Abstract
Prospective birth cohorts offer unprecedented opportunities to investigate the pathogenesis of complex disorders such as autism, in which gene–environment interactions must be appreciated in a temporal context. This Perspective article considers the history of autism research, including missteps that reflected an incomplete understanding of the epidemiology of autistic spectrum disorders, the effects of advocacy and philanthropy on the trajectory of scientific inquiry, and the current and future roles of prospective birth cohort research in illuminating the pathology of these and other complex disorders wherein exposures during gestation might not manifest until later in life.
Similar content being viewed by others
Introduction
Autism spectrum disorders (ASDs) are characterized by impairments in social skills and communication and the presence of repetitive behaviours. Affecting approximately 1 in 50 children, ASDs are four times more common in boys than in girls1. As there are no objective laboratory tests for ASDs, diagnosis requires expert clinical assessment. This situation impedes early identification of ASDs at a time when the brain is highly plastic and amenable to positive reinforcement exercises that can mitigate their presentation2. The social and economic burdens of ASD for those with these disorders, their families and their communities are lifelong and substantial. Accordingly, understanding the pathophysiology of ASD is an urgent unmet clinical need, as are developing methods for early diagnosis and treatment and ways to support those with ASD as they navigate their life course. Figure 1 shows a timeline of seminal events in the history of biomedical research in autism.
Although initially characterized as psychosis74,75,76, from the 1970s onwards autism was reframed as a developmental disorder with a heritable component15,77,78,79,80,81. Various exposures were subsequently identified as contributing to the risk of autism spectrum disorder, the understanding of which continues to evolve5,16,17,19,20,25,82,83,84,85,86,87,88,89. Special projects initiated and supported by The National Alliance for Autism Research and Cure Autism Now include the Autism Tissue Program brain bank, the Autism Genetic Resource Exchange and the High-Risk Baby Siblings Consortium.
In this Perspective article, we consider how cohort studies have contributed to our understanding of the epidemiology and pathogenesis of ASDs, place cohort research into the context of complementary work conducted in animal models, and speculate on the promise and potential challenges of cohort research for dissecting the causes of ASDs and developing interventional strategies for these and other chronic disorders.
Changes in reported prevalence
The public perception of autism and its pathogenesis has been strongly affected by changes in its reported prevalence. In the 1960s and 1970s, worldwide autism prevalence estimates ranged from 0.04 to 2.00 cases per 1,000 of the population3. These rates rose tenfold in the 1980s and 1990s. A lack of clarity on the basis for these prevalence estimates prompted a search for environmental factors that had been introduced into paediatric care at around the time of this increase. Two came to the fore: vaccines and acetaminophen (paracetamol).
The measles, mumps and rubella (MMR) vaccine, which contains a mixture of three live attenuated viruses, was licensed in 1971. This vaccine is administered in two doses, the first given at 9–15 months of age and the second between 15 months and 6 years of age. Killed bacterial vaccines, including diphtheria toxin, pertussis bacteria and tetanus toxin (DPT vaccine), that were first introduced in the 1950s also came to attention because they contained the preservative thimerosal (ethyl mercury), which raised the concern that even the low levels of mercury present in DPT vaccine doses (which were given at 2 months, 4 months, 6 months, 15–18 months and 4–6 years of age) might be neurotoxic. In 1980, epidemiologists at the US Centers for Disease Control reported a link between the use of salicylates for fever management in paediatric patients and Reye syndrome, a fatal hepatic encephalopathy4, which led to the substitution of acetaminophen for salicylates. In each of these instances, temporal associations between the introduction of a vaccine or treatment and the apparent increase in the prevalence of autism (a condition that was only rarely diagnosed before the age of 2 years) led to popular concerns that there was a causal link. No causal relationships have been found, despite intensive investigation. Nonetheless, spurious links between ASDs and the MMR vaccine5 or the vaccine preservative thimerosal6 continue to have a profound effect on public acceptance and uptake of MMR and DPT7,8 — as well as other vaccines, including polio and SARS-CoV-2 vaccines9.
Prevalence is very challenging to estimate accurately for complex syndromes such as autism, in which changes over time in diagnostic criteria and in access to specialists with the expertise required to diagnose the condition can result in large changes in case ascertainment that mimic or obscure bona fide changes in prevalence. One analysis attributed 33% of the increase in reported ASD prevalence among children born in Denmark during the period 1980–1991 to changes in diagnostic criteria alone, 42% to increased case ascertainment alone and 60% to the combination of changes in diagnostic criteria and increased case ascertainment10. This analysis not only suggested that such artefacts played an important role in the increased prevalence of ASDs but also confirmed that a substantial part of the increase could not be explained by them.
Philanthropy and parental advocacy
It is difficult to overstate the importance of philanthropy and parental advocacy in the course of autism research and treatment (Fig. 1). An influential early advocate was Bernard Rimland, a clinical psychologist and father of a son with autism, who rejected the psychogenic hypothesis. Rimland founded the Autism Society of America, the Autism Research Institute and the Defeat Autism Now Foundation. He also served as the technical adviser on the film Rain Man, in which Dustin Hoffman’s portrayal of a man with Asperger syndrome brought attention to ASDs. Cure Autism Now and the National Alliance for Autism Research were private foundations initiated by parents to support peer-reviewed research projects and physician training programmes. Each foundation recruited strong scientific advisory boards and funded investigators who would not otherwise have had the resources to begin autism research. Cure Autism Now also focused on the collection of clinical materials to enable genetic analyses (Autism Genetic Research Exchange). These two organizations were subsequently assimilated into the Autism Speaks Foundation established by the Chief Executive Officer of NBC Universal, grandparent of an affected child, who leveraged his considerable influence to promote funding for autism research and treatment programmes. Amongst those programmes is the Baby Sibs Research Consortium, which was founded in 2003 to expedite the discovery of insights into the diagnosis and biology of ASDs by conducting prospective studies of the siblings of children with autism (who have a 20-fold increased risk of themselves receiving a diagnosis of ASD). The largest organization to date is the Simons Foundation Autism Research Initiative (SFARI), which was established by mathematicians Jim and Marilyn Simons to support basic and clinical research into the neuroscience of autism. This organization has provided more than US $200 million to more than 150 investigators worldwide since 2007, including funding to study the immunopathology of disease in the Norwegian Autism Birth Cohort (ABC).
Prospective birth cohorts
The pathogenesis of ASD in most individuals is unknown. The first description of autism as a rare disorder in 1943 suggested a role for socially distant caregivers — the ‘refrigerator parent’ hypothesis11. Although not unchallenged, this model remained dominant until the 1970s, when an increased risk of autism was reported in children with congenital rubella12 and other researchers reported finding evidence of heritability in a study of monozygotic and dizygotic twin pairs in which at least one twin had autism13. In this study, the concordance rate for autism was 36% in monozygotic twins, whereas none of the dizygotic twins were concordant for this disease13. For comparison purposes, a meta-analysis published in 2016 found heritability estimates for ASDs ranging from 38% to 90%14. The reasons for the variation in heritability estimates remain unclear.
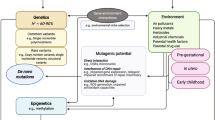
This substantial heritability has sometimes been misinterpreted to imply that environmental contributions to ASDs are minimal. However, these data are also consistent with genetic vulnerability to shared environmental exposures. This distinction can be illustrated by comparison with phenylketonuria, in which virtually all affected individuals have the genetic cause but the disease manifests only in response to a specific (but ubiquitous) nutritional exposure that can, however, be eliminated from the diet. Thus, the heritability of phenylketonuria approaches 100% and the phenotype is ubiquitous without intervention, and abrogating exposure to a specific environmental factor is key to its prevention. Therefore, the existence of seminal papers suggesting contributions of genetic, nutritional, immunological and toxic environmental factors to ASDs15,16,17,18,19,20 does not eliminate the social environment as having a critical role in neural development. Importantly, 50–70% of children with ASD have comorbid attention deficit hyperactivity disorder (ADHD)21, and Romanian infants confined to institutions early in life who had only minimal contact with caregivers developed behaviours consistent with ADHD22. Epigenetic analyses indicate that the ADHD-like behaviours in these children might be associated with altered DNA methylation patterns versus those of children raised in family settings23. Of note, genetic and environmental models of ASD pathogenesis are not mutually exclusive. Specific genetic defects, such as those that occur in patients with fragile X syndrome or are caused by gestational exposure to antiepileptic medications or thalidomide, are sufficient to induce behaviours associated with autism. In the majority of patients with ASDs, no links to a specific genetic or environmental factor can be identified. However, genetic vulnerability and environmental triggers almost certainly act together to cause ASD in some individuals.
The observation that maternal use of anticonvulsants24 or thalidomide19 and rubella infection12,25 during pregnancy are all associated with an increased risk of autism suggested the presence of windows of developmental vulnerability to ASDs prior to birth. Rigorous testing of this hypothesis requires the recruitment of population-based birth cohorts in which biological samples and environmental exposure data are collected at multiple time points during gestation. These early data then need to be combined with data on development, social determinants and phenotypes collected during gestation and across childhood. Taken together, birth cohorts can (at least in theory) be used to analyse omics data and environmental exposures, and determine which developmentally sensitive periods are most relevant. Some birth cohorts include almost all individuals with ASDs among population samples of >100,000 individuals and enable longitudinal follow-up of the children with ASDs. These cohorts can be used to analyse the relationships between genomic findings, environmental exposures and sensitive developmental periods. All existing cohorts, however, have limitations. The ABC (nested within the Norwegian Mother, Father and Child Cohort Study (MoBA)) is arguably the largest such cohort and has most, although not all, of the features required for such analyses. Yet it is still subject to selection bias; only 40% of the pregnant women approached chose to participate.
Although population-based birth cohorts are potentially the most informative, other cohort designs (for example, those focused on children resulting from subsequent pregnancies in mothers of a child with ASD) can provide complementary data. Table 1 presents one exemplar for each of six types of cohort study, and notes the typical features of each design as used in ASD research26,27,28,29,30. Given the variety of characteristics among cohort studies that share the same basic design and the large number of cohort studies now established, ‘typical’ as used here does not imply uniformity. Presentation of all current ASD cohorts and in-depth analysis of the differences between them are beyond the scope of this paper. Accordingly, we focus on how these six cohort designs offer complementary data with respect to three gestational exposures linked to ASD: maternal folate intake, maternal immune response and preterm birth.
Folate and folic acid
The ABC study was the first to show specifically that maternal folic acid supplementation is associated with a reduced risk of language delay31 and ASD in offspring17. Although these initial findings were based on early data in a subset of individuals, the same associations have now been found in subsequent larger studies, in both this and other cohorts32. A study conducted in California had previously suggested that increased maternal folate intake overall was associated with a reduced risk of ASD in offspring33. However, the Norwegian study was distinctive in several ways. Perhaps the most important is that, unlike many countries (including the USA and UK), Norway does not fortify foods with folic acid. Accordingly, the folate levels of women who did and did not take supplements during gestation were markedly different. A related strength of the ABC study is that supplementation usually involved small doses, such as 400 μg of folic acid included in a multivitamin pill. This factor is relevant because some studies suggest that high levels of synthetic folic acid (as opposed to natural folate in foods) might have adverse effects34,35. Further analysis of data from a study of baby siblings of children with an ASD, which examined the effect of folic acid supplementation on the risk of ASD recurrence in high-risk families, showed that supplementation in the first month of pregnancy halved the risk of autism36. Complementary evidence has also been provided by registry studies that demonstrates an increased risk of ASD associated with short interpregnancy intervals37,38. One potential explanation for this finding is that folate depleted during pregnancy might not be fully restored by the start of a subsequent pregnancy.
Folate is fundamental to brain development. For example, folate is a critical donor of the methyl groups used in DNA and histone methylation. Although early gestation and/or periconception have been identified as sensitive periods with regard to the effects of folic acid supplementation, the epigenetic activity of folate might extend the period of folate-dependent vulnerability to ASD. Animal studies suggest that maternal early life exposure to either low or high levels of folate, including the mother’s own gestational exposure (which is when her oocytes are developing) as well as paternal lifetime exposure to folic acid deficiency and supplementation, all pose a theoretical risk to offspring by altering DNA and histone methylation patterns39,40. The findings of several birth cohort studies that the characteristics of a child’s maternal grandmother (such as smoking during pregnancy) are linked to the child’s risk of autistic traits and autism41,42,43 might be tied to this intergenerational mechanism.
Immune dysregulation
Activation of dysregulated maternal immune responses during pregnancy has been implicated in the pathogenesis of ASD and other neurodevelopmental disorders. We set aside the question of whether the observed immune dysregulation is due to infection, medication, fever, autoimmunity or genetic or other factors. Instead, we consider the evidence that certain kinds of immune dysregulation could be involved in the pathogenesis of ASD and/or be an early indicator of heightened risk of ASD.
Robust evidence for immune dysregulation in mothers midway through their pregnancy and in neonatal cord blood has been developed in the ABC substudy of the MoBA cohort in Norway, which was designed to interrogate biological processes occurring at multiple points in pregnancy. Immune profiling of blood samples collected from mothers at 17–21 weeks of pregnancy and cord blood samples collected at birth found differences consistent with systemic inflammation in a wide range of analytes in samples from both boys and girls with ASDs20. The number of elevated pro-inflammatory cytokines, chemokines and adhesion molecules associated with ASD and the mean effect sizes of these elevations were larger in girls than in boys. These effects of systemic inflammation, and the difference in effect sizes between girls and boys, were most pronounced in mid-gestation20. Previous research examining immune signatures associated with ASD was limited: only a few studies had examined maternal immune molecules in mid-gestation44,45,46 and a handful had examined immune molecules in dried blood spots47,48,49,50,51. Most of these studies examined only a small number of analytes, and although most had controlled for sex, sex-specific differences were not detailed44,45,46,47,48,49,50,51.
The strength of these findings in the ABC study prompted further scrutiny of the biomarker potential of immune signatures in maternal mid-gestation and cord blood samples. Five predictive models were built for girls and boys in relation to each sample type, and tested in 80% of the ABC population. Model validation was conducted in test sets composed of the remaining 20%20. The predictive models were able to distinguish children with ASD from control children without ASD among both boys and girls in the two sample types: model average areas under the receiver operating characteristic curve were 0.848 in mid-gestation and 0.846 in cord blood for boys, and 0.965 in mid-gestation and 0.917 in cord blood for girls20. Ultimately this work might result in the development of early biomarker(s) for ASD52. As yet, other cohorts have produced only limited complementary data, but we expect that additional data will be reported in the near future.
Preterm birth
Although infant survival following preterm birth has improved exponentially over the past 40 years, prematurity is not benign. Indeed, linked registry studies provided the first evidence of an association between preterm birth and ASD53,54,55. The association is particularly strong in brain imaging studies conducted during infancy, which reveal evidence of structural abnormalities, including ventricular enlargement and brain volume overgrowth56,57. The addition of genetic analyses and fetal imaging to studies in pregnant women at high risk of premature delivery might ultimately help to untangle the factors underlying these associations. An example of such a study that measures autistic traits is the population-based prospective Generation R cohort study58, which aims to identify early environmental and genetic causes of normal and abnormal growth, development and health in participants monitored from fetal life until young adulthood.
Animal models
Animal models cannot fully reproduce the complex spectrum of behavioural and social deficits that occur in people with ASDs. Nonetheless, features such as repetitive movements and impaired social interaction can be used to investigate the plausible possibility that genetic, microbial, immune, toxic and nutritional factors contribute to the risk of ASD, and to explore mechanisms of ASD pathogenesis and potential interventions. The majority of animal models use genetically modified mice, in which overexpression or knockdown of specific genes has been used to assess their contributions and those of associated pathways to development and behaviour. Some animal models focus on the environmental exposures implicated in ASD by epidemiological research. We highlight some examples of these animal models; however, readers will find more details in recent reviews59,60.
Gestational exposure to thalidomide and valproic acid are both associated with an increased risk of autism19,61. Accordingly, rodent models were established to dissect the developmental consequences of exposure to these agents. In studies of pregnant rats, exposure to thalidomide resulted in auditory hypersensitivity in offspring62. Auditory hypersensitivity is common in individuals with autism, in whom this feature has been linked to electrophysiological abnormalities in the primary auditory cortex63. Gestational exposure of rats to valproic acid resulted in reduced social interaction and exploratory activity reminiscent of features described in autism, accompanied by reduced numbers of myelinated axons and aberrant myelin sheath ultrastructure in the corpus callosum64. These histopathological findings are consistent with corpus callosum abnormalities reported in individuals with autism and with conceptual models of the disorder that focus on impaired brain connectivity65.
Rodent and primate models of gestational exposure to bacteria and viruses and their gene products, as well as cytokines and antibodies, have also been established66,67,68,69. Influenza virus infection in pregnant mice resulted in offspring with deficient prepulse inhibition of their acoustic startle response, as well as deficits in exploratory behaviours and social interaction. These findings did not reflect a direct effect of exposure to this infectious virus because they could be reproduced by exposure to the viral mimic, synthetic double-stranded RNA polyinosinic-polycytidylic acid (poly I:C), or to the pro-inflammatory cytokine IL-6 (refs. 66,70). Other researchers found additional support for an indirect, immune-mediated mechanism of behavioural deficits in the offspring of pregnant mice exposed to bacterial superantigens71 and mitigation of the behavioural deficits in animals gestationally exposed to anti-inflammatory drug treatment following immune stimulation with poly I:C68. Animal models have also been used to examine the role of the gastrointestinal microbiome in ASD pathogenesis. Intestinal dendritic cells from pregnant mice colonized with segmented filamentous bacteria secrete cytokines that promote the differentiation of intestinal T helper 17 cells, which results in inflammation in their offspring as well as cortical and behavioural abnormalities72.
Germ-free mice colonized with microbiota from human donors with autism show autistic behaviours and have reduced levels of two GABA agonists, 5-aminovaleric acid and taurine, in their brain and faeces. The autistic behaviours of these mice normalized when these two GABA agonists were provided through oral supplementation73. The extent to which the results of these individual studies can be replicated and the fidelity of animal models in reproducing the cardinal features of autism are unclear. Nonetheless, in concert, they confirm that gestational exposures to toxins and inflammation can result in neurodevelopmental damage that culminates in social deficits and/or neurodiversity (the term neurodiversity acknowledges that not all differences are deficits).
Conclusions
For millennia, philosophers and theologians have debated the relative importance of nature and nurture as determinants of biology and behaviour. With the advent of prospective birth cohorts, we have an opportunity to move from abstract discussions to rigorous dissection of the interactions of genetic and environmental factors in health and disease. Several important points that must be considered in the design of future prospective cohorts are highlighted in the following paragraphs.
The effect of an environmental factor could vary according to when (for example, in which trimester of gestation) and to whom (for instance, mother, father or grandparent) it is introduced; thus, our ability to identify its consequences depends, at minimum, on whether we have data that bracket critical intervals. Several windows exist during development wherein the absence of a nutrient (such as folate), lack of social interaction or the presence of toxins (such as alcohol, heavy metals, anticonvulsant drugs and by-products of infection) can profoundly influence brain structure and function. The effect of those same factors might be qualitatively or quantitatively different at a different time. For example, lead intoxication in children can result in cognitive dysfunction, whereas in adults it is more commonly associated with peripheral neuropathy.
The value of a specific cohort as a resource is determined by the size and composition of its population, the onset and frequency of collection of data and samples, the types of data and samples collected, the duration of follow-up and the commitment of cohort members to continued engagement with the project. In the Norwegian ABC, the parental questionnaire completion rate was 95% at the 18-month time point but decreased to 61% at the 36-month time point74. Fortunately, the National Patient Registry (NPR) allowed us to identify additional individuals with ASDs who were not found through questionnaire responses, either because parents did not respond or diagnosis was delayed. Without the NPR we would not have been able to use the gestational questionnaires and patient samples that were required to find links between outcomes and exposures. A thoughtful experimental design should limit participant fatigue and maximize retention by reducing the frequency and complexity of data and sample collection.
The validity and generalizability of findings from individual cohorts must be tested against those from other cohorts. Such comparisons can be challenging, not only because of differences in methods of case ascertainment and sampling, but also because genetic and environmental factors can vary. For example, in the ABC, the strength of the mitigating effect of folic acid supplements on the risk of autism was considerably increased with respect to some other studies because foods are not supplemented with folate in Norway. As another example, cytokine levels in plasma samples stored at ultra-low temperatures differ from those in whole blood collected on filter paper and stored at room temperature. Findings that remain consistent across cohorts despite differences in sampling methods are considered particularly robust; however, failures to replicate findings across different cohorts must be considered in this context. Although DNA, serum and plasma samples are readily collected and their storage at −20 °C is adequate for genetic analyses or serology, the materials required for transcriptomics, metabolomics, and proteomics are more labile. In the ABC, we stored plasma and RNA samples at −80 °C and learned to our chagrin that this was insufficient to maintain their quality for use in some assays. Accordingly, at least a portion of each sample should be stored at −140 °C for future use, including with platforms that do not yet exist. We also did not collect faecal or vaginal samples in the ABC that could have been used in microbiome studies. However, sample biobanking is resource-intensive and expensive. Therefore, despite our best efforts to future-proof data collection, regret for opportunities missed can be anticipated as laboratory methods continually evolve and require different sample types or management.
Ethical challenges will almost certainly emerge as population-based cohort research reveals links between health outcomes and genetic as well as environmental factors. In some instances, ASD biomarkers might be found that enable early intervention to prevent or ameliorate the condition. In other situations, the information found might only predict an individual’s risk of ASD. Leadership teams should consider whether and how such sensitive data will be released to cohort participants, and should include ethicists who can appropriately manage its communication.
Finally, the future of cohort research will almost certainly entail the use of machine learning methods for integrating genetic, epigenetic and multiomics datasets across populations in meta-analyses. In concert, these approaches aim to generate important insights into the pathology of a wide range of acute and chronic diseases and might lead to biomarkers for early diagnosis of individuals at risk, as well as new methods for mitigating risk. In this new era of ASD research, investigators will have an opportunity to use birth cohorts to embrace the complexity of interactions of genetic and environmental factors in a temporal context.
References
Lai, M. C., Lombardo, M. V. & Baron-Cohen, S. Autism. Lancet 383, 896–910 (2014).
Orinstein, A. J. et al. Intervention for optimal outcome in children and adolescents with a history of autism. J. Dev. Behav. Pediatr. 35, 247–256 (2014).
Fombonne, E. Editorial: The rising prevalence of autism. J. Child. Psychol. Psychiatry 59, 717–720 (2018).
Starko, K. M., Ray, C. G., Dominguez, L. B., Stromberg, W. L. & Woodall, D. F. Reye’s syndrome and salicylate use. Pediatrics 66, 859–864 (1980).
Wakefield, A. J. et al. Ileal-lymphoid-nodular hyperplasia, non-specific colitis, and pervasive developmental disorder in children. Lancet 351, 637–641 (1998); retraction 375, 445 (2010).
Geier, D. A. & Geier, M. R. A comparative evaluation of the effects of MMR immunization and mercury doses from thimerosal-containing childhood vaccines on the population prevalence of autism. Med. Sci. Monit. 10, PI33–PI39 (2004).
Hornig, M. et al. Lack of association between measles virus vaccine and autism with enteropathy: a case-control study. PLoS ONE 3, e3140 (2008).
Gerber, J. S. & Offit, P. A. Vaccines and autism: a tale of shifting hypotheses. Clin. Infect. Dis. 48, 456–461 (2009).
Velasquez-Manoff, M. The anti-vaccine movement’s new frontier. New York Times https://www.nytimes.com/2022/05/25/magazine/anti-vaccine-movement.html (2022).
Hansen, S. N., Schendel, D. E. & Parner, E. T. Explaining the increase in the prevalence of autism spectrum disorders: the proportion attributable to changes in reporting practices. JAMA Pediatr. 169, 56–62 (2015).
Kanner, L. Problems of nosology and psychodynamics of early infantile autism. Am. J. Orthopsychiatry 19, 416–426 (1949).
Chess, S. Autism in children with congenital rubella. J. Autism Child. Schizophr. 1, 33–47 (1971).
Folstein, S. & Rutter, M. Infantile autism: a genetic study of 21 twin pairs. J. Child. Psychol. Psychiatry 18, 297–321 (1977).
Tick, B., Bolton, P., Happe, F., Rutter, M. & Rijsdijk, F. Heritability of autism spectrum disorders: a meta-analysis of twin studies. J. Child. Psychol. Psychiatry 57, 585–595 (2016).
Folstein, S. & Rutter, M. Genetic influences and infantile autism. Nature 265, 726–728 (1977).
Sebat, J. et al. Strong association of de novo copy number mutations with autism. Science 316, 445–449 (2007).
Suren, P. et al. Association between maternal use of folic acid supplements and risk of autism spectrum disorders in children. JAMA 309, 570–577 (2013).
Zhou, X. et al. Integrating de novo and inherited variants in 42,607 autism cases identifies mutations in new moderate-risk genes. Nat. Genet. 54, 1305–1319 (2022).
Stromland, K., Nordin, V., Miller, M., Akerstrom, B. & Gillberg, C. Autism in thalidomide embryopathy: a population study. Dev. Med. Child. Neurol. 36, 351–356 (1994).
Che, X. et al. Maternal mid-gestational and child cord blood immune signatures are strongly associated with offspring risk of ASD. Mol. Psychiatry https://doi.org/10.1038/s41380-021-01415-4 (2022).
Hours, C., Recasens, C. & Baleyte, J. M. ASD and ADHD comorbidity: what are we talking about? Front. Psychiatry 13, 837424 (2022).
Kennedy, M. et al. Early severe institutional deprivation is associated with a persistent variant of adult attention-deficit/hyperactivity disorder: clinical presentation, developmental continuities and life circumstances in the English and Romanian Adoptees study. J. Child. Psychol. Psychiatry 57, 1113–1125 (2016).
Kumsta, R. et al. Severe psychosocial deprivation in early childhood is associated with increased DNA methylation across a region spanning the transcription start site of CYP2E1. Transl. Psychiatry 6, e830 (2016).
Moore, S. J. et al. A clinical study of 57 children with fetal anticonvulsant syndromes. J. Med. Genet. 37, 489–497 (2000).
Chess, S. Follow-up report on autism in congenital rubella. J. Autism Child. Schizophr. 7, 69–81 (1977).
Hertz-Picciotto, I. et al. A prospective study of environmental exposures and early biomarkers in autism spectrum disorder: design, protocols, and preliminary data from the MARBLES study. Env. Health Perspect. 126, 117004 (2018).
Pedersen, C. B. et al. The iPSYCH2012 case-cohort sample: new directions for unravelling genetic and environmental architectures of severe mental disorders. Mol. Psychiatry 23, 6–14 (2018).
Schendel, D. E. et al. The International Collaboration for Autism Registry Epidemiology (iCARE): multinational registry-based investigations of autism risk factors and trends. J. Autism Dev. Disord. 43, 2650–2663 (2013).
Boyd, A. et al. Cohort profile: the ‘children of the 90s’ – the index offspring of the Avon Longitudinal Study of Parents and Children. Int. J. Epidemiol. 42, 111–127 (2013).
Pinto-Martin, J. et al. The central New Jersey neonatal brain haemorrhage study: design of the study and reliability of ultrasound diagnosis. Paediatr. Perinat. Epidemiol. 6, 273–284 (1992).
Roth, C. et al. Folic acid supplements in pregnancy and severe language delay in children. JAMA 306, 1566–1573 (2011).
Liu, X., Zou, M., Sun, C., Wu, L. & Chen, W. X. Prenatal folic acid supplements and offspring’s autism spectrum disorder: a meta-analysis and meta-regression. J. Autism Dev. Disord. 52, 522–539 (2022).
Schmidt, R. J. et al. Maternal periconceptional folic acid intake and risk of autism spectrum disorders and developmental delay in the CHARGE (Childhood Autism Risks from Genetics and Environment) case-control study. Am. J. Clin. Nutr. 96, 80–89 (2012).
Maruvada, P. et al. Knowledge gaps in understanding the metabolic and clinical effects of excess folates/folic acid: a summary, and perspectives, from an NIH workshop. Am. J. Clin. Nutr. 112, 1390–1403 (2020).
Naderi, N. & House, J. D. Recent developments in folate nutrition. Adv. Food Nutr. Res. 83, 195–213 (2018).
Schmidt, R. J., Iosif, A. M., Guerrero Angel, E. & Ozonoff, S. Association of maternal prenatal vitamin use with risk for autism spectrum disorder recurrence in young siblings. JAMA Psychiatry 76, 391–398 (2019).
Cheslack-Postava, K. et al. Increased risk of autism spectrum disorders at short and long interpregnancy intervals in Finland. J. Am. Acad. Child. Adolesc. Psychiatry 53, 1074–1081.e4 (2014).
Gunnes, N. et al. Interpregnancy interval and risk of autistic disorder. Epidemiology 24, 906–912 (2013).
Ly, L. et al. Impact of mothers’ early life exposure to low or high folate on progeny outcome and DNA methylation patterns. Environ. Epigenet. 6, dvaa018 (2020).
Ly, L. et al. Intergenerational impact of paternal lifetime exposures to both folic acid deficiency and supplementation on reproductive outcomes and imprinted gene methylation. Mol. Hum. Reprod. 23, 461–477 (2017).
Golding, J. et al. Ancestral smoking and developmental outcomes: a review of publications from a population birth cohort. Biol. Reprod. 105, 625–631 (2021).
Golding, J., Steer, C. & Pembrey, M. Parental and grandparental ages in the autistic spectrum disorders: a birth cohort study. PLoS ONE 5, e9939 (2010).
Golding, J. et al. Grand-maternal smoking in pregnancy and grandchild’s autistic traits and diagnosed autism. Sci. Rep. 7, 46179 (2017).
Goines, P. E. et al. Increased midgestational IFN-γ, IL-4 and IL-5 in women bearing a child with autism: a case-control study. Mol. Autism 2, 13 (2011).
Jones, K. L. et al. Autism with intellectual disability is associated with increased levels of maternal cytokines and chemokines during gestation. Mol. Psychiatry 22, 273–279 (2017).
Casey, S. et al. Maternal mid-gestation cytokine dysregulation in mothers of children with autism spectrum disorder. J. Autism Dev. Disord. https://doi.org/10.1007/s10803-021-05271-7 (2021).
Krakowiak, P. et al. Neonatal cytokine profiles associated with autism spectrum disorder. Biol. Psychiatry 81, 442–451 (2017).
Abdallah, M. W. et al. Amniotic fluid chemokines and autism spectrum disorders: an exploratory study utilizing a Danish historic birth cohort. Brain Behav. Immun. 26, 170–176 (2012).
Heuer, L. S. et al. An exploratory examination of neonatal cytokines and chemokines as predictors of autism risk: the early markers for autism study. Biol. Psychiatry 86, 255–264 (2019).
Zerbo, O. et al. Neonatal cytokines and chemokines and risk of autism spectrum disorder: the early markers for autism (EMA) study: a case-control study. J. Neuroinflammation 11, 113 (2014).
Abdallah, M. W. et al. Neonatal levels of cytokines and risk of autism spectrum disorders: an exploratory register-based historic birth cohort study utilizing the Danish newborn screening biobank. J. Neuroimmunol. 252, 75–82 (2012).
Walsh, P., Elsabbagh, M., Bolton, P. & Singh, I. In search of biomarkers for autism: scientific, social and ethical challenges. Nat. Rev. Neurosci. 12, 603–612 (2011).
Hultman, C. M., Sparen, P. & Cnattingius, S. Perinatal risk factors for infantile autism. Epidemiology 13, 417–423 (2002).
Eaton, W. W., Mortensen, P. B., Thomsen, P. H. & Frydenberg, M. Obstetric complications and risk for severe psychopathology in childhood. J. Autism Dev. Disord. 31, 279–285 (2001).
Schendel, D. & Bhasin, T. K. Birth weight and gestational age characteristics of children with autism, including a comparison with other developmental disabilities. Pediatrics 121, 1155–1164 (2008).
Movsas, T. Z. et al. Autism spectrum disorder is associated with ventricular enlargement in a low birth weight population. J. Pediatr. 163, 73–78 (2013).
Hazlett, H. C. et al. Early brain development in infants at high risk for autism spectrum disorder. Nature 542, 348–351 (2017).
Jaddoe, V. W. et al. The Generation R study: design and cohort profile. Eur. J. Epidemiol. 21, 475–484 (2006).
Kazdoba, T. M. et al. Translational mouse models of autism: advancing toward pharmacological therapeutics. Curr. Top. Behav. Neurosci. 28, 1–52 (2016).
Ergaz, Z., Weinstein-Fudim, L. & Ornoy, A. Genetic and non-genetic animal models for autism spectrum disorders (ASD). Reprod. Toxicol. 64, 116–140 (2016).
Christensen, J. et al. Prenatal valproate exposure and risk of autism spectrum disorders and childhood autism. JAMA 309, 1696–1703 (2013).
Tsugiyama, L. E., Ida-Eto, M., Ohkawara, T., Noro, Y. & Narita, M. Altered neuronal activity in the auditory brainstem following sound stimulation in thalidomide-induced autism model rats. Congenit. Anom. 60, 82–86 (2020).
Matsuzaki, J. et al. Differential responses of primary auditory cortex in autistic spectrum disorder with auditory hypersensitivity. Neuroreport 23, 113–118 (2012).
Uccelli, N. A. et al. Neurobiological substrates underlying corpus callosum hypoconnectivity and brain metabolic patterns in the valproic acid rat model of autism spectrum disorder. J. Neurochem. 159, 128–144 (2021).
Frith, C. Is autism a disconnection disorder? Lancet Neurol. 3, 577 (2004).
Shi, L., Fatemi, S. H., Sidwell, R. W. & Patterson, P. H. Maternal influenza infection causes marked behavioral and pharmacological changes in the offspring. J. Neurosci. 23, 297–302 (2003).
Bauman, M. D. et al. Activation of the maternal immune system during pregnancy alters behavioral development of rhesus monkey offspring. Biol. Psychiatry 75, 332–341 (2014).
De Miranda, J. et al. Induction of Toll-like receptor 3-mediated immunity during gestation inhibits cortical neurogenesis and causes behavioral disturbances. mBio 1, e00176-10 (2010).
Bauman, M. D. et al. Maternal antibodies from mothers of children with autism alter brain growth and social behavior development in the rhesus monkey. Transl. Psychiatry 3, e278 (2013).
Hsiao, E. Y. & Patterson, P. H. Activation of the maternal immune system induces endocrine changes in the placenta via IL-6. Brain Behav. Immun. 25, 604–615 (2011).
Glass, R., Norton, S., Fox, N. & Kusnecov, A. W. Maternal immune activation with staphylococcal enterotoxin A produces unique behavioral changes in C57BL/6 mouse offspring. Brain Behav. Immun. 75, 12–25 (2019).
Kim, S. et al. Maternal gut bacteria promote neurodevelopmental abnormalities in mouse offspring. Nature 549, 528–532 (2017).
Sharon, G. et al. Human gut microbiota from autism spectrum disorder promote behavioral symptoms in mice. Cell 177, 1600–1618.e17 (2019).
Stoltenberg, C. et al. The Autism Birth Cohort: a paradigm for gene-environment-timing research. Mol. Psychiatry 15, 676–680 (2010).
Kanner, L. Autistic disturbances of affective contact. Nerv. Child. 2, 217 (1943).
Asperger, H. Die “Autistischen Psychopathen” im Kindesalter. Arch. Psychiatr. Nervenkr. 117, 76–136 (1944).
Wing, L. Asperger’s syndrome: a clinical account. Psychol. Med. 11, 115–129 (1981).
Stein, Z., Susser, M., Saenger, G. & Marolla, F. Famine and Human Development: The Dutch Hunger Winter of 1944–1945 (Oxford Univ. Press, 1975).
Wing, L. & Gould, J. Severe impairments of social interaction and associated abnormalities in children: epidemiology and classification. J. Autism Dev. Disord. 9, 11–29 (1979).
Schopler, E., Rutter, M. & Chess, S. Editorial: Change of journal scope and title. J. Autism Dev. Disord. 9, 1–10 (1979).
Meryash, D. L., Szymanski, L. S. & Gerald, P. S. Infantile autism associated with the fragile-X syndrome. J. Autism Dev. Disord. 12, 295–301 (1982).
MRC Vitamin Study Research Group. Prevention of neural tube defects: results of the Medical Research Council Vitamin Study. Lancet 338, 131–137 (1991).
Lord, C., Rutter, M. & Le Couteur, A. Autism diagnostic interview-revised: a revised version of a diagnostic interview for caregivers of individuals with possible pervasive developmental disorders. J. Autism Dev. Disord. 24, 659–685 (1994).
Rodier, P. M., Ingram, J. L., Tisdale, B., Nelson, S. & Romano, J. Embryological origin for autism: developmental anomalies of the cranial nerve motor nuclei. J. Comp. Neurol. 370, 247–261 (1996).
Rutter, M. et al. Quasi-autistic patterns following severe early global privation. English and Romanian Adoptees (ERA) Study Team. J. Child. Psychol. Psychiatry 40, 537–549 (1999).
Lord, C. et al. The autism diagnostic observation schedule-generic: a standard measure of social and communication deficits associated with the spectrum of autism. J. Autism Dev. Disord. 30, 205–223 (2000).
Courchesne, E., Carper, R. & Akshoomoff, N. Evidence of brain overgrowth in the first year of life in autism. JAMA 290, 337–344 (2003).
Reichenberg, A. et al. Advancing paternal age and autism. Arch. Gen. Psychiatry 63, 1026–1032 (2006).
Sadik, A. et al. Parental inflammatory bowel disease and autism in children. Nat. Med. 28, 1406–1411 (2022).
Author information
Authors and Affiliations
Contributions
All authors contributed to researching data for the article, writing the manuscript, discussions of its content and review or editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Neurology thanks Catherine Lord and the other, anonymous, reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
SFARI: https://www.sfari.org
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lipkin, W.I., Bresnahan, M. & Susser, E. Cohort-guided insights into gene–environment interactions in autism spectrum disorders. Nat Rev Neurol 19, 118–125 (2023). https://doi.org/10.1038/s41582-022-00764-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41582-022-00764-0
This article is cited by
-
Validation of plasma protein glycation and oxidation biomarkers for the diagnosis of autism
Molecular Psychiatry (2023)