Abstract
In a range of neurological conditions, including movement disorders, sex-related differences are emerging not only in brain anatomy and function, but also in pathogenesis, clinical features and response to treatment. In Parkinson disease (PD), for example, oestrogens can influence the severity of motor symptoms, whereas elevation of androgens can exacerbate tic disorders. Nevertheless, the real impact of sex differences in movement disorders remains under-recognized. In this article, we provide an up-to-date review of sex-related differences in PD and the most common hyperkinetic movement disorders, namely, essential tremor, dystonia, Huntington disease and other chorea syndromes, and Tourette syndrome and other chronic tic disorders. We highlight the most relevant clinical aspects of movement disorders that differ between men and women. Increased recognition of these differences and their impact on patient care could aid the development of tailored approaches to the management of movement disorders and enable the optimization of preclinical research and clinical studies.
Key points
Sex differences in epidemiology, clinical features and/or response to treatment have been reported in several movement disorders, including Parkinson disease (PD), essential tremor, dystonia, Huntington disease, Sydenham chorea and tic disorders.
In the case of PD, male sex is associated with higher incidence and prevalence, earlier disease onset, more severe motor symptoms and progression, and more frequent cognitive decline compared with female sex.
Few data are available on sex differences in hyperkinetic movement disorders, although craniocervical dystonia is more prevalent in women, whereas most focal task-specific dystonias and tics are more frequent in men.
Prospective studies specifically addressing sex differences in risk factors, symptomatology, disease progression, biomarkers and response to treatment are needed to develop tailored management strategies for patients with movement disorders.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ferretti, M. T. et al. Sex differences in Alzheimer disease — the gateway to precision medicine. Nat. Rev. Neurol. 14, 457–469 (2018).
Cordonnier, C. et al. Stroke in women — from evidence to inequalities. Nat. Rev. Neurol. 13, 521–532 (2017).
Vetvik, K. G. & MacGregor, E. A. Sex differences in the epidemiology, clinical features, and pathophysiology of migraine. Lancet Neurol. 16, 76–87 (2017).
Rabin, M. L., Stevens-Haas, C., Havrilla, E., Devi, T. & Kurlan, R. Movement disorders in women: a review. Mov. Disord. 29, 177–183 (2014).
Smith, K. M. & Dahodwala, N. Sex differences in Parkinson’s disease and other movement disorders. Exp. Neurol. 259, 44–56 (2014).
Picillo, M. et al. The relevance of gender in Parkinson’s disease: a review. J. Neurol. 264, 1583–1607 (2017).
Fahn, S. & Jankovic, J. Principles and Practice of Movement Disorders (Churchill Livingstone, 2007).
Cantuti-Castelvetri, I. et al. Effects of gender on nigral gene expression and parkinson disease. Neurobiol. Dis. 26, 606–614 (2007).
Simunovic, F., Yi, M., Wang, Y., Stephens, R. & Sonntag, K. C. Evidence for gender-specific transcriptional profiles of nigral dopamine neurons in Parkinson disease. PLOS ONE 5, e8856 (2010).
Rijpkema, M. et al. Normal sexual dimorphism in the human basal ganglia. Hum. Brain Mapp. 33, 1246–1252 (2012).
Xu, J. et al. Gender effects on age-related changes in brain structure. Am. J. Neuroradiol. 21, 112–118 (2000).
Gillies, G. E., Pienaar, I. S., Vohra, S. & Qamhawi, Z. Sex differences in Parkinson’s disease. Front. Neuroendocrinol. 35, 370–384 (2014).
Litim, N., Morissette, M. & Di Paolo, T. Neuroactive gonadal drugs for neuroprotection in male and female models of Parkinson’s disease. Neurosci. Biobehav. Rev. 67, 79–88 (2016).
Greene, N., Lassen, C. F., Rugbjerg, K. & Ritz, B. Reproductive factors and Parkinson’s disease risk in Danish women. Eur. J. Neurol. 21, 1168–1177 (2014).
Liu, R. et al. Female reproductive factors, menopausal hormone use, and Parkinson’s disease. Mov. Disord. 29, 889–896 (2014).
Gatto, N. M. et al. Lifetime exposure to estrogens and Parkinson’s disease in California teachers. Parkinsonism Relat. Disord. 20, 1149–1156 (2014).
Strafella, C. et al. Application of precision medicine in neurodegenerative diseases. Front. Neurol. 9, 701 (2018).
Institute of Medicine (eds Wizemann, T. M. & Pardue. M.-L.). Exploring the Biological Contributions to Human Health: Does Sex Matter? (National Academies Press, 2001).
Goedert, M., Spillantini, M. G., Del Tredici, K. & Braak, H. 100 years of Lewy pathology. Nat. Rev. Neurol. 9, 13–24 (2013).
Dickson, D. W. et al. Neuropathological assessment of Parkinson’s disease: refining the diagnostic criteria. Lancet Neurol. 8, 1150–1157 (2009).
Postuma, R. B. et al. Identifying prodromal Parkinson’s disease: pre-motor disorders in Parkinson’s disease. Mov. Disord. 27, 617–626 (2012).
Moore, K. L., Boscardin, W. J., Steinman, M. A. & Schwartz, J. B. Age and sex variation in prevalence of chronic medical conditions in older residents of US nursing homes. J. Am. Geriatr. Soc. 60, 756–764 (2012).
Pringsheim, T., Jette, N., Frolkis, A. & Steeves, T. D. L. The prevalence of Parkinson’s disease: a systematic review and meta-analysis. Mov. Disord. 29, 1583–1590 (2014).
Abbas, M. M., Xu, Z. & Tan, L. C. S. Epidemiology of Parkinson’s disease — East versus West. Mov. Disord. Clin. Pract. 5, 14–28 (2018).
Xu, J., Gong, D. D., Man, C. F. & Fan, Y. Parkinson’s disease and risk of mortality: meta-analysis and systematic review. Acta Neurol. Scand. 129, 71–79 (2014).
Maeda, T. et al. Clinical manifestations of nonmotor symptoms in 1021 Japanese Parkinson’s disease patients from 35 medical centers. Parkinsonism Relat. Disord. 38, 54–60 (2017).
Moisan, F. et al. Parkinson disease male-to-female ratios increase with age: French nationwide study and meta-analysis. J. Neurol. Neurosurg. Psychiatry 87, 952–957 (2016).
de Lau, L. M. L., Verbaan, D., Marinus, J. & van Hilten, J. J. Survival in Parkinson’s disease. Relation with motor and non-motor features. Parkinsonism Relat. Disord. 20, 613–616 (2014).
Pinter, B. et al. Mortality in Parkinson’s disease: a 38-year follow-up study. Mov. Disord. 30, 266–269 (2015).
Taylor, K. S. M., Cook, J. A. & Counsell, C. E. Heterogeneity in male to female risk for Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 78, 905–906 (2007).
Wooten, G. F., Currie, L. J., Bovbjerg, V. E., Lee, J. K. & Patrie, J. Are men at greater risk for Parkinson’s disease than women? J. Neurol. Neurosurg. Psychiatry 75, 637–639 (2004).
Hirsch, L., Jette, N., Frolkis, A., Steeves, T. & Pringsheim, T. The incidence of Parkinson’s disease: a systematic review and meta-analysis. Neuroepidemiology 46, 292–300 (2016).
Ascherio, A. & Schwarzschild, M. A. The epidemiology of Parkinson’s disease: risk factors and prevention. Lancet Neurol. 15, 1257–1272 (2016).
GBD 2016 Neurology Collaborators. Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the Global Burden Of Disease Study 2016. Lancet Neurol. 18, 459–480 (2019).
Diem-Zangerl, A. et al. Mortality in Parkinson’s disease: a 20-year follow-up study. Mov. Disord. 24, 819–825 (2009).
Haaxma, C. A. et al. Gender differences in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 78, 819–824 (2007).
Alves, G. et al. Incidence of Parkinson’s disease in Norway: the Norwegian ParkWest study. J. Neurol. Neurosurg. Psychiatry 80, 851–857 (2009).
Twelves, D., Perkins, K. S. M. & Counsell, C. Systematic review of incidence studies of Parkinson’s disease. Mov. Disord. 18, 19–31 (2003).
Simunovic, F. et al. Gene expression profiling of substantia nigra dopamine neurons: further insights into Parkinson’s disease pathology. Brain J. Neurol. 132, 1795–1809 (2009).
Bae, Y. H., Hwang, J. Y., Kim, Y. H. & Koh, J. Y. Anti-oxidative neuroprotection by estrogens in mouse cortical cultures. J. Korean Med. Sci. 15, 327–336 (2000).
Sawada, H. & Shimohama, S. Estrogens and Parkinson disease: novel approach for neuroprotection. Endocrine 21, 77–79 (2003).
Benedetti, M. D. et al. Hysterectomy, menopause, and estrogen use preceding Parkinson’s disease: an exploratory case–control study. Mov. Disord. 16, 830–837 (2001).
Popat, R. A. et al. Effect of reproductive factors and postmenopausal hormone use on the risk of Parkinson disease. Neurology 65, 383–390 (2005).
Ascherio, A. et al. Caffeine, postmenopausal estrogen, and risk of Parkinson’s disease. Neurology 60, 790–795 (2003).
Nicoletti, A. et al. Reproductive factors and Parkinson’s disease: a multicenter case-control study. Mov. Disord. 26, 2563–2566 (2011).
Martignoni, E. et al. Parkinson’s disease and reproductive life events. Neurol. Sci. 23 (Suppl. 2), S85–S86 (2002).
Currie, L. J., Harrison, M. B., Trugman, J. M., Bennett, J. P. & Wooten, G. F. Postmenopausal estrogen use affects risk for Parkinson disease. Arch. Neurol. 61, 886–888 (2004).
Ragonese, P. et al. Risk of Parkinson disease in women: effect of reproductive characteristics. Neurology 62, 2010–2014 (2004).
Rocca, W. A. et al. Increased risk of parkinsonism in women who underwent oophorectomy before menopause. Neurology 70, 200–209 (2008).
Simon, K. C., Chen, H., Gao, X., Schwarzschild, M. A. & Ascherio, A. Reproductive factors, exogenous estrogen use, and risk of Parkinson’s disease. Mov. Disord. 24, 1359–1365 (2009).
Frigerio, R. et al. Number of children and risk of Parkinson’s disease. Mov. Disord. 22, 632–639 (2007).
Rugbjerg, K., Christensen, J., Tjonneland, A. & Olsen, J. H. Exposure to estrogen and women’s risk for Parkinson’s disease: a prospective cohort study in Denmark. Parkinsonism Relat. Disord. 19, 457–460 (2013).
Cereda, E., Barichella, M., Cassani, E., Caccialanza, R. & Pezzoli, G. Reproductive factors and clinical features of Parkinson’s disease. Parkinsonism Relat. Disord. 19, 1094–1099 (2013).
Wang, P., Li, J., Qiu, S., Wen, H. & Du, J. Hormone replacement therapy and Parkinson’s disease risk in women: a meta-analysis of 14 observational studies. Neuropsychiatr. Dis. Treat. 11, 59–66 (2015).
Palacios, N. et al. Caffeine and risk of Parkinson’s disease in a large cohort of men and women. Mov. Disord. 27, 1276–1282 (2012).
Lv, M. et al. Reproductive factors and risk of Parkinson’s disease in women: a meta-analysis of observational studies. Behav. Brain Res. 335, 103–110 (2017).
Savica, R., Grossardt, B. R., Bower, J. H., Ahlskog, J. E. & Rocca, W. A. Risk factors for Parkinson’s disease may differ in men and women: an exploratory study. Horm. Behav. 63, 308–314 (2013).
Chahine, L. M. & Stern, M. B. Parkinson’s disease biomarkers: where are we and where do we go next? Mov. Disord. Clin. Pract. 4, 796–805 (2017).
Ikeda, K. et al. Serological profiles of urate, paraoxonase-1, ferritin and lipid in Parkinson’s disease: changes linked to disease progression. Neurodegener. Dis. 8, 252–258 (2011).
Caranci, G. et al. Gender differences in Parkinson’s disease: focus on plasma α-synuclein. J. Neural Transm. 120, 1209–1215 (2013).
Ho, D. H., Yi, S., Seo, H., Son, I. & Seol, W. Increased DJ-1 in urine exosome of Korean males with Parkinson’s disease. Biomed. Res. Int. 2014, 704678 (2014).
Chen, H., Mosley, T. H., Alonso, A. & Huang, X. Plasma urate and Parkinson’s disease in the Atherosclerosis Risk in Communities (ARIC) study. Am. J. Epidemiol. 169, 1064–1069 (2009).
Gao, X., O’Reilly, E. J., Schwarzschild, M. A. & Ascherio, A. Prospective study of plasma urate and risk of Parkinson disease in men and women. Neurology 86, 520–526 (2016).
Schwarzschild, M. A. et al. Serum urate as a predictor of clinical and radiographic progression in Parkinson disease. Arch. Neurol. 65, 716–723 (2008).
Ascherio, A. et al. Urate as a predictor of the rate of clinical decline in Parkinson disease. Arch. Neurol. 66, 1460–1468 (2009).
Schwarzschild, M. A. et al. Serum urate and probability of dopaminergic deficit in early ‘Parkinson’s disease’. Mov. Disord. 26, 1864–1868 (2011).
Jesus, S. et al. Low serum uric acid concentration in Parkinson’s disease in southern Spain. Eur. J. Neurol. 20, 208–210 (2013).
McFarland, N. R., Burdett, T., Desjardins, C. A., Frosch, M. P. & Schwarzschild, M. A. Postmortem brain levels of urate and precursors in Parkinson’s disease and related disorders. Neurodegener. Dis. 12, 189–198 (2013).
Yadav, S. K. et al. Gender-based analysis of cortical thickness and structural connectivity in Parkinson’s disease. J. Neurol. 263, 2308–2318 (2016).
De Micco, R. et al. Sex-related pattern of intrinsic brain connectivity in drug-naive Parkinson’s disease patients. Mov. Disord. 34, 997–1005 (2019).
Kaasinen, V., Joutsa, J., Noponen, T., Johansson, J. & Seppanen, M. Effects of aging and gender on striatal and extrastriatal [123I]FP-CIT binding in Parkinson’s disease. Neurobiol. Aging 36, 1757–1763 (2015).
Kaasinen, V. et al. Increased frontal [18F]fluorodopa uptake in early Parkinson’s disease: sex differences in the prefrontal cortex. Brain J. Neurol. 124, 1125–1130 (2001).
Mariani, S. et al. Association between sex, systemic iron variation and probability of Parkinson’s disease. Int. J. Neurosci. 126, 354–360 (2016).
Palacios, N. et al. Polymorphisms of caffeine metabolism and estrogen receptor genes and risk of Parkinson’s disease in men and women. Parkinsonism Relat. Disord. 16, 370–375 (2010).
San Luciano, M. et al. Gender differences in the IL6 -174G>C and ESR2 1730G>A polymorphisms and the risk of Parkinson’s disease. Neurosci. Lett. 506, 312–316 (2012).
Clark, L. N. et al. Frequency of LRRK2 mutations in early- and late-onset Parkinson disease. Neurology 67, 1786–1791 (2006).
Agalliu, I. et al. Higher frequency of certain cancers in LRRK2 G2019S mutation carriers with Parkinson disease: a pooled analysis. JAMA Neurol. 72, 58–65 (2015).
Cilia, R. et al. LRRK2 mutations in Parkinson’s disease: confirmation of a gender effect in the Italian population. Parkinsonism Relat. Disord. 20, 911–914 (2014).
Orr-Urtreger, A. et al. The LRRK2 G2019S mutation in Ashkenazi Jews with Parkinson disease: is there a gender effect? Neurology 69, 1595–1602 (2007).
Saunders-Pullman, R. et al. Gender differences in the risk of familial parkinsonism: beyond LRRK2? Neurosci. Lett. 496, 125–128 (2011).
San Luciano, M. et al. Sex differences in LRRK2 G2019S and idiopathic Parkinson’s disease. Ann. Clin. Transl. Neurol. 4, 801–810 (2017).
Gan-Or, Z. et al. LRRK2 mutations in Parkinson disease; a sex effect or lack thereof? A meta-analysis. Parkinsonism Relat. Disord. 21, 778–782 (2015).
Shu, L. et al. Clinical heterogeneity among LRRK2 variants in Parkinson’s disease: a meta-analysis. Front. Aging Neurosci. 10, 283 (2018).
Gan-Or, Z. et al. LRRK2 and GBA mutations differentially affect the initial presentation of Parkinson disease. Neurogenetics 11, 121–125 (2010).
Swan, M. et al. Neuropsychiatric characteristics of GBA-associated Parkinson disease. J. Neurol. Sci. 370, 63–69 (2016).
Nalls, M. A. et al. Diagnosis of Parkinson’s disease on the basis of clinical and genetic classification: a population-based modelling study. Lancet Neurol. 14, 1002–1009 (2015).
Lavalaye, J., Booij, J., Reneman, L., Habraken, J. B. & van Royen, E. A. Effect of age and gender on dopamine transporter imaging with [123I]FP-CIT SPET in healthy volunteers. Eur. J. Nucl. Med. 27, 867–869 (2000).
Saunders-Pullman, R., Wang, C., Stanley, K. & Bressman, S. B. Diagnosis and referral delay in women with Parkinson’s disease. Gend. Med. 8, 209–217 (2011).
Adamson, J., Ben-Shlomo, Y., Chaturvedi, N. & Donovan, J. Ethnicity, socio-economic position and gender — do they affect reported health-care seeking behaviour? Soc. Sci. Med. 57, 895–904 (2003).
Latourelle, J. C. et al. Large-scale identification of clinical and genetic predictors of motor progression in patients with newly diagnosed Parkinson’s disease: a longitudinal cohort study and validation. Lancet Neurol. 16, 908–916 (2017).
Scott, N. W., Macleod, A. D. & Counsell, C. E. Motor complications in an incident Parkinson’s disease cohort. Eur. J. Neurol. 23, 304–312 (2016).
Sato, K. et al. Prognosis of Parkinson’s disease: time to stage III, IV, V, and to motor fluctuations. Mov. Disord. 21, 1384–1395 (2006).
Bjornestad, A. et al. Risk and course of motor complications in a population-based incident Parkinson’s disease cohort. Parkinsonism Relat. Disord. 22, 48–53 (2016).
Colombo, D. et al. The “gender factor” in wearing-off among patients with Parkinson’s disease: a post hoc analysis of DEEP study. Sci. World J. 2015, 787451 (2015).
Zappia, M. et al. Sex differences in clinical and genetic determinants of levodopa peak-dose dyskinesias in Parkinson disease: an exploratory study. Arch. Neurol. 62, 601–605 (2005).
Sharma, J. C., Bachmann, C. G. & Linazasoro, G. Classifying risk factors for dyskinesia in Parkinson’s disease. Parkinsonism Relat. Disord. 16, 490–497 (2010).
Hassin-Baer, S. et al. Gender effect on time to levodopa-induced dyskinesias. J. Neurol. 258, 2048–2053 (2011).
Lubomski, M., Louise Rushworth, R., Lee, W., Bertram, K. L. & Williams, D. R. Sex differences in Parkinson’s disease. J. Clin. Neurosci. 21, 1503–1506 (2014).
De Pablo-Fernandez, E., Lees, A. J., Holton, J. L. & Warner, T. T. Prognosis and neuropathologic correlation of clinical subtypes of Parkinson disease. JAMA Neurol. 76, 470–479 (2019).
Horstink, M. W. I. M., Strijks, E. & Dluzen, D. E. Estrogen and Parkinson’s disease. Adv. Neurol. 91, 107–114 (2003).
Quinn, N. P. & Marsden, C. D. Menstrual-related fluctuations in Parkinson’s disease. Mov. Disord. 1, 85–87 (1986).
Sandyk, R. Estrogens and the pathophysiology of Parkinson’s disease. Int. J. Neurosci. 45, 119–122 (1989).
Castrioto, A., Hulliger, S., Poon, Y.-Y., Lang, A. E. & Moro, E. A survey on the impact of the menstrual cycle on movement disorders severity. Can. J. Neurol. Sci. 37, 478–481 (2010).
Saunders-Pullman, R. et al. The effect of estrogen replacement on early Parkinson’s disease. Neurology 52, 1417–1421 (1999).
Tsang, K. L., Ho, S. L. & Lo, S. K. Estrogen improves motor disability in parkinsonian postmenopausal women with motor fluctuations. Neurology 54, 2292–2298 (2000).
Shulman, L. M., Minagar, A. & Weiner, W. J. The effect of pregnancy in Parkinson’s disease. Mov. Disord. 15, 132–135 (2000).
Golbe, L. I. Pregnancy and movement disorders. Neurol. Clin. 12, 497–508 (1994).
Hagell, P., Odin, P. & Vinge, E. Pregnancy in Parkinson’s disease: a review of the literature and a case report. Mov. Disord. 13, 34–38 (1998).
Seier, M. & Hiller, A. Parkinson’s disease and pregnancy: an updated review. Parkinsonism Relat. Disord. 40, 11–17 (2017).
Shulman, L. M. Gender differences in Parkinson’s disease. Gend. Med. 4, 8–18 (2007).
Sauerbier, A., Lenka, A., Aris, A. & Pal, P. K. Nonmotor symptoms in Parkinson’s disease: gender and ethnic differences. Int. Rev. Neurobiol. 133, 417–446 (2017).
Guo, X. et al. Gender and onset age-related features of non-motor symptoms of patients with Parkinson’s disease — a study from Southwest China. Parkinsonism Relat. Disord. 19, 961–965 (2013).
Martinez-Martin, P. et al. Gender-related differences in the burden of non-motor symptoms in Parkinson’s disease. J. Neurol. 259, 1639–1647 (2012).
Nicoletti, A. et al. Gender effect on non-motor symptoms in Parkinson’s disease: are men more at risk? Parkinsonism Relat. Disord. 35, 69–74 (2017).
Solla, P. et al. Gender differences in motor and non-motor symptoms among Sardinian patients with Parkinson’s disease. J. Neurol. Sci. 323, 33–39 (2012).
Song, Y., Gu, Z., An, J. & Chan, P. Gender differences on motor and non-motor symptoms of de novo patients with early Parkinson’s disease. Neurol. Sci. 35, 1991–1996 (2014).
Leentjens, A. F. G. et al. Symptomatology and markers of anxiety disorders in Parkinson’s disease: a cross-sectional study. Mov. Disord. 26, 484–492 (2011).
Leentjens, A. F. G. et al. Modeling depression in Parkinson disease: disease-specific and nonspecific risk factors. Neurology 81, 1036–1043 (2013).
Szewczyk-Krolikowski, K. et al. The influence of age and gender on motor and non-motor features of early Parkinson’s disease: initial findings from the Oxford Parkinson Disease Center (OPDC) discovery cohort. Parkinsonism Relat. Disord. 20, 99–105 (2014).
Kovacs, M. et al. Impact of sex on the nonmotor symptoms and the health-related quality of life in Parkinson’s disease. Parkinsons Dis. 2016, 7951840 (2016).
Picillo, M. et al. Gender differences in non-motor symptoms in early, drug naive Parkinson’s disease. J. Neurol. 260, 2849–2855 (2013).
Picillo, M. et al. The PRIAMO study: urinary dysfunction as a marker of disease progression in early Parkinson’s disease. Eur. J. Neurol. 24, 788–795 (2017).
Erro, R. et al. Non-motor symptoms in early Parkinson’s disease: a 2-year follow-up study on previously untreated patients. J. Neurol. Neurosurg. Psychiatry 84, 14–17 (2013).
Liu, R. et al. Potential sex differences in nonmotor symptoms in early drug-naive Parkinson disease. Neurology 84, 2107–2115 (2015).
Picillo, M. et al. Gender differences in non-motor symptoms in early Parkinson’s disease: a 2-years follow-up study on previously untreated patients. Parkinsonism Relat. Disord. 20, 850–854 (2014).
Warren Olanow, C. et al. Factors predictive of the development of levodopa-induced dyskinesia and wearing-off in Parkinson’s disease. Mov. Disord. 28, 1064–1071 (2013).
Picillo, M. et al. Gender and non motor fluctuations in Parkinson’s disease: a prospective study. Parkinsonism Relat. Disord. 27, 89–92 (2016).
Heller, J., Dogan, I., Schulz, J. B. & Reetz, K. Evidence for gender differences in cognition, emotion and quality of life in Parkinson’s disease? Aging Dis. 5, 63–75 (2014).
Yoon, J.-E. et al. Gender differences of nonmotor symptoms affecting quality of life in Parkinson disease. Neurodegener. Dis. 17, 276–280 (2017).
Liu, G. et al. Prediction of cognition in Parkinson’s disease with a clinical-genetic score: a longitudinal analysis of nine cohorts. Lancet Neurol. 16, 620–629 (2017).
Curtis, A. F., Masellis, M., Camicioli, R., Davidson, H. & Tierney, M. C. Cognitive profile of non-demented Parkinson’s disease: meta-analysis of domain and sex-specific deficits. Parkinsonism Relat. Disord. 60, 32–42 (2019).
Miller, I. N. & Cronin-Golomb, A. Gender differences in Parkinson’s disease: clinical characteristics and cognition. Mov. Disord. 25, 2695–2703 (2010).
Uc, E. Y. et al. Incidence of and risk factors for cognitive impairment in an early Parkinson disease clinical trial cohort. Neurology 73, 1469–1477 (2009).
Anang, J. B. M. et al. Predictors of dementia in Parkinson disease: a prospective cohort study. Neurology 83, 1253–1260 (2014).
Pigott, K. et al. Longitudinal study of normal cognition in Parkinson disease. Neurology 85, 1276–1282 (2015).
Anang, J. B. M. et al. Dementia predictors in Parkinson disease: a validation study. J. Parkinsons Dis. 7, 159–162 (2017).
Augustine, E. F. et al. Sex differences in clinical features of early, treated Parkinson’s disease. PLOS ONE 10, e0133002 (2015).
Cholerton, B. et al. Sex differences in progression to mild cognitive impairment and dementia in Parkinson’s disease. Parkinsonism Relat. Disord. 50, 29–36 (2018).
Cereda, E. et al. Dementia in Parkinson’s disease: is male gender a risk factor? Parkinsonism Relat. Disord. 26, 67–72 (2016).
Baba, Y., Putzke, J. D., Whaley, N. R., Wszolek, Z. K. & Uitti, R. J. Gender and the Parkinson’s disease phenotype. J. Neurol. 252, 1201–1205 (2005).
Nyholm, D., Karlsson, E., Lundberg, M. & Askmark, H. Large differences in levodopa dose requirement in Parkinson’s disease: men use higher doses than women. Eur. J. Neurol. 17, 260–266 (2010).
Sharma, J. C., Macnamara, L., Hasoon, M., Vassallo, M. & Ross, I. Cascade of levodopa dose and weight-related dyskinesia in Parkinson’s disease (LD-WD-PD cascade). Parkinsonism Relat. Disord. 12, 499–505 (2006).
Sharma, J. C., Ross, I. N., Rascol, O. & Brooks, D. Relationship between weight, levodopa and dyskinesia: the significance of levodopa dose per kilogram body weight. Eur. J. Neurol. 15, 493–496 (2008).
Kompoliti, K. et al. Gender and pramipexole effects on levodopa pharmacokinetics and pharmacodynamics. Neurology 58, 1418–1422 (2002).
Arabia, G. et al. Body weight, levodopa pharmacokinetics and dyskinesia in Parkinson’s disease. Neurol. Sci. 23 (Suppl. 2), 53–54 (2002).
Kumagai, T. et al. Sex differences in the pharmacokinetics of levodopa in elderly patients with Parkinson disease. Clin. Neuropharmacol. 37, 173–176 (2014).
Martinelli, P. et al. Levodopa pharmacokinetics and dyskinesias: are there sex-related differences? Neurol. Sci. 24, 192–193 (2003).
Montaurier, C. et al. Mechanisms of body weight gain in patients with Parkinson’s disease after subthalamic stimulation. Brain J. Neurol. 130, 1808–1818 (2007).
Sampaio, T. F. et al. MAO-B and COMT genetic variations associated with levodopa treatment response in patients with Parkinson’s disease. J. Clin. Pharmacol. 58, 920–926 (2018).
Fox, S. H. et al. International Parkinson and Movement Disorder Society evidence-based medicine review: update on treatments for the motor symptoms of Parkinson’s disease. Mov. Disord. 33, 1248–1266 (2018).
Seppi, K. et al. Update on treatments for nonmotor symptoms of Parkinson’s disease-an evidence-based medicine review. Mov. Disord. 34, 180–198 (2019).
Chan, A. K. et al. Disparities in access to deep brain stimulation surgery for Parkinson disease: interaction between African American race and Medicaid use. JAMA Neurol. 71, 291–299 (2014).
Willis, A. W. et al. Disparities in deep brain stimulation surgery among insured elders with Parkinson disease. Neurology 82, 163–171 (2014).
Hariz, G.-M. et al. Gender distribution of patients with Parkinson’s disease treated with subthalamic deep brain stimulation; a review of the 2000-2009 literature. Parkinsonism Relat. Disord. 17, 146–149 (2011).
Hariz, G.-M. et al. Gender differences in quality of life following subthalamic stimulation for Parkinson’s disease. Acta Neurol. Scand. 128, 281–285 (2013).
Romito, L. M., Contarino, F. M. & Albanese, A. Transient gender-related effects in Parkinson’s disease patients with subthalamic stimulation. J. Neurol. 257, 603–608 (2010).
Accolla, E. et al. Gender differences in patients with Parkinson’s disease treated with subthalamic deep brain stimulation. Mov. Disord. 22, 1150–1156 (2007).
Scelzo, E. et al. Deep brain stimulation during pregnancy and delivery: experience from a series of ‘DBS babies’. Front. Neurol. 6, 191 (2015).
Bhatia, K. P. et al. Consensus statement on the classification of tremors, from the Task Force on Tremor of the International Parkinson and Movement Disorder Society. Mov. Disord. 33, 75–87 (2018).
Louis, E. D. & Ferreira, J. J. How common is the most common adult movement disorder? Update on the worldwide prevalence of essential tremor. Mov. Disord. 25, 534–541 (2010).
Louis, E. D., Thawani, S. P. & Andrews, H. F. Prevalence of essential tremor in a multiethnic, community-based study in northern Manhattan, New York, NY. Neuroepidemiology 32, 208–214 (2009).
Glik, A. et al. Essential tremor might be less frequent than Parkinson’s disease in North Israel Arab villages. Mov. Disord. 24, 119–122 (2009).
Mancini, M. L. et al. Prevalence of essential tremor in the territory of lake Trasimeno, Italy: results of a population-based study. Mov. Disord. 22, 540–545 (2007).
Dotchin, C. L. & Walker, R. W. The prevalence of essential tremor in rural northern Tanzania. J. Neurol. Neurosurg. Psychiatry 79, 1107–1109 (2008).
Tan, L. C. S., Venketasubramanian, N., Ramasamy, V., Gao, W. & Saw, S.-M. Prevalence of essential tremor in Singapore: a study on three races in an Asian country. Parkinsonism Relat. Disord. 11, 233–239 (2005).
Louis, E. D. & Vonsattel, J. P. G. The emerging neuropathology of essential tremor. Mov. Disord. 23, 174–182 (2008).
Louis, E. D., Fernandez-Alvarez, E., Dure, L. S. 4th, Frucht, S. & Ford, B. Association between male gender and pediatric essential tremor. Mov. Disord. 20, 904–906 (2005).
Rajput, A. H., Offord, K. P., Beard, C. M. & Kurland, L. T. Essential tremor in Rochester, Minnesota: a 45-year study. J. Neurol. Neurosurg. Psychiatry 47, 466–470 (1984).
Larsson, T. & Sjogren, T. Essential tremor: a clinical and genetic population study. Acta Psychiatr. Scand. Suppl. 36, 1–176 (1960).
Hubble, J. P., Busenbark, K. L., Pahwa, R., Lyons, K. & Koller, W. C. Clinical expression of essential tremor: effects of gender and age. Mov. Disord. 12, 969–972 (1997).
Hardesty, D. E., Maraganore, D. M., Matsumoto, J. Y. & Louis, E. D. Increased risk of head tremor in women with essential tremor: longitudinal data from the Rochester Epidemiology Project. Mov. Disord. 19, 529–533 (2004).
Chen, W. et al. Topography of essential tremor. Parkinsonism Relat. Disord. 40, 58–63 (2017).
Veroniki, A. A. et al. Comparative safety of antiepileptic drugs for neurological development in children exposed during pregnancy and breast feeding: a systematic review and network meta-analysis. BMJ Open. 7, e017248 (2017).
Bordelon, Y. M. & Smith, M. Movement disorders in pregnancy. Semin. Neurol. 27, 467–475 (2007).
Kranick, S. M., Mowry, E. M., Colcher, A., Horn, S. & Golbe, L. I. Movement disorders and pregnancy: a review of the literature. Mov. Disord. 25, 665–671 (2010).
Blomstedt, P. et al. Influence of age, gender and severity of tremor on outcome after thalamic and subthalamic DBS for essential tremor. Parkinsonism Relat. Disord. 17, 617–620 (2011).
Albanese, A. et al. Phenomenology and classification of dystonia: a consensus update. Mov. Disord. 28, 863–873 (2013).
Jinnah, H. A. et al. The focal dystonias: current views and challenges for future research. Mov. Disord. 28, 926–943 (2013).
Steeves, T. D., Day, L., Dykeman, J., Jette, N. & Pringsheim, T. The prevalence of primary dystonia: a systematic review and meta-analysis. Mov. Disord. 27, 1789–1796 (2012).
Defazio, G. The epidemiology of primary dystonia: current evidence and perspectives. Eur. J. Neurol. 17 (Suppl. 1), 9–14 (2010).
Defazio, G., Abbruzzese, G., Livrea, P. & Berardelli, A. Epidemiology of primary dystonia. Lancet Neurol. 3, 673–678 (2004).
Williams, L. et al. Epidemiological, clinical and genetic aspects of adult onset isolated focal dystonia in Ireland. Eur. J. Neurol. 24, 73–81 (2017).
Hintze, J. M., Ludlow, C. L., Bansberg, S. F., Adler, C. H. & Lott, D. G. Spasmodic dysphonia: a review. Part 1: pathogenic factors. Otolaryngol. Head. Neck Surg. 157, 551–557 (2017).
Pandey, S. & Sharma, S. Meige’s syndrome: history, epidemiology, clinical features, pathogenesis and treatment. J. Neurol. Sci. 372, 162–170 (2017).
Soland, V. L., Bhatia, K. P. & Marsden, C. D. Sex prevalence of focal dystonias. J. Neurol. Neurosurg. Psychiatry 60, 204–205 (1996).
Defazio, G. et al. The Italian Dystonia Registry: rationale, design and preliminary findings. Neurol. Sci. 38, 819–825 (2017).
The Epidemiological Study of Dystonia in Europe (ESDE) Collaborative Group. A prevalence study of primary dystonia in eight European countries. J. Neurol. 247, 787–792 (2000).
Torres-Russotto, D. & Perlmutter, J. S. Task-specific dystonias: a review. Ann. N. Y. Acad. Sci. 1142, 179–199 (2008).
Dhungana, S. & Jankovic, J. Yips and other movement disorders in golfers. Mov. Disord. 28, 576–581 (2013).
Adler, C. H. et al. Are the yips a task-specific dystonia or ‘golfer’s cramp’? Mov. Disord. 26, 1993–1996 (2011).
Defazio, G., Berardelli, A. & Hallett, M. Do primary adult-onset focal dystonias share aetiological factors? Brain J. Neurol. 130, 1183–1193 (2007).
Ham, J. H. et al. A prognostic factor in focal hand dystonia: typist’s cramp cases and literature review. J. Neurol. Sci. 371, 85–87 (2016).
Wijemanne, S. & Jankovic, J. Dopa-responsive dystonia — clinical and genetic heterogeneity. Nat. Rev. Neurol. 11, 414–424 (2015).
Cossu, G. & Colosimo, C. Hyperkinetic movement disorder emergencies. Curr. Neurol. Neurosci. Rep. 17, 6 (2017).
Savitt, D. & Jankovic, J. Tardive syndromes. J. Neurol. Sci. 389, 35–42 (2018).
Groen, J. L. et al. Phenotypes and genetic architecture of focal primary torsion dystonia. J. Neurol. Neurosurg. Psychiatry 83, 1006–1011 (2012).
Fiorio, M. et al. Tactile temporal discrimination in patients with blepharospasm. J. Neurol. Neurosurg. Psychiatry 79, 796–798 (2008).
Kimmich, O. et al. Temporal discrimination, a cervical dystonia endophenotype: penetrance and functional correlates. Mov. Disord. 29, 804–811 (2014).
Fiorio, M. et al. Defective temporal processing of sensory stimuli in DYT1 mutation carriers: a new endophenotype of dystonia? Brain J. Neurol. 130, 134–142 (2007).
Scontrini, A. et al. Somatosensory temporal discrimination in patients with primary focal dystonia. J. Neurol. Neurosurg. Psychiatry 80, 1315–1319 (2009).
Kimmich, O. et al. Sporadic adult onset primary torsion dystonia is a genetic disorder by the temporal discrimination test. Brain J. Neurol. 134, 2656–2663 (2011).
Hutchinson, M. et al. Cervical dystonia: a disorder of the midbrain network for covert attentional orienting. Front. Neurol. 5, 54 (2014).
Green, C. D. & Han, J.-D. J. Epigenetic regulation by nuclear receptors. Epigenomics 3, 59–72 (2011).
Gwinn-Hardy, K. A., Adler, C. H., Weaver, A. L., Fish, N. M. & Newman, S. J. Effect of hormone variations and other factors on symptom severity in women with dystonia. Mayo Clin. Proc. 75, 235–240 (2000).
Rogers, J. D. & Fahn, S. Movement disorders and pregnancy. Adv. Neurol. 64, 163–178 (1994).
Martino, D. et al. Menopause and menarche in patients with primary blepharospasm: an exploratory case-control study. Eur. Neurol. 47, 161–164 (2002).
Zimprich, A. et al. Mutations in the gene encoding epsilon-sarcoglycan cause myoclonus-dystonia syndrome. Nat. Genet. 29, 66–69 (2001).
Lohmann, K. & Klein, C. Genetics of dystonia: What’s known? What’s new? What’s next? Mov. Disord. 28, 899–905 (2013).
Lee, L. V. et al. The unique phenomenology of sex-linked dystonia parkinsonism (XDP, DYT3, ‘Lubag’). Int. J. Neurosci. 121 (Suppl. 1), 3–11 (2011).
Marras, C. et al. Minimum incidence of primary cervical dystonia in a multiethnic health care population. Neurology 69, 676–680 (2007).
Matsumoto, S., Nishimura, M., Shibasaki, H. & Kaji, R. Epidemiology of primary dystonias in Japan: comparison with Western countries. Mov. Disord. 18, 1196–1198 (2003).
Peall, K. J., Kuiper, A., de Koning, T. J. & Tijssen, M. A. J. Non-motor symptoms in genetically defined dystonia: homogenous groups require systematic assessment. Parkinsonism Relat. Disord. 21, 1031–1040 (2015).
Conte, A. et al. Non-motor symptoms in patients with adult-onset focal dystonia: sensory and psychiatric disturbances. Parkinsonism Relat. Disord. 22 (Suppl. 1), 111–114 (2016).
Eggink, H. et al. Motor and non-motor determinants of health-related quality of life in young dystonia patients. Parkinsonism Relat. Disord. 58, 50–55 (2019).
Novaretti, N. et al. The prevalence and correlation of non-motor symptoms in adult patients with idiopathic focal or segmental dystonia. Tremor Other Hyperkinetic Mov. 9, 596 (2019).
Pirio Richardson, S., Wegele, A. R., Skipper, B., Deligtisch, A. & Jinnah, H. A. Dystonia treatment: patterns of medication use in an international cohort. Neurology 88, 543–550 (2017).
Contarino, M. F. et al. Clinical practice: evidence-based recommendations for the treatment of cervical dystonia with botulinum toxin. Front. Neurol. 8, 35 (2017).
Ziman, N. et al. Pregnancy in a series of dystonia patients treated with deep brain stimulation: outcomes and management recommendations. Stereotact. Funct. Neurosurg. 94, 60–65 (2016).
Paluzzi, A. et al. Pregnancy in dystonic women with in situ deep brain stimulators. Mov. Disord. 21, 695–698 (2006).
Bates, G. P. et al. Huntington disease. Nat. Rev. Dis. Prim. 1, 15005 (2015).
MacDonald, M. E. et al. A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington’s disease chromosomes. Cell 72, 971–983 (1993).
Padovan-Neto, F. E. et al. Age- and sex-related changes in cortical and striatal nitric oxide synthase in the Q175 mouse model of Huntington’s disease. Nitric Oxide 83, 40–50 (2019).
Dorner, J. L., Miller, B. R., Barton, S. J., Brock, T. J. & Rebec, G. V. Sex differences in behavior and striatal ascorbate release in the 140 CAG knock-in mouse model of Huntington’s disease. Behav. Brain Res. 178, 90–97 (2007).
Bode, F. J. et al. Sex differences in a transgenic rat model of Huntington’s disease: decreased 17beta-estradiol levels correlate with reduced numbers of DARPP32+ neurons in males. Hum. Mol. Genet. 17, 2595–2609 (2008).
Fisher, E. R. & Hayden, M. R. Multisource ascertainment of Huntington disease in Canada: prevalence and population at risk. Mov. Disord. 29, 105–114 (2014).
Pringsheim, T. et al. The incidence and prevalence of Huntington’s disease: a systematic review and meta-analysis. Mov. Disord. 27, 1083–1091 (2012).
Bruzelius, E. et al. Huntington’s disease in the United States: variation by demographic and socioeconomic factors. Mov. Disord. 34, 858–865 (2019).
Duyao, M. et al. Trinucleotide repeat length instability and age of onset in Huntington’s disease. Nat. Genet. 4, 387–392 (1993).
Kehoe, P., Krawczak, M., Harper, P. S., Owen, M. J. & Jones, A. L. Age of onset in Huntington disease: sex specific influence of apolipoprotein E genotype and normal CAG repeat length. J. Med. Genet. 36, 108–111 (1999).
Lee, J. K. et al. Sex-specific effects of the Huntington gene on normal neurodevelopment. J. Neurosci. Res. 95, 398–408 (2017).
Aziz, N. A., van Belzen, M. J., Coops, I. D., Belfroid, R. D. M. & Roos, R. A. C. Parent-of-origin differences of mutant HTT CAG repeat instability in Huntington’s disease. Eur. J. Med. Genet. 54, e413–e418 (2011).
Ranen, N. G. et al. Anticipation and instability of IT-15 (CAG)n repeats in parent-offspring pairs with Huntington disease. Am. J. Hum. Genet. 57, 593–602 (1995).
Kremer, B. et al. Sex-dependent mechanisms for expansions and contractions of the CAG repeat on affected Huntington disease chromosomes. Am. J. Hum. Genet. 57, 343–350 (1995).
Farrer, L. A., Cupples, L. A., Kiely, D. K., Conneally, P. M. & Myers, R. H. Inverse relationship between age at onset of Huntington disease and paternal age suggests involvement of genetic imprinting. Am. J. Hum. Genet. 50, 528–535 (1992).
Zielonka, D. et al. The influence of gender on phenotype and disease progression in patients with Huntington’s disease. Parkinsonism Relat. Disord. 19, 192–197 (2013).
Foroud, T., Gray, J., Ivashina, J. & Conneally, P. M. Differences in duration of Huntington’s disease based on age at onset. J. Neurol. Neurosurg. Psychiatry 66, 52–56 (1999).
Myers, R. H. et al. Factors associated with slow progression in Huntington’s disease. Arch. Neurol. 48, 800–804 (1991).
Zielonka, D. et al. The contribution of gender differences in motor, behavioral and cognitive features to functional capacity, independence and quality of life in patients with Huntington’s disease. Parkinsonism Relat. Disord. 49, 42–47 (2018).
Markianos, M., Panas, M., Kalfakis, N. & Vassilopoulos, D. Plasma testosterone, dehydroepiandrosterone sulfate, and cortisol in female patients with Huntington’s disease. Neuro Endocrinol. Lett. 28, 199–203 (2007).
Markianos, M., Panas, M., Kalfakis, N. & Vassilopoulos, D. Plasma testosterone in male patients with Huntington’s disease: relations to severity of illness and dementia. Ann. Neurol. 57, 520–525 (2005).
van Duijn, E. et al. Neuropsychiatric symptoms in a European Huntington’s disease cohort (REGISTRY). J. Neurol. Neurosurg. Psychiatry 85, 1411–1418 (2014).
Dale, M., Maltby, J., Shimozaki, S., Cramp, R. & Rickards, H. Disease stage, but not sex, predicts depression and psychological distress in Huntington’s disease: a European population study. J. Psychosom. Res. 80, 17–22 (2016).
Costa de Miranda, R. et al. Body composition and bone mineral density in Huntington’s disease. Nutrition 59, 145–149 (2019).
Huntington Study Group. Tetrabenazine as antichorea therapy in Huntington disease: a randomized controlled trial. Neurology 66, 366–372 (2006).
Frank, S. et al. A study of chorea after tetrabenazine withdrawal in patients with Huntington disease. Clin. Neuropharmacol. 31, 127–133 (2008).
Frank, S. Huntington Study Group/TETRA-HD Investigators. Tetrabenazine as anti-chorea therapy in Huntington disease: an open-label continuation study. BMC Neurol. 9, 62 (2009).
Schultz, J. L., Kamholz, J. A., Nopoulos, P. C. & Killoran, A. Comparing risperidone and olanzapine to tetrabenazine for the management of chorea in Huntington disease: an analysis from the Enroll-HD database. Mov. Disord. Clin. Pract. 6, 132–138 (2019).
Rowe, K. C. et al. Patterns of serotonergic antidepressant usage in prodromal Huntington disease. Psychiatry Res. 196, 309–314 (2012).
Damkier, P. & Videbech, P. The safety of second-generation antipsychotics during pregnancy: a clinically focused review. CNS Drugs 32, 351–366 (2018).
Committee on Drugs. Use of psychoactive medication during pregnancy and possible effects on the fetus and newborn. Pediatrics 105, 880–887 (2000).
Beier, K. & Pratt, D. P. Sydenham chorea. StatPearls https://www.statpearls.com/as/genetics/29792/ (2019).
Cardoso, F. Chorea gravidarum. Arch. Neurol. 59, 868–870 (2002).
Maia, D. P. et al. Pregnancy in patients with Sydenham’s chorea. Parkinsonism Relat. Disord. 18, 458–461 (2012).
Martino, D., Ganos, C. & Pringsheim, T. M. Tourette syndrome and chronic tic disorders: the clinical spectrum beyond tics. Int. Rev. Neurobiol. 134, 1461–1490 (2017).
Erenberg, G., Cruse, R. P. & Rothner, A. D. The natural history of Tourette syndrome: a follow-up study. Ann. Neurol. 22, 383–385 (1987).
Jankovic, J. & Rohaidy, H. Motor, behavioral and pharmacologic findings in Tourette’s syndrome. Can. J. Neurol. Sci. 14, 541–546 (1987).
Comings, D. E. & Comings, B. G. Tourette syndrome: clinical and psychological aspects of 250 cases. Am. J. Hum. Genet. 37, 435–450 (1985).
Eapen, V., Fox-Hiley, P., Banerjee, S. & Robertson, M. Clinical features and associated psychopathology in a Tourette syndrome cohort. Acta Neurol. Scand. 109, 255–260 (2004).
Freeman, R. D. et al. An international perspective on Tourette syndrome: selected findings from 3,500 individuals in 22 countries. Dev. Med. Child. Neurol. 42, 436–447 (2000).
Lapouse, R. & Monk, M. A. Behavior deviations in a representative sample of children: variation by sex, age, race, social class and family size. Am. J. Orthopsychiatry 34, 436–446 (1964).
Robertson, M. M. Diagnosing Tourette syndrome: is it a common disorder? J. Psychosom. Res. 55, 3–6 (2003).
Scahill, L., Sukhodolsky, D. G., Williams, S. K. & Leckman, J. F. Public health significance of tic disorders in children and adolescents. Adv. Neurol. 96, 240–248 (2005).
Robertson, M. M. The prevalence and epidemiology of Gilles de la Tourette syndrome. Part 1: the epidemiological and prevalence studies. J. Psychosom. Res. 65, 461–472 (2008).
Kerbeshian, J. & Burd, L. Epidemiology and comorbidity. The North Dakota prevalence studies of Tourette syndrome and other developmental disorders. Adv. Neurol. 58, 67–74 (1992).
Schlander, M., Schwarz, O., Rothenberger, A. & Roessner, V. Tic disorders: administrative prevalence and co-occurrence with attention-deficit/hyperactivity disorder in a German community sample. Eur. Psychiatry 26, 370–374 (2011).
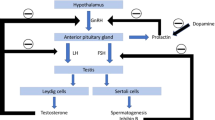
Martino, D., Macerollo, A. & Leckman, J. F. Neuroendocrine aspects of Tourette syndrome. Int. Rev. Neurobiol. 112, 239–279 (2013).
Robertson, M. M. The Gilles de la Tourette syndrome: the current status. Arch. Dis. Child. Educ. Pract. Ed. 97, 166–175 (2012).
Motlagh, M. G. et al. Severe psychosocial stress and heavy cigarette smoking during pregnancy: an examination of the pre- and perinatal risk factors associated with ADHD and Tourette syndrome. Eur. Child. Adolesc. Psychiatry 19, 755–764 (2010).
Dalsgaard, S., Waltoft, B. L., Leckman, J. F. & Mortensen, P. B. Maternal history of autoimmune disease and later development of Tourette syndrome in offspring. J. Am. Acad. Child. Adolesc. Psychiatry 54, 495–501.e1 (2015).
Fahim, C. et al. Somatosensory-motor bodily representation cortical thinning in Tourette: effects of tic severity, age and gender. Cortex 46, 750–760 (2010).
Lichter, D. G., Jackson, L. A. & Schachter, M. Clinical evidence of genomic imprinting in Tourette’s syndrome. Neurology 45, 924–928 (1995).
Mataix-Cols, D. et al. Familial risks of Tourette syndrome and chronic tic disorders. A population-based cohort study. JAMA Psychiatry 72, 787–793 (2015).
Zilhao, N. R. et al. Heritability of tic disorders: a twin-family study. Psychol. Med. 47, 1085–1096 (2017).
Yu, D. et al. Interrogating the genetic determinants of Tourette’s syndrome and other tic disorders through genome-wide association studies. Am. J. Psychiatry 176, 217–227 (2019).
Schwabe, M. J. & Konkol, R. J. Menstrual cycle-related fluctuations of tics in Tourette syndrome. Pediatr. Neurol. 8, 43–46 (1992).
Martino, D. & Hedderly, T. Tics and stereotypies: a comparative clinical review. Parkinsonism Relat. Disord. 59, 117–124 (2019).
Santangelo, S. L. et al. Tourette’s syndrome: what are the influences of gender and comorbid obsessive–compulsive disorder? J. Am. Acad. Child. Adolesc. Psychiatry 33, 795–804 (1994).
Rodgers, S. et al. Sex-related and non-sex-related comorbidity subtypes of tic disorders: a latent class approach. Eur. J. Neurol. 21, 700–707 (2014).
Lewin, A. B. et al. A phenomenological investigation of women with Tourette or other chronic tic disorders. Compr. Psychiatry 53, 525–534 (2012).
Schuerholz, L. J., Cutting, L., Mazzocco, M. M., Singer, H. S. & Denckla, M. B. Neuromotor functioning in children with Tourette syndrome with and without attention deficit hyperactivity disorder. J. Child. Neurol. 12, 438–442 (1997).
Kompoliti, K., Goetz, C. G., Leurgans, S., Raman, R. & Comella, C. L. Estrogen, progesterone, and tic severity in women with Gilles de la Tourette syndrome. Neurology 57, 1519 (2001).
Peterson, B. S. et al. Steroid hormones and CNS sexual dimorphisms modulate symptom expression in Tourette’s syndrome. Psychoneuroendocrinology 17, 553–563 (1992).
Pauls, D. L., Leckman, J. F., Towbin, K. E., Zahner, G. E. & Cohen, D. J. A possible genetic relationship exists between Tourette’s syndrome and obsessive-compulsive disorder. Psychopharmacol. Bull. 22, 730–733 (1986).
Burd, L. et al. Long-term follow-up of an epidemiologically defined cohort of patients with Tourette syndrome. J. Child. Neurol. 16, 431–437 (2001).
Author information
Authors and Affiliations
Contributions
E.M. and S.M. conceived the paper. All authors contributed to the literature search and to the writing. S.M. designed the figures. E.M. provided guidance for specific areas of competence and the overall manuscript outline.
Corresponding author
Ethics declarations
Competing interests
E.M. has received honoraria for lecturing from Medtronic and for acting as a consultant from Medtronic and Newronika. She has received research grants from Merz and educational grants from Boston, Homeperf and LVL. The other authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Neurology thanks G. Defazio and other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Meoni, S., Macerollo, A. & Moro, E. Sex differences in movement disorders. Nat Rev Neurol 16, 84–96 (2020). https://doi.org/10.1038/s41582-019-0294-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41582-019-0294-x
This article is cited by
-
Association between sex and Huntington’s disease: an updated review on symptomatology and prognosis of neurodegenerative disorders
Wiener Medizinische Wochenschrift (2024)
-
The correlation between prenatal maternal active smoking and neurodevelopmental disorders in children: a systematic review and meta-analysis
BMC Public Health (2023)
-
Pattern and frequency of involuntary movements: hospital-based study
The Egyptian Journal of Neurology, Psychiatry and Neurosurgery (2023)
-
The experience of Anxiety for people with Parkinson’s disease
npj Parkinson's Disease (2023)
-
Sex and gender in neurodevelopmental conditions
Nature Reviews Neurology (2023)