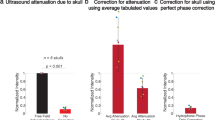
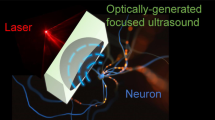
Abstract
Focused ultrasound (FUS) is a disruptive medical technology, and its implementation in the clinic represents the culmination of decades of research. Lying at the convergence of physics, engineering, imaging, biology and neuroscience, FUS offers the ability to non-invasively and precisely intervene in key circuits that drive common and challenging brain conditions. The actions of FUS in the brain take many forms, ranging from transient blood–brain barrier opening and neuromodulation to permanent thermoablation. Over the past 5 years, we have seen a dramatic expansion of indications for and experience with FUS in humans, with a resultant exponential increase in academic and public interest in the technology. Applications now span the clinical spectrum in neurological and psychiatric diseases, with insights still emerging from preclinical models and human trials. In this Review, we provide a comprehensive overview of therapeutic ultrasound and its current and emerging indications in the brain. We examine the potential impact of FUS on the landscape of brain therapies as well as the challenges facing further advancement and broader adoption of this promising minimally invasive therapeutic alternative.
Key points
-
Recent advances have led to a surge of interest in focused ultrasound (FUS) as a non-invasive, potentially disruptive tool for the most intractable neurological conditions.
-
Magnetic resonance-guided FUS thermoablation has been approved for the treatment of essential tremor and tremor-dominant Parkinson disease and is being investigated in psychiatric applications as well as in chronic pain and epilepsy.
-
Transient opening of the blood–brain barrier for drug delivery is a burgeoning field, with early human studies demonstrating a favourable safety profile as well as versatility across and scalability within a range of clinical indications.
-
Future studies will investigate the delivery of established pharmaceuticals and novel therapies in combination with FUS blood–brain barrier opening.
-
Emerging applications are also harnessing the myriad of ways in which FUS can interact with the CNS, including immune modulation and neuromodulation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Aubry, J.-F. et al. The road to clinical use of high-intensity focused ultrasound for liver cancer: technical and clinical consensus. J. Ther. Ultrasound 1, 13 (2013).
Tempany, C. M. C., McDannold, N. J., Hynynen, K. & Jolesz, F. A. Focused ultrasound surgery in oncology: overview and principles. Radiology 259, 39–56 (2011).
El-Hayek, Y. H. et al. Tip of the iceberg: assessing the global socioeconomic costs of Alzheimer’s disease and related dementias and strategic implications for stakeholders. J. Alzheimers Dis. 70, 323–341 (2019).
Aldape, K. et al. Challenges to curing primary brain tumours. Nat. Rev. Clin. Oncol. 16, 509–520 (2019).
Makin, S. The amyloid hypothesis on trial. Nature 559, S4–S7 (2018).
Lozano, A. M. et al. A phase II study of fornix deep brain stimulation in mild Alzheimer’s disease. J. Alzheimers Dis. 54, 777–787 (2016).
Elias, W. J. et al. A randomized trial of focused ultrasound thalamotomy for essential tremor. N. Engl. J. Med. 375, 730–739 (2016). This pivotal study led to regulatory approval of the first approved indication for MRgFUS thermoablation in the treatment of essential tremor.
Bond, A. E. et al. Safety and efficacy of focused ultrasound thalamotomy for patients with medication-refractory, tremor-dominant Parkinson disease: a randomized clinical trial. JAMA Neurol. 74, 1412–1418 (2017). This pivotal study led to regulatory approval of the second — and, to date, only other — approved clinical indication for MRgFUS thermoablation in the treatment of TDPD.
Leinenga, G., Langton, C., Nisbet, R. & Götz, J. Ultrasound treatment of neurological diseases — current and emerging applications. Nat. Rev. Neurol. 12, 161–174 (2016).
Gandaglia, G. et al. Effect of minimally invasive surgery on the risk for surgical site infections: results from the National Surgical Quality Improvement Program (NSQIP) Database. JAMA Surg. 149, 1039–1044 (2014).
Hynynen, K. & Jones, R. M. Image-guided ultrasound phased arrays are a disruptive technology for non-invasive therapy. Phys. Med. Biol. 61, R206–R248 (2016).
Raymond, S. B. & Hynynen, K. Acoustic transmission losses and field alterations due to human scalp hair. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 52, 1415–1419 (2005).
Meyers, R. et al. Early experiences with ultrasonic irradiation of the pallidofugal and nigral complexes in hyperkinetic and hypertonic disorders. J. Neurosurg. 16, 32–54 (1959).
Nelson, E., Lindstrom, P. A. & Haymaker, W. Pathological effects of ultrasound on the human brain: a study of 25 cases in which ultrasonic irradiation was used as a lobotomy procedure. J. Neuropathol. Exp. Neurol. 18, 489–508 (1959).
Leksell, L. Echo-encephalography. I. Detection of intracranial complications following head injury. Acta Chir. Scand. 110, 301–315 (1956).
Jagannathan, J. et al. High-intensity focused ultrasound surgery of the brain: part 1 — a historical perspective with modern applications. Neurosurgery 64, 201–210 (2009).
Guthkelch, A. N. et al. Treatment of malignant brain tumors with focused ultrasound hyperthermia and radiation: results of a phase I trial. J. Neurooncol. 10, 271–284 (1991).
Ram, Z. et al. Magnetic resonance imaging-guided, high-intensity focused ultrasound for brain tumor therapy. Neurosurgery 59, 949–955 (2006).
Hynynen, K. et al. Pre-clinical testing of a phased array ultrasound system for MRI-guided noninvasive surgery of the brain — a primate study. Eur. J. Radiol. 59, 149–156 (2006).
Clement, G. T. & Hynynen, K. A non-invasive method for focusing ultrasound through the human skull. Phys. Med. Biol. 47, 1219–1236 (2002).
Aubry, J.-F. & Tanter, M. MR-guided transcranial focused ultrasound. Adv. Exp. Med. Biol. 880, 97–111 (2016).
Haworth, K. J., Fowlkes, J. B., Carson, P. L. & Kripfgans, O. D. Towards aberration correction of transcranial ultrasound using acoustic droplet vaporization. Ultrasound Med. Biol. 34, 435–445 (2008).
Hynynen, K., Darkazanli, A., Unger, E. & Schenck, J. F. MRI-guided noninvasive ultrasound surgery. Med. Phys. 20, 107–115 (1993).
Jeanmonod, D. et al. Transcranial magnetic resonance imaging-guided focused ultrasound: noninvasive central lateral thalamotomy for chronic neuropathic pain. Neurosurg. Focus 32, E1 (2012).
Carpentier, A. et al. Clinical trial of blood–brain barrier disruption by pulsed ultrasound. Sci. Transl. Med. 8, 343re2 (2016).
Maimbourg, G., Houdouin, A., Deffieux, T., Tanter, M. & Aubry, J.-F. 3D-printed adaptive acoustic lens as a disruptive technology for transcranial ultrasound therapy using single-element transducers. Phys. Med. Biol. 63, 025026 (2018).
Haar, G. T. & Coussios, C. High intensity focused ultrasound: physical principles and devices. Int. J. Hyperth. 23, 89–104 (2007).
Mouratidis, P. X. E., Rivens, I., Civale, J., Symonds-Tayler, R. & Ter Haar, G. ‘Relationship between thermal dose and cell death for “rapid” ablative and “slow” hyperthermic heating’. Int. J. Hyperth. 36, 228–242 (2019).
Hynynen, K., McDannold, N., Vykhodtseva, N. & Jolesz, F. A. Noninvasive MR imaging-guided focal opening of the blood–brain barrier in rabbits. Radiology 220, 640–646 (2001).
Sukovich, J. R. et al. In vivo histotripsy brain treatment. J. Neurosurg. 131, 1331–1338 (2019).
Lozano, A. M. et al. Deep brain stimulation: current challenges and future directions. Nat. Rev. Neurol. 15, 148–160 (2019).
Deuschl, G. et al. A randomized trial of deep-brain stimulation for Parkinson’s disease. N. Engl. J. Med. 355, 896–908 (2006).
McDannold, N., Clement, G. T., Black, P., Jolesz, F. & Hynynen, K. Transcranial magnetic resonance imaging-guided focused ultrasound surgery of brain tumors: initial findings in 3 patients. Neurosurgery 66, 323–332 (2010).
Coluccia, D. et al. First noninvasive thermal ablation of a brain tumor with MR-guided focused ultrasound. J. Ther. Ultrasound 2, 17 (2014).
Jung, N. Y. et al. Factors related to successful energy transmission of focused ultrasound through a skull: a study in human cadavers and its comparison with clinical experiences. J. Korean Neurosurg. Soc. 62, 712–722 (2019).
Benito-León, J. & Louis, E. D. Essential tremor: emerging views of a common disorder. Nat. Rev. Neurol. 2, 666–678 (2006).
Elble, R. J. The essential tremor syndromes. Curr. Opin. Neurol. 29, 507–512 (2016).
Elble, R. J. Mechanisms of deep brain stimulation for essential tremor. Brain 137, 4–6 (2014).
Sharifi, S., Nederveen, A. J., Booij, J. & van Rootselaar, A.-F. Neuroimaging essentials in essential tremor: a systematic review. Neuroimage Clin. 5, 217–231 (2014).
Dallapiazza, R. F. et al. Outcomes from stereotactic surgery for essential tremor. J. Neurol. Neurosurg. Psychiatry 90, 474–482 (2019).
Lipsman, N. et al. MR-guided focused ultrasound thalamotomy for essential tremor: a proof-of-concept study. Lancet Neurol. 12, 462–468 (2013).
Elias, W. J. et al. A pilot study of focused ultrasound thalamotomy for essential tremor. N. Engl. J. Med. 369, 640–648 (2013).
Scantlebury, N. et al. Change in some quality of life domains mimics change in tremor severity after ultrasound thalamotomy. Mov. Disord. 34, 1400–1401 (2019).
Chang, J. W. et al. A prospective trial of magnetic resonance guided focused ultrasound thalamotomy for essential tremor: results at the 2-year follow-up. Ann. Neurol. 83, 107–114 (2017).
Meng, Y. et al. Magnetic resonance-guided focused ultrasound thalamotomy for treatment of essential tremor: a 2-year outcome study: MRgFUS thalamotomy for ET: 2-year outcome. Mov. Disord. 33, 1647–1650 (2018).
Park, Y.-S., Jung, N. Y., Na, Y. C. & Chang, J. W. Four-year follow-up results of magnetic resonance-guided focused ultrasound thalamotomy for essential tremor. Mov. Disord. 34, 727–734 (2019).
Weidman, E. K., Kaplitt, M. G., Strybing, K. & Chazen, J. L. Repeat magnetic resonance imaging-guided focused ultrasound thalamotomy for recurrent essential tremor: case report and review of MRI findings. J. Neurosurg. 132, 211–216 (2020).
Fishman, P. S. et al. Neurological adverse event profile of magnetic resonance imaging-guided focused ultrasound thalamotomy for essential tremor. Mov. Disord. 33, 843–847 (2018).
Boutet, A. et al. Focused ultrasound thalamotomy location determines clinical benefits in patients with essential tremor. Brain 141, 3405–3414 (2018).
Pineda-Pardo, J. A. et al. Transcranial magnetic resonance-guided focused ultrasound thalamotomy in essential tremor: a comprehensive lesion characterization. Neurosurgery 87, 256–265 (2019).
Wintermark, M. et al. Thalamic connectivity in patients with essential tremor treated with MR imaging-guided focused ultrasound: in vivo fiber tracking by using diffusion-tensor MR imaging. Radiology 272, 202–209 (2014).
Pineda-Pardo, J. A. et al. Microstructural changes of the dentato-rubro-thalamic tract after transcranial MR guided focused ultrasound ablation of the posteroventral VIM in essential tremor. Hum. Brain Mapp. 40, 2933–2942 (2019).
Pouratian, N., Baltuch, G., Elias, W. J. & Gross, R. American Society for Stereotactic and Functional Neurosurgery position statement on magnetic resonance-guided focused ultrasound for the management of essential tremor. Neurosurgery 87, E126–E129 (2020).
Ravikumar, V. K. et al. Cost-effectiveness of focused ultrasound, radiosurgery, and DBS for essential tremor. Mov. Disord. 32, 1165–1173 (2017).
Li, C. et al. Cost-effectiveness of magnetic resonance-guided focused ultrasound for essential tremor. Mov. Disord. 34, 735–743 (2019).
Horisawa, S. et al. A single case of MRI-guided focused ultrasound ventro-oral thalamotomy for musician’s dystonia. J. Neurosurg. 131, 384–386 (2018).
Meng, Y., Suppiah, S., Scantlebury, N., Lipsman, N. & Schwartz, M. L. Treatment of a patient with task-specific writing tremor using magnetic resonance-guided focused ultrasound. Can. J. Neurol. Sci. 45, 474–477 (2018).
Fasano, A. et al. MRI-guided focused ultrasound thalamotomy in non-ET tremor syndromes. Neurology 89, 771–775 (2017).
Poewe, W. et al. Parkinson disease. Nat. Rev. Dis. Primers 3, 17013 (2017).
Kalia, S. K., Sankar, T. & Lozano, A. M. Deep brain stimulation for Parkinson’s disease and other movement disorders. Curr. Opin. Neurol. 26, 374–380 (2013).
Prasad, S. et al. Spinal cord stimulation for very advanced Parkinson’s disease: a 1-year prospective trial. Mov. Disord. 35, 1082–1083 (2020).
Stefani, A. et al. Bilateral deep brain stimulation of the pedunculopontine and subthalamic nuclei in severe Parkinson’s disease. Brain 130, 1596–1607 (2007).
López-Azcárate, J. et al. Coupling between beta and high-frequency activity in the human subthalamic nucleus may be a pathophysiological mechanism in Parkinson’s disease. J. Neurosci. 30, 6667–6677 (2010).
Martínez-Fernández, R. et al. Focused ultrasound subthalamotomy in patients with asymmetric Parkinson’s disease: a pilot study. Lancet Neurol. 17, 54–63 (2018). This small open-label trial showed that unilateral MRgFUS subthalamotomy was technically feasible, effective and associated with a relatively low risk of hemichorea–ballism.
Jung, N. Y. et al. The efficacy and limits of magnetic resonance-guided focused ultrasound pallidotomy for Parkinson’s disease: a phase I clinical trial. J. Neurosurg. 130, 1853–1861 (2018).
Gallay, M. N. et al. MRgFUS pallidothalamic tractotomy for chronic therapy-resistant Parkinson’s disease in 51 consecutive patients: single center experience. Front. Surg. 6, 76 (2020).
Alvarez, L. Bilateral subthalamotomy in Parkinson’s disease: initial and long-term response. Brain 128, 570–583 (2005).
Meng, Y. et al. Cost-effectiveness analysis of MR-guided focused ultrasound thalamotomy for tremor-dominant Parkinson’s disease. J. Neurosurg. https://doi.org/10.3171/2020.5.JNS20692 (2020).
Stein, D. J. et al. Obsessive–compulsive disorder. Nat. Rev. Dis. Primers 5, 52 (2019).
Garnaat, S. L. et al. Who qualifies for deep brain stimulation for OCD? Data from a naturalistic clinical sample. J. Neuropsychiatry Clin. Neurosci. 26, 81–86 (2014).
Pauls, D. L., Abramovitch, A., Rauch, S. L. & Geller, D. A. Obsessive–compulsive disorder: an integrative genetic and neurobiological perspective. Nat. Rev. Neurosci. 15, 410–424 (2014).
Whiteside, S. P., Port, J. D. & Abramowitz, J. S. A meta-analysis of functional neuroimaging in obsessive–compulsive disorder. Psychiatry Res. 132, 69–79 (2004).
Hamani, C. et al. Deep brain stimulation for obsessive–compulsive disorder. Neurosurgery 75, 327–333 (2014).
Mallet, L. et al. Subthalamic nucleus stimulation in severe obsessive–compulsive disorder. N. Engl. J. Med. 359, 2121–2134 (2008).
Denys, D. et al. Deep brain stimulation of the nucleus accumbens for treatment-refractory obsessive–compulsive disorder. Arch. Gen. Psychiatry 67, 1061–1068 (2010).
Rück, C. et al. Capsulotomy for obsessive–compulsive disorder: long-term follow-up of 25 patients. Arch. Gen. Psychiatry 65, 914–921 (2008).
Alonso, P. et al. Deep brain stimulation for obsessive–compulsive disorder: a meta-analysis of treatment outcome and predictors of response. PLoS ONE 10, e0133591 (2015).
Jung, H. H. et al. Bilateral thermal capsulotomy with MR-guided focused ultrasound for patients with treatment-refractory obsessive–compulsive disorder: a proof-of-concept study. Mol. Psychiatry 20, 1205–1211 (2015). The first use of MRgFUS thermoablation for bilateral anterior capsulotomy to treat a psychiatric disorder.
Kim, S. J. et al. A study of novel bilateral thermal capsulotomy with focused ultrasound for treatment-refractory obsessive–compulsive disorder: 2-year follow-up. J. Psychiatry Neurosci. 43, 170188 (2018).
Brown, L. T. et al. Dorsal anterior cingulotomy and anterior capsulotomy for severe, refractory obsessive–compulsive disorder: a systematic review of observational studies. J. Neurosurg. 124, 77–89 (2016).
Davidson, B. et al. Magnetic resonance-guided focused ultrasound capsulotomy for refractory obsessive compulsive disorder and major depressive disorder: clinical and imaging results from two phase I trials. Mol. Psychiatry 25, 1946–1957 (2020).
Otte, C. et al. Major depressive disorder. Nat. Rev. Dis. Primers 2, 16065 (2016).
Kim, M., Kim, C.-H., Jung, H. H., Kim, S. J. & Chang, J. W. Treatment of major depressive disorder via magnetic resonance-guided focused ultrasound surgery. Biol. Psychiatry 83, e17–e18 (2018).
Treede, R.-D. et al. A classification of chronic pain for ICD-11. Pain 156, 1003–1007 (2015).
The Lancet Neurology. Novel ways to manage chronic pain are needed. Lancet Neurol. 17, 829 (2018).
Burchiel, K. J. & Raslan, A. M. Contemporary concepts of pain surgery. J. Neurosurg. 130, 1039–1049 (2019).
Martin, E., Jeanmonod, D., Morel, A., Zadicario, E. & Werner, B. High-intensity focused ultrasound for noninvasive functional neurosurgery. Ann. Neurol. 66, 858–861 (2009). The first report of incisionless surgery using MRgFUS thermoablation in humans, undertaken in patients with chronic pain.
Clary, A., Tyler, W. J. & Wetmore, D. Z. Abstract #45: ultrasound neuromodulation for the treatment of peripheral nerve compression syndromes. Brain Stimul. 12, e16 (2019).
Sweeney, M. D., Sagare, A. P. & Zlokovic, B. V. Blood–brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nat. Rev. Neurol. 14, 133–150 (2018).
Obermeier, B., Verma, A. & Ransohoff, R. M. The blood–brain barrier. Handb. Clin. Neurol. 133, 39–59 (2016).
Galea, I., Bechmann, I. & Perry, V. H. What is immune privilege (not)? Trends Immunol. 28, 12–18 (2007).
van Tellingen, O. et al. Overcoming the blood–brain tumor barrier for effective glioblastoma treatment. Drug Resist. Updat. 19, 1–12 (2015).
Arvanitis, C. D., Ferraro, G. B. & Jain, R. K. The blood–brain barrier and blood–tumour barrier in brain tumours and metastases. Nat. Rev. Cancer 20, 26–41 (2020).
Garbuzova-Davis, S., Thomson, A., Kurien, C., Shytle, R. D. & Sanberg, P. R. Potential new complication in drug therapy development for amyotrophic lateral sclerosis. Expert Rev. Neurother. 16, 1397–1405 (2016).
Pardridge, W. M. The blood–brain barrier: bottleneck in brain drug development. NeuroRX 2, 3–14 (2005).
Jablonski, M. R. et al. Inhibiting drug efflux transporters improves efficacy of ALS therapeutics. Ann. Clin. Transl. Neurol. 1, 996–1005 (2014).
Meng, Y. et al. Safety and efficacy of focused ultrasound induced blood–brain barrier opening, an integrative review of animal and human studies. J. Control. Release 309, 25–36 (2019).
O’Reilly, M. A., Waspe, A. C., Chopra, R. & Hynynen, K. MRI-guided disruption of the blood–brain barrier using transcranial focused ultrasound in a rat model. J. Vis. Exp. 61, 3555 (2012).
Pelekanos, M. et al. Establishing sheep as an experimental species to validate ultrasound-mediated blood–brain barrier opening for potential therapeutic interventions. Theranostics 8, 2583–2602 (2018).
Kovacs, Z. I. et al. Disrupting the blood–brain barrier by focused ultrasound induces sterile inflammation. Proc. Natl Acad. Sci. USA 114, E75–E84 (2017).
Poon, C. T. et al. Time course of focused ultrasound effects on β-amyloid plaque pathology in the TgCRND8 mouse model of Alzheimer’s disease. Sci. Rep. 8, 14061 (2018).
Jordão, J. F. et al. Amyloid-β plaque reduction, endogenous antibody delivery and glial activation by brain-targeted, transcranial focused ultrasound. Exp. Neurol. 248, 16–29 (2013).
McMahon, D., Bendayan, R. & Hynynen, K. Acute effects of focused ultrasound-induced increases in blood–brain barrier permeability on rat microvascular transcriptome. Sci. Rep. 7, 45657 (2017).
McMahon, D. & Hynynen, K. Acute inflammatory response following increased blood–brain barrier permeability induced by focused ultrasound is dependent on microbubble dose. Theranostics 7, 3989–4000 (2017).
Olumolade, O. O., Wang, S., Samiotaki, G. & Konofagou, E. E. Longitudinal motor and behavioral assessment of blood–brain barrier opening with transcranial focused ultrasound. Ultrasound Med. Biol. 42, 2270–2282 (2016).
Horodyckid, C. et al. Safe long-term repeated disruption of the blood–brain barrier using an implantable ultrasound device: a multiparametric study in a primate model. J. Neurosurg. 126, 1351–1361 (2017).
Kinoshita, M., McDannold, N., Jolesz, F. A. & Hynynen, K. Noninvasive localized delivery of Herceptin to the mouse brain by MRI-guided focused ultrasound-induced blood–brain barrier disruption. Proc. Natl Acad. Sci. USA 103, 11719–11723 (2006).
Wu, S.-K. et al. Characterization of different microbubbles in assisting focused ultrasound-induced blood–brain barrier opening. Sci. Rep. 7, 46689 (2017).
McDannold, N., Vykhodtseva, N. & Hynynen, K. Effects of acoustic parameters and ultrasound contrast agent dose on focused-ultrasound induced blood–brain barrier disruption. Ultrasound Med. Biol. 34, 930–937 (2008).
Chen, H. & Konofagou, E. E. The size of blood–brain barrier opening induced by focused ultrasound is dictated by the acoustic pressure. J. Cereb. Blood Flow Metab. 34, 1197–1204 (2014).
Jordão, J. F. et al. Antibodies targeted to the brain with image-guided focused ultrasound reduces amyloid-beta plaque load in the TgCRND8 mouse model of Alzheimer’s disease. PLoS ONE 5, e10549 (2010).
Kobus, T., Zervantonakis, I. K., Zhang, Y. & McDannold, N. J. Growth inhibition in a brain metastasis model by antibody delivery using focused ultrasound-mediated blood–brain barrier disruption. J. Control. Release 238, 281–288 (2016).
Liu, H.-L. et al. Focused ultrasound enhances central nervous system delivery of bevacizumab for malignant glioma treatment. Radiology 281, 99–108 (2016).
Alecou, T., Giannakou, M. & Damianou, C. Amyloid β plaque reduction with antibodies crossing the blood–brain barrier, which was opened in 3 sessions of focused ultrasound in a rabbit model. J. Ultrasound Med. 36, 2257–2270 (2017).
Alli, S. et al. Brainstem blood brain barrier disruption using focused ultrasound: a demonstration of feasibility and enhanced doxorubicin delivery. J. Control. Release 281, 29–41 (2018).
Coluccia, D. et al. Enhancing glioblastoma treatment using cisplatin-gold-nanoparticle conjugates and targeted delivery with magnetic resonance-guided focused ultrasound. Nanomedicine 14, 1137–1148 (2018).
Thévenot, E. et al. Targeted delivery of self-complementary adeno-associated virus serotype 9 to the brain, using magnetic resonance imaging-guided focused ultrasound. Hum. Gene Ther. 23, 1144–1155 (2012).
Burgess, A. et al. Targeted delivery of neural stem cells to the brain using MRI-guided focused ultrasound to disrupt the blood–brain barrier. PLoS ONE 6, e27877 (2011).
Alkins, R. et al. Focused ultrasound delivers targeted immune cells to metastatic brain tumors. Cancer Res. 73, 1892–1899 (2013).
Alkins, R., Burgess, A., Kerbel, R., Wels, W. S. & Hynynen, K. Early treatment of HER2-amplified brain tumors with targeted NK-92 cells and focused ultrasound improves survival. Neuro Oncol. 18, 974–981 (2016).
Noroozian, Z. et al. MRI-guided focused ultrasound for targeted delivery of rAAV to the brain. Methods Mol. Biol. 1950, 177–197 (2019).
Lipsman, N. et al. Blood–brain barrier opening in Alzheimer’s disease using MR-guided focused ultrasound. Nat. Commun. 9, 2336 (2018). The first report that transcranial FUS BBBO is safe in people with mild-to-moderate AD.
Pouliopoulos, A. N. et al. A clinical system for non-invasive blood–brain barrier opening using a neuronavigation-guided single-element focused ultrasound transducer. Ultrasound Med. Biol. 46, 73–89 (2020).
Asquier, N. et al. Blood–brain barrier disruption in humans using an implantable ultrasound device: quantification with MR images and correlation with local acoustic pressure. J. Neurosurg. 132, 875–883 (2019).
Beccaria, K. et al. Blood–brain barrier disruption with low-intensity pulsed ultrasound for the treatment of pediatric brain tumors: a review and perspectives. Neurosurg. Focus. 48, E10 (2020).
Alzheimer’s Association. 2017 Alzheimer’s disease facts and figures. Alzheimers Dement. 13, 325–373 (2017).
Masters, C. L. et al. Alzheimer’s disease. Nat. Rev. Dis. Primers 1, 15056 (2015).
Greenberg, S. M. et al. Cerebral amyloid angiopathy and Alzheimer disease — one peptide, two pathways. Nat. Rev. Neurol. 16, 30–42 (2020).
Nisbet, R. M. et al. Combined effects of scanning ultrasound and a tau-specific single chain antibody in a tau transgenic mouse model. Brain 140, 1220–1230 (2017).
Xhima, K. et al. Focused ultrasound delivery of a selective TrkA agonist rescues cholinergic function in a mouse model of Alzheimer’s disease. Sci. Adv. 6, eaax6646 (2020).
Burgess, A. et al. Alzheimer disease in a mouse model: MR imaging-guided focused ultrasound targeted to the hippocampus opens the blood–brain barrier and improves pathologic abnormalities and behavior. Radiology 273, 736–745 (2014).
Leinenga, G. & Götz, J. Scanning ultrasound removes amyloid-β and restores memory in an Alzheimer’s disease mouse model. Sci. Transl. Med. 7, 278ra33 (2015).
Leinenga, G. & Götz, J. Safety and efficacy of scanning ultrasound treatment of aged APP23 mice. Front. Neurosci. 12, 55 (2018).
Scarcelli, T. et al. Stimulation of hippocampal neurogenesis by transcranial focused ultrasound and microbubbles in adult mice. Brain Stimul. 7, 304–307 (2014).
Mooney, S. J. et al. Focused ultrasound-induced neurogenesis requires an increase in blood–brain barrier permeability. PLoS ONE 11, e0159892 (2016).
Nation, D. A. et al. Blood–brain barrier breakdown is an early biomarker of human cognitive dysfunction. Nat. Med. 25, 270–276 (2019).
Rezai, A. R. et al. Noninvasive hippocampal blood−brain barrier opening in Alzheimer’s disease with focused ultrasound. Proc. Natl Acad. Sci. USA 117, 9180–9182 (2020).
Panza, F., Lozupone, M., Logroscino, G. & Imbimbo, B. P. A critical appraisal of amyloid-β-targeting therapies for Alzheimer disease. Nat. Rev. Neurol. 15, 73–88 (2019).
Hardiman, O. et al. Amyotrophic lateral sclerosis. Nat. Rev. Dis. Primers 3, 17085 (2017).
Hobson, E. V. & McDermott, C. J. Supportive and symptomatic management of amyotrophic lateral sclerosis. Nat. Rev. Neurol. 12, 526–538 (2016).
Hetz, C. & Saxena, S. ER stress and the unfolded protein response in neurodegeneration. Nat. Rev. Neurol. 13, 477–491 (2017).
Lacomblez, L., Bensimon, G., Meininger, V., Leigh, P. N. & Guillet, P. Dose-ranging study of riluzole in amyotrophic lateral sclerosis. Lancet 347, 1425–1431 (1996).
Edaravone (MCI-186) ALS 19 Study Group. Safety and efficacy of edaravone in well defined patients with amyotrophic lateral sclerosis: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 16, 505–512 (2017).
Geevasinga, N., Menon, P., Özdinler, P. H., Kiernan, M. C. & Vucic, S. Pathophysiological and diagnostic implications of cortical dysfunction in ALS. Nat. Rev. Neurol. 12, 651–661 (2016).
Thomsen, G. M. et al. Delayed disease onset and extended survival in the SOD1G93A rat model of amyotrophic lateral sclerosis after suppression of mutant SOD1 in the motor cortex. J. Neurosci. 34, 15587–15600 (2014).
Thomsen, G. M. et al. Transplantation of neural progenitor cells expressing glial cell line-derived neurotrophic factor into the motor cortex as a strategy to treat amyotrophic lateral sclerosis. Stem Cell 36, 1122–1131 (2018).
Abrahao, A. et al. First-in-human trial of blood–brain barrier opening in amyotrophic lateral sclerosis using MR-guided focused ultrasound. Nat. Commun. 10, 4373 (2019). The first study of transcranial FUS BBBO in eloquent cortex in people with ALS, showing that the procedure was safe and technically successful.
Axelsen, T. M. & Woldbye, D. P. D. Gene therapy for Parkinson’s disease, an update. J. Parkinsons Dis. 8, 195–215 (2018).
Xhima, K., Nabbouh, F., Hynynen, K., Aubert, I. & Tandon, A. Noninvasive delivery of an α-synuclein gene silencing vector with magnetic resonance-guided focused ultrasound. Mov. Disord. 33, 1567–1579 (2018).
Fan, C.-H. et al. Noninvasive, targeted, and non-viral ultrasound-mediated GDNF-plasmid delivery for treatment of Parkinson’s disease. Sci. Rep. 6, 19579 (2016).
Fan, C.-H., Lin, C.-Y., Liu, H.-L. & Yeh, C.-K. Ultrasound targeted CNS gene delivery for Parkinson’s disease treatment. J. Control. Release 261, 246–262 (2017).
Stupp, R. et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 352, 987–996 (2005).
Mainprize, T. et al. Blood–brain barrier opening in primary brain tumors with non-invasive MR-guided focused ultrasound: a clinical safety and feasibility study. Sci. Rep. 9, 321 (2019).
Idbaih, A. et al. Safety and feasibility of repeated and transient blood–brain barrier disruption by pulsed ultrasound in patients with recurrent glioblastoma. Clin. Cancer Res. 25, 3793–3801 (2019). This study indicated that the use of an implanted ultrasound device to deliver repeated BBBO during carboplatin administration for recurrent glioblastoma is safe and potentially enhances progression-free survival.
Razavi, S.-M. et al. Immune evasion strategies of glioblastoma. Front. Surg. 3, 11 (2016).
Chen, P.-Y. et al. Focused ultrasound-induced blood–brain barrier opening to enhance interleukin-12 delivery for brain tumor immunotherapy: a preclinical feasibility study. J. Transl. Med. 13, 93 (2015).
Sheybani, N. D., Witter, A. R., Stevens, A. D., Bullock, T. N. & Price, R. J. Focused ultrasound ablation as an immunomodulatory strategy for metastatic breast cancer therapy. J. Immunol. 200, 178.39 (2018).
Sperling, R. A. et al. Amyloid related imaging abnormalities (ARIA) in amyloid modifying therapeutic trials: recommendations from the Alzheimer’s Association Research Roundtable Workgroup. Alzheimers Dement. 7, 367–385 (2011).
Schneider, S., Potthast, S., Komminoth, P., Schwegler, G. & Böhm, S. PD-1 checkpoint inhibitor associated autoimmune encephalitis. Case Rep. Oncol. 10, 473–478 (2017).
Meng, Y. et al. Glymphatics visualization after focused ultrasound-induced blood–brain barrier opening in humans. Ann. Neurol. 86, 975–980 (2019). Localized and transient BBB opening using FUS allowed the first in vivo visualization of the glymphatic system in humans.
Engelhardt, B., Vajkoczy, P. & Weller, R. O. The movers and shapers in immune privilege of the CNS. Nat. Immunol. 18, 123–131 (2017).
Park, E.-J., Zhang, Y.-Z., Vykhodtseva, N. & McDannold, N. Ultrasound-mediated blood–brain/blood-tumor barrier disruption improves outcomes with trastuzumab in a breast cancer brain metastasis model. J. Control. Release 163, 277–284 (2012).
Park, S. H. et al. Safety and feasibility of multiple blood–brain barrier disruptions for the treatment of glioblastoma in patients undergoing standard adjuvant chemotherapy. J. Neurosurg. https://doi.org/10.3171/2019.10.JNS192206 (2020).
O’Reilly, M. A. et al. Preliminary investigation of focused ultrasound-facilitated drug delivery for the treatment of leptomeningeal metastases. Sci. Rep. 8, 9013 (2018).
Blumberger, D. M. et al. Effectiveness of theta burst versus high-frequency repetitive transcranial magnetic stimulation in patients with depression (THREE-D): a randomised non-inferiority trial. Lancet 391, 1683–1692 (2018).
Deisseroth, K. Optogenetics: 10 years of microbial opsins in neuroscience. Nat. Neurosci. 18, 1213–1225 (2015).
Dallapiazza, R. F. et al. Noninvasive neuromodulation and thalamic mapping with low-intensity focused ultrasound. J. Neurosurg. 128, 875–884 (2018).
Folloni, D. et al. Manipulation of subcortical and deep cortical activity in the primate brain using transcranial focused ultrasound stimulation. Neuron 101, 1109–1116.e5 (2019).
Nicodemus, N. E. et al. Focused transcranial ultrasound for treatment of neurodegenerative dementia. Alzheimers Dement. 5, 374–381 (2019).
Legon, W. et al. Transcranial focused ultrasound modulates the activity of primary somatosensory cortex in humans. Nat. Neurosci. 17, 322–329 (2014).
Verhagen, L. et al. Offline impact of transcranial focused ultrasound on cortical activation in primates. eLife 8, e40541 (2019). Demonstration that the neuromodulatory effect of FUS extends beyond the immediate period of sonication, which increases the translational potential of this technology.
Khalighinejad, N. et al. A basal forebrain–cingulate circuit in macaques decides it is time to act. Neuron 105, 370–384.e8 (2020).
Wattiez, N. et al. Transcranial ultrasonic stimulation modulates single-neuron discharge in macaques performing an antisaccade task. Brain Stimul. 10, 1024–1031 (2017).
Younan, Y. et al. Influence of the pressure field distribution in transcranial ultrasonic neurostimulation. Med. Phys. 40, 082902 (2013).
Sato, T., Shapiro, M. G. & Tsao, D. Y. Ultrasonic neuromodulation causes widespread cortical activation via an indirect auditory mechanism. Neuron 98, 1031–1041.e5 (2018).
Guo, H. et al. Ultrasound produces extensive brain activation via a cochlear pathway. Neuron 98, 1020–1030.e4 (2018).
Constans, C., Mateo, P., Tanter, M. & Aubry, J.-F. Potential impact of thermal effects during ultrasonic neurostimulation: retrospective numerical estimation of temperature elevation in seven rodent setups. Phys. Med. Biol. 63, 025003 (2018).
Deffieux, T. et al. Low-intensity focused ultrasound modulates monkey visuomotor behavior. Curr. Biol. 23, 2430–2433 (2013).
Yoon, K. et al. Effects of sonication parameters on transcranial focused ultrasound brain stimulation in an ovine model. PLoS ONE 14, e0224311 (2019).
Darrow, D. P., O’Brien, P., Richner, T. J., Netoff, T. I. & Ebbini, E. S. Reversible neuroinhibition by focused ultrasound is mediated by a thermal mechanism. Brain Stimul. 12, 1439–1447 (2019).
Oh, S.-J. et al. Ultrasonic neuromodulation via astrocytic TRPA1. Curr. Biol. 29, 3386–3401.e8 (2019).
Chen, S.-G. et al. Transcranial focused ultrasound pulsation suppresses pentylenetetrazol induced epilepsy in vivo. Brain Stimul. 13, 35–46 (2020).
Cui, Z. et al. Enhanced neuronal activity in mouse motor cortex with microbubbles’ oscillations by transcranial focused ultrasound stimulation. Ultrason. Sonochem. 59, 104745 (2019).
Cho, H. et al. Localized down-regulation of P-glycoprotein by focused ultrasound and microbubbles induced blood–brain barrier disruption in rat brain. Sci. Rep. 6, 31201 (2016).
Meng, Y. et al. Resting state functional connectivity changes after MR-guided focused ultrasound mediated blood–brain barrier opening in patients with Alzheimer’s disease. Neuroimage 200, 275–280 (2019).
Todd, N., Zhang, Y., Livingstone, M., Borsook, D. & McDannold, N. The neurovascular response is attenuated by focused ultrasound-mediated disruption of the blood–brain barrier. Neuroimage 201, 116010 (2019).
Wang, Z., Yan, J., Wang, X., Yuan, Y. & Li, X. Transcranial ultrasound stimulation directly influences the cortical excitability of the motor cortex in Parkinsonian mice. Mov. Disord. 35, 693–698 (2020).
Lee, W. et al. Transcranial focused ultrasound stimulation of human primary visual cortex. Sci. Rep. 6, 34026 (2016).
Salminen-Vaparanta, N., Noreika, V., Revonsuo, A., Koivisto, M. & Vanni, S. Is selective primary visual cortex stimulation achievable with TMS? Hum. Brain Mapp. 33, 652–665 (2012).
Beisteiner, R. et al. Transcranial pulse stimulation with ultrasound in Alzheimer’s disease — a new navigated focal brain therapy. Adv. Sci. 7, 1902583 (2020).
Cotero, V. et al. Noninvasive sub-organ ultrasound stimulation for targeted neuromodulation. Nat. Commun. 10, 952 (2019).
Lea-Banks, H., O’Reilly, M. A., Hamani, C. & Hynynen, K. Localized anesthesia of a specific brain region using ultrasound-responsive barbiturate nanodroplets. Theranostics 10, 2849–2858 (2020).
Todd, N. et al. Modulation of brain function by targeted delivery of GABA through the disrupted blood–brain barrier. Neuroimage 189, 267–275 (2019).
Wu, X. et al. Sono-optogenetics facilitated by a circulation-delivered rechargeable light source for minimally invasive optogenetics. Proc. Natl Acad. Sci. USA 116, 26332–26342 (2019).
Wang, S. et al. Non-invasive, focused ultrasound-facilitated gene delivery for optogenetics. Sci. Rep. 7, 39955 (2017).
Wang, J. B., Aryal, M., Zhong, Q., Vyas, D. B. & Airan, R. D. Noninvasive ultrasonic drug uncaging maps whole-brain functional networks. Neuron 100, 728–738.e7 (2018).
Szablowski, J. O., Lee-Gosselin, A., Lue, B., Malounda, D. & Shapiro, M. G. Acoustically targeted chemogenetics for the non-invasive control of neural circuits. Nat. Biomed. Eng. 2, 475–484 (2018).
Constans, C. et al. Non-invasive ultrasonic modulation of visual evoked response by GABA delivery through the blood brain barrier. J. Control. Release 318, 223–231 (2020).
Frey, B. et al. Old and new facts about hyperthermia-induced modulations of the immune system. Int. J. Hyperth. 28, 528–542 (2012).
Cohen-Inbar, O., Xu, Z. & Sheehan, J. P. Focused ultrasound-aided immunomodulation in glioblastoma multiforme: a therapeutic concept. J. Ther. Ultrasound 4, 2 (2016).
Man, J. et al. Hyperthermia sensitizes glioma stem-like cells to radiation by inhibiting AKT signaling. Cancer Res. 75, 1760–1769 (2015).
Zhu, L. et al. Ultrasound hyperthermia technology for radiosensitization. Ultrasound Med. Biol. 45, 1025–1043 (2019).
Yoshida, M. et al. Sonodynamic therapy for malignant glioma using 220-kHz transcranial magnetic resonance imaging-guided focused ultrasound and 5-aminolevulinic acid. Ultrasound Med. Biol. 45, 526–538 (2019).
Ricci, S. et al. Sonothrombolysis for acute ischaemic stroke. Cochrane Database Syst. Rev. 10, CD008348 (2012).
Alexandrov, A. V. et al. Safety and efficacy of sonothrombolysis for acute ischaemic stroke: a multicentre, double-blind, phase 3, randomised controlled trial. Lancet Neurol. 18, 338–347 (2019). The addition of an operator-independent ultrasound device to alteplase was safe but did not improve functional outcomes at 90 days after ischaemic stroke.
Alexandrov, A. V. et al. Endovascular equipoise shift in a phase III randomized clinical trial of sonothrombolysis for acute ischemic stroke. Ther. Adv. Neurol. Disord. 12, 1756286419860652 (2019).
Gerhardson, T. et al. Histotripsy clot liquefaction in a porcine intracerebral hemorrhage model. Neurosurgery 86, 429–436 (2020).
Burgess, A. et al. High-intensity focused ultrasound (HIFU) for dissolution of clots in a rabbit model of embolic stroke. PLoS ONE 7, e42311 (2012).
Chang, W. S. et al. Factors associated with successful magnetic resonance-guided focused ultrasound treatment: efficiency of acoustic energy delivery through the skull. J. Neurosurg. 124, 411–416 (2016).
D’Souza, M. et al. Impact of skull density ratio on efficacy and safety of magnetic resonance-guided focused ultrasound treatment of essential tremor. J. Neurosurg. 132, 1392–1397 (2019).
Schwartz, M. L. et al. Skull bone marrow injury caused by MR-guided focused ultrasound for cerebral functional procedures. J. Neurosurg. 130, 758–762 (2019).
Hughes, A. & Hynynen, K. Design of patient-specific focused ultrasound arrays for non-invasive brain therapy with increased trans-skull transmission and steering range. Phys. Med. Biol. 62, L9–L19 (2017).
Arvanitis, C. D., Vykhodtseva, N., Jolesz, F., Livingstone, M. & McDannold, N. Cavitation-enhanced nonthermal ablation in deep brain targets: feasibility in a large animal model. J. Neurosurg. 124, 1450–1459 (2015).
Macklin, R. The ethical problems with sham surgery in clinical research. N. Engl. J. Med. 341, 992–996 (1999).
Whone, A. et al. Randomized trial of intermittent intraputamenal glial cell line-derived neurotrophic factor in Parkinson’s disease. Brain 142, 512–525 (2019).
Alonso, A. et al. Focal delivery of AAV2/1-transgenes into the rat brain by localized ultrasound-induced BBB opening. Mol. Ther. Nucleic Acids 2, e73 (2013).
Acknowledgements
We acknowledge Hang Yu Lin for her artistic contribution to the figures in this article. N. L. acknowledges and is grateful for the generous philanthropic gifts to the Sunnybrook Foundation, Sunnybrook Research Institute and the Harquail Centre for Neuromodulation as well as the support of the Focused Ultrasound Foundation.
Author information
Authors and Affiliations
Contributions
Y.M. researched data for the article. Y.M. and N.L. wrote the article. All authors made substantial contributions to discussions of the content and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
K.H. is an inventor on intellectual property owned by Brigham and Women’s Hospital in Boston, MA, USA, and Sunnybrook Research Institute in Toronto, Canada, related to intracranial focused ultrasound technology. N.L. has received an honorarium from the Focused Ultrasound Foundation, a not-for-profit funding agency, for serving on an expert steering committee on focused ultrasound in Alzheimer disease. Y.M. declares no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- Blood–brain barrier
-
(BBB). A structural and functional border along the capillaries in the brain that tightly regulates paracellular and transcellular transport.
- Craniectomy
-
A surgical procedure in which a piece of the skull is removed and the overlying skin flap is replaced to create a window for ultrasound propagation.
- Sonication
-
The active delivery of ultrasound. Currently, each typical sonication lasts ~0.5 min for thermoablation and ~1 min for blood–brain barrier (BBB) opening. A rest period allows scalp cooling in thermoablation and systemic clearance of microbubbles in BBB opening.
- Cavitation
-
The change of a liquid to a gas state when subjected to reduced pressures and/or interactions of ultrasound with gas bubbles.
- Treatment envelopes
-
The spatial extent of the brain regions where the desired biological effect (for example, thermoablation) can be successfully achieved with FUS.
- Beta frequency
-
Sustained beta frequency oscillations (12.5–30 Hz) in the cortex and subthalamic nucleus are a characteristic of Parkinson disease and related motor impairments.
- P-glycoprotein
-
A member of the ATP-binding cassette transporter B subfamily that pumps a wide range of foreign substances out of cells and is important in multidrug resistance.
- Stereotactic frame
-
A stereotactic frame is fixed to the head to provide a reference for precise targeting. Common examples include the Leksell (polar coordinate) and Cosman–Roberts–Wells (Cartesian coordinate) frames.
- Common Terminology Criteria for Adverse Events
-
The Common Terminology Criteria for Adverse Events allows the standardized classification of adverse events with condition-specific severity designations. Generally, grade 1 denotes a mild adverse event and grade 5 denotes death.
- Optogenetics
-
A neuromodulation technology that uses specific wavelengths of light to excite or inhibit neurons through light-sensitive ion channels, which can be introduced through viral transfection.
- Skull density ratio
-
(SDR). The ratio of cancellous to cortical bone density in the skull.
Rights and permissions
About this article
Cite this article
Meng, Y., Hynynen, K. & Lipsman, N. Applications of focused ultrasound in the brain: from thermoablation to drug delivery. Nat Rev Neurol 17, 7–22 (2021). https://doi.org/10.1038/s41582-020-00418-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41582-020-00418-z
This article is cited by
-
Ultrasound and antibodies — a potentially powerful combination for Alzheimer disease therapy
Nature Reviews Neurology (2024)
-
Scanning ultrasound-mediated memory and functional improvements do not require amyloid-β reduction
Molecular Psychiatry (2024)
-
The future of brain circuit-targeted therapeutics
Neuropsychopharmacology (2024)
-
Controlled noninvasive modulation of deep brain regions in humans
Communications Engineering (2024)
-
Strategies to Improve Drug Delivery Across the Blood–Brain Barrier for Glioblastoma
Current Neurology and Neuroscience Reports (2024)