Abstract
Advances in the molecular understanding of facioscapulohumeral muscular dystrophy (FSHD) have revealed that FSHD results from epigenetic de-repression of the DUX4 gene in skeletal muscle, which encodes a transcription factor that is active in early embryonic development but is normally silenced in almost all somatic tissues. These advances also led to the identification of targets for disease-altering therapies for FSHD, as well as an improved understanding of the molecular mechanism of the disease and factors that influence its progression. Together, these developments led the FSHD research community to shift its focus towards the development of disease-modifying treatments for FSHD. This Review presents advances in the molecular and clinical understanding of FSHD, discusses the potential targeted therapies that are currently being explored, some of which are already in clinical trials, and describes progress in the development of FSHD-specific outcome measures and assessment tools for use in future clinical trials.
Key points
-
Facioscapulohumeral muscular dystrophy (FSHD), a disorder for which there currently is no cure, is characterized by muscle weakness, predominantly affecting muscles in the face, shoulder girdle and upper arms.
-
FSHD is associated with epigenetic de-repression of the DUX4 gene, which leads to aberrant expression of the transcription factor DUX4 and cytotoxicity in skeletal muscle cells.
-
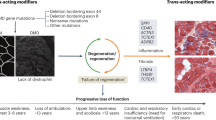
Clinical disease progression occurs in a nonlinear and muscle-by-muscle fashion with phases of muscle inflammation preceding rapid fatty replacement of muscle tissue and muscle wasting.
-
Consensus has been reached on the pathogenetic mechanism of FSHD, and the field is entering a new era of targeted therapy development.
-
Disease-altering therapies currently in development range from proof-of-principle gene-editing technologies focusing on reducing DUX4 expression to clinical trials of DUX4-blocking agents.
-
Clinical trials in FSHD require the development of meaningful patient outcome measures, identification of reliable biomarkers and accurate methods of measuring disease progression, such as MRI and ultrasonography.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Deenen, J. C. et al. Population-based incidence and prevalence of facioscapulohumeral dystrophy. Neurology 83, 1056–1059 (2014).
Lemmers, R. J. et al. A unifying genetic model for facioscapulohumeral muscular dystrophy. Science 329, 1650–1653 (2010).
Lemmers, R. J. et al. Digenic inheritance of an SMCHD1 mutation and an FSHD-permissive D4Z4 allele causes facioscapulohumeral muscular dystrophy type 2. Nat. Genet. 44, 1370–1374 (2012).
Tawil, R. et al. Clinical trial preparedness in facioscapulohumeral muscular dystrophy: clinical, tissue, and imaging outcome measures 29–30 May 2015, Rochester, New York. Neuromuscul. Disord. 26, 181–186 (2016).
Mul, K. et al. What’s in a name? The clinical features of facioscapulohumeral muscular dystrophy. Pract. Neurol. 16, 201–207 (2016).
Tawil, R. & Van Der Maarel, S. M. Facioscapulohumeral muscular dystrophy. Muscle Nerve 34, 1–15 (2006).
Statland, J. M. & Tawil, R. Risk of functional impairment in facioscapulohumeral muscular dystrophy. Muscle Nerve 49, 520–527 (2014).
Lemmers, R. J. et al. Somatic mosaicism in FSHD often goes undetected. Ann. Neurol. 55, 845–850 (2004).
Tonini, M. M. et al. Asymptomatic carriers and gender differences in facioscapulohumeral muscular dystrophy (FSHD). Neuromuscul. Disord. 14, 33–38 (2004).
Ricci, G. et al. Large scale genotype–phenotype analyses indicate that novel prognostic tools are required for families with facioscapulohumeral muscular dystrophy. Brain https://doi.org/10.1093/brain/awt226 (2013).
Salort-Campana, E. et al. Low penetrance in facioscapulohumeral muscular dystrophy type 1 with large pathological D4Z4 alleles: a cross-sectional multicenter study. Orphanet J. Rare Dis. 10, 2 (2015).
Wohlgemuth, M. et al. A family-based study into penetrance in facioscapulohumeral muscular dystrophy type 1. Neurology 91, e444–e454 (2018).
Zatz, M. et al. The facioscapulohumeral muscular dystrophy (FSHD1) gene affects males more severely and more frequently than females. Am. J. Med. Genet. 77, 155–161 (1998).
van der Maarel, S. M. et al. De novo facioscapulohumeral muscular dystrophy: frequent somatic mosaicism, sex-dependent phenotype, and the role of mitotic transchromosomal repeat interaction between chromosomes 4 and 10. Am. J. Hum. Genet. 66, 26–35 (2000).
Sakellariou, P. et al. Mutation spectrum and phenotypic manifestation in FSHD Greek patients. Neuromuscul. Disord. 22, 339–349 (2012).
Mul, K. et al. Phenotype–genotype relations in facioscapulohumeral muscular dystrophy type 1. Clin. Genet. 94, 521–527 (2018).
Monforte, M. et al. Tracking muscle wasting and disease activity in facioscapulohumeral muscular dystrophy by qualitative longitudinal imaging. J. Cachexia Sarcopenia Muscle 10, 1258–1265 (2019).
Katz, N. K. et al. Predictors of functional outcomes in patients with facioscapulohumeral muscular dystrophy. Brain 144, 3451–3460 (2021).
Teveroni, E. et al. Estrogens enhance myoblast differentiation in facioscapulohumeral muscular dystrophy by antagonizing DUX4 activity. J. Clin. Invest. 127, 1531–1545 (2017).
Mul, K., Horlings, C. G. C., Voermans, N. C., Schreuder, T. H. A. & van Engelen, B. G. M. Lifetime endogenous estrogen exposure and disease severity in female patients with facioscapulohumeral muscular dystrophy. Neuromuscul. Disord. 28, 508–511 (2018).
Goselink, R. J. M. et al. Early onset as a marker for disease severity in facioscapulohumeral muscular dystrophy. Neurology 92, e378–e385 (2019).
Klinge, L. et al. Severe phenotype in infantile facioscapulohumeral muscular dystrophy. Neuromuscul. Disord. 16, 553–558 (2006).
Brouwer, O. F., Padberg, G. W., Wijmenga, C. & Frants, R. R. Facioscapulohumeral muscular dystrophy in early childhood. Arch. Neurol. 51, 387–394 (1994).
Goselink, R. J. M. et al. Early onset facioscapulohumeral dystrophy — a systematic review using individual patient data. Neuromuscul. Disord. 27, 1077–1083 (2017).
Tawil, R. et al. Evidence-based guideline summary: evaluation, diagnosis, and management of facioscapulohumeral muscular dystrophy: report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology and the Practice Issues Review Panel of the American Association of Neuromuscular and Electrodiagnostic Medicine. Neurology 85, 357–364 (2015).
Aprile, I. et al. Balance and walking involvement in facioscapulohumeral dystrophy: a pilot study on the effects of custom lower limb orthoses. Eur. J. Phys. Rehab. Med. 49, 169–178 (2013).
Voet, N. et al. Both aerobic exercise and cognitive-behavioral therapy reduce chronic fatigue in FSHD: an RCT. Neurology 83, 1914–1922 (2014).
Andersen, G., Prahm, K. P., Dahlqvist, J. R., Citirak, G. & Vissing, J. Aerobic training and postexercise protein in facioscapulohumeral muscular dystrophy: RCT study. Neurology 85, 396–403 (2015).
Andersen, G., Heje, K., Buch, A. E. & Vissing, J. High-intensity interval training in facioscapulohumeral muscular dystrophy type 1: a randomized clinical trial. J. Neurol. 264, 1099–1106 (2017).
Wijmenga, C. et al. Location of facioscapulohumeral muscular dystrophy gene on chromosome 4. Lancet 336, 651–653 (1990).
Wijmenga, C. et al. Mapping of facioscapulohumeral muscular dystrophy gene to chromosome 4q35-qter by multipoint linkage analysis and in situ hybridization. Genomics 9, 570–575 (1991).
Wijmenga, C. et al. Chromosome 4q DNA rearrangements associated with facioscapulohumeral muscular dystrophy. Nat. Genet. 2, 26–30 (1992).
van Deutekom, J. C. et al. FSHD associated DNA rearrangements are due to deletions of integral copies of a 3.2 kb tandemly repeated unit. Hum. Mol. Genet. 2, 2037–2042 (1993).
Hewitt, J. E. et al. Analysis of the tandem repeat locus D4Z4 associated with facioscapulohumeral muscular dystrophy. Hum. Mol. Genet. 3, 1287–1295 (1994).
van Deutekom, J. C. et al. Identification of the first gene (FRG1) from the FSHD region on human chromosome 4q35. Hum. Mol. Genet. 5, 581–590 (1996).
van Geel, M. et al. The FSHD region on human chromosome 4q35 contains potential coding regions among pseudogenes and a high density of repeat elements. Genomics 61, 55–65 (1999).
van Geel, M. et al. Identification of a novel β-tubulin subfamily with one member (TUBB4Q) located near the telomere of chromosome region 4q35. Cytogenet. Cell Genet. 88, 316–321 (2000).
Rijkers, T. et al. FRG2, an FSHD candidate gene, is transcriptionally upregulated in differentiating primary myoblast cultures of FSHD patients. J. Med. Genet. 41, 826–836 (2004).
Tupler, R. et al. Monosomy of distal 4q does not cause facioscapulohumeral muscular dystrophy. J. Med. Genet. 33, 366–370 (1996).
Gabellini, D., Green, M. R. & Tupler, R. Inappropriate gene activation in FSHD: a repressor complex binds a chromosomal repeat deleted in dystrophic muscle. Cell 110, 339–348 (2002).
Bodega, B. et al. Remodeling of the chromatin structure of the facioscapulohumeral muscular dystrophy (FSHD) locus and upregulation of FSHD-related gene 1 (FRG1) expression during human myogenic differentiation. BMC Biol. 7, 41 (2009).
Klooster, R. et al. Comprehensive expression analysis of FSHD candidate genes at the mRNA and protein level. Eur. J. Hum. Genet. 17, 1615–1624 (2009).
Jiang, G. et al. Testing the position-effect variegation hypothesis for facioscapulohumeral muscular dystrophy by analysis of histone modification and gene expression in subtelomeric 4q. Hum. Mol. Genet. 12, 2909–2921 (2003).
Cheli, S. et al. Expression profiling of FSHD-1 and FSHD-2 cells during myogenic differentiation evidences common and distinctive gene dysregulation patterns. PLoS ONE 6, e20966 (2011).
Thijssen, P. E. et al. DUX4 promotes transcription of FRG2 by directly activating its promoter in facioscapulohumeral muscular dystrophy. Skelet. Muscle 4, 19 (2014).
Ferri, G., Huichalaf, C. H., Caccia, R. & Gabellini, D. Direct interplay between two candidate genes in FSHD muscular dystrophy. Hum. Mol. Genet. 24, 1256–1266 (2015).
Lemmers, R. et al. Chromosome 10q-linked FSHD identifies DUX4 as principal disease gene. J. Med. Genet. 59, 180–188 (2021).
Gabriels, J. et al. Nucleotide sequence of the partially deleted D4Z4 locus in a patient with FSHD identifies a putative gene within each 3.3 kb element. Gene 236, 25–32 (1999).
van Geel, M. et al. Genomic analysis of human chromosome 10q and 4q telomeres suggests a common origin. Genomics 79, 210–217 (2002).
Lemmers, R. J. et al. Facioscapulohumeral muscular dystrophy is uniquely associated with one of the two variants of the 4q subtelomere. Nat. Genet. 32, 235–236 (2002).
Lemmers, R. J. et al. Contractions of D4Z4 on 4qB subtelomeres do not cause facioscapulohumeral muscular dystrophy. Am. J. Hum. Genet. 75, 1124–1130 (2004).
Dixit, M. et al. DUX4, a candidate gene of facioscapulohumeral muscular dystrophy, encodes a transcriptional activator of PITX1. Proc. Natl Acad. Sci. USA 104, 18157–18162 (2007).
Snider, L. et al. RNA transcripts, miRNA-sized fragments and proteins produced from D4Z4 units: new candidates for the pathophysiology of facioscapulohumeral dystrophy. Hum. Mol. Genet. 18, 2414–2430 (2009).
Bakker, E. et al. The FSHD-linked locus D4F104S1 (p13E-11) on 4q35 has a homologue on 10qter. Muscle Nerve. 2, S39–S44 (1995).
Deidda, G. et al. Physical mapping evidence for a duplicated region on chromosome 10qter showing high homology with the facioscapulohumeral muscular dystrophy locus on chromosome 4qter. Eur. J. Hum. Genet. 3, 155–167 (1995).
Lemmers, R. J. et al. Worldwide population analysis of the 4q and 10q subtelomeres identifies only four discrete interchromosomal sequence transfers in human evolution. Am. J. Hum. Genet. 86, 364–377 (2010).
Jones, T. I. et al. Facioscapulohumeral muscular dystrophy family studies of DUX4 expression: evidence for disease modifiers and a quantitative model of pathogenesis. Hum. Mol. Genet. 21, 4419–4430 (2012).
Snider, L. et al. Facioscapulohumeral dystrophy: incomplete suppression of a retrotransposed gene. PLoS Genet. 6, e1001181 (2010).
Huichalaf, C., Micheloni, S., Ferri, G., Caccia, R. & Gabellini, D. DNA methylation analysis of the macrosatellite repeat associated with FSHD muscular dystrophy at single nucleotide level. PLoS ONE 9, e115278 (2014).
Das, S. & Chadwick, B. P. Influence of repressive histone and DNA methylation upon D4Z4 transcription in non-myogenic cells. PLoS ONE 11, e0160022 (2016).
Zeng, W. et al. Specific loss of histone H3 lysine 9 trimethylation and HP1γ/cohesin binding at D4Z4 repeats is associated with facioscapulohumeral dystrophy (FSHD). PLoS Genet 5, e1000559 (2009).
Boros, J., Arnoult, N., Stroobant, V., Collet, J. F. & Decottignies, A. Polycomb repressive complex 2 and H3K27me3 cooperate with H3K9 methylation to maintain heterochromatin protein 1α at chromatin. Mol. Cell Biol. 34, 3662–3674 (2014).
Chen, K. et al. Genome-wide binding and mechanistic analyses of Smchd1-mediated epigenetic regulation. Proc. Natl Acad. Sci. USA 112, E3535–E3544 (2015).
Campbell, A. E. et al. NuRD and CAF-1-mediated silencing of the D4Z4 array is modulated by DUX4-induced MBD3L proteins. eLife 7, e31023 (2018).
Casa, V. et al. Polycomb repressive complex 1 provides a molecular explanation for repeat copy number dependency in FSHD muscular dystrophy. Hum. Mol. Genet. 26, 753–767 (2017).
Haynes, P., Bomsztyk, K. & Miller, D. G. Sporadic DUX4 expression in FSHD myocytes is associated with incomplete repression by the PRC2 complex and gain of H3K9 acetylation on the contracted D4Z4 allele. Epigenet. Chromatin 11, 47 (2018).
Sacconi, S. et al. FSHD1 and FSHD2 form a disease continuum. Neurology 92, e2273–e2285 (2019).
van Overveld, P. G. et al. Hypomethylation of D4Z4 in 4q-linked and non-4q-linked facioscapulohumeral muscular dystrophy. Nat. Genet. 35, 315–317 (2003).
Calandra, P. et al. Allele-specific DNA hypomethylation characterises FSHD1 and FSHD2. J. Med. Genet. 53, 348–355 (2016).
Rieken, A., Bossler, A. D., Mathews, K. D. & Moore, S. A. CLIA Laboratory testing for facioscapulohumeral dystrophy: a retrospective analysis. Neurology 96, e1054–e1062 (2021).
Lemmers, R. J. et al. Hemizygosity for SMCHD1 in facioscapulohumeral muscular dystrophy type 2: consequences for 18p deletion syndrome. Hum. Mutat. 36, 679–683 (2015).
van den Boogaard, M. L. et al. Mutations in DNMT3B modify epigenetic repression of the D4Z4 repeat and the penetrance of facioscapulohumeral dystrophy. Am. J. Hum. Genet. 98, 1020–1029 (2016).
Hamanaka, K. et al. Homozygous nonsense variant in LRIF1 associated with facioscapulohumeral muscular dystrophy. Neurology 94, e2441–e2447 (2020).
Sacconi, S. et al. The FSHD2 gene SMCHD1 is a modifier of disease severity in families affected by FSHD1. Am. J. Hum. Genet. 93, 744–751 (2013).
van Deutekom, J. C. et al. Evidence for subtelomeric exchange of 3.3 kb tandemly repeated units between chromosomes 4q35 and 10q26: implications for genetic counselling and etiology of FSHD1. Hum. Mol. Genet. 5, 1997–2003 (1996).
Nguyen, K. et al. Deciphering the complexity of the 4q and 10q subtelomeres by molecular combing in healthy individuals and patients with facioscapulohumeral dystrophy. J. Med. Genet. 56, 590–601 (2019).
Goossens, R. et al. Intronic SMCHD1 variants in FSHD: testing the potential for CRISPR-Cas9 genome editing. J. Med. Genet. 56, 828–837 (2019).
Lemmers, R. J. L. F. et al. Cis D4Z4 repeat duplications associated with facioscapulohumeral muscular dystrophy type 2. Hum. Mol. Genet. 27, 3488–3497 (2018).
Lemmers, R. et al. High-resolution breakpoint junction mapping of proximally extended D4Z4 deletions in FSHD1 reveals evidence for a founder effect. Hum. Mol. Genet. 31, 748–760 (2022).
Lemmers, R. J. et al. D4F104S1 deletion in facioscapulohumeral muscular dystrophy: phenotype, size, and detection. Neurology 61, 178–183 (2003).
van den Boogaard, M. L. et al. Double SMCHD1 variants in FSHD2: the synergistic effect of two SMCHD1 variants on D4Z4 hypomethylation and disease penetrance in FSHD2. Eur. J. Hum. Genet. 24, 78–85 (2016).
Mocciaro, E., Runfola, V., Ghezzi, P., Pannese, M. & Gabellini, D. DUX4 role in normal physiology and in FSHD muscular dystrophy. Cells https://doi.org/10.3390/cells10123322 (2021).
Geng, L. N. et al. DUX4 activates germline genes, retroelements, and immune mediators: implications for facioscapulohumeral dystrophy. Dev. Cell 22, 38–51 (2012).
Hendrickson, P. G. et al. Conserved roles of mouse DUX and human DUX4 in activating cleavage-stage genes and MERVL/HERVL retrotransposons. Nat. Genet. 49, 925–934 (2017).
De Iaco, A. et al. DUX-family transcription factors regulate zygotic genome activation in placental mammals. Nat. Genet. 49, 941–994 (2017).
Choi, S. H. et al. DUX4 recruits p300/CBP through its C-terminus and induces global H3K27 acetylation changes. Nucleic Acids Res. 44, 5161–5173 (2016).
Resnick, R. et al. DUX4-induced histone variants H3.X and H3.Y mark DUX4 target genes for expression. Cell Rep. 29, 1812–1820.e5 (2019).
Gannon, O. M., Merida de Long, L. & Saunders, N. A. DUX4 is derepressed in late-differentiating keratinocytes in conjunction with loss of H3K9me3 epigenetic repression. J. Invest. Dermatol. 136, 1299–1302 (2016).
Kowaljow, V. et al. The DUX4 gene at the FSHD1A locus encodes a pro-apoptotic protein. Neuromuscul. Disord. 17, 611–623 (2007).
Knopp, P. et al. DUX4 induces a transcriptome more characteristic of a less-differentiated cell state and inhibits myogenesis. J. Cell Sci. 129, 3816–3831 (2016).
Wallace, L. M. et al. DUX4, a candidate gene for facioscapulohumeral muscular dystrophy, causes p53-dependent myopathy in vivo. Ann. Neurol. 69, 540–552 (2011).
Lek, A. et al. Applying genome-wide CRISPR–Cas9 screens for therapeutic discovery in facioscapulohumeral muscular dystrophy. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aay0271 (2020).
Bosnakovski, D. et al. p53-independent DUX4 pathology in cell and animal models of facioscapulohumeral muscular dystrophy. Dis. Model. Mech. 10, 1211–1216 (2017).
Shadle, S. C. et al. DUX4-induced dsRNA and MYC mRNA stabilization activate apoptotic pathways in human cell models of facioscapulohumeral dystrophy. PLoS Genet. 13, e1006658 (2017).
Grow, E. J. et al. p53 convergently activates Dux/DUX4 in embryonic stem cells and in facioscapulohumeral muscular dystrophy cell models. Nat. Genet. 53, 1207–1220 (2021).
Vogelstein, B., Lane, D. & Levine, A. J. Surfing the p53 network. Nature 408, 307–310 (2000).
Winokur, S. T. et al. Expression profiling of FSHD muscle supports a defect in specific stages of myogenic differentiation. Hum. Mol. Genet. 12, 2895–2907 (2003).
Sasaki-Honda, M. et al. A patient-derived iPSC model revealed oxidative stress increases facioscapulohumeral muscular dystrophy-causative DUX4. Hum. Mol. Genet. 27, 4024–4035 (2018).
Tsumagari, K. et al. Gene expression during normal and FSHD myogenesis. BMC Med. Genom. 4, 67 (2011).
Jagannathan, S., Ogata, Y., Gafken, P. R., Tapscott, S. J. & Bradley, R. K. Quantitative proteomics reveals key roles for post-transcriptional gene regulation in the molecular pathology of facioscapulohumeral muscular dystrophy. eLife 8, e41740 (2019).
Beermann, M. L., Homma, S. & Miller, J. B. Proximity ligation assay to detect DUX4 protein in FSHD1 muscle: a pilot study. BMC Res. Notes 15, 163 (2022).
Tassin, A. et al. DUX4 expression in FSHD muscle cells: how could such a rare protein cause a myopathy. J. Cell. Mol. Med. 17, 76–89 (2013).
Rickard, A. M., Petek, L. M. & Miller, D. G. Endogenous DUX4 expression in FSHD myotubes is sufficient to cause cell death and disrupts RNA splicing and cell migration pathways. Hum. Mol. Genet. 24, 5901–5914 (2015).
Yao, Z. et al. DUX4-induced gene expression is the major molecular signature in FSHD skeletal muscle. Hum. Mol. Genet. 23, 5342–5352 (2014).
Jagannathan, S. et al. Model systems of DUX4 expression recapitulate the transcriptional profile of FSHD cells. Hum. Mol. Genet. 25, 4419–4431 (2016).
Heuvel, A. V. D. et al. Single-cell RNA-sequencing in facioscapulohumeral muscular dystrophy disease etiology and development. Hum. Mol. Genet. 28, 1064–1075 (2018).
Jiang, S. et al. Single-nucleus RNA-seq identifies divergent populations of FSHD2 myotube nuclei. PLoS Genet 16, e1008754 (2020).
Nikolic, A. et al. Clinical expression of facioscapulohumeral muscular dystrophy in carriers of 1-3 D4Z4 reduced alleles: experience of the FSHD Italian National Registry. BMJ Open 6, e007798 (2016).
Statland, J. M. et al. Milder phenotype in facioscapulohumeral dystrophy with 7–10 residual D4Z4 repeats. Neurology 85, 2147–2150 (2015).
Lemmers, R. J. et al. Inter-individual differences in CpG methylation at D4Z4 correlate with clinical variability in FSHD1 and FSHD2. Hum. Mol. Genet. 24, 659–669 (2014).
Scionti, I. et al. Large-scale population analysis challenges the current criteria for the molecular diagnosis of fascioscapulohumeral muscular dystrophy. Am. J. Hum. Genet. 90, 628–635 (2012).
Lemmers, R. J. et al. Specific sequence variations within the 4q35 region are associated with facioscapulohumeral muscular dystrophy. Am. J. Hum. Genet. 81, 884–894 (2007).
Nikolic, A. et al. Interpretation of the epigenetic signature of facioscapulohumeral muscular dystrophy in light of genotype–phenotype studies. Int. J. Mol. Sci. 21, 2635 (2020).
Salsi, V., Magdinier, F. & Tupler, R. Does DNA methylation matter in FSHD. Genes 11, 258 (2020).
van Overveld, P. G. et al. Variable hypomethylation of D4Z4 in facioscapulohumeral muscular dystrophy. Ann. Neurol. 58, 569–576 (2005).
de Greef, J. C. et al. Hypomethylation is restricted to the D4Z4 repeat array in phenotypic FSHD. Neurology 69, 1018–1026 (2007).
de Greef, J. C. et al. Common epigenetic changes of D4Z4 in contraction-dependent and contraction-independent FSHD. Hum. Mutat. 30, 1449–1459 (2009).
Gaillard, M. C. et al. Differential DNA methylation of the D4Z4 repeat in patients with FSHD and asymptomatic carriers. Neurology 83, 733–742 (2014).
Larsen, M. et al. Diagnostic approach for FSHD revisited: SMCHD1 mutations cause FSHD2 and act as modifiers of disease severity in FSHD1. Eur. J. Hum. Genet. 23, 808–816 (2014).
Jones, T. I. et al. Individual epigenetic status of the pathogenic D4Z4 macrosatellite correlates with disease in facioscapulohumeral muscular dystrophy. Clin. Epigenet. 7, 37 (2015).
Roche, S. et al. Methylation hotspots evidenced by deep sequencing in patients with facioscapulohumeral dystrophy and mosaicism. Neurol. Genet. 5, e372–e372 (2019).
Jones, T. I. et al. Identifying diagnostic DNA methylation profiles for facioscapulohumeral muscular dystrophy in blood and saliva using bisulfite sequencing. Clin. Epigenet. 6, 23 (2014).
Erdmann, H. et al. Methylation of the 4q35 D4Z4 repeat defines disease status in facioscapulohumeral muscular dystrophy. Brain https://doi.org/10.1093/brain/awac336 (2022).
Mason, A. G. et al. SMCHD1 regulates a limited set of gene clusters on autosomal chromosomes. Skelet. Muscle 7, 12 (2017).
Hartweck, L. M. et al. A focal domain of extreme demethylation within D4Z4 in FSHD2. Neurology 80, 392–399 (2013).
Lemmers, R. J. L. F. et al. Inter-individual differences in CpG methylation at D4Z4 correlate with clinical variability in FSHD1 and FSHD2. Hum. Mol. Genet. 24, 659–669 (2015).
Balog, J. et al. Increased DUX4 expression during muscle differentiation correlates with decreased SMCHD1 protein levels at D4Z4. Epigenetics 10, 1133–1142 (2015).
Balog, J. et al. Monosomy 18p is a risk factor for facioscapulohumeral dystrophy. J. Med. Genet. 55, 469–478 (2018).
Gordon, C. T. et al. De novo mutations in SMCHD1 cause bosma arhinia microphthalmia syndrome and abrogate nasal development. Nat. Genet. 49, 249–255 (2017).
Shaw, N. D. et al. SMCHD1 mutations associated with a rare muscular dystrophy can also cause isolated arhinia and bosma arhinia microphthalmia syndrome. Nat. Genet. 49, 238–248 (2017).
Kinjo, K. et al. Rare variant of the epigenetic regulator SMCHD1 in a patient with pituitary hormone deficiency. Sci. Rep. 10, 10985 (2020).
Lemmers, R. J. L. F. et al. SMCHD1 mutation spectrum for facioscapulohumeral muscular dystrophy type 2 (FSHD2) and bosma arhinia microphthalmia syndrome (BAMS) reveals disease-specific localisation of variants in the ATPase domain. J. Med. Genet. 56, 693–700 (2019).
Gurzau, A. D. et al. FSHD2- and BAMS-associated mutations confer opposing effects on SMCHD1 function. J. Biol. Chem. 293, 9841–9853 (2018).
Dion, C. et al. SMCHD1 is involved in de novo methylation of the DUX4-encoding D4Z4 macrosatellite. Nucleic Acids Res 47, 2822–2839 (2019).
Mul, K. et al. FSHD type 2 and bosma arhinia microphthalmia syndrome: two faces of the same mutation. Neurology 91, e562–e570 (2018).
Mohassel, P. et al. Cross-sectional neuromuscular phenotyping study of patients with arhinia with SMCHD1 variants. Neurology 98, e1384–e1396 (2022).
Hansen, R. S. et al. The DNMT3B DNA methyltransferase gene is mutated in the ICF immunodeficiency syndrome. Proc. Natl Acad. Sci. USA 96, 14412–14417 (1999).
Xu, G. L. et al. Chromosome instability and immunodeficiency syndrome caused by mutations in a DNA methyltransferase gene. Nature 402, 187–191 (1999).
Gendrel, A. V. et al. Smchd1-dependent and -independent pathways determine developmental dynamics of CpG island methylation on the inactive X chromosome. Dev. Cell 23, 265–279 (2012).
Thijssen, P. E. et al. Mutations in CDCA7 and HELLS cause immunodeficiency-centromeric instability-facial anomalies syndrome. Nat. Commun. 6, 7870 (2015).
Jeanpierre, M. et al. An embryonic-like methylation pattern of classical satellite DNA is observed in ICF syndrome. Hum. Mol. Genet. 2, 731–735 (1993).
Kondo, T. et al. Whole-genome methylation scan in ICF syndrome: hypomethylation of non-satellite DNA repeats D4Z4 and NBL2. Hum. Mol. Genet. 9, 597–604 (2000).
Nozawa, R. S. et al. Human inactive X chromosome is compacted through a PRC2-independent SMCHD1–HBiX1 pathway. Nat. Struct. Mol. Biol. 20, 566–573 (2013).
Brideau, N. J. et al. Independent mechanisms target SMCHD1 to H3K9me3-modified chromatin and the inactive X chromosome. Mol. Cell Biol. 35, 4053–4068 (2015).
Balog, J. et al. Correlation analysis of clinical parameters with epigenetic modifications in the DUX4 promoter in FSHD. Epigenetics 7, 579–584 (2012).
Cabianca, D. S. et al. A long ncRNA links copy number variation to a polycomb/trithorax epigenetic switch in FSHD muscular dystrophy. Cell 149, 819–831 (2012).
Himeda, C. L. et al. Identification of epigenetic regulators of DUX4-fl for targeted therapy of facioscapulohumeral muscular dystrophy. Mol. Ther. 26, 1797–1807 (2018).
Olsen, D. B., Ørngreen, M. C. & Vissing, J. Aerobic training improves exercise performance in facioscapulohumeral muscular dystrophy. Neurology 64, 1064–1066 (2005).
Janssen, B., Voet, N., Geurts, A., van Engelen, B. & Heerschap, A. Quantitative MRI reveals decelerated fatty infiltration in muscles of active FSHD patients. Neurology 86, 1700–1707 (2016).
Wang, L. H. et al. MRI-informed muscle biopsies correlate MRI with pathology and DUX4 target gene expression in FSHD. Hum. Mol. Genet. 28, 476–486 (2019).
Dahlqvist, J. R. et al. Evaluation of inflammatory lesions over 2 years in facioscapulohumeral muscular dystrophy. Neurology 95, e1211–e1221 (2020).
van den Heuvel, A. et al. Facioscapulohumeral dystrophy transcriptome signatures correlate with different stages of disease and are marked by different MRI biomarkers. Sci. Rep. 12, 1426 (2022).
Mul, K. et al. Quantitative muscle MRI and ultrasound for facioscapulohumeral muscular dystrophy: complementary imaging biomarkers. J. Neurol. 265, 2646–2655 (2018).
Lassche, S. et al. Correlation between quantitative MRI and muscle histopathology in muscle biopsies from healthy controls and patients with IBM, FSHD and OPMD. J. Neuromuscul. Dis. 7, 495–504 (2020).
Wong, C. J. et al. Longitudinal measures of RNA expression and disease activity in FSHD muscle biopsies. Hum. Mol. Genet. 29, 1030–1043 (2020).
Andersen, G. et al. MRI as outcome measure in facioscapulohumeral muscular dystrophy: 1-year follow-up of 45 patients. J. Neurol. 264, 438–447 (2017).
Fatehi, F. et al. Long-term follow-up of MRI changes in thigh muscles of patients with facioscapulohumeral dystrophy: a quantitative study. PLoS One 12, e0183825 (2017).
Mul, K. et al. Adding quantitative muscle MRI to the FSHD clinical trial toolbox. Neurology 89, 2057–2065 (2017).
Frisullo, G. et al. CD8+ T cells in facioscapulohumeral muscular dystrophy patients with inflammatory features at muscle MRI. J. Clin. Immunol. 31, 155–166 (2011).
Tasca, G. et al. Different molecular signatures in magnetic resonance imaging-staged facioscapulohumeral muscular dystrophy muscles. PLoS ONE 7, e38779 (2012).
Ferguson, M. R. et al. MRI change metrics of facioscapulohumeral muscular dystrophy: STIR and T1. Muscle Nerve 57, 905–912 (2018).
Dahlqvist, J. R. et al. Relationship between muscle inflammation and fat replacement assessed by MRI in facioscapulohumeral muscular dystrophy. J. Neurol. 266, 1127–1135 (2019).
Friedman, S. D. et al. Longitudinal features of STIR bright signal in FSHD. Muscle Nerve 49, 257–260 (2014).
Janssen, B. H. et al. Distinct disease phases in muscles of facioscapulohumeral dystrophy patients identified by MR detected fat infiltration. PLoS ONE 9, e85416 (2014).
Cohen, J., DeSimone, A., Lek, M. & Lek, A. Therapeutic approaches in facioscapulohumeral muscular dystrophy. Trends Mol. Med. 27, 123–137 (2020).
Kissel, J. T. et al. Randomized, double-blind, placebo-controlled trial of albuterol in facioscapulohumeral dystrophy. Neurology 57, 1434–1440 (2001).
van der Kooi, E. L. et al. Strength training and albuterol in facioscapulohumeral muscular dystrophy. Neurology 63, 702–708 (2004).
Campbell, A. E. et al. BET bromodomain inhibitors and agonists of the β2 adrenergic receptor identified in screens for compounds that inhibit DUX4 expression in FSHD muscle cells. Skelet. Muscle 7, 16 (2017).
Oliva, J. et al. Clinically advanced p38 inhibitors suppress DUX4 expression in cellular and animal models of facioscapulohumeral muscular dystrophy. J. Pharmacol. Exp. Ther. 370, 219–223 (2019).
Rojas, L. A. et al. p38α regulates expression of DUX4 in a model of facioscapulohumeral muscular dystrophy. J. Pharmacol. Exp. Ther. 374, 489–498 (2020).
Mellion, M. L. et al. Phase 1 clinical trial of losmapimod in facioscapulohumeral dystrophy: safety, tolerability, pharmacokinetics, and target engagement. Br. J. Clin. Pharmacol. 87, 4658–4669 (2021).
Tawil, R. & Wagner, K. Clinical research: O.5 A phase 2, randomized, double-blind, placebo-controlled, 48-week study of the efficacy and safety of losmapimod in subjects with FSHD: ReDUX4. Neuromuscul. Disord. 31, S48–S49 (2021).
Jagannathan, S. et al. Meeting report: the 2021 FSHD International Research Congress. Skelet. Muscle 12, 1 (2022).
Wang, N., Wu, R., Tang, D. & Kang, R. The BET family in immunity and disease. Signal. Transduct. Target. Ther. 6, 23 (2021).
Yang, H., Wei, L., Xun, Y., Yang, A. & You, H. BRD4: an emerging prospective therapeutic target in glioma. Mol. Ther. Oncolyt. 21, 1–14 (2021).
Lim, J. W. et al. DICER/AGO-dependent epigenetic silencing of D4Z4 repeats enhanced by exogenous siRNA suggests mechanisms and therapies for FSHD. Hum. Mol. Genet. 24, 4817–4828 (2015).
Saad, N. Y. et al. Human miRNA miR-675 inhibits DUX4 expression and may be exploited as a potential treatment for facioscapulohumeral muscular dystrophy. Nat. Commun. 12, 7128 (2021).
Kuijper, E. C., Bergsma, A. J., Pijnappel, W. W. M. P. & Aartsma-Rus, A. Opportunities and challenges for antisense oligonucleotide therapies. J. Inherit. Metab. Dis. 44, 72–87 (2021).
Marsollier, A. C. et al. Antisense targeting of 3ʹ end elements involved in DUX4 mRNA processing is an efficient therapeutic strategy for facioscapulohumeral dystrophy: a new gene-silencing approach. Hum. Mol. Genet. 25, 1468–1478 (2016).
Chen, J. C. et al. Morpholino-mediated knockdown of DUX4 toward facioscapulohumeral muscular dystrophy therapeutics. Mol. Ther. 24, 1405–1411 (2016).
Ansseau, E. et al. Antisense oligonucleotides used to target the DUX4 mRNA as therapeutic approaches in faciosscapulohumeral muscular dystrophy (FSHD). Genes 8, 93 (2017).
Lu-Nguyen, N., Dickson, G., Malerba, A. & Popplewell, L. Long-term systemic treatment of a mouse model displaying chronic FSHD-like pathology with antisense therapeutics that inhibit DUX4 expression. Biomedicines 10, 1623 (2022).
Lu-Nguyen, N., Malerba, A., Antoni Pineda, M., Dickson, G. & Popplewell, L. Improving molecular and histopathology in diaphragm muscle of the double transgenic ACTA1-MCM/FLExDUX4 mouse model of FSHD with systemic antisense therapy. Hum. Gene Ther. 33, 923–935 (2022).
Lu-Nguyen, N., Malerba, A., Herath, S., Dickson, G. & Popplewell, L. Systemic antisense therapeutics inhibiting DUX4 expression ameliorates FSHD-like pathology in an FSHD mouse model. Hum. Mol. Genet. 30, 1398–1412 (2021).
Bouwman, L. F. et al. Systemic delivery of a DUX4-targeting antisense oligonucleotide to treat facioscapulohumeral muscular dystrophy. Mol. Ther. Nucleic Acids 26, 813–827 (2021).
George, L. A. et al. Long-term follow-up of the first in human intravascular delivery of AAV for gene transfer: AAV2-hFIX16 for severe hemophilia B. Mol. Ther. 28, 2073–2082 (2020).
Wallace, L. M. et al. RNA interference inhibits DUX4-induced muscle toxicity in vivo: implications for a targeted FSHD therapy. Mol. Ther. 20, 1417–1423 (2012).
Wallace, L. M. et al. Pre-clinical safety and off-target studies to support translation of AAV-mediated RNAi therapy for FSHD. Mol. Ther. Methods Clin. Dev. 8, 121–130 (2018).
Rashnonejad, A., Amini-Chermahini, G., Taylor, N. K., Wein, N. & Harper, S. Q. Designed U7 snRNAs inhibit DUX4 expression and improve FSHD-associated outcomes in DUX4 overexpressing cells and FSHD patient myotubes. Mol. Ther. Nucleic Acids 23, 476–486 (2021).
Bosnakovski, D. et al. A novel P300 inhibitor reverses DUX4-mediated global histone H3 hyperacetylation, target gene expression, and cell death. Sci. Adv. 5, eaaw7781 (2019).
Mariot, V. et al. A deoxyribonucleic acid decoy trapping DUX4 for the treatment of facioscapulohumeral muscular dystrophy. Mol. Ther. Nucleic Acids 22, 1191–1199 (2020).
Klingler, C. et al. DNA aptamers against the DUX4 protein reveal novel therapeutic implications for FSHD. FASEB J. https://doi.org/10.1096/fj.201902696 (2020).
Ran, F. A. et al. Genome engineering using the CRISPR–Cas9 system. Nat. Protoc. 8, 2281–2308 (2013).
Cong, L. et al. Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819–823 (2013).
Richardson, C. D., Ray, G. J., DeWitt, M. A., Curie, G. L. & Corn, J. E. Enhancing homology-directed genome editing by catalytically active and inactive CRISPR-Cas9 using asymmetric donor DNA. Nat. Biotechnol. 34, 339–344 (2016).
Larson, M. H. et al. CRISPR interference (CRISPRi) for sequence-specific control of gene expression. Nat. Protoc. 8, 2180–2196 (2013).
Himeda, C. L., Jones, T. I. & Jones, P. L. CRISPR/dCas9-mediated transcriptional inhibition ameliorates the epigenetic dysregulation at D4Z4 and represses DUX4-fl in FSH muscular dystrophy. Mol. Ther. 24, 527–535 (2016).
Joubert, R., Mariot, V., Charpentier, M., Concordet, J. P. & Dumonceaux, J. Gene editing targeting the DUX4 polyadenylation signal: a therapy for FSHD? J. Pers. Med. https://doi.org/10.3390/jpm11010007 (2020).
Das, S. & Chadwick, B. P. CRISPR mediated targeting of DUX4 distal regulatory element represses DUX4 target genes dysregulated in facioscapulohumeral muscular dystrophy. Sci. Rep. 11, 12598 (2021).
Gaudelli, N. M. et al. Programmable base editing of A*T to G*C in genomic DNA without DNA cleavage. Nature 551, 464–471 (2017).
Komor, A. C., Kim, Y. B., Packer, M. S., Zuris, J. A. & Liu, D. R. Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature 533, 420–424 (2016).
Ran, F. A. et al. Double nicking by RNA-guided CRISPR Cas9 for enhanced genome editing specificity. Cell 154, 1380–1389 (2013).
Šikrová, D. et al. Adenine base editing of the DUX4 polyadenylation signal for targeted genetic therapy in facioscapulohumeral muscular dystrophy. Mol. Ther. Nucleic Acids 25, 342–354 (2021).
Himeda, C. L., Jones, T. I. & Jones, P. L. Targeted epigenetic repression by CRISPR/dSaCas9 suppresses pathogenic DUX4-fl expression in FSHD. Mol. Ther. Methods Clin. Dev. 20, 298–311 (2021).
Tawil, R., Shaw, D. W., van der Maarel, S. M. & Tapscott, S. J. Clinical trial preparedness in facioscapulohumeral dystrophy: outcome measures and patient access: 8–9 April 2013, Leiden, The Netherlands. Neuromuscul. Disord. 24, 79–85 (2014).
The FSH-DY group. A prospective, quantitative study of the natural history of facioscapulohumeral muscular dystrophy (FSHD): implications for therapeutic trials. Neurology 48, 38–46 (1997).
Kinoshita, J., Magdinier, F. & Padberg, G. W. 26th Annual Facioscapulohumeral Dystrophy International Research Congress Marseille, France, 19–20 June 2019. Neuromuscul. Disord. 29, 811–817 (2019).
Mul, K. et al. 225th ENMC international workshop: a global FSHD registry framework, 18–20 November 2016, Heemskerk, The Netherlands. Neuromuscul. Disord. 27, 782–790 (2017).
Tassin, A. et al. FSHD myotubes with different phenotypes exhibit distinct proteomes. PLoS ONE 7, e51865 (2012).
Jones, T. I. et al. Transgenic mice expressing tunable levels of DUX4 develop characteristic facioscapulohumeral muscular dystrophy-like pathophysiology ranging in severity. Skelet. Muscle 10, 8 (2020).
Jones, T. & Jones, P. L. A cre-inducible DUX4 transgenic mouse model for investigating facioscapulohumeral muscular dystrophy. PLoS One 13, e0192657 (2018).
Banerji, C. R. S. et al. PAX7 target genes are globally repressed in facioscapulohumeral muscular dystrophy skeletal muscle. Nat. Commun. 8, 2152 (2017).
Bosnakovski, D. et al. An isogenetic myoblast expression screen identifies DUX4-mediated FSHD-associated molecular pathologies. EMBO J. 27, 2766–2779 (2008).
Banerji, C. R. S. & Zammit, P. S. PAX7 target gene repression is a superior FSHD biomarker than DUX4 target gene activation, associating with pathological severity and identifying FSHD at the single-cell level. Hum. Mol. Genet. 28, 2224–2236 (2019).
Banerji, C. R. S. PAX7 target gene repression associates with FSHD progression and pathology over one year. Hum. Mol. Genet. 29, 2124–2133 (2020).
Petek, L. M. et al.A cross sectional study of two independent cohorts identifies serum biomarkers for facioscapulohumeral muscular dystrophy (FSHD). Neuromuscul. Disord. 26, 405–413 (2016).
Statland, J., Donlin-Smith, C. M., Tapscott, S. J., van der Maarel, S. & Tawil, R. Multiplex screen of serum biomarkers in facioscapulohumeral muscular dystrophy. J. Neuromuscul. Dis. 1, 181–190 (2014).
Signorelli, M. et al. Evaluation of blood gene expression levels in facioscapulohumeral muscular dystrophy patients. Sci. Rep. 10, 17547 (2020).
Heier, C. R. et al. Multi-omics identifies circulating miRNA and protein biomarkers for facioscapulohumeral dystrophy. J. Pers. Med. 10, 236 (2020).
Corasolla Carregari, V. et al. Proteomics of muscle microdialysates identifies potential circulating biomarkers in facioscapulohumeral muscular dystrophy. Int. J. Mol. Sci. 22, 290 (2020).
Gros, M. et al. Identification of serum interleukin 6 levels as a disease severity biomarker in facioscapulohumeral muscular dystrophy. J. Neuromuscul. Dis. 9, 83–93 (2022).
Wong, C. J. et al. Elevated plasma complement components in facioscapulohumeral dystrophy. Hum. Mol. Genet. 31, 1821–1829 (2022).
Nunes, A. M., Ramirez, M., Jones, T. I. & Jones, P. L. Identification of candidate miRNA biomarkers for facioscapulohumeral muscular dystrophy using DUX4-based mouse models. Dis. Model. Mech. 14, dmm049016 (2021).
Tasca, G. et al. Magnetic resonance imaging in a large cohort of facioscapulohumeral muscular dystrophy patients: pattern refinement and implications for clinical trials. Ann. Neurol. 79, 854–864 (2016).
Regula, J. U. et al. Clinical muscle testing compared with whole-body magnetic resonance imaging in facio-scapulo-humeral muscular dystrophy. Clin. Neuroradiol. 26, 445–455 (2016).
Olsen, D. B., Gideon, P., Jeppesen, T. D. & Vissing, J. Leg muscle involvement in facioscapulohumeral muscular dystrophy assessed by MRI. J. Neurol. 253, 1437–1441 (2006).
Leung, D. G., Carrino, J. A., Wagner, K. R. & Jacobs, M. A. Whole-body magnetic resonance imaging evaluation of facioscapulohumeral muscular dystrophy. Muscle Nerve 52, 512–520 (2015).
Lareau-Trudel, E. et al. Muscle quantitative MR imaging and clustering analysis in patients with facioscapulohumeral muscular dystrophy type 1. PLoS ONE 10, e0132717 (2015).
Dahlqvist, J. R., Vissing, C. R., Thomsen, C. & Vissing, J. Severe paraspinal muscle involvement in facioscapulohumeral muscular dystrophy. Neurology 83, 1178–1183 (2014).
Goselink, R. J. M. et al. Muscle ultrasound is a responsive biomarker in facioscapulohumeral dystrophy. Neurology 94, e1488–e1494 (2020).
Vera, K. A., McConville, M., Kyba, M. & Keller-Ross, M. L. Sarcopenic obesity in facioscapulohumeral muscular dystrophy. Front. Physiol. 11, 1008 (2020).
U.S. Food and Drug Administration. Guidance document: patient-reported outcome measures: use in medical product development to support labeling claims. Guidance for Industry (docket number FDA-2006-D-0362). https://www.fda.gov/regulatory-information/search-fda-guidance-documents/patient-reported-outcome-measures-use-medical-product-development-support-labeling-claims (2009).
European Medicines Agency. Appendix 2 to the guideline on the evaluation of anticancer medicinal products in man. The use of patient-reported outcome (PRO) measures in oncology studies (EMA/CHMP/292464/2014). https://www.ema.europa.eu/en/documents/other/appendix-2-guideline-evaluation-anticancer-medicinal-products-man_en.pdf (2016).
Mul, K., Horlings, C. G. C., Faber, C. G., van Engelen, B. G. M. & Merkies, I. S. J. Rasch analysis to evaluate the motor function measure for patients with facioscapulohumeral muscular dystrophy. Int. J. Rehabil. Res. 44, 38–44 (2020).
Eichinger, K. et al. Facioscapulohumeral muscular dystrophy functional composite outcome measure. Muscle Nerve 58, 72–78 (2018).
Han, J. J. et al. Reachable workspace reflects dynamometer-measured upper extremity strength in facioscapulohumeral muscular dystrophy. Muscle Nerve 52, 948–955 (2015).
Hatch, M. N. et al. Longitudinal study of upper extremity reachable workspace in fascioscapulohumeral muscular dystrophy. Neuromuscul. Disord. 29, 503–513 (2019).
Hatch, M. N., Kurillo, G., Chan, V. & Han, J. J. Motion sensor-acquired reachable workspace correlates with patient-reported upper extremity activities of daily living (ADL) function in facioscapulohumeral dystrophy. Muscle Nerve 63, 250–257 (2021).
Hamel, J. et al. Patient-reported symptoms in facioscapulohumeral muscular dystrophy (PRISM-FSHD). Neurology 93, e1180–e1192 (2019).
Acknowledgements
The authors apologize to those whose work was not cited or insufficiently cited because of size constraints. Our work is supported by the Prinses Beatrix Spierfonds (W.OR19-06, W.OR21-04), public–private partnerships allowances made available by Health-Holland, Top Sector Life Sciences and Health, The Netherlands, the Medical Research Council UK, Stichting Spieren voor Spieren, the National Institute of Neurological Disorders and Stroke (Grant No. P01NS069539) and the National Institute of Arthritis and Musculoskeletal and Skin Diseases (Grant No. P50AR065139). Some of the authors are members of the Radboud University Medical Centre (UMC), Center of Expertise for Neuromuscular Disorders (Radboud-NMD), Netherlands Neuromuscular Center (NL-NMD) and the European Reference Network for Rare Neuromuscular Diseases [ERN EURO-NMD].
Author information
Authors and Affiliations
Contributions
M.S.T. and K.M. contributed equally to all aspects of the article. S.M.v.d.M., S.J.T., R.T. and J.M.S. contributed substantially to discussions of the article content and to review and/or editing of manuscript before submission. J.B. researched data for the article and J.C.d.G. contributed to review and/or editing of the article before submission.
Corresponding author
Ethics declarations
Competing interests
K.M. declares that she has acted as a consultant for Avidity Biosciences. S.J.T. declares that he has acted as a consultant for Avidity Biosciences and is a Board member for Renogenyx. R.T. declares that he has acted as a consultant and/or is a member of the advisory board for Arrowhead Pharma, Avidity Biosciences, Dyne Therapeutics, Fulcrum Therapeutics, Mitsubishi Tanabe Pharma, miRecule Biotech and Roche. J.M.S. declares that he has acted as a consultant and/or is a member of the advisory board for Acceleron, Avidity Biosciences, Dyne Therapeutics, Fulcrum Therapeutics, Ionis, ML Bio Solutions, Mitsubishi Tanabe Pharma, Roche and Sarepta. S.M.v.d.M. declares that he has acted as consultant and/or is a member of the advisory board for Avidity Biosciences, Dyne Therapeutics and Fulcrum Therapeutics and is a Board member for Renogenyx. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Neurology thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- cis-spreading
-
Spreading of epigenetic chromatin modifications from a locus to neighbouring loci on the same chromatin strand.
- FSHD Composite Outcome Measure
-
(FSHD-COM) An evaluator-administered instrument composed of individually validated functional motor tasks that assess leg, shoulder and arm, trunk and hand function and balance.
- FSHD Health Index
-
(FSHD-HI) A patient-reported outcome measures to assess total FSHD health-related quality of life and 14 separate subdomains, each identified by FSHD patients as having the greatest importance to their specific population.
- FSHD-Rasch-built Overall Disability Scale
-
(FSHD-RODS) A linear-weighted patient-reported outcome measure for detecting activity and participation restrictions in patients with FSHD.
- PAX7 score
-
The ratio of genes induced to genes repressed indicated by the satellite cell marker PAX7, which is closely related to DUX4.
- Perdurance
-
Lasting target gene expression followed after a short DUX4 transcriptional burst.
- pLAM
-
A DNA region flanking the most distal D4Z4 unit that contains the polyadenylation signal (PAS) of the DUX4 gene, named after the cosmid clone used for its isolation.
- Proximity ligation assay
-
An antibody assay based on rolling-circle amplification of a DNA loop formed from ligation of oligonucleotide-labelled probes bound to distinct primary antibodies, used for detection of proteins at single-molecule resolution in unmodified cells and tissues.
- Pseudotime trajectory modelling
-
A computational method used in single-cell transcriptomics to establish the pattern of a dynamic cellular process; followed by ordering of cells based on their progression through the process.
- Reachable Workspace
-
(RWS) A depth-ranging sensor (Kinect)-based upper extremity motion analysis system that is applied to determine the spectrum of reachable workspace encountered with the arms and shoulders.
- Retrogene
-
A segment of DNA reverse-transcribed from mRNA and randomly inserted into a genome.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tihaya, M.S., Mul, K., Balog, J. et al. Facioscapulohumeral muscular dystrophy: the road to targeted therapies. Nat Rev Neurol 19, 91–108 (2023). https://doi.org/10.1038/s41582-022-00762-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41582-022-00762-2
This article is cited by
-
Genetisch-basierte Therapien bei Muskelkrankheiten
InFo Neurologie + Psychiatrie (2023)