Abstract
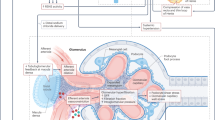
Excessive adiposity raises blood pressure and accounts for 65–75% of primary hypertension, which is a major driver of cardiovascular and kidney diseases. In obesity, abnormal kidney function and associated increases in tubular sodium reabsorption initiate hypertension, which is often mild before the development of target organ injury. Factors that contribute to increased sodium reabsorption in obesity include kidney compression by visceral, perirenal and renal sinus fat; increased renal sympathetic nerve activity (RSNA); increased levels of anti-natriuretic hormones, such as angiotensin II and aldosterone; and adipokines, particularly leptin. The renal and neurohormonal pathways of obesity and hypertension are intertwined. For example, leptin increases RSNA by stimulating the central nervous system proopiomelanocortin–melanocortin 4 receptor pathway, and kidney compression and RSNA contribute to renin–angiotensin–aldosterone system activation. Glucocorticoids and/or oxidative stress may also contribute to mineralocorticoid receptor activation in obesity. Prolonged obesity and progressive renal injury often lead to the development of treatment-resistant hypertension. Patient management therefore often requires multiple antihypertensive drugs and concurrent treatment of dyslipidaemia, insulin resistance, diabetes and inflammation. If more effective strategies for the prevention and control of obesity are not developed, cardiorenal, metabolic and other obesity-associated diseases could overwhelm health-care systems in the future.
Key points
-
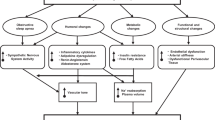
Obesity is associated with chronic diseases, including hypertension, which is a major risk factor for chronic kidney disease and cardiovascular diseases such as stroke, myocardial infarction and heart failure.
-
Excessive weight gain, especially when associated with visceral obesity, raises blood pressure and is the most important known risk factor for primary (essential) hypertension.
-
Abnormal kidney function, which is associated with increased tubular sodium reabsorption, has a key role in initiating obesity-associated hypertension.
-
Mechanisms that initiate obesity-induced sodium retention include kidney compression by visceral, perirenal and renal sinus fat, stimulation of the renin–angiotensin–aldosterone system, aldosterone-independent mineralocorticoid receptor activation and activation of the sympathetic nervous system.
-
Sympathetic activation in obesity may be mediated by hypoxia, chemoreceptor activation, baroreflex dysfunction and adipokines, including leptin, which activates the central nervous system melanocortin pathway.
-
Chronic obesity may gradually amplify hypertension, resulting in resistance to antihypertensive treatment and initiating a pathophysiological cascade of factors that exacerbate target organ injury.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
NCD Risk Factor Collaboration. Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128.9 million children, adolescents, and adults. Lancet 390, 2627–2642 (2017).
Afshin, A., Reitsma, M. B. & Murray, C. J. L. Health effects of overweight and obesity in 195 countries. N. Engl. J. Med. 377, 1496–1497 (2017).
Gregg, E. W. & Shaw, J. E. Global health effects of overweight and obesity. N. Engl. J. Med. 377, 80–81 (2017).
World Health Organization. Obesity and overweight — key facts. WHO http://www.who.int/mediacentre/factsheets/fs311/en/ (2018).
Benjamin, E. J. et al. Heart disease and stroke statistics-2018 update: a report from the American Heart Association. Circulation 137, e67–e492 (2018).
Hall, J. E., do Carmo, J. M., da Silva, A. A., Wang, Z. & Hall, M. E. Obesity-induced hypertension: interaction of neurohumoral and renal mechanisms. Circ. Res. 116, 991–1006 (2015).
Garrison, R. J., Kannel, W. B., Stokes, J. III & Castelli, W. P. Incidence and precursors of hypertension in young adults: the Framingham Offspring Study. Prev. Med. 16, 235–251 (1987).
USRDS Coordinating Center. United States Renal Data System Annual Report. USRDS https://www.usrds.org/adr.aspx (2017).
Hall, J. E. et al. Is obesity a major cause of chronic kidney disease? Adv. Ren. Replace. Ther. 11, 41–54 (2004).
Hall, M. E. et al. Obesity, hypertension, and chronic kidney disease. Int. J. Nephrol. Renovasc. Dis. 7, 75–88 (2014).
Whaley-Connell, A. & Sowers, J. R. Obesity and kidney disease: from population to basic science and the search for new therapeutic targets. Kidney Int. 92, 313–323 (2017).
DeMarco, V. G., Aroor, A. R. & Sowers, J. R. The pathophysiology of hypertension in patients with obesity. Nat. Rev. Endocrinol. 10, 364–376 (2014).
Neeland, I. J., Poirier, P. & Despres, J. P. Cardiovascular and metabolic heterogeneity of obesity: clinical challenges and implications for management. Circulation 137, 1391–1406 (2018).
Tchernof, A. & Despres, J. P. Pathophysiology of human visceral obesity: an update. Physiol. Rev. 93, 359–404 (2013).
Mohammed, B. S., Cohen, S., Reeds, D., Young, V. L. & Klein, S. Long-term effects of large-volume liposuction on metabolic risk factors for coronary heart disease. Obesity 16, 2648–2651 (2008).
Schiavon, C. A. et al. Effects of bariatric surgery in obese patients with hypertension: the GATEWAY randomized trial (gastric bypass to treat obese patients with steady hypertension). Circulation 137, 1132–1142 (2018).
Schauer, P. R. et al. Bariatric surgery versus intensive medical therapy for diabetes — 5-year outcomes. N. Engl. J. Med. 376, 641–651 (2017).
Piche, M. E., Poirier, P., Lemieux, I. & Despres, J. P. Overview of epidemiology and contribution of obesity and body fat distribution to cardiovascular disease: an update. Prog. Cardiovasc. Dis. 61, 103–113 (2018).
Chughtai, H. L. et al. Renal sinus fat and poor blood pressure control in middle-aged and elderly individuals at risk for cardiovascular events. Hypertension 56, 901–906 (2010).
Foster, M. C. et al. Fatty kidney, hypertension, and chronic kidney disease: the Framingham Heart Study. Hypertension 58, 784–790 (2011).
Chandra, A. et al. The relationship of body mass and fat distribution with incident hypertension: observations from the dallas heart study. J. Am. Coll. Cardiol. 64, 997–1002 (2014).
Ortega, F. B., Lavie, C. J. & Blair, S. N. Obesity and cardiovascular disease. Circ. Res. 118, 1752–1770 (2016).
Cui, Z., Truesdale, K. P., Bradshaw, P. T., Cai, J. & Stevens, J. Three-year weight change and cardiometabolic risk factors in obese and normal weight adults who are metabolically healthy: the atherosclerosis risk in communities study. Int. J. Obes. 39, 1203–1208 (2015).
Eckel, N. et al. Transition from metabolic healthy to unhealthy phenotypes and association with cardiovascular disease risk across BMI categories in 90 257 women (the Nurses’ Health Study): 30 year follow-up from a prospective cohort study. Lancet Diabetes Endocrinol. 6, 714–724 (2018).
Jones, D. W., Kim, J. S., Andrew, M. E., Kim, S. J. & Hong, Y. P. Body mass index and blood pressure in Korean men and women: the Korean National Blood Pressure Survey. J. Hypertens. 12, 1433–1437 (1994).
Bramlage, P. et al. Hypertension in overweight and obese primary care patients is highly prevalent and poorly controlled. Am. J. Hypertens. 17, 904–910 (2004).
Whelton, P. K. et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension 71, 1269–1324 (2018).
Nyamdorj, R. et al. Comparison of body mass index with waist circumference, waist-to-hip ratio, and waist-to-stature ratio as a predictor of hypertension incidence in Mauritius. J. Hypertens. 26, 866–870 (2008).
Field, A. E. et al. Impact of overweight on the risk of developing common chronic diseases during a 10-year period. Arch. Intern. Med. 161, 1581–1586 (2001).
Shihab, H. M. et al. Body mass index and risk of incident hypertension over the life course: the Johns Hopkins Precursors Study. Circulation 126, 2983–2989 (2012).
Harrap, S. B. & Charchar, F. J. Genetics of blood pressure: time to curate the collection. J. Hypertens. 35, 1360–1362 (2017).
Saad, M. F. et al. Racial differences in the relation between blood pressure and insulin resistance. N. Engl. J. Med. 324, 733–739 (1991).
Weyer, C. et al. Ethnic differences in insulinemia and sympathetic tone as links between obesity and blood pressure. Hypertension 36, 531–537 (2000).
de Courten, M. P., Pettitt, D. J. & Knowler, W. C. Hypertension in Pima Indians: prevalence and predictors. Public Health Rep. 111 (Suppl. 2), 40–43 (1996).
Hall, J. E., Crook, E. D., Jones, D. W., Wofford, M. R. & Dubbert, P. M. Mechanisms of obesity-associated cardiovascular and renal disease. Am. J. Med. Sci. 324, 127–137 (2002).
Alpert, M. A., Karthikeyan, K., Abdullah, O. & Ghadban, R. Obesity and cardiac remodeling in adults: mechanisms and clinical implications. Prog. Cardiovasc. Dis. 61, 114–123 (2018).
Van Vliet, B. N., Hall, J. E., Mizelle, H. L., Montani, J. P. & Smith, M. J. Jr. Reduced parasympathetic control of heart rate in obese dogs. Am. J. Physiol. 269, H629–H637 (1995).
Raitakari, M. et al. Weight reduction with very-low-caloric diet and endothelial function in overweight adults: role of plasma glucose. Arterioscler. Thromb. Vasc. Biol. 24, 124–128 (2004).
Wasserman, D. H., Wang, T. J. & Brown, N. J. The vasculature in prediabetes. Circ. Res. 122, 1135–1150 (2018).
Wildman, R. P., Mackey, R. H., Bostom, A., Thompson, T. & Sutton-Tyrrell, K. Measures of obesity are associated with vascular stiffness in young and older adults. Hypertension 42, 468–473 (2003).
Lavie, C. J., Arena, R., Alpert, M. A., Milani, R. V. & Ventura, H. O. Management of cardiovascular diseases in patients with obesity. Nat. Rev. Cardiol. 15, 45–56 (2018).
Tonneijck, L. et al. Glomerular hyperfiltration in diabetes: mechanisms, clinical significance, and treatment. J. Am. Soc. Nephrol. 28, 1023–1039 (2017).
Carlstrom, M., Wilcox, C. S. & Arendshorst, W. J. Renal autoregulation in health and disease. Physiol. Rev. 95, 405–511 (2015).
Woods, L. L., Mizelle, H. L. & Hall, J. E. Control of renal hemodynamics in hyperglycemia: possible role of tubuloglomerular feedback. Am. J. Physiol. 252, F65–F73 (1987).
Hall, J. E. et al. Obesity-associated hypertension and kidney disease. Curr. Opin. Nephrol. Hypertens. 12, 195–200 (2003).
Woods, L. L., Mizelle, H. L., Montani, J. P. & Hall, J. E. Mechanisms controlling renal hemodynamics and electrolyte excretion during amino acids. Am. J. Physiol. 251, F303–F312 (1986).
de Paula, R. B., da Silva, A. A. & Hall, J. E. Aldosterone antagonism attenuates obesity-induced hypertension and glomerular hyperfiltration. Hypertension 43, 41–47 (2004).
Hall, J. E., Granger, J. P., Smith, M. J. Jr & Premen, A. J. Role of renal hemodynamics and arterial pressure in aldosterone “escape”. Hypertension 6, I183–I192 (1984).
Fu, Y. et al. Aldosterone blunts tubuloglomerular feedback by activating macula densa mineralocorticoid receptors. Hypertension 59, 599–606 (2012).
Granger, J. P. et al. Role of nitric oxide in modulating renal function and arterial pressure during chronic aldosterone excess. Am. J. Physiol. 276, R197–R202 (1999).
Hall, J. E. Renal dysfunction, rather than nonrenal vascular dysfunction, mediates salt-induced hypertension. Circulation 133, 894–906 (2016).
Sugerman, H., Windsor, A., Bessos, M. & Wolfe, L. Intra-abdominal pressure, sagittal abdominal diameter and obesity comorbidity. J. Intern. Med. 241, 71–79 (1997).
Hall, J. E. Louis, K. Dahl Memorial Lecture. Renal and cardiovascular mechanisms of hypertension in obesity. Hypertension 23, 381–394 (1994).
Alonso-Galicia, M., Dwyer, T. M., Herrera, G. A. & Hall, J. E. Increased hyaluronic acid in the inner renal medulla of obese dogs. Hypertension 25, 888–892 (1995).
Dwyer, T. M. et al. Distribution of renal medullary hyaluronan in lean and obese rabbits. Kidney Int. 58, 721–729 (2000).
Hall, J. E., Brands, M. W. & Henegar, J. R. Mechanisms of hypertension and kidney disease in obesity. Ann. NY Acad. Sci. 892, 91–107 (1999).
Adeosun, S. O. et al. Loss of biliverdin reductase-A promotes lipid accumulation and lipotoxicity in mouse proximal tubule cells. Am. J. Physiol. Renal Physiol. 315, F323–F331 (2018).
Unger, R. H. & Scherer, P. E. Gluttony, sloth and the metabolic syndrome: a roadmap to lipotoxicity. Trends Endocrinol. Metab. 21, 345–352 (2010).
Engeli, S. & Sharma, A. M. The renin-angiotensin system and natriuretic peptides in obesity-associated hypertension. J. Mol. Med. 79, 21–29 (2001).
Cabandugama, P. K., Gardner, M. J. & Sowers, J. R. The renin angiotensin aldosterone system in obesity and hypertension: roles in the cardiorenal metabolic syndrome. Med. Clin. North Am. 101, 129–137 (2017).
Schutten, M. T., Houben, A. J., de Leeuw, P. W. & Stehouwer, C. D. The link between adipose tissue renin-angiotensin-aldosterone system signaling and obesity-associated hypertension. Physiology 32, 197–209 (2017).
Alonso-Galicia, M., Brands, M. W., Zappe, D. H. & Hall, J. E. Hypertension in obese Zucker rats. Role of angiotensin II and adrenergic activity. Hypertension 28, 1047–1054 (1996).
Hall, J. E. et al. Hypertension: physiology and pathophysiology. Compr. Physiol. 2, 2393–2442 (2012).
Dorresteijn, J. A. et al. Differential effects of renin-angiotensin-aldosterone system inhibition, sympathoinhibition and diuretic therapy on endothelial function and blood pressure in obesity-related hypertension: a double-blind, placebo-controlled cross-over trial. J. Hypertens. 31, 393–403 (2013).
Grassi, G. et al. Comparative effects of candesartan and hydrochlorothiazide on blood pressure, insulin sensitivity, and sympathetic drive in obese hypertensive individuals: results of the CROSS study. J. Hypertens. 21, 1761–1769 (2003).
Reisin, E. et al. Lisinopril versus hydrochlorothiazide in obese hypertensive patients: a multicenter placebo-controlled trial. Treatment in Obese Patients With Hypertension (TROPHY) study group. Hypertension 30, 140–145 (1997).
Brenner, B. M. et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N. Engl. J. Med. 345, 861–869 (2001).
Lewis, E. J. et al. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N. Engl. J. Med. 345, 851–860 (2001).
Dinh Cat, A. N., Friederich-Persson, M., White, A. & Touyz, R. M. Adipocytes, aldosterone and obesity-related hypertension. J. Mol. Endocrinol. 57, F7–F21 (2016).
Davel, A. P., Jaffe, I. Z., Tostes, R. C., Jaisser, F. & Belin de Chantemele, E. J. New roles of aldosterone and mineralocorticoid receptors in cardiovascular disease: translational and sex-specific effects. Am. J. Physiol. Heart Circ. Physiol. 315, H989–H999 (2018).
Faulkner, J. L., Bruder-Nascimento, T. & Belin de Chantemele, E. J. The regulation of aldosterone secretion by leptin: implications in obesity-related cardiovascular disease. Curr. Opin. Nephrol. Hypertens. 27, 63–69 (2018).
Bomback, A. S., Muskala, P., Bald, E., Chwatko, G. & Nowicki, M. Low-dose spironolactone, added to long-term ACE inhibitor therapy, reduces blood pressure and urinary albumin excretion in obese patients with hypertensive target organ damage. Clin. Nephrol. 72, 449–456 (2009).
Dudenbostel, T. & Calhoun, D. A. Use of aldosterone antagonists for treatment of uncontrolled resistant hypertension. Am. J. Hypertens. 30, 103–109 (2017).
de Souza, F., Muxfeldt, E., Fiszman, R. & Salles, G. Efficacy of spironolactone therapy in patients with true resistant hypertension. Hypertension 55, 147–152 (2010).
Hwang, M. H. et al. Mineralocorticoid receptors modulate vascular endothelial function in human obesity. Clin. Sci. 125, 513–520 (2013).
Kosmala, W., Przewlocka-Kosmala, M., Szczepanik-Osadnik, H., Mysiak, A. & Marwick, T. H. Fibrosis and cardiac function in obesity: a randomised controlled trial of aldosterone blockade. Heart 99, 320–326 (2013).
Andersen, K. Aldosterone synthase inhibition in hypertension. Curr. Hypertens. Rep. 15, 484–488 (2013).
Namsolleck, P. & Unger, T. Aldosterone synthase inhibitors in cardiovascular and renal diseases. Nephrol. Dial. Transplant. 29, i62–i68 (2014).
Calhoun, D. A. et al. Effects of a novel aldosterone synthase inhibitor for treatment of primary hypertension: results of a randomized, double-blind, placebo- and active-controlled phase 2 trial. Circulation 124, 1945–1955 (2011).
Karns, A. D., Bral, J. M., Hartman, D., Peppard, T. & Schumacher, C. Study of aldosterone synthase inhibition as an add-on therapy in resistant hypertension. J. Clin. Hypertens. 15, 186–192 (2013).
Liao, W. H. et al. Aldosterone deficiency in mice burdens respiration and accentuates diet-induced hyperinsulinemia and obesity. JCI Insight 3, 99015 (2018).
Fujita, T. Mechanism of salt-sensitive hypertension: focus on adrenal and sympathetic nervous systems. J. Am. Soc. Nephrol. 25, 1148–1155 (2014).
Chapman, K., Holmes, M. & Seckl, J. 11beta-hydroxysteroid dehydrogenases: intracellular gate-keepers of tissue glucocorticoid action. Physiol. Rev. 93, 1139–1206 (2013).
Funder, J. W. Apparent mineralocorticoid excess. J. Steroid Biochem. Mol. Biol. 165, 151–153 (2017).
Williams, B. et al. Endocrine and haemodynamic changes in resistant hypertension, and blood pressure responses to spironolactone or amiloride: the PATHWAY-2 mechanisms substudies. Lancet Diabetes Endocrinol. 6, 464–475 (2018).
Williams, B. et al. Spironolactone versus placebo, bisoprolol, and doxazosin to determine the optimal treatment for drug-resistant hypertension (PATHWAY-2): a randomised, double-blind, crossover trial. Lancet 386, 2059–2068 (2015).
Carey, R. M. et al. Resistant hypertension: detection, evaluation, and management: a scientific statement from the American Heart Association. Hypertension 72, e53–e90 (2018).
Calhoun, D. A. Fluid retention, aldosterone excess, and treatment of resistant hypertension. Lancet Diabetes Endocrinol. 6, 431–433 (2018).
Jordan, J., Birkenfeld, A. L., Melander, O. & Moro, C. Natriuretic peptides in cardiovascular and metabolic crosstalk: implications for hypertension management. Hypertension 72, 270–276 (2018).
Asferg, C. L. et al. Relative atrial natriuretic peptide deficiency and inadequate renin and angiotensin II suppression in obese hypertensive men. Hypertension 62, 147–153 (2013).
Asferg, C. L., Andersen, U. B., Linneberg, A., Goetze, J. P. & Jeppesen, J. L. Obese hypertensive men have lower circulating proatrial natriuretic peptide concentrations despite greater left atrial size. Am. J. Hypertens. 31, 645–650 (2018).
Savoia, C., Volpe, M., Alonzo, A., Rossi, C. & Rubattu, S. Natriuretic peptides and cardiovascular damage in the metabolic syndrome: molecular mechanisms and clinical implications. Clin. Sci. 118, 231–240 (2010).
Wang, T. J. et al. Impact of obesity on plasma natriuretic peptide levels. Circulation 109, 594–600 (2004).
Grandi, A. M. et al. Natriuretic peptides as markers of preclinical cardiac disease in obesity. Eur. J. Clin. Invest. 34, 342–348 (2004).
Abdulle, A. M. et al. Plasma N terminal pro-brain natriuretic peptide levels and its determinants in a multi-ethnic population. J. Hum. Hypertens. 21, 647–653 (2007).
Birkenfeld, A. L. et al. Lipid mobilization with physiological atrial natriuretic peptide concentrations in humans. J. Clin. Endocrinol. Metab. 90, 3622–3628 (2005).
Birkenfeld, A. L. et al. Atrial natriuretic peptide induces postprandial lipid oxidation in humans. Diabetes 57, 3199–3204 (2008).
Miyashita, K. et al. Natriuretic peptides/cGMP/cGMP-dependent protein kinase cascades promote muscle mitochondrial biogenesis and prevent obesity. Diabetes 58, 2880–2892 (2009).
Bordicchia, M. et al. Cardiac natriuretic peptides act via p38 MAPK to induce the brown fat thermogenic program in mouse and human adipocytes. J. Clin. Invest. 122, 1022–1036 (2012).
Canfora, E. E., Meex, R. C. R., Venema, K. & Blaak, E. E. Gut microbial metabolites in obesity, NAFLD and T2DM. Nat. Rev. Endocrinol. https://doi.org/10.1038/s41574-019-0156-z (2019).
Aydin, O., Nieuwdorp, M. & Gerdes, V. The gut microbiome as a target for the treatment of type 2 diabetes. Curr. Diab Rep. 18, 55 (2018).
Ridaura, V. K. et al. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science 341, 1241214 (2013).
Yang, T., Richards, E. M., Pepine, C. J. & Raizada, M. K. The gut microbiota and the brain-gut-kidney axis in hypertension and chronic kidney disease. Nat. Rev. Nephrol. 14, 442–456 (2018).
Wilck, N. et al. Salt-responsive gut commensal modulates TH17 axis and disease. Nature 551, 585–589 (2017).
Santisteban, M. M. et al. Hypertension-linked pathophysiological alterations in the gut. Circ. Res. 120, 312–323 (2017).
Riedl, R. A., Atkinson, S. N., Burnett, C. M. L., Grobe, J. L. & Kirby, J. R. The gut microbiome, energy homeostasis, and implications for hypertension. Curr. Hypertens. Rep. 19, 27 (2017).
Richards, E. M., Pepine, C. J., Raizada, M. K. & Kim, S. The gut, its microbiome, and hypertension. Curr. Hypertens. Rep. 19, 36 (2017).
Afsar, B., Vaziri, N. D., Aslan, G., Tarim, K. & Kanbay, M. Gut hormones and gut microbiota: implications for kidney function and hypertension. J. Am. Soc. Hypertens. 10, 954–961 (2016).
Muskiet, M. H. A. et al. GLP-1 and the kidney: from physiology to pharmacology and outcomes in diabetes. Nat. Rev. Nephrol. 13, 605–628 (2017).
Zhu, Q. & Scherer, P. E. Immunologic and endocrine functions of adipose tissue: implications for kidney disease. Nat. Rev. Nephrol. 14, 105–120 (2018).
Abraham, T. M., Pedley, A., Massaro, J. M., Hoffmann, U. & Fox, C. S. Association between visceral and subcutaneous adipose depots and incident cardiovascular disease risk factors. Circulation 132, 1639–1647 (2015).
D’Souza, K., Nzirorera, C. & Kienesberger, P. C. Lipid metabolism and signaling in cardiac lipotoxicity. Biochim. Biophys. Acta 1861, 1513–1524 (2016).
Yamauchi, T. et al. Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat. Med. 8, 1288–1295 (2002).
Ohashi, K., Ouchi, N. & Matsuzawa, Y. Adiponectin and hypertension. Am. J. Hypertens. 24, 263–269 (2011).
Wang, Z. V. & Scherer, P. E. Adiponectin, cardiovascular function, and hypertension. Hypertension 51, 8–14 (2008).
Sabbatini, A. R., Fontana, V., Laurent, S. & Moreno, H. An update on the role of adipokines in arterial stiffness and hypertension. J. Hypertens. 33, 435–444 (2015).
Ohashi, K. et al. Adiponectin replenishment ameliorates obesity-related hypertension. Hypertension 47, 1108–1116 (2006).
Kim, D. H., Kim, C., Ding, E. L., Townsend, M. K. & Lipsitz, L. A. Adiponectin levels and the risk of hypertension: a systematic review and meta-analysis. Hypertension 62, 27–32 (2013).
Pan, W. W. & Myers, M. G. Jr. Leptin and the maintenance of elevated body weight. Nat. Rev. Neurosci. 19, 95–105 (2018).
Schwartz, M. W. et al. Central nervous system control of food intake. Nature 404, 661–671 (2000).
Hall, J. E. et al. Obesity-induced hypertension: role of sympathetic nervous system, leptin, and melanocortins. J. Biol. Chem. 285, 17271–17276 (2010).
do Carmo, J. M. et al. Control of metabolic and cardiovascular function by the leptin-brain melanocortin pathway. IUBMB Life 65, 692–698 (2013).
Snitker, S., Pratley, R. E., Nicolson, M., Tataranni, P. A. & Ravussin, E. Relationship between muscle sympathetic nerve activity and plasma leptin concentration. Obes. Res. 5, 338–340 (1997).
Haynes, W. G., Morgan, D. A., Walsh, S. A., Mark, A. L. & Sivitz, W. I. Receptor-mediated regional sympathetic nerve activation by leptin. J. Clin. Invest. 100, 270–278 (1997).
Shek, E. W., Brands, M. W. & Hall, J. E. Chronic leptin infusion increases arterial pressure. Hypertension 31, 409–414 (1998).
Mark, A. L. Selective leptin resistance revisited. Am. J. Physiol. Regul. Integr. Comp. Physiol. 305, R566–R581 (2013).
Kuo, J. J., Jones, O. B. & Hall, J. E. Inhibition of NO synthesis enhances chronic cardiovascular and renal actions of leptin. Hypertension 37, 670–676 (2001).
Carlyle, M., Jones, O. B., Kuo, J. J. & Hall, J. E. Chronic cardiovascular and renal actions of leptin: role of adrenergic activity. Hypertension 39, 496–501 (2002).
Lim, K., Burke, S. L. & Head, G. A. Obesity-related hypertension and the role of insulin and leptin in high-fat-fed rabbits. Hypertension 61, 628–634 (2013).
Mark, A. L. et al. Contrasting blood pressure effects of obesity in leptin-deficient ob/ob mice and agouti yellow obese mice. J. Hypertens. 17, 1949–1953 (1999).
Aizawa-Abe, M. et al. Pathophysiological role of leptin in obesity-related hypertension. J. Clin. Invest. 105, 1243–1252 (2000).
Machleidt, F. et al. Experimental hyperleptinemia acutely increases vasoconstrictory sympathetic nerve activity in healthy humans. J. Clin. Endocrinol. Metab. 98, E491–E496 (2013).
Zelissen, P. M. et al. Effect of three treatment schedules of recombinant methionyl human leptin on body weight in obese adults: a randomized, placebo-controlled trial. Diabetes Obes. Metab. 7, 755–761 (2005).
Ozata, M., Ozdemir, I. C. & Licinio, J. Human leptin deficiency caused by a missense mutation: multiple endocrine defects, decreased sympathetic tone, and immune system dysfunction indicate new targets for leptin action, greater central than peripheral resistance to the effects of leptin, and spontaneous correction of leptin-mediated defects. J. Clin. Endocrinol. Metab. 84, 3686–3695 (1999).
Munzberg, H. & Myers, M. G. Jr. Molecular and anatomical determinants of central leptin resistance. Nat. Neurosci. 8, 566–570 (2005).
Rahmouni, K., Morgan, D. A., Morgan, G. M., Mark, A. L. & Haynes, W. G. Role of selective leptin resistance in diet-induced obesity hypertension. Diabetes 54, 2012–2018 (2005).
Dubinion, J. H., da Silva, A. A. & Hall, J. E. Chronic blood pressure and appetite responses to central leptin infusion in rats fed a high fat diet. J. Hypertens. 29, 758–762 (2011).
Mark, A. L. et al. Leptin signaling in the nucleus tractus solitarii increases sympathetic nerve activity to the kidney. Hypertension 53, 375–380 (2009).
Harlan, S. M. et al. Ablation of the leptin receptor in the hypothalamic arcuate nucleus abrogates leptin-induced sympathetic activation. Circ. Res. 108, 808–812 (2011).
do Carmo, J. M. et al. Control of blood pressure, appetite, and glucose by leptin in mice lacking leptin receptors in proopiomelanocortin neurons. Hypertension 57, 918–926 (2011).
Gao, Q. et al. Disruption of neural signal transducer and activator of transcription 3 causes obesity, diabetes, infertility, and thermal dysregulation. Proc. Natl Acad. Sci. USA 101, 4661–4666 (2004).
Dubinion, J. H. et al. Role of proopiomelanocortin neuron Stat3 in regulating arterial pressure and mediating the chronic effects of leptin. Hypertension 61, 1066–1074 (2013).
Rahmouni, K., Haynes, W. G., Morgan, D. A. & Mark, A. L. Intracellular mechanisms involved in leptin regulation of sympathetic outflow. Hypertension 41, 763–767 (2003).
do Carmo, J. M., da Silva, A. A. & Hall, J. E. Leptin reduces food intake but fails to raise blood pressure in mice with deficiency of insulin receptor substrate 2 (IRS2) in the entire brain or specifically in POMC neurons. Hypertension 60, A27 (2012).
Krajewska, M. et al. Development of diabesity in mice with neuronal deletion of Shp2 tyrosine phosphatase. Am. J. Pathol. 172, 1312–1324 (2008).
do Carmo, J. M. et al. Shp2 signaling in POMC neurons is important for leptin’s actions on blood pressure, energy balance, and glucose regulation. Am. J. Physiol. Regul. Integr. Comp. Physiol. 307, R1438–R1447 (2014).
do Carmo, J. M. et al. Role of Shp2 in forebrain neurons in regulating metabolic and cardiovascular functions and responses to leptin. Int. J. Obes. 38, 775–783 (2014).
Belin de Chantemele, E. J. et al. Protein tyrosine phosphatase 1B, a major regulator of leptin-mediated control of cardiovascular function. Circulation 120, 753–763 (2009).
Aberdein, N. et al. Role of PTP1B in POMC neurons during chronic high-fat diet: sex differences in regulation of liver lipids and glucose tolerance. Am. J. Physiol. Regul. Integr. Comp. Physiol. 314, R478–R488 (2018).
Mori, H. et al. Socs3 deficiency in the brain elevates leptin sensitivity and confers resistance to diet-induced obesity. Nat. Med. 10, 739–743 (2004).
do Carmo, J. M. et al. Neuronal suppressor of cytokine signaling 3: role in modulating chronic metabolic and cardiovascular effects of leptin. Hypertension 71, 1248–1257 (2018).
Grassi, G., Mark, A. & Esler, M. The sympathetic nervous system alterations in human hypertension. Circ. Res. 116, 976–990 (2015).
Gentile, C. L., Orr, J. S., Davy, B. M. & Davy, K. P. Modest weight gain is associated with sympathetic neural activation in nonobese humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 292, R1834–R1838 (2007).
Esler, M. et al. Obesity paradox in hypertension: is this because sympathetic activation in obesity-hypertension takes a benign form? Hypertension 71, 22–33 (2018).
Armitage, J. A. et al. Rapid onset of renal sympathetic nerve activation in rabbits fed a high-fat diet. Hypertension 60, 163–171 (2012).
Davy, K. P. & Hall, J. E. Obesity and hypertension: two epidemics or one? Am. J. Physiol. Regul. Integr. Comp. Physiol. 286, R803–R813 (2004).
Davy, K. P. & Orr, J. S. Sympathetic nervous system behavior in human obesity. Neurosci. Biobehav. Rev. 33, 116–124 (2009).
Wofford, M. R. et al. Antihypertensive effect of alpha- and beta-adrenergic blockade in obese and lean hypertensive subjects. Am. J. Hypertens. 14, 694–698 (2001).
Kassab, S. et al. Endothelin-A receptor antagonism attenuates the hypertension and renal injury in Dahl salt-sensitive rats. Hypertension 31, 397–402 (1998).
Lohmeier, T. E. et al. Systemic and renal-specific sympathoinhibition in obesity hypertension. Hypertension 59, 331–338 (2012).
Henegar, J. R. et al. Catheter-based radiorefrequency renal denervation lowers blood pressure in obese hypertensive dogs. Am. J. Hypertens. 27, 1285–1292 (2014).
Iliescu, R., Lohmeier, T. E., Tudorancea, I., Laffin, L. & Bakris, G. L. Renal denervation for the treatment of resistant hypertension: review and clinical perspective. Am. J. Physiol. Renal Physiol. 309, F583–F594 (2015).
Asirvatham-Jeyaraj, N. et al. Renal denervation normalizes arterial pressure with no effect on glucose metabolism or renal inflammation in obese hypertensive mice. Hypertension 68, 929–936 (2016).
Hering, D. et al. Sustained sympathetic and blood pressure reduction 1 year after renal denervation in patients with resistant hypertension. Hypertension 64, 118–124 (2014).
Bhatt, D. L. et al. A controlled trial of renal denervation for resistant hypertension. N. Engl. J. Med. 370, 1393–1401 (2014).
Azizi, M. et al. Endovascular ultrasound renal denervation to treat hypertension (RADIANCE-HTN SOLO): a multicentre, international, single-blind, randomised, sham-controlled trial. Lancet 391, 2335–2345 (2018).
Henegar, J. R. et al. Catheter-based radiofrequency renal denervation: location effects on renal norepinephrine. Am. J. Hypertens. 28, 909–914 (2015).
Kandzari, D. E. et al. Effect of renal denervation on blood pressure in the presence of antihypertensive drugs: 6-month efficacy and safety results from the SPYRAL HTN-ON MED proof-of-concept randomised trial. Lancet 391, 2346–2355 (2018).
DiBona, G. F. Sympathetic nervous system and hypertension. Hypertension 61, 556–560 (2013).
Schlaich, M. P., Sobotka, P. A., Krum, H., Lambert, E. & Esler, M. D. Renal sympathetic-nerve ablation for uncontrolled hypertension. N. Engl. J. Med. 361, 932–934 (2009).
Hering, D. et al. Substantial reduction in single sympathetic nerve firing after renal denervation in patients with resistant hypertension. Hypertension 61, 457–464 (2013).
Brinkmann, J. et al. Catheter-based renal nerve ablation and centrally generated sympathetic activity in difficult-to-control hypertensive patients: prospective case series. Hypertension 60, 1485–1490 (2012).
Zappe, D. H. et al. Role of renal afferent nerves in obesity-induced hypertension. Am. J. Hypertens. 9, 20A (1996).
Dewan, N. A., Nieto, F. J. & Somers, V. K. Intermittent hypoxemia and OSA: implications for comorbidities. Chest 147, 266–274 (2015).
Lim, K., Jackson, K. L., Sata, Y. & Head, G. A. Factors responsible for obesity-related hypertension. Curr. Hypertens. Rep. 19, 53 (2017).
Hall, J. E., Hildebrandt, D. A. & Kuo, J. Obesity hypertension: role of leptin and sympathetic nervous system. Am. J. Hypertens. 14, 103S–115S (2001).
Hall, J. E. Hyperinsulinemia: a link between obesity and hypertension? Kidney Int. 43, 1402–1417 (1993).
Hall, J. E., Brands, M. W., Hildebrandt, D. A. & Mizelle, H. L. Obesity-associated hypertension. Hyperinsulinemia and renal mechanisms. Hypertension 19, I45–I55 (1992).
Iliescu, R., Tudorancea, I., Irwin, E. D. & Lohmeier, T. E. Chronic baroreflex activation restores spontaneous baroreflex control and variability of heart rate in obesity-induced hypertension. Am. J. Physiol. Heart Circ. Physiol. 305, H1080–H1088 (2013).
Seravalle, G. et al. Long-term sympathoinhibitory effects of surgically induced weight loss in severe obese patients. Hypertension 64, 431–437 (2014).
Grassi, G. et al. Body weight reduction, sympathetic nerve traffic, and arterial baroreflex in obese normotensive humans. Circulation 97, 2037–2042 (1998).
Mansukhani, M. P., Kara, T., Caples, S. M. & Somers, V. K. Chemoreflexes, sleep apnea, and sympathetic dysregulation. Curr. Hypertens. Rep. 16, 476 (2014).
Mansukhani, M. P., Wang, S. & Somers, V. K. Chemoreflex physiology and implications for sleep apnoea: insights from studies in humans. Exp. Physiol. 100, 130–135 (2015).
Floras, J. S. Sleep apnea and cardiovascular disease: an enigmatic risk factor. Circ. Res. 122, 1741–1764 (2018).
Javaheri, S. et al. Sleep apnea: types, mechanisms, and clinical cardiovascular consequences. J. Am. Coll. Cardiol. 69, 841–858 (2017).
Iturriaga, R., Oyarce, M. P. & Dias, A. C. R. Role of carotid body in intermittent hypoxia-related hypertension. Curr. Hypertens. Rep. 19, 38 (2017).
Ciriello, J. & Moreau, J. M. Leptin signaling in the nucleus of the solitary tract alters the cardiovascular responses to activation of the chemoreceptor reflex. Am. J. Physiol. Regul. Integr. Comp. Physiol. 303, R727–R736 (2012).
Lohmeier, T. E. et al. Chronic interactions between carotid baroreceptors and chemoreceptors in obesity hypertension. Hypertension 68, 227–235 (2016).
do Carmo, J. M. et al. Role of the brain melanocortins in blood pressure regulation. Biochim. Biophys. Acta 1863, 2508–2514 (2017).
da Silva, A. A., do Carmo, J. M., Wang, Z. & Hall, J. E. The brain melanocortin system, sympathetic control, and obesity hypertension. Physiology 29, 196–202 (2014).
Tao, Y. X. The melanocortin-4 receptor: physiology, pharmacology, and pathophysiology. Endocr. Rev. 31, 506–543 (2010).
Cone, R. D. Studies on the physiological functions of the melanocortin system. Endocr. Rev. 27, 736–749 (2006).
Girardet, C. & Butler, A. A. Neural melanocortin receptors in obesity and related metabolic disorders. Biochim. Biophys. Acta 1842, 482–494 (2014).
Vaisse, C. et al. Melanocortin-4 receptor mutations are a frequent and heterogeneous cause of morbid obesity. J. Clin. Invest. 106, 253–262 (2000).
Kuo, J. J., Silva, A. A. & Hall, J. E. Hypothalamic melanocortin receptors and chronic regulation of arterial pressure and renal function. Hypertension 41, 768–774 (2003).
Kuo, J. J., da Silva, A. A., Tallam, L. S. & Hall, J. E. Role of adrenergic activity in pressor responses to chronic melanocortin receptor activation. Hypertension 43, 370–375 (2004).
da Silva, A. A. et al. Endogenous melanocortin system activity contributes to the elevated arterial pressure in spontaneously hypertensive rats. Hypertension 51, 884–890 (2008).
Tallam, L. S., da Silva, A. A. & Hall, J. E. Melanocortin-4 receptor mediates chronic cardiovascular and metabolic actions of leptin. Hypertension 48, 58–64 (2006).
Greenfield, J. R. et al. Modulation of blood pressure by central melanocortinergic pathways. N. Engl. J. Med. 360, 44–52 (2009).
Greenfield, J. R. Melanocortin signalling and the regulation of blood pressure in human obesity. J. Neuroendocrinol. 23, 186–193 (2011).
do Carmo, J. M., da Silva, A. A., Rushing, J. S. & Hall, J. E. Activation of the central melanocortin system contributes to the increased arterial pressure in obese Zucker rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 302, R561–R567 (2012).
Ward, K. R., Bardgett, J. F., Wolfgang, L. & Stocker, S. D. Sympathetic response to insulin is mediated by melanocortin 3/4 receptors in the hypothalamic paraventricular nucleus. Hypertension 57, 435–441 (2011).
Yosten, G. L., Pate, A. T. & Samson, W. K. Neuronostatin acts in brain to biphasically increase mean arterial pressure through sympatho-activation followed by vasopressin secretion: the role of melanocortin receptors. Am. J. Physiol. Regul. Integr. Comp. Physiol. 300, R1194–R1199 (2011).
Yosten, G. L. & Samson, W. K. Neural circuitry underlying the central hypertensive action of nesfatin-1: melanocortins, corticotropin-releasing hormone, and oxytocin. Am. J. Physiol. Regul. Integr. Comp. Physiol. 306, R722–R727 (2014).
da Silva, A. A. et al. Chronic central nervous system MC3/4R blockade attenuates hypertension induced by nitric oxide synthase inhibition but not by angiotensin II infusion. Hypertension 65, 171–177 (2014).
Sayk, F. et al. Sympathetic function in human carriers of melanocortin-4 receptor gene mutations. J. Clin. Endocrinol. Metab. 95, 1998–2002 (2010).
Rossi, J. et al. Melanocortin-4 receptors expressed by cholinergic neurons regulate energy balance and glucose homeostasis. Cell Metab. 13, 195–204 (2011).
Sohn, J. W. et al. Melanocortin 4 receptors reciprocally regulate sympathetic and parasympathetic preganglionic neurons. Cell 152, 612–619 (2013).
do Carmo, J. M., da Silva, A. A., Rushing, J. S., Pace, B. & Hall, J. E. Differential control of metabolic and cardiovascular functions by melanocortin-4 receptors in proopiomelanocortin neurons. Am. J. Physiol. Regul. Integr. Comp. Physiol. 305, R359–R368 (2013).
Nicholson, J. R., Peter, J. C., Lecourt, A. C., Barde, Y. A. & Hofbauer, K. G. Melanocortin-4 receptor activation stimulates hypothalamic brain-derived neurotrophic factor release to regulate food intake, body temperature and cardiovascular function. J. Neuroendocrinol. 19, 974–982 (2007).
Bariohay, B. et al. Brain-derived neurotrophic factor/tropomyosin-related kinase receptor type B signaling is a downstream effector of the brainstem melanocortin system in food intake control. Endocrinology 150, 2646–2653 (2009).
Hall, J. E., Brands, M. W., Dixon, W. N. & Smith, M. J. Jr. Obesity-induced hypertension. Renal function and systemic hemodynamics. Hypertension 22, 292–299 (1993).
Carroll, J. F., Huang, M., Hester, R. L., Cockrell, K. & Mizelle, H. L. Hemodynamic alterations in hypertensive obese rabbits. Hypertension 26, 465–470 (1995).
da Silva, A. A., Kuo, J. J., Tallam, L. S. & Hall, J. E. Role of endothelin-1 in blood pressure regulation in a rat model of visceral obesity and hypertension. Hypertension 43, 383–387 (2004).
Mills, E., Kuhn, C. M., Feinglos, M. N. & Surwit, R. Hypertension in CB57BL/6J mouse model of non-insulin-dependent diabetes mellitus. Am. J. Physiol. 264, R73–R78 (1993).
Aronne, L. J., Mackintosh, R., Rosenbaum, M., Leibel, R. L. & Hirsch, J. Autonomic nervous system activity in weight gain and weight loss. Am. J. Physiol. 269, R222–R225 (1995).
Contreras, R. J. & Williams, V. L. Dietary obesity and weight cycling: effects on blood pressure and heart rate in rats. Am. J. Physiol. 256, R1209–R1219 (1989).
Wang, Z. et al. Role of Socs3 in Pomc neurons in metabolic and cardiovascular regulation. Am. J. Physiol. Regul. Integr. Comp. Physiol. https://doi.org/10.1152/ajpregu.00163.2018 (2019).
Messerli, F. H. et al. Obesity and essential hypertension. Hemodynamics, intravascular volume, sodium excretion, and plasma renin activity. Arch. Intern. Med. 141, 81–85 (1981).
Crandall, D. L., Goldstein, B. M., Lizzo, F. H., Gabel, R. A. & Cervoni, P. Hemodynamics of obesity: influence of pattern of adipose tissue cellularity. Am. J. Physiol. 251, R314–R319 (1986).
Adolphe, J. L., Silver, T. I., Childs, H., Drew, M. D. & Weber, L. P. Short-term obesity results in detrimental metabolic and cardiovascular changes that may not be reversed with weight loss in an obese dog model. Br. J. Nutr. 112, 647–656 (2014).
Philip-Couderc, P. et al. Cardiac transcriptome analysis in obesity-related hypertension. Hypertension 41, 414–421 (2003).
Carroll, J. F., Braden, D. S., Cockrell, K. & Mizelle, H. L. Obese hypertensive rabbits develop concentric and eccentric hypertrophy and diastolic filling abnormalities. Am. J. Hypertens. 10, 230–233 (1997).
Nagarajan, V. et al. Cardiac function and lipid distribution in rats fed a high-fat diet: in vivo magnetic resonance imaging and spectroscopy. Am. J. Physiol. Heart Circ. Physiol. 304, H1495–H1504 (2013).
Mendes-Junior, L. G. et al. The usefulness of short-term high-fat/high salt diet as a model of metabolic syndrome in mice. Life Sci. 209, 341–348 (2018).
Carroll, J. F. et al. Hypertension, cardiac hypertrophy, and neurohumoral activity in a new animal model of obesity. Am. J. Physiol. 271, H373–H378 (1996).
Koncsos, G. et al. Diastolic dysfunction in prediabetic male rats: role of mitochondrial oxidative stress. Am. J. Physiol. Heart Circ. Physiol. 311, H927–H943 (2016).
Rocchini, A. P., Moorehead, C., Katch, V., Key, J. & Finta, K. M. Forearm resistance vessel abnormalities and insulin resistance in obese adolescents. Hypertension 19, 615–620 (1992).
Franco, R. L. et al. Forearm blood flow response to acute exercise in obese and non-obese males. Eur. J. Appl. Physiol. 113, 2015–2023 (2013).
Chagnac, A. et al. Obesity-induced glomerular hyperfiltration: its involvement in the pathogenesis of tubular sodium reabsorption. Nephrol. Dial. Transplant. 23, 3946–3952 (2008).
D’Agati, V. D. et al. Obesity-related glomerulopathy: clinical and pathologic characteristics and pathogenesis. Nat. Rev. Nephrol. 12, 453–471 (2016).
Antic, V., Tempini, A. & Montani, J. P. Serial changes in cardiovascular and renal function of rabbits ingesting a high-fat, high-calorie diet. Am. J. Hypertens. 12, 826–829 (1999).
Polichnowski, A. J. et al. Glomerulosclerosis in the diet-induced obesity model correlates with sensitivity to nitric oxide inhibition but not glomerular hyperfiltration or hypertrophy. Am. J. Physiol. Renal Physiol. 309, F791–F799 (2015).
Riazi, S., Tiwari, S., Sharma, N., Rash, A. & Ecelbarger, C. M. Abundance of the Na-K-2Cl cotransporter NKCC2 is increased by high-fat feeding in Fischer 344 X Brown Norway (F1) rats. Am. J. Physiol. Renal Physiol. 296, F762–F770 (2009).
Nizar, J. M. et al. Na+-sensitive elevation in blood pressure is ENaC independent in diet-induced obesity and insulin resistance. Am. J. Physiol. Renal Physiol. 310, F812–F820 (2016).
Dwyer, T. M., Bigler, S. A., Moore, N. A., Carroll, J. F. & Hall, J. E. The altered structure of renal papillary outflow tracts in obesity. Ultrastruct. Pathol. 24, 251–257 (2000).
Hall, J. E. et al. Hemodynamic and renal responses to chronic hyperinsulinemia in obese, insulin-resistant dogs. Hypertension 25, 994–1002 (1995).
Eppel, G. A., Armitage, J. A., Eikelis, N., Head, G. A. & Evans, R. G. Progression of cardiovascular and endocrine dysfunction in a rabbit model of obesity. Hypertens. Res. 36, 588–595 (2013).
do Carmo, J. M. et al. Inhibition of soluble epoxide hydrolase reduces food intake and increases metabolic rate in obese mice. Nutr. Metab. Cardiovasc. Dis. 22, 598–604 (2012).
Carroll, J. F. & Kyser, C. K. Exercise training in obesity lowers blood pressure independent of weight change. Med. Sci. Sports Exerc. 34, 596–601 (2002).
Northcott, C. A. et al. The development of hypertension and hyperaldosteronism in a rodent model of life-long obesity. Endocrinology 153, 1764–1773 (2012).
Li, C. et al. High-fat diet amplifies renal renin angiotensin system expression, blood pressure elevation, and renal dysfunction caused by Ceacam1 null deletion. Am. J. Physiol. Endocrinol. Metab. 309, E802–E810 (2015).
Schafer, N. et al. Endothelial mineralocorticoid receptor activation mediates endothelial dysfunction in diet-induced obesity. Eur. Heart J. 34, 3515–3524 (2013).
Khan, S. A. et al. Obesity depresses baroreflex control of renal sympathetic nerve activity and heart rate in Sprague Dawley rats: role of the renal innervation. Acta Physiol. 214, 390–401 (2015).
Fardin, N. M., Oyama, L. M. & Campos, R. R. Changes in baroreflex control of renal sympathetic nerve activity in high-fat-fed rats as a predictor of hypertension. Obesity 20, 1591–1597 (2012).
Acknowledgements
The authors’ research was supported by grants from the US National Heart, Lung, and Blood Institute (P01 HL51971), the US National Institute of General Medical Sciences (P20 GM104357 and U54 GM115428) and the US National Institute of Diabetes and Digestive and Kidney Diseases (1K08DK099415-01A1) of the US National Institutes of Health.
Reviewer information
Nature Reviews Nephrology thanks the anonymous reviewers for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
J.E.H. wrote the manuscript. All authors researched the data for the article, contributed to discussions of the content and reviewed or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Lipotoxicity
-
The toxic effect of lipids that accumulate in non-adipose tissue and cause cellular dysfunction.
- Renal sinus fat
-
(RSF). The adipose tissue that accumulates in the renal sinuses, which are cavities within the kidneys that are occupied by the renal pelvis, renal calyces, blood vessels and nerves.
- Perirenal fat
-
(PRF). Also called the adipose capsule of the kidney. The perirenal fat is a structure located between the renal fascia and renal capsule.
- Blood flow reserve
-
The maximum increase in blood flow above the resting level of blood flow.
- Hypoxaemia
-
The condition of abnormally low oxygen concentration in the blood.
- Baroreflex
-
Often called the baroreceptor reflex. The reflex mechanism by which stretch receptors (baroreceptors), located especially in the carotid sinuses and aortic arch, regulate blood pressure.
- Hypercapnia
-
The condition of excessive carbon dioxide concentration in the blood.
- Eucapnic
-
The condition of having normal carbon dioxide concentration in the blood.
Rights and permissions
About this article
Cite this article
Hall, J.E., do Carmo, J.M., da Silva, A.A. et al. Obesity, kidney dysfunction and hypertension: mechanistic links. Nat Rev Nephrol 15, 367–385 (2019). https://doi.org/10.1038/s41581-019-0145-4
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41581-019-0145-4
This article is cited by
-
Obesity-and lipid-related indices as a risk factor of hypertension in mid-aged and elderly Chinese: a cross-sectional study
BMC Geriatrics (2024)
-
Obesity and the kidney: mechanistic links and therapeutic advances
Nature Reviews Endocrinology (2024)
-
Body Fat Depletion: the Yin Paradigm for Treating Type 2 Diabetes
Current Atherosclerosis Reports (2024)
-
Pretreatment body mass index affects achievement of target blood pressure with sodium-glucose cotransporter 2 inhibitors in patients with type 2 diabetes mellitus and chronic kidney disease
Hypertension Research (2024)
-
Identifying a target group for selenium supplementation in high-risk cardiac surgery: a secondary analysis of the SUSTAIN CSX trial
Intensive Care Medicine Experimental (2023)