Abstract
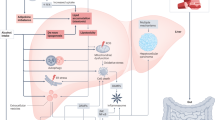
Non-alcoholic fatty liver disease (NAFLD) is characterized by the accumulation of fat in more than 5% of hepatocytes in the absence of excessive alcohol consumption and other secondary causes of hepatic steatosis. In 2020, the more inclusive term metabolic (dysfunction)-associated fatty liver disease (MAFLD) — defined by broader diagnostic criteria — was proposed to replace the term NAFLD. The new terminology and revised definition better emphasize the pathogenic role of metabolic dysfunction and uses a set of definitive, inclusive criteria for diagnosis. Diagnosis of MAFLD is based on evidence of hepatic steatosis (as assessed by liver biopsy, imaging techniques or blood biomarkers and scores) in persons who are overweight or obese and have type 2 diabetes mellitus or metabolic dysregulation, regardless of the coexistence of other liver diseases or excessive alcohol consumption. The known association between NAFLD and chronic kidney disease (CKD) and our understanding that CKD can occur as a consequence of metabolic dysfunction suggests that individuals with MAFLD — who by definition have fatty liver and metabolic comorbidities — are at increased risk of CKD. In this Perspective article, we discuss the clinical associations between MAFLD and CKD, the pathophysiological mechanisms by which MAFLD may increase the risk of CKD and the potential drug treatments that may benefit both conditions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Chalasani, N. et al. The diagnosis and management of non-alcoholic fatty liver disease: practice guideline by the American Gastroenterological Association, American Association for the Study of Liver Diseases, and American College of Gastroenterology. Gastroenterology 142, 1592–1609 (2012).
Younossi, Z. M. et al. Global epidemiology of nonalcoholic fatty liver disease — meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 64, 73–84 (2016).
Sarin, S. K. et al. Liver diseases in the Asia-Pacific region: a Lancet Gastroenterology & Hepatology Commission. Lancet Gastroenterol. Hepatol. 5, 167–228 (2020).
Targher, G., Chonchol, M. B. & Byrne, C. D. CKD and nonalcoholic fatty liver disease. Am. J. Kidney Dis. 64, 638–652 (2014).
Musso, G. et al. Association of non-alcoholic fatty liver disease with chronic kidney disease: a systematic review and meta-analysis. PLoS Med. 11, e1001680 (2014).
Mantovani, A. et al. Nonalcoholic fatty liver disease increases risk of incident chronic kidney disease: a systematic review and meta-analysis. Metabolism 79, 64–76 (2018).
Wijarnpreecha, K. et al. Nonalcoholic fatty liver disease and albuminuria: a systematic review and meta-analysis. Eur. J. Gastroenterol. Hepatol. 30, 986–994 (2018).
Mantovani, A. et al. Non-alcoholic fatty liver disease and risk of incident chronic kidney disease: an updated meta-analysis. Gut https://doi.org/10.1136/gutjnl-2020-323082 (2020).
Sun, D. Q. et al. Higher liver stiffness scores are associated with early kidney dysfunction in patients with histologically proven non-cirrhotic NAFLD. Diabetes Metab. 46, 288–295 (2020).
Paik, J. et al. Chronic kidney disease is independently associated with increased mortality in patients with nonalcoholic fatty liver disease. Liver Int. 39, 342–352 (2019).
Yilmaz, Y. et al. Microalbuminuria in nondiabetic patients with nonalcoholic fatty liver disease: association with liver fibrosis. Metabolism 59, 1327–1330 (2010).
Onnerhag, K., Dreja, K., Nilsson, P. M. & Lindgren, S. Increased mortality in non-alcoholic fatty liver disease with chronic kidney disease is explained by metabolic comorbidities. Clin. Res. Hepatol. Gastroenterol. 43, 542–550 (2019).
Eslam, M. et al. A new definition for metabolic dysfunction-associated fatty liver disease: an international expert consensus statement. J. Hepatol. 73, 202–209 (2020).
Brunt, E. M. et al. Concurrence of histologic features of steatohepatitis with other forms of chronic liver disease. Mod. Pathol. 16, 49–56 (2003).
Eslam, M., Sanyal, A. J. & George, J., International Consensus Panel. MAFLD: a consensus-driven proposed nomenclature for metabolic associated fatty liver disease. Gastroenterology 158, 1999–2014.e1 (2020).
Wong, V. W. et al. Impact of the new definition of metabolic associated fatty liver disease on the epidemiology of the disease. Clin. Gastroenterol. Hepatol. 19, 2161–2171.e5 (2021).
Sun, D. Q. et al. MAFLD and risk of CKD. Metabolism 115, 154433 (2021).
Targher, G. Concordance between MAFLD and NAFLD diagnostic criteria in ‘real-world’ data. Liver Int. 40, 2879–2880 (2020).
Schaffner, F. & Thaler, H. Nonalcoholic fatty liver disease. Prog. Liver Dis. 8, 283–298 (1986).
Zelman, S. The liver in obesity. AMA 90, 141–156 (1952).
Cortez-Pinto, H., Camilo, M. E., Baptista, A., De Oliveira, A. G. & De Moura, M. C. Non-alcoholic fatty liver: another feature of the metabolic syndrome? Clin. Nutr. 18, 353–358 (1999).
Marceau, P. et al. Liver pathology and the metabolic syndrome X in severe obesity. J. Clin. Endocrinol. Metab. 84, 1513–1517 (1999).
Dunn, W. et al. Modest alcohol consumption is associated with decreased prevalence of steatohepatitis in patients with non-alcoholic fatty liver disease (NAFLD). J. Hepatol. 57, 384–391 (2012).
Kwon, H. K., Greenson, J. K. & Conjeevaram, H. S. Effect of lifetime alcohol consumption on the histological severity of non-alcoholic fatty liver disease. Liver Int. 34, 129–135 (2014).
Ascha, M. S. et al. The incidence and risk factors of hepatocellular carcinoma in patients with nonalcoholic steatohepatitis. Hepatology 51, 1972–1978 (2010).
Mitchell, T. et al. Type and pattern of alcohol consumption is associated with liver fibrosis in patients with non-alcoholic fatty liver disease. Am. J. Gastroenterol. 113, 1484–1493 (2018).
Mohanty, S. R. et al. Influence of ethnicity on histological differences in non-alcoholic fatty liver disease. J. Hepatol. 50, 797–804 (2009).
Hashimoto, E. & Tokushige, K. Prevalence, gender, ethnic variations, and prognosis of NASH. J. Gastroenterol. 46(Suppl 1), 63–69 (2011).
Wang, T. Y., George, J. & Zheng, M. H. Metabolic (dysfunction) associated fatty liver: more evidence and a bright future. Hepatobiliary Surg. Nutr. https://doi.org/10.21037/hbsn-21-352 (2021).
Zheng, K. I. et al. From NAFLD to MAFLD: a “redefining” moment for fatty liver disease. Chin. Med. J. 133, 2271–2273 (2020).
Younossi, Z. M. et al. From NAFLD to MAFLD: Implications of a premature change in terminology. Hepatology 73, 1194–1198 (2020).
Chen, F. et al. Lean NAFLD: a distinct entity shaped by differential metabolic adaptation. Hepatology 71, 1213–1227 (2020).
Frey, S. et al. Prevalence of NASH/NAFLD in people with obesity who are currently classified as metabolically healthy. Surg. Obes. Relat. Dis. 16, 2050–2057 (2020).
Yamamura, S. et al. MAFLD identifies patients with significant hepatic fibrosis better than NAFLD. Liver Int. 40, 3018–3030 (2020).
Huang, S. C. et al. Clinical and histologic features of patients with biopsy-proven metabolic dysfunction-associated fatty liver disease. Gut Liver 15, 451–458 (2021).
Lin, S. et al. Comparison of MAFLD and NAFLD diagnostic criteria in real world. Liver Int. 40, 2082–2089 (2020).
Mak, L. Y., Yuen, M. F. & Seto, W. K. Letter regarding “A new definition for metabolic dysfunction-associated fatty liver disease: an international expert consensus statement”. J. Hepatol. 73, 1573–1574 (2020).
Zheng, K. I., Sun, D. Q., Jin, Y., Zhu, P. W. & Zheng, M. H. Clinical utility of the MAFLD definition. J. Hepatol. 74, 989–991 (2021).
Yilmaz, Y., Byrne, C. D. & Musso, G. A single-letter change in an acronym: signals, reasons, promises, challenges, and steps ahead for moving from NAFLD to MAFLD. Expert Rev. Gastroenterol. Hepatol. 15, 345–352 (2021).
Angulo, P. et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology 149, 389–397.e10 (2015).
Hagstrom, H. et al. Fibrosis stage but not NASH predicts mortality and time to development of severe liver disease in biopsy-proven NAFLD. J. Hepatol. 67, 1265–1273 (2017).
Yeung, M. W. et al. Advanced liver fibrosis but not steatosis is independently associated with albuminuria in Chinese patients with type 2 diabetes. J. Hepatol. 68, 147–156 (2017).
Zhang, H. J., Wang, Y. Y., Chen, C., Lu, Y. L. & Wang, N. J. Cardiovascular and renal burdens of metabolic associated fatty liver disease from serial US national surveys, 1999–2016. Chin. Med. J. 134, 1593–1601 (2021).
Deng, Y., Zhao, Q. & Gong, R. Association between metabolic associated fatty liver disease and chronic kidney disease: a cross-sectional study from NHANES 2017-2018. Diabetes Metab. Syndr. Obes. 14, 1751–1761 (2021).
Vespasiani-Gentilucci, U. et al. Promoting genetics in non-alcoholic fatty liver disease: combined risk score through polymorphisms and clinical variables. World J. Gastroenterol. 24, 4835–4845 (2018).
Pirazzi, C. et al. PNPLA3 has retinyl-palmitate lipase activity in human hepatic stellate cells. Hum. Mol. Genet. 23, 4077–4085 (2014).
Xu, R., Tao, A., Zhang, S., Deng, Y. & Chen, G. Association between patatin-like phospholipase domain containing 3 gene (PNPLA3) polymorphisms and nonalcoholic fatty liver disease: a HuGE review and meta-analysis. Sci. Rep. 5, 9284 (2015).
Tilson, S. G. et al. Modeling PNPLA3-Associated NAFLD using human-induced pluripotent stem cells. Hepatology 74, 2998–3017 (2021).
Pirazzi, C. et al. Patatin-like phospholipase domain-containing 3 (PNPLA3) I148M (rs738409) affects hepatic VLDL secretion in humans and in vitro. J. Hepatol. 57, 1276–1282 (2012).
Hellemans, K., Grinko, I., Rombouts, K., Schuppan, D. & Geerts, A. All-trans and 9-cis retinoic acid alter rat hepatic stellate cell phenotype differentially. Gut 45, 134–142 (1999).
Bruschi, F. V. et al. The PNPLA3 I148M variant modulates the fibrogenic phenotype of human hepatic stellate cells. Hepatology 65, 1875–1890 (2017).
Targher, G. et al. Relationship between PNPLA3 rs738409 polymorphism and decreased kidney function in children With NAFLD. Hepatology 70, 142–153 (2019).
Oniki, K. et al. Influence of the PNPLA3 rs738409 polymorphism on non-alcoholic fatty liver disease and renal function among normal weight subjects. PLoS One 10, e0132640 (2015).
Marzuillo, P. et al. Nonalcoholic fatty liver disease and eGFR levels could be linked by the PNPLA3 I148M polymorphism in children with obesity. Pediatr. Obes. 14, e12539 (2019).
Mantovani, A. et al. Association between PNPLA3rs738409 polymorphism decreased kidney function in postmenopausal type 2 diabetic women with or without non-alcoholic fatty liver disease. Diabetes Metab. 45, 480–487 (2019).
Sun, D. Q. et al. PNPLA3 rs738409 is associated with renal glomerular and tubular injury in NAFLD patients with persistently normal ALT levels. Liver Int. 40, 107–119 (2020).
Mantovani, A. et al. PNPLA3 I148M gene variant and chronic kidney disease in type 2 diabetic patients with NAFLD: clinical and experimental findings. Liver Int. 40, 1130–1141 (2020).
Gellert-Kristensen, H., Nordestgaard, B. G., Tybjaerg-Hansen, A. & Stender, S. High risk of fatty liver disease amplifies the alanine transaminase-lowering effect of a HSD17B13 variant. Hepatology 71, 56–66 (2020).
Luukkonen, P. K. et al. Hydroxysteroid 17-beta dehydrogenase 13 variant increases phospholipids and protects against fibrosis in nonalcoholic fatty liver disease. JCI Insight 5, e132158 (2020).
Di Sessa, A. et al. Pediatric non-alcoholic fatty liver disease and kidney function: effect of HSD17B13 variant. World J. Gastroenterol. 26, 5474–5483 (2020).
Sun, D. Q. et al. The HSD17B13 rs72613567 variant is associated with lower levels of albuminuria in patients with biopsy-proven nonalcoholic fatty liver disease. Nutr. Metab. Cardiovasc. Dis. 31, 1822–1831 (2021).
Luo, F., Oldoni, F. & Das, A. TM6SF2: a novel genetic player in nonalcoholic fatty liver and cardiovascular disease. Hepatol. Commun. https://doi.org/10.1002/hep4.1822 (2021).
Musso, G., Cassader, M., Paschetta, E. & Gambino, R. TM6SF2 may drive postprandial lipoprotein cholesterol toxicity away from the vessel walls to the liver in NAFLD. J. Hepatol. 64, 979–981 (2016).
Musso, G., Cassader, M. & Gambino, R. PNPLA3 rs738409 and TM6SF2 rs58542926 gene variants affect renal disease and function in nonalcoholic fatty liver disease. Hepatology 62, 658–659 (2015).
Marzuillo, P. et al. Transmembrane 6 superfamily member 2 167K allele improves renal function in children with obesity. Pediatr. Res. 88, 300–304 (2020).
Mancina, R. M. et al. The MBOAT7-TMC4 Variant rs641738 increases risk of nonalcoholic fatty liver disease in individuals of European descent. Gastroenterology 150, 1219–1230.e6 (2016).
Thabet, K. et al. The membrane-bound O-acyltransferase domain-containing 7 variant rs641738 increases inflammation and fibrosis in chronic hepatitis B. Hepatology 65, 1840–1850 (2017).
Koo, B. K. et al. Association between a polymorphism in MBOAT7 and chronic kidney disease in patients with biopsy-confirmed nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 18, 2837–2839.e2 (2020).
Sliz, E. et al. NAFLD risk alleles in PNPLA3, TM6SF2, GCKR and LYPLAL1 show divergent metabolic effects. Hum. Mol. Genet. 27, 2214–2223 (2018).
Boger, C. A. et al. Association of eGFR-related loci identified by GWAS with incident CKD and ESRD. PLoS Genet. 7, e1002292 (2011).
Hishida, A. et al. GCK, GCKR polymorphisms and risk of chronic kidney disease in Japanese individuals: data from the J-MICC Study. J. Nephrol. 27, 143–149 (2014).
Simons, P. et al. Association of common gene variants in glucokinase regulatory protein with cardiorenal disease: a systematic review and meta-analysis. PLoS One 13, e0206174 (2018).
Di Costanzo, A. et al. Nonalcoholic Fatty Liver Disease (NAFLD), but not its susceptibility gene variants, influences the decrease of kidney function in overweight/obese children. Int. J. Mol. Sci. 20, 4444 (2019).
Xia, M., Zeng, H., Wang, S., Tang, H. & Gao, X. Insights into contribution of genetic variants towards the susceptibility of MAFLD revealed by the NMR-based lipoprotein profiling. J. Hepatol. 74, 974–977 (2021).
Raj, D., Tomar, B., Lahiri, A. & Mulay, S. R. The gut-liver-kidney axis: Novel regulator of fatty liver associated chronic kidney disease. Pharmacol. Res. 152, 104617 (2020).
Meijers, B., Evenepoel, P. & Anders, H. J. Intestinal microbiome and fitness in kidney disease. Nat. Rev. Nephrol. 15, 531–545 (2019).
Mafra, D. et al. Food as medicine: targeting the uraemic phenotype in chronic kidney disease. Nat. Rev. Nephrol. 17, 153–171 (2021).
Tan, X. et al. Trimethylamine N-oxide aggravates liver steatosis through modulation of bile acid metabolism and inhibition of farnesoid X receptor signaling in nonalcoholic fatty liver disease. Mol. Nutr. Food Res. 63, e1900257 (2019).
Ravid, J. D., Kamel, M. H. & Chitalia, V. C. Uraemic solutes as therapeutic targets in CKD-associated cardiovascular disease. Nat. Rev. Nephrol. 17, 402–416 (2021).
Tang, W. H. et al. Gut microbiota-dependent trimethylamine N-oxide (TMAO) pathway contributes to both development of renal insufficiency and mortality risk in chronic kidney disease. Circ. Res. 116, 448–455 (2015).
Herman-Edelstein, M., Weinstein, T. & Levi, M. Bile acid receptors and the kidney. Curr. Opin. Nephrol. Hypertens. 27, 56–62 (2018).
Wang, X. X. et al. A dual agonist of farnesoid X receptor (FXR) and the G protein-coupled receptor TGR5, INT-767, reverses age-related kidney disease in mice. J. Biol. Chem. 292, 12018–12024 (2017).
Jiao, N. et al. Suppressed hepatic bile acid signalling despite elevated production of primary and secondary bile acids in NAFLD. Gut 67, 1881–1891 (2018).
Morrison, D. J. & Preston, T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes 7, 189–200 (2016).
Wang, S. et al. Quantitative reduction in short-chain fatty acids, especially butyrate, contributes to the progression of chronic kidney disease. Clin. Sci. 133, 1857–1870 (2019).
Andrade-Oliveira, V. et al. Gut bacteria products prevent AKI induced by ischemia-reperfusion. J. Am. Soc. Nephrol. 26, 1877–1888 (2015).
Pluznick, J. L. et al. Olfactory receptor responding to gut microbiota-derived signals plays a role in renin secretion and blood pressure regulation. Proc. Natl Acad. Sci. USA 110, 4410–4415 (2013).
Chambers, E. S. et al. Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults. Gut 64, 1744–1754 (2015).
Zhou, D. et al. Sodium butyrate reduces high-fat diet-induced non-alcoholic steatohepatitis through upregulation of hepatic GLP-1R expression. Exp. Mol. Med. 50, 1–12 (2018).
Chambers, E. S. et al. The effects of dietary supplementation with inulin and inulin-propionate ester on hepatic steatosis in adults with non-alcoholic fatty liver disease. Diabetes Obes. Metab. 21, 372–376 (2019).
Zhao, Z. H. et al. Sodium butyrate supplementation inhibits hepatic steatosis by stimulating liver kinase B1 and insulin-induced gene. Cell Mol. Gastroenterol. Hepatol. 12, 857–871 (2021).
Chambers, E. S., Morrison, D. J. & Frost, G. Control of appetite and energy intake by SCFA: what are the potential underlying mechanisms? Proc. Nutr. Soc. 74, 328–336 (2015).
Zhang, S. et al. Dietary fiber-derived short-chain fatty acids: a potential therapeutic target to alleviate obesity-related nonalcoholic fatty liver disease. Obes. Rev. 22, e13316 (2021).
Giorgio, V. et al. Intestinal permeability is increased in children with non-alcoholic fatty liver disease, and correlates with liver disease severity. Dig. Liver Dis. 46, 556–560 (2014).
Shi, K. et al. Gut bacterial translocation may aggravate microinflammation in hemodialysis patients. Dig. Dis. Sci. 59, 2109–2117 (2014).
Sanchez-Lozada, L. G. et al. Uric acid activates aldose reductase and the polyol pathway for endogenous fructose and fat production causing development of fatty liver in rats. J. Biol. Chem. 294, 4272–4281 (2019).
Xu, K. et al. Gender effect of hyperuricemia on the development of nonalcoholic fatty liver disease (NAFLD): a clinical analysis and mechanistic study. Biomed. Pharmacother. 117, 109158 (2019).
Ding, R. B., Bao, J. & Deng, C. X. Emerging roles of SIRT1 in fatty liver diseases. Int. J. Biol. Sci. 13, 852–867 (2017).
Hasegawa, K. et al. Renal tubular Sirt1 attenuates diabetic albuminuria by epigenetically suppressing Claudin-1 overexpression in podocytes. Nat. Med. 19, 1496–1504 (2013).
Ralto, K. M., Rhee, E. P. & Parikh, S. M. NAD+ homeostasis in renal health and disease. Nat. Rev. Nephrol. 16, 99–111 (2020).
Sato, Y. et al. The case for uric acid-lowering treatment in patients with hyperuricaemia and CKD. Nat. Rev. Nephrol. 15, 767–775 (2019).
Steiger, S., Ma, Q. & Anders, H. J. The case for evidence-based medicine for the association between hyperuricaemia and CKD. Nat. Rev. Nephrol. 16, 422 (2020).
Kurella, M., Lo, J. C. & Chertow, G. M. Metabolic syndrome and the risk for chronic kidney disease among nondiabetic adults. J. Am. Soc. Nephrol. 16, 2134–2140 (2005).
Singh, A. K. & Kari, J. A. Metabolic syndrome and chronic kidney disease. Curr. Opin. Nephrol. Hypertens. 22, 198–203 (2013).
Chen, J. et al. The metabolic syndrome and chronic kidney disease in U.S. adults. Ann. Intern. Med. 140, 167–174 (2004).
Stojsavljevic, S., Gomercic Palcic, M., Virovic Jukic, L., Smircic Duvnjak, L. & Duvnjak, M. Adipokines and proinflammatory cytokines, the key mediators in the pathogenesis of nonalcoholic fatty liver disease. World J. Gastroenterol. 20, 18070–18091 (2014).
Ix, J. H. & Sharma, K. Mechanisms linking obesity, chronic kidney disease, and fatty liver disease: the roles of fetuin-A, adiponectin, and AMPK. J. Am. Soc. Nephrol. 21, 406–412 (2010).
Mantovani, A. et al. Non-alcoholic fatty liver disease and risk of incident diabetes mellitus: an updated meta-analysis of 501 022 adult individuals. Gut 70, 962–969 (2021).
Spoto, B., Pisano, A. & Zoccali, C. Insulin resistance in chronic kidney disease: a systematic review. Am. J. Physiol. Renal Physiol. 311, F1087–F1108 (2016).
Guerreiro, G. T. S., Longo, L., Fonseca, M. A., de Souza, V. E. G. & Alvares-da-Silva, M. R. Does the risk of cardiovascular events differ between biopsy-proven NAFLD and MAFLD? Hepatol. Int. 15, 380–391 (2021).
Gupta, A. & Quigg, R. J. Glomerular diseases associated with hepatitis B and C. Adv. Chronic Kidney Dis. 22, 343–351 (2015).
Eslam, M. et al. The Asian Pacific Association for the Study of the Liver clinical practice guidelines for the diagnosis and management of metabolic associated fatty liver disease. Hepatol. Int. 14, 889–919 (2020).
Alicic, R. Z., Cox, E. J., Neumiller, J. J. & Tuttle, K. R. Incretin drugs in diabetic kidney disease: biological mechanisms and clinical evidence. Nat. Rev. Nephrol. 17, 227–244 (2021).
Kang, A. & Jardine, M. J. SGLT2 inhibitors may offer benefit beyond diabetes. Nat. Rev. Nephrol. 17, 83–84 (2021).
DeFronzo, R. A., Reeves, W. B. & Awad, A. S. Pathophysiology of diabetic kidney disease: impact of SGLT2 inhibitors. Nat. Rev. Nephrol. 17, 319–334 (2021).
Marton, A. et al. Organ protection by SGLT2 inhibitors: role of metabolic energy and water conservation. Nat. Rev. Nephrol. 17, 65–77 (2021).
Sloan, L. A. Review of glucagon-like peptide-1 receptor agonists for the treatment of type 2 diabetes mellitus in patients with chronic kidney disease and their renal effects. J. Diabetes 11, 938–948 (2019).
Patel Chavez, C., Cusi, K. & Kadiyala, S. The emerging role of glucagon-like peptide-1 receptor agonists for the management of NAFLD. J. Clin. Endocrinol. Metab. https://doi.org/10.1210/clinem/dgab578 (2021).
American Diabetes, A. 9. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes-2021. Diabetes Care 44, S111–S124 (2021).
Acknowledgements
The authors’ work was supported by grants from the National Natural Science Foundation of China (82070588, 82000690), High Level Creative Talents from the Department of Public Health in Zhejiang Province, Project of New Century 551 Talent Nurturing in Wenzhou and a Project of Science and Technology Development Fund in Wuxi (N20202001). D.-Q.S. is supported in part by grants from the Youth Research Project Fund from Wuxi Municipal Health Commission (Q201932), Top-notch Talents from Young and Middle-Age Health Care in Wuxi (BJ2020026). G.T. is supported in part by grants from the University School of Medicine of Verona, Verona, Italy. C.D.B. is supported in part by the Southampton NIHR Biomedical Research Centre (IS-BRC-20004), UK.
Author information
Authors and Affiliations
Contributions
R.-F.W., Z.-Y.B. and D.-Q.S. researched data for the article. T.-Y.W., G.T., C.D.B., D.-Q.S. and M.-H.Z. wrote the manuscript. All authors contributed substantially to the discussion of the content and reviewed and/or edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Nephrology thanks Manuel Praga, Yusuf Yilmaz and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, TY., Wang, RF., Bu, ZY. et al. Association of metabolic dysfunction-associated fatty liver disease with kidney disease. Nat Rev Nephrol 18, 259–268 (2022). https://doi.org/10.1038/s41581-021-00519-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41581-021-00519-y
This article is cited by
-
MAFLD as part of systemic metabolic dysregulation
Hepatology International (2024)
-
Comparative effectiveness of multiple different treatment regimens for nonalcoholic fatty liver disease with type 2 diabetes mellitus: a systematic review and Bayesian network meta-analysis of randomised controlled trials
BMC Medicine (2023)
-
Tripartite motif containing 26 prevents steatohepatitis progression by suppressing C/EBPδ signalling activation
Nature Communications (2023)
-
A comprehensive risk factor analysis using association rules in people with diabetic kidney disease
Scientific Reports (2023)
-
MAFLD and NAFLD in the prediction of incident chronic kidney disease
Scientific Reports (2023)