whole transcriptome profiling. J. Transl. Med. 17, 205 (2019).
Kriek, N. et al. Spinal cord stimulation in patients with complex regional pain syndrome: a possible target for immunomodulation. Neuromodulation 21, 77–86 (2018).
Das, B., Conroy, M., Moore, D., Lysaght, J. & McCrory, C. Human dorsal root ganglion pulsed radiofrequency treatment modulates cerebrospinal fluid lymphocytes and neuroinflammatory markers in chronic radicular pain. Brain Behav. Immun. 70, 157–165 (2018).
Moore, D. et al. Characterisation of the effects of pulsed radio frequency treatment of the dorsal root ganglion on cerebrospinal fluid cellular and peptide constituents in patients with chronic radicular pain: a randomised, triple-blinded, controlled trial. J. Neuroimmunol. 343, 577219 (2020).
Chatterjee, P. et al. Effect of deep tissue laser therapy treatment on peripheral neuropathic pain in older adults with type 2 diabetes: a pilot randomized clinical trial. BMC Geriatr. 19, 218 (2019).
Choi, E. M. et al. Efficacy of intermittent epidural dexamethasone bolus for zoster-associated pain beyond the acute phase. Int. J. Med. Sci. 17, 1811–1818 (2020).
Kikuchi, A. et al. Comparative therapeutic evaluation of intrathecal versus epidural methylprednisolone for long-term analgesia in patients with intractable postherpetic neuralgia. Reg. Anesth. Pain Med. 24, 287–293 (1999).
Bayry, J. et al. Mechanisms of action of intravenous immunoglobulin in autoimmune and inflammatory diseases. Transfus. Clin. Biol. 10, 165–169 (2003).
Hartung, H. P. et al. Patient-reported outcomes with subcutaneous immunoglobulin in chronic inflammatory demyelinating polyneuropathy: the PATH study. Eur. J. Neurol. 27, 196–203 (2020).
Huang, Y. H. et al. Intravenous immunoglobulin for postpolio syndrome: a systematic review and meta-analysis. BMC Neurol. 15, 39 (2015).
Sumitani, M. et al. Minocycline does not decrease intensity of neuropathic pain, but does improve its affective dimension. J. Pain Palliat. Care Pharmacother. 30, 31–35 (2016).
Nishida, T. et al. Involvement of high mobility group Box 1 in the development and maintenance of chemotherapy-induced peripheral neuropathy in rats. Toxicology 365, 48–58 (2016).
Kotaka, M. et al. A placebo-controlled, double-blind, randomized study of recombinant thrombomodulin (ART-123) to prevent oxaliplatin-induced peripheral neuropathy. Cancer Chemother. Pharmacol. 86, 607–618 (2020).
Araki, M. et al. Efficacy of the anti-IL-6 receptor antibody tocilizumab in neuromyelitis optica: a pilot study. Neurology 82, 1302–1306 (2014).
Ohtori, S. et al. Epidural administration of spinal nerves with the tumor necrosis factor-α inhibitor, etanercept, compared with dexamethasone for treatment of sciatica in patients with lumbar spinal stenosis: a prospective randomized study. Spine 37, 439–444 (2012).
Cohen, S. P. et al. Randomized, double-blind, placebo-controlled, dose-response, and preclinical safety study of transforaminal epidural etanercept for the treatment of sciatica. Anesthesiology 110, 1116–1126 (2009).
Ohtori, S. et al. Efficacy of epidural administration of anti-interleukin-6 receptor antibody onto spinal nerve for treatment of sciatica. Eur. Spine J. 21, 2079–2084 (2012).
Genevay, S. et al. Adalimumab in severe and acute sciatica: a multicenter, randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 62, 2339–2346 (2010).
Genevay, S. et al. Adalimumab in acute sciatica reduces the long-term need for surgery: a 3-year follow-up of a randomised double-blind placebo-controlled trial. Ann. Rheum. Dis. 71, 560–562 (2012).
Karppinen, J. et al. Tumor necrosis factor-α monoclonal antibody, infliximab, used to manage severe sciatica. Spine 28, 750–754 (2003).
Williams, N. H. et al. A systematic review and meta-analysis of biological treatments targeting tumour necrosis factor α for sciatica. Eur. Spine J. 22, 1921–1935 (2013).
Watkins, L. R. et al. Targeted interleukin-10 plasmid DNA therapy in the treatment of osteoarthritis: toxicology and pain efficacy assessments. Brain Behav. Immun. 90, 155–166 (2020).
Rosenzwajg, M. et al. Immunological and clinical effects of low-dose interleukin-2 across 11 autoimmune diseases in a single, open clinical trial. Ann. Rheum. Dis. 78, 209–217 (2019).
Duffy, S. S., Keating, B. A., Perera, C. J. & Moalem-Taylor, G. The role of regulatory T cells in nervous system pathologies. J. Neurosci. Res. 96, 951–968 (2018).
Qu, G. et al. Current status and perspectives of regulatory T cell-based therapy. J. Genet. Genom. 49, 599–611 (2022).
Chernykh, E. R. et al. Safety and therapeutic potential of M2 macrophages in stroke treatment. Cell Transpl. 25, 1461–1471 (2016).
Mia, S., Warnecke, A., Zhang, X. M., Malmström, V. & Harris, R. A. An optimized protocol for human M2 macrophages using M-CSF and IL-4/IL-10/TGF-β yields a dominant immunosuppressive phenotype. Scand. J. Immunol. 79, 305–314 (2014).
Miller, J. S. et al. Successful adoptive transfer and in vivo expansion of human haploidentical NK cells in patients with cancer. Blood 105, 3051–3057 (2005).
Lin, B. et al. Gut microbiota regulates neuropathic pain: potential mechanisms and therapeutic strategy. J. Headache Pain 21, 103 (2020).
Pimentel, M. et al. Effects of rifaximin treatment and retreatment in nonconstipated IBS subjects. Dig. Dis. Sci. 56, 2067–2072 (2011).
Sorbara, M. T. & Pamer, E. G. Microbiome-based therapeutics. Nat. Rev. Microbiol. 20, 365–380 (2022).
Hungin, A. P. S. et al. Systematic review: probiotics in the management of lower gastrointestinal symptoms — an updated evidence-based international consensus. Aliment. Pharmacol. Ther. 47, 1054–1070 (2018).
Mandel, D. R., Eichas, K. & Holmes, J. Bacillus coagulans: a viable adjunct therapy for relieving symptoms of rheumatoid arthritis according to a randomized, controlled trial. BMC Complement. Altern. Med. 10, 1 (2010).
Vaghef-Mehrabany, E. et al. Probiotic supplementation improves inflammatory status in patients with rheumatoid arthritis. Nutrition 30, 430–435 (2014).
Zhou, S. Y. et al. FODMAP diet modulates visceral nociception by lipopolysaccharide-mediated intestinal inflammation and barrier dysfunction. J. Clin. Invest. 128, 267–280 (2018).
Singh, P. et al. High FODMAP diet causes barrier loss via lipopolysaccharide-mediated mast cell activation. JCI Insight https://doi.org/10.1172/jci.insight.146529 (2021).
Allison, D. J., Thomas, A., Beaudry, K. & Ditor, D. S. Targeting inflammation as a treatment modality for neuropathic pain in spinal cord injury: a randomized clinical trial. J. Neuroinflammation 13, 152 (2016).
Casini, I. et al. Food-specific IgG4 antibody-guided exclusion diet improves conditions of patients with chronic pain. Pain Ther. 11, 873–906 (2022).
Vendrik, K. E. W. et al. Fecal microbiota transplantation in neurological disorders. Front. Cell Infect. Microbiol. 10, 98 (2020).
DeFilipp, Z. et al. Drug-resistant E. coli bacteremia transmitted by fecal microbiota transplant. N. Engl. J. Med. 381, 2043–2050 (2019).
Thurm, T., Ablin, J., Buskila, D. & Maharshak, N. Fecal microbiota transplantation for fibromyalgia: a case report and review of the literature. Open J. Gastroenterol. 7, 131–139 (2017).
Schebb, N. H. et al. Formation, signaling and occurrence of specialized pro-resolving lipid mediators-what is the evidence so far? Front. Pharmacol. 13, 838782 (2022).
Duran, A. M. et al. Effects of omega-3 polyunsaturated fatty-acid supplementation on neuropathic pain symptoms and sphingosine levels in Mexican-Americans with type 2 diabetes. Diabetes Metab. Syndr. Obes. 12, 109–120 (2019).
Duran, A. M., Beeson, W. L., Firek, A., Cordero-MacIntyre, Z. & De Leon, M. Dietary omega-3 polyunsaturated fatty-acid supplementation upregulates protective cellular pathways in patients with type 2 diabetes exhibiting improvement in painful diabetic neuropathy. Nutrients 14, 761 (2022).
Ko, G. D., Nowacki, N. B., Arseneau, L., Eitel, M. & Hum, A. Omega-3 fatty acids for neuropathic pain: case series. Clin. J. Pain 26, 168–172 (2010).
Barden, A. E. et al. Specialised pro-resolving mediators of inflammation in inflammatory arthritis. Prostaglandins Leukot. Essent. Fat. Acids 107, 24–29 (2016).
Ramsden, C. E. et al. Targeted alterations in dietary n-3 and n-6 fatty acids improve life functioning and reduce psychological distress among patients with chronic headache: a secondary analysis of a randomized trial. Pain 156, 587–596 (2015).
Tajmirriahi, M. et al. The effects of sodium valproate with fish oil supplementation or alone in migraine prevention: a randomized single-blind clinical trial. Iran. J. Neurol. 11, 21–24 (2012).
Levine, J. D., Gooding, J., Donatoni, P., Borden, L. & Goetzl, E. J. The role of the polymorphonuclear leukocyte in hyperalgesia. J. Neurosci. 5, 3025–3029 (1985).
Perkins, N. M. & Tracey, D. J. Hyperalgesia due to nerve injury: role of neutrophils. Neuroscience 101, 745–757 (2000).
Zuo, Y., Perkins, N. M., Tracey, D. J. & Geczy, C. L. Inflammation and hyperalgesia induced by nerve injury in the rat: a key role of mast cells. Pain 105, 467–479 (2003).
Carreira, E. U. et al. Neutrophils recruited by CXCR1/2 signalling mediate post-incisional pain. Eur. J. Pain 17, 654–663 (2013).
Harada, Y. et al. Cathepsin E in neutrophils contributes to the generation of neuropathic pain in experimental autoimmune encephalomyelitis. Pain 160, 2050–2062 (2019).
Caxaria, S. B. et al. Neutrophils infiltrate sensory ganglia and mediate chronic 1 widespread pain in fibromyalgia. Preprint at bioRxiv https://doi.org/10.1101/2022.06.29.498149 (2022).
Morin, N. et al. Neutrophils invade lumbar dorsal root ganglia after chronic constriction injury of the sciatic nerve. J. Neuroimmunol. 184, 164–171 (2007).
Kim, C. F. & Moalem-Taylor, G. Detailed characterization of neuro-immune responses following neuropathic injury in mice. Brain Res. 1405, 95–108 (2011).
Lindborg, J. A., Mack, M. & Zigmond, R. E. Neutrophils are critical for myelin removal in a peripheral nerve injury model of Wallerian degeneration. J. Neurosci. 37, 10258–10277 (2017).
Cui, J. G., Holmin, S., Mathiesen, T., Meyerson, B. A. & Linderoth, B. Possible role of inflammatory mediators in tactile hypersensitivity in rat models of mononeuropathy. Pain 88, 239–248 (2000).
Vivier, E., Tomasello, E., Baratin, M., Walzer, T. & Ugolini, S. Functions of natural killer cells. Nat. Immunol. 9, 503–510 (2008).
Shinotsuka, N. & Denk, F. Fibroblasts: the neglected cell type in peripheral sensitisation and chronic pain? A review based on a systematic search of the literature. BMJ Open Sci. 6, e100235 (2022).
Singhmar, P. et al. The fibroblast-derived protein PI16 controls neuropathic pain. Proc. Natl Acad. Sci. USA 117, 5463–5471 (2020).
Silva, C. E. A., Guimaraes, R. M. & Cunha, T. M. Sensory neuron-associated macrophages as novel modulators of neuropathic pain. Pain Rep. 6, e873 (2021).
Yu, X. et al. Dorsal root ganglion macrophages contribute to both the initiation and persistence of neuropathic pain. Nat. Commun. 11, 264 (2020).
Msheik, Z., El Massry, M., Rovini, A., Billet, F. & Desmouliere, A. The macrophage: a key player in the pathophysiology of peripheral neuropathies. J. Neuroinflammation 19, 97 (2022).
Inoue, K. & Tsuda, M. Microglia in neuropathic pain: cellular and molecular mechanisms and therapeutic potential. Nat. Rev. Neurosci. 19, 138–152 (2018).
Tsuda, M. et al. P2X4 receptors induced in spinal microglia gate tactile allodynia after nerve injury. Nature 424, 778–783 (2003).
Butovsky, O. et al. Identification of a unique TGF-β-dependent molecular and functional signature in microglia. Nat. Neurosci. 17, 131–143 (2014).
Tansley, S. et al. Microglia-mediated degradation of perineuronal nets promotes pain. Science 377, 80–86 (2022).
Kobayashi, Y. et al. Macrophage-T cell interactions mediate neuropathic pain through the glucocorticoid-induced tumor necrosis factor ligand system. J. Biol. Chem. 290, 12603–12613 (2015).
Draleau, K. et al. Phenotypic identification of spinal cord-infiltrating CD4+ T Lymphocytes in a murine model of neuropathic pain. J. Pain Relief 2014, 003 (2014).
Jaggi, A. S., Jain, V. & Singh, N. Animal models of neuropathic pain. Fundam. Clin. Pharmacol. 25, 1–28 (2011).
Serhan, C. N. & Levy, B. D. Resolvins in inflammation: emergence of the pro-resolving superfamily of mediators. J. Clin. Invest. 128, 2657–2669 (2018).
Chen, G., Zhang, Y. Q., Qadri, Y. J., Serhan, C. N. & Ji, R. R. Microglia in pain: detrimental and protective roles in pathogenesis and resolution of pain. Neuron 100, 1292–1311 (2018).
Serhan, C. N., Chiang, N. & Dalli, J. The resolution code of acute inflammation: novel pro-resolving lipid mediators in resolution. Semin. Immunol. 27, 200–215 (2015).
Serhan, C. N. Lipoxins and aspirin-triggered 15-epi-lipoxins are the first lipid mediators of endogenous anti-inflammation and resolution. Prostaglandins Leukot. Essent. Fat. Acids 73, 141–162 (2005).
Buckley, C. D., Gilroy, D. W., Serhan, C. N., Stockinger, B. & Tak, P. P. The resolution of inflammation. Nat. Rev. Immunol. 13, 59–66 (2013).
Serhan, C. N. et al. Resolution of inflammation: state of the art, definitions and terms. FASEB J. 21, 325–332 (2007).
Abboud, C. et al. Animal models of pain: diversity and benefits. J. Neurosci. Methods 348, 108997 (2021).
Challa, S. R. Surgical animal models of neuropathic pain: pros and cons. Int. J. Neurosci. 125, 170–174 (2015).
Khan, N. & Smith, M. T. Multiple sclerosis-induced neuropathic pain: pharmacological management and pathophysiological insights from rodent EAE models. Inflammopharmacology 22, 1–22 (2014).
Liu, Y. et al. Animal models of complex regional pain syndrome type I. J. Pain Res. 14, 3711–3721 (2021).
Mirabelli, E. & Elkabes, S. Neuropathic pain in multiple sclerosis and its animal models: focus on mechanisms, knowledge gaps and future directions. Front. Neurol. 12, 793745 (2021).
Yanow, J., Pappagallo, M. & Pillai, L. Complex regional pain syndrome (CRPS/RSD) and neuropathic pain: role of intravenous bisphosphonates as analgesics. Sci. World J. 8, 229–236 (2008).
Bouali-Benazzouz, R., Landry, M., Benazzouz, A. & Fossat, P. Neuropathic pain modeling: focus on synaptic and ion channel mechanisms. Prog. Neurobiol. 201, 102030 (2021).
Brum, E. S., Becker, G., Fialho, M. F. P. & Oliveira, S. M. Animal models of fibromyalgia: what is the best choice? Pharmacol. Ther. 230, 107959 (2022).
Matsuda, M., Huh, Y. & Ji, R. R. Roles of inflammation, neurogenic inflammation, and neuroinflammation in pain. J. Anesth. 33, 131–139 (2019).
Basbaum, A. I., Bautista, D. M., Scherrer, G. & Julius, D. Cellular and molecular mechanisms of pain. Cell 139, 267–284 (2009).
Acknowledgements
The authors gratefully acknowledge support from the National Health and Medical Research Council (NHMRC) of Australia (ID APP1162060 and ID APP1187416) awarded to G.M.-T. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. The authors thank Timothy Lee for providing professional medical illustration services used to generate the original figures.
Review criteria
The authors searched PubMed and Google Scholar for original research articles and reviews published in English, using the keywords or search terms “neuropathic pain” or “pain resolution” and “neutrophils”, “natural killer cells’, “fibroblasts”, “microglia”, “T cells’, “B cells”, “gut microbiota”, “microbiome”, “specialized pro-resolving mediators”, “animal models”, “inflammation”, “inflammatory response” and “immune response”. Relevant studies on other pain conditions were also included. CENTRAL, MEDLINE, EMBASE and ClinicalTrials.gov were searched for additional relevant clinical articles, using the following search terms and keywords: “inflammation” or “immune” and “pain” and “neuropathic” or “chronic”. Additional relevant articles were selected from the reference lists of publications identified in these searches. The final reference list was generated based on relevance to the topics covered, focusing on publications within the past 10 years.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Neurology thanks Claudia Sommer, who co-reviewed with Patricia García Fernández; Francesca Guida; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fiore, N.T., Debs, S.R., Hayes, J.P. et al. Pain-resolving immune mechanisms in neuropathic pain. Nat Rev Neurol 19, 199–220 (2023). https://doi.org/10.1038/s41582-023-00777-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41582-023-00777-3
This article is cited by
-
SAFit2 ameliorates paclitaxel-induced neuropathic pain by reducing spinal gliosis and elevating pro-resolving lipid mediators
Journal of Neuroinflammation (2023)
-
AQP4 is an Emerging Regulator of Pathological Pain: A Narrative Review
Cellular and Molecular Neurobiology (2023)