Abstract
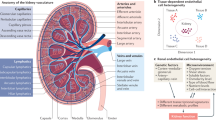
In the dense circulatory system of the kidney, as in all vascularized tissues, pericytes enwrap capillaries and microvessels to regulate angiogenesis, stabilize microvascular networks and control blood flow by vasoconstriction. Specialized renal pericytes known as mesangial cells provide physical support to glomerular capillaries, whereas a subset of juxtaglomerular arteriolar pericytes control the local blood pressure in the glomerulus via contraction and influence systemic blood pressure by secreting renin. Similar to pericytes from many other organs, cultured human renal pericytes give rise to mesenchymal stem/stromal cells, suggesting a role of perivascular cells in renal homeostasis and regeneration. On the other hand, pericytes directly contribute to renal fibrosis, and mesangial cells may have an essential role in the development of glomerulosclerosis and other nephropathies. From their early emergence in the renal embryonic rudiment to their distribution in diverse perivascular niches in the adult organ, we review the anatomy and function of pericytes in the healthy and diseased kidney. Many aspects of the ontogeny, specification and functional specialization of renal pericytes remain elusive. The development of powerful models in the easily accessible and genetically tractable zebrafish will help to uncover the multiple facets of these cells.
Key points
-
Perivascular niches in the kidney are highly heterogeneous, leading to multiple subtypes of perivascular cells with distinct functional roles.
-
Although shown to arise from a forkhead box protein D1 (FOXD1)+ progenitor pool, the precise embryonic origins and developmental signals that determine renal perivascular subpopulations remain undefined or ambiguous.
-
Renal pericytes have key roles in homeostasis; for example, they can regulate blood pressure either directly by their contraction or indirectly by the secretion of renin by certain subpopulations.
-
Renal pericytes can give rise to mesenchymal stem and/or stromal cells, which have shown promise for the treatment of acute kidney injury and chronic kidney disease.
-
Renal pericytes are important in the pathogenesis of kidney disease; they are key to vascular survival, can interact with the immune system and can contribute to glomerular and interstitial fibrosis.
-
Powerful molecular tools for the study of renal pericytes are beginning to emerge in the zebrafish model organism.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kennedy-Lydon, T. M., Crawford, C., Wildman, S. S. P. & Peppiatt-Wildman, C. M. Renal pericytes: regulators of medullary blood flow. Acta Physiol. 207, 212–225 (2013).
Lindblom, P. et al. Endothelial PDGF-B retention is required for proper investment of pericytes in the microvessel wall. Genes Dev. 17, 1835–1840 (2003).
Kramann, R. et al. Perivascular Gli1+ progenitors are key contributors to injury-induced organ fibrosis. Cell Stem Cell 16, 51–66 (2015).
Murray, I. R. et al. αv integrins on mesenchymal cells regulate skeletal and cardiac muscle fibrosis. Nat. Commun. 8, 1118 (2017).
Corselli, M. et al. Perivascular support of human hematopoietic stem/progenitor cells. Blood 121, 2891–2901 (2013).
Volz, K. S. et al. Pericytes are progenitors for coronary artery smooth muscle. eLife 4, e10036 (2015).
Crisan, M. et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 3, 301–313 (2008).
Chen, C.-W. et al. Human pericytes for ischemic heart repair. Stem Cells 31, 305–316 (2013).
Meyers, C. A. et al. Early immunomodulatory effects of implanted human perivascular stromal cells during bone formation. Tissue Eng. Part A 24, 448–457 (2017).
Tawonsawatruk, T. et al. Adipose derived pericytes rescue fractures from a failure of healing-non-union. Sci. Rep. 6, 22779 (2016).
Stefanska, A. et al. Human kidney pericytes produce renin. Kidney Int. 90, 1251–1261 (2016).
Johnson, R. J. et al. Expression of smooth muscle cell phenotype by rat mesangial cells in immune complex nephritis. Alpha-smooth muscle actin is a marker of mesangial cell proliferation. J. Clin. Invest. 87, 847–858 (1991).
Armulik, A., Genové, G. & Betsholtz, C. Pericytes: developmental, physiological, and pathological perspectives, problems, and promises. Dev. Cell 21, 193–215 (2011).
Sims, D. E. The pericyte-a review. Tissue Cell 18, 153–174 (1986).
Lin, S.-L., Kisseleva, T., Brenner, D. A. & Duffield, J. S. Pericytes and perivascular fibroblasts are the primary source of collagen-producing cells in obstructive fibrosis of the kidney. Am. J. Pathol. 173, 1617–1627 (2008).
Turley, S. J., Cremasco, V. & Astarita, J. L. Immunological hallmarks of stromal cells in the tumour microenvironment. Nat. Rev. Immunol. 15, 669–682 (2015).
Ribatti, D., Nico, B. & Crivellato, E. The role of pericytes in angiogenesis. Int. J. Dev. Biol. 55, 261–268 (2011).
Navar, L. G. et al. The renal microcirculation. Microcirculation https://doi.org/10.1016/B978-0-12-374530-9.00015-2 (2008).
Tonelli, M. et al. Chronic kidney disease and mortality risk: a systematic review. J. Am. Soc. Nephrol. 17, 2034–2047 (2006).
Foley, R. N. & Ibrahim, H. N. Long-term outcomes of kidney donors. Curr. Opin. Nephrol. Hypertens. 19, 129–133 (2010).
Sakai, T., Billo, R. & Kriz, W. The structural organization of the kidney of typhlonectes compressicaudus (Amphibia. Gymnophiona). Anat. Embryol. 174, 243–252 (1986).
Pallone, T. L., Turner, M. R., Edwards, A. & Jamison, R. L. Countercurrent exchange in the renal medulla. Am. J. Physiol. Regul. Integr. Comp. Physiol. 284, R1153–R1175 (2003).
Dinour, D. & Brezis, M. Effects of adenosine on intrarenal oxygenation. Am. J. Physiol. 261, F787–F791 (1991).
Beeuwkes, R. & Bonventre, J. V. Tubular organization and vascular-tubular relations in the dog kidney. Am. J. Physiol. 229, 695–713 (1975).
Short, K. M. & Smyth, I. M. The contribution of branching morphogenesis to kidney development and disease. Nat. Rev. Nephrol. 12, 754–767 (2016).
Osathanondh, V. & Potter, E. L. Development of human kidney as shown by microdissection. I. Preparation of tissue with reasons for possible misinterpretation of observations. Arch. Pathol. 76, 271–276 (1963).
Short, K. M. et al. Global quantification of tissue dynamics in the developing mouse kidney. Dev. Cell 29, 188–202 (2014).
Oliver, J. Nephrons and Kidneys: A Quantitative Study of Development and Evolutionary Mammalian Renal Architectonics (Harper & Row, 1968).
Kobayashi, A. et al. Six2 defines and regulates a multipotent self-renewing nephron progenitor population throughout mammalian kidney development. Cell Stem Cell 3, 169–181 (2008).
Kobayashi, A. et al. Identification of a multipotent self-renewing stromal progenitor population during mammalian kidney organogenesis. Stem Cell Rep. 3, 650–662 (2014).
Humphreys, B. D. et al. Fate tracing reveals the pericyte and not epithelial origin of myofibroblasts in kidney fibrosis. Am. J. Pathol. 176, 85–97 (2010).
Bohnenpoll, T. et al. Tbx18 expression demarcates multipotent precursor populations in the developing urogenital system but is exclusively required within the ureteric mesenchymal lineage to suppress a renal stromal fate. Dev. Biol. 380, 25–36 (2013).
Asada, N. et al. Dysfunction of fibroblasts of extrarenal origin underlies renal fibrosis and renal anemia in mice. J. Clin. Invest. 121, 3981–3990 (2011).
Guillaume, R., Bressan, M. & Herzlinger, D. Paraxial mesoderm contributes stromal cells to the developing kidney. Dev. Biol. 329, 169–175 (2009).
Lemos, D. R. et al. Maintenance of vascular integrity by pericytes is essential for normal kidney function. Am. J. Physiol. Renal Physiol. 311, F1230–F1242 (2016).
Das, A. et al. Stromal–epithelial crosstalk regulates kidney progenitor cell differentiation. Nat. Cell Biol. 15, 1035–1044 (2013).
Hum, S., Rymer, C., Schaefer, C., Bushnell, D. & Sims-Lucas, S. Ablation of the renal stroma defines its critical role in nephron progenitor and vasculature patterning. PLoS ONE 9, e88400 (2014).
Raj, B. et al. Simultaneous single-cell profiling of lineages and cell types in the vertebrate brain by scGESTALT. Nat. Biotechnol. 36, 442–450 (2017).
Fetting, J. L. et al. FOXD1 promotes nephron progenitor differentiation by repressing decorin in the embryonic kidney. Development 141, 17–27 (2014).
Hatini, V., Huh, S. O., Herzlinger, D., Soares, V. C. & Lai, E. Essential role of stromal mesenchyme in kidney morphogenesis revealed by targeted disruption of Winged Helix transcription factor BF-2. Genes Dev. 10, 1467–1478 (1996).
Karner, C. M. et al. Canonical Wnt9b signaling balances progenitor cell expansion and differentiation during kidney development. Development 138, 1247–1257 (2011).
Ohmori, T., Tanigawa, S., Kaku, Y., Fujimura, S. & Nishinakamura, R. Sall1 in renal stromal progenitors non-cell autonomously restricts the excessive expansion of nephron progenitors. Sci. Rep. 5, 15676 (2015).
Bagherie-Lachidan, M. et al. Stromal Fat4 acts non-autonomously with Dchs1/2 to restrict the nephron progenitor pool. Development 142, 2564–2573 (2015).
Mao, Y., Francis-West, P. & Irvine, K. D. Fat4/Dchs1 signaling between stromal and cap mesenchyme cells influences nephrogenesis and ureteric bud branching. Development 142, 2574–2585 (2015).
Boivin, F. J. et al. Stromally expressed β-catenin modulates Wnt9b signaling in the ureteric epithelium. PLoS ONE 10, e0120347 (2015).
Li, W., Hartwig, S. & Rosenblum, N. D. Developmental origins and functions of stromal cells in the normal and diseased mammalian kidney. Dev. Dyn. 243, 853–863 (2014).
Lindahl, P. et al. Paracrine PDGF-B/PDGF-Rβ signaling controls mesangial cell development in kidney glomeruli. Development 125, 3313–3322 (1998).
Lin, E. E., Sequeira-Lopez, M. L. S. & Gomez, R. A. RBP-J in FOXD1+ renal stromal progenitors is crucial for the proper development and assembly of the kidney vasculature and glomerular mesangial cells. Am. J. Physiol. Renal Physiol. 306, F249–F258 (2014).
McCright, B. et al. Defects in development of the kidney, heart and eye vasculature in mice homozygous for a hypomorphic Notch2 mutation. Development 128, 491–502 (2001).
Hickmann, L. et al. Persistent and inducible neogenesis repopulates progenitor renin lineage cells in the kidney. Kidney Int. 92, 1419–1432 (2017).
Sequeira Lopez, M. L., Pentz, E. S., Robert, B., Abrahamson, D. R. & Gomez, R. A. Embryonic origin and lineage of juxtaglomerular cells. Am. J. Physiol. Renal Physiol. 281, F345–F356 (2001).
Sequeira López, M. L. S., Pentz, E. S., Nomasa, T., Smithies, O. & Gomez, R. A. Renin cells are precursors for multiple cell types that switch to the renin phenotype when homeostasis is threatened. Dev. Cell 6, 719–728 (2004).
Sequeira-Lopez, M. L. S. et al. The MicroRNA-processing enzyme dicer maintains juxtaglomerular cells. J. Am. Soc. Nephrol. 21, 460–467 (2010).
Brunskill, E. W. et al. Genes that confer the identity of the renin cell. J. Am. Soc. Nephrol. 22, 2213–2225 (2011).
Berg, A. C. Pericytes synthesize renin. World J. Nephrol. 2, 11 (2013).
Song, R., Lopez, M. L. S. S. & Yosypiv, I. V. Foxd1 is an upstream regulator of the renin–angiotensin system during metanephric kidney development. Pediatr. Res. 82, 855–862 (2017).
Sequeira-Lopez, M. L. S. et al. The earliest metanephric arteriolar progenitors and their role in kidney vascular development. Am. J. Physiol. Regul. Integr. Comp. Physiol. 308, R138–R149 (2015).
Hyink, D. P. et al. Endogenous origin of glomerular endothelial and mesangial cells in grafts of embryonic kidneys. Am. J. Physiol. 270, F886–F899 (1996).
Abrahamson, D. R., Robert, B., Hyink, D. P., St John, P. L. & Daniel, T. O. Origins and formation of microvasculature in the developing kidney. Kidney Int. 67, S7–S11 (1998).
Robert, B., St John, P. L. & Abrahamson, D. R. Direct visualization of renal vascular morphogenesis in Flk1 heterozygous mutant mice. Am. J. Physiol. 275, F164–F172 (1998).
Gattone II, V. H. & Goldowitz, D. The renal glomerulus and vasculature in ‘aggregation’ chimeric mice. Nephron 90, 267–272 (2002).
Sequeira Lopez, M. L. S. & Gomez, R. A. Development of the renal arterioles. J. Am. Soc. Nephrol. 22, 2156–2165 (2011).
Stolz, D. B. & Sims-Lucas, S. Unwrapping the origins and roles of the renal endothelium. Pediatr. Nephrol. 30, 865–872 (2015).
Herzlinger, D. & Hurtado, R. Patterning the renal vascular bed. Semin. Cell Dev. Biol. 36, 50–56 (2014).
Robert, B., St John, P. L., Hyink, D. P. & Abrahamson, D. R. Evidence that embryonic kidney cells expressing flk-1 are intrinsic, vasculogenic angioblasts. Am. J. Physiol. 271, F744–F753 (1996).
Munro, D. A. D., Hohenstein, P. & Davies, J. A. Cycles of vascular plexus formation within the nephrogenic zone of the developing mouse kidney. Sci. Rep. 7, 3273 (2017).
Sims-Lucas, S. et al. Endothelial progenitors exist within the kidney and lung mesenchyme. PLoS ONE 8, e65993 (2013).
Halt, K. J. et al. CD146+ cells are essential for kidney vasculature development. Kidney Int. 90, 311–324 (2016).
Gao, X. et al. Angioblast-mesenchyme induction of early kidney development is mediated by Wt1 and Vegfa. Development 132, 5437–5449 (2005).
Naruse, K. et al. An immunohistochemical study of developing glomeruli in human fetal kidneys, see editorial by Oliver and Al-Awqati, 2167. Kidney Int. 57, 1836–1846 (2000).
Terry, P., Y., Dumont, D. J., Conlon, R. A., Breitman, M. L. & Rossant, J. flk-1, an flt-related receptor tyrosine kinase is an early marker for endothelial cell precursors. Development 118, 489–498 (1993).
Dumont, D. J. et al. Vascularization of the mouse embryo: a study of flk-1, tek, tie, and vascular endothelial growth factor expression during development. Dev. Dyn. 203, 80–92 (1995).
Satchell, S. C. et al. Human podocytes express angiopoietin 1, a potential regulator of glomerular vascular endothelial growth factor. J. Am. Soc. Nephrol. 13, 544–550 (2002).
Rosselot, C. et al. Non-cell-autonomous retinoid signaling is crucial for renal development. Development 137, 283–292 (2010).
Mendelsohn, C., Batourina, E., Fung, S., Gilbert, T. & Dodd, J. Stromal cells mediate retinoid-dependent functions essential for renal development. Development 126, 1139–1148 (1999).
Paroly, S. S. et al. Stromal protein Ecm1 regulates ureteric bud patterning and branching. PLoS ONE 8, e84155 (2013).
Munro, D. A. D., Hohenstein, P., Coate, T. M. & Davies, J. A. Refuting the hypothesis that semaphorin-3f/neuropilin-2 exclude blood vessels from the cap mesenchyme in the developing kidney. Dev. Dyn. 246, 1047–1056 (2017).
Reddi, V., Zaglul, A., Pentz, E. S. & Gomez, R. A. Renin-expressing cells are associated with branching of the developing kidney vasculature. J. Am. Soc. Nephrol. 9, 63–71 (1998).
Oliverio, M. I. et al. Reduced growth, abnormal kidney structure, and type 2 (AT(2)) angiotensin receptor-mediated blood pressure regulation in mice lacking both AT(1A) and AT(1B) receptors for angiotensin II. Proc. Natl Acad. Sci. USA 95, 15496–15501 (1998).
Daïkha-Dahmane, F., Levy-Beff, E., Jugie, M. & Lenclen, R. Foetal kidney maldevelopment in maternal use of angiotensin II type I receptor antagonists. Pediatr. Nephrol. 21, 729–732 (2006).
Matsui, T. et al. Redundant roles of Sox17 and Sox18 in postnatal angiogenesis in mice. J. Cell Sci. 119, 3513–3526 (2006).
Atlas, S. A. The renin-angiotensin aldosterone system: pathophysiological role and pharmacologic inhibition. J. Manag. Care Pharm. 13, 9–20 (2007).
Peppiatt-Wildman, C. M. The evolving role of renal pericytes. Curr. Opin. Nephrol. Hypertens. 22, 10–16 (2013).
Stefanska, A. M., Peault, B. & Mullins, J. J. Renal pericytes: multifunctional cells of the kidneys. Eur. J. Physiol. 465, 767–773 (2013).
Zhang, Q. et al. Membrane current oscillations in descending vasa recta pericytes. Am. J. Physiol. Renal Physiol. 294, F656–F666 (2008).
Crawford, C., Wildman, S. S. P., Kelly, M. C., Kennedy-Lydon, T. M. & Peppiatt-Wildman, C. M. Sympathetic nerve-derived ATP regulates renal medullary vasa recta diameter via pericyte cells: a role for regulating medullary blood flow? Front. Physiol. 4, 307 (2013).
Dickhout, J. G., Mori, T. & Cowley, A. W. Tubulovascular nitric oxide crosstalk: buffering of angiotensin II-induced medullary vasoconstriction. Circ. Res. 91, 487–493 (2002).
Kennedy-Lydon, T. M. et al. Inhibition of medullary prostaglandin E2 (PGE2) by non-steroidal anti-inflammatory drugs (NSAIDs) adversely affects medullary blood flow. FASEB J. 27, 984.4–984.4 (2013).
Zhang, Z., Payne, K. & Pallone, T. L. Descending vasa recta endothelial membrane potential response requires pericyte communication. PLoS ONE 11, e0154948 (2016).
Zhang, Z., Lin, H., Cao, C., Payne, K. & Pallone, T. L. Descending vasa recta endothelial cells and pericytes form mural syncytia. Am. J. Physiol. 306, F751–F763 (2014).
Baumann, M. et al. Renal medullary effects of transient prehypertensive treatment in young spontaneously hypertensive rats. Acta Physiol. 196, 231–237 (2008).
Murray, I. R. & Péault, B. Q&A: mesenchymal stem cells - where do they come from and is it important? BMC Biol. 13, 99 (2015).
Schwab, K. E. & Gargett, C. E. Co-expression of two perivascular cell markers isolates mesenchymal stem-like cells from human endometrium. Hum. Reprod. 22, 2903–2911 (2007).
Traktuev, D. O. et al. A population of multipotent CD34-positive adipose stromal cells share pericyte and mesenchymal surface markers, reside in a periendothelial location, and stabilize endothelial networks. Circ. Res. 102, 77–85 (2008).
da Silva Meirelles, L., Caplan, A. I. & Nardi, N. B. In search of the in vivo identity of mesenchymal stem cells. Stem Cells 26, 2287–2299 (2008).
Corselli, M. et al. The tunica adventitia of human arteries and veins as a source of mesenchymal stem cells. Stem Cells Dev. 21, 1299–1308 (2012).
Eirin, A. & Lerman, L. O. Mesenchymal stem cell treatment for chronic renal failure. Stem Cell Res. Ther. 5, 83 (2014).
Choi, S. et al. The role of mesenchymal stem cells in the functional improvement of chronic renal failure. Stem Cells Dev. 18, 521–530 (2009).
Eirin, A. et al. Adipose tissue-derived mesenchymal stem cells improve revascularization outcomes to restore renal function in swine atherosclerotic renal artery stenosis. Stem Cells 30, 1030–1041 (2012).
Ebrahimi, B. et al. Mesenchymal stem cells improve medullary inflammation and fibrosis after revascularization of swine atherosclerotic renal artery stenosis. PLoS ONE 8, e67474 (2013).
Reinders, M. E. J. et al. Autologous bone marrow-derived mesenchymal stromal cells for the treatment of allograft rejection after renal transplantation: results of a phase I study. Stem Cells Transl. Med. 2, 107–111 (2013).
Reinders, M. E. J. et al. Safety of allogeneic bone marrow derived mesenchymal stromal cell therapy in renal transplant recipients: the neptune study. J. Transl. Med. 13, 344 (2015).
Kramann, R. et al. Adventitial MSC-like cells are progenitors of vascular smooth muscle cells and drive vascular calcification in chronic kidney disease. Cell Stem Cell 19, 628–642 (2016).
Morigi, M. et al. Life-sparing effect of human cord blood-mesenchymal stem cells in experimental acute kidney injury. Stem Cells 28, 513–522 (2010).
Leuning, D. G. et al. Clinical-grade isolated human kidney perivascular stromal cells as an organotypic cell source for kidney regenerative medicine. Stem Cells Transl. Med. 6, 405–418 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02166489 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02966717 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02585622 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01840540 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03325322 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02266394 (2017).
Emamian, S. A., Nielsen, M. B., Pedersen, J. F. & Ytte, L. Kidney dimensions at sonography: correlation with age, sex, and habitus in 665 adult volunteers. AJR Am. J. Roentgenol. 160, 83–86 (1993).
Oyuela-Carrasco, J., Rodríguez-Castellanos, F., Kimura, E., Delgado-Hernández, R. & Herrera-Félix, J. P. Renal length measured by ultrasound in adult mexican population. Nefrologia 29, 30–34 (2009).
Denic, A. et al. The substantial loss of nephrons in healthy human kidneys with aging. J. Am. Soc. Nephrol. 28, 313–320 (2017).
Nyengaard, J. R. & Bendtsen, T. F. Glomerular number and size in relation to age, kidney weight, and body surface in normal man. Anat. Rec. 232, 194–201 (1992).
Wetzels, J. F. M., Kiemeney, L. A. L. M., Swinkels, D. W., Willems, H. L. & den Heijer, M. Age- and gender-specific reference values of estimated GFR in Caucasians: the Nijmegen Biomedical Study. Kidney Int. 72, 632–637 (2007).
Choudhury, D., Raj, D. S. C. & Levi, M. Effect of Aging on Renal Function and Disease. The Kidney (Elsevier, 2004).
Macedo, E., Zanetta, D. M. T. & Abdulkader, R. C. R. M. Long-term follow-up of patients after acute kidney injury: patterns of renal functional recovery. PLoS ONE 7, e36388 (2012).
Forni, L. G. et al. Renal recovery after acute kidney injury. Intensive Care Med. 43, 855–866 (2017).
Schmitt, R. et al. Recovery of kidney function after acute kidney injury in the elderly: a systematic review and meta-analysis. Am. J. Kidney Dis. 5203, 262–271 (2008).
Ferenbach, Da. et al. The induction of macrophage hemeoxygenase-1 is protective during acute kidney injury in aging mice. Kidney Int. 79, 966–976 (2011).
O’Sullivan, E. D., Hughes, J. & Ferenbach, D. A. Renal aging: causes and consequences. J. Am. Soc. Nephrol. 28, 407–420 (2017).
Lin, C. H. S. et al. Endostatin and kidney fibrosis in aging: a case for antagonistic pleiotropy? Am. J. Physiol. Heart Circ. Physiol. 306, H1692–H1699 (2014).
Kang, D. H. et al. Role of the microvascular endothelium in progressive renal disease. J. Am. Soc. Nephrol. 13, 806–816 (2002).
Kang, D.-H. et al. Impaired angiogenesis in the aging kidney: vascular endothelial growth factor and Thrombospondin-1 in renal disease. Am. J. Kidney Dis. 37, 601–611 (2001).
Stefanska, A. et al. Interstitial pericytes decrease in aged mouse kidneys. Aging 7, 370–382 (2015).
Rodríguez-Iturbe, B., Johnson, R. J. & Herrera-Acosta, J. Tubulointerstitial damage & progression of renal failure. Kidney Int. 68, S82–S86 Elsevier, (2005).
Berger, K. & Moeller, M. J. Mechanisms of epithelial repair and regeneration after acute kidney injury. Semin. Nephrol. 34, 394–403 (2014).
Bidani, A. K. & Griffin, K. A. Pathophysiology of hypertensive renal damage: implications for therapy. Hypertension 44, 595–601 (2004).
Robertson, C., Deen, W., Troy, J. & Brenner, B. Dynamics of glomerular ultrafiltration in the rat. 3. Hemodynamics and autoregulation. Am. J. Physiol. 223, 1191–1200 (1972).
Berger, K. et al. Origin of regenerating tubular cells after acute kidney injury. Proc. Natl Acad. Sci. USA 111, 1533–1538 (2014).
Nath, K. A. Tubulointerstitial changes as a major determinant in the progression of renal damage. Am. J. Kidney Dis. 20, 1–17 (1992).
Menn-Josephy, H. et al. Renal interstitial fibrosis: an imperfect predictor of kidney disease progression in some patient cohorts HHS public access. Am. J. Nephrol. 44, 289–299 (2016).
Nugent, M. M., Lee, K. & He, J. C. HIPK2 is a new drug target for anti-fibrosis therapy in kidney disease. Front. Physiol. 6, 132 (2015).
Declèves, A.-E. & Sharma, K. Novel targets of antifibrotic and anti-inflammatory treatment in CKD. Nat. Rev. Nephrol. 10, 257–267 (2014).
Breyer, M. D. & Susztak, K. The next generation of therapeutics for chronic kidney disease. Nat. Rev. Drug Discov. 15, 568–588 (2016).
Zeisberg, M. & Neilson, E. G. Mechanisms of tubulointerstitial fibrosis. J. Am. Soc. Nephrol. 21, 1819–1834 (2010).
Zeisberg, E. M., Potenta, S. E., Sugimoto, H., Zeisberg, M. & Kalluri, R. Fibroblasts in kidney fibrosis emerge via endothelial-to-mesenchymal transition. J. Am. Soc. Nephrol. 19, 2282–2287 (2008).
LeBleu, V. S. et al. Origin and function of myofibroblasts in kidney fibrosis. Nat. Med. 19, 1047–1053 (2013).
Faulkner, J. L., Szcykalski, L. M., Springer, F. & Barnes, J. L. Origin of interstitial fibroblasts in an accelerated model of angiotensin ii-induced renal fibrosis. Am. J. Pathol. 167, 1193–1205 (2005).
Kramann, R., Wongboonsin, J., Chang-Panesso, M., Machado, F. G. & Humphreys, B. D. Gli1 + pericyte loss induces capillary rarefaction and proximal tubular injury. J. Am. Soc. Nephrol. 28, 776–784 (2017).
Wiggins, R., Goyal, M., Merritt, S. & Killen, P. D. Vascular adventitial cell expression of collagen I messenger ribonucleic acid in anti-glomerular basement membrane antibody-induced crescentic nephritis in the rabbit. A cellular source for interstitial collagen synthesis in inflammatory renal disease. Lab. Invest. 68, 557–565 (1993).
Habibi, J., Hayden, M. R., Ferrario, C. M., Sowers, J. R. & Whaley-Connell, A. T. Salt loading promotes kidney injury via fibrosis in young female Ren2 rats. Cardiorenal Med. 4, 43–52 (2014).
Hardy, W. R. et al. Transcriptional networks in single perivascular cells sorted from human adipose tissue reveal a hierarchy of mesenchymal stem cells. Stem Cells 35, 1273–1289 (2017).
Fabian, S. L. et al. Hedgehog-Gli pathway activation during kidney fibrosis. Am. J. Pathol. 180, 1441–1453 (2012).
Xiao, L. et al. Sustained activation of Wnt/β-catenin signaling drives AKI to CKD progression. J. Am. Soc. Nephrol. 27, 1727–1740 (2015).
Maarouf, O. H. et al. Paracrine Wnt1 drives interstitial fibrosis without inflammation by tubulointerstitial cross-talk. J. Am. Soc. Nephrol. 27, 781–790 (2016).
Grgic, I. et al. Translational profiles of medullary myofibroblasts during kidney fibrosis. J. Am. Soc. Nephrol. 25, 1979–1990 (2014).
Schrimpf, C., Teebken, O. E., Wilhelmi, M. & Duffield, J. S. The role of pericyte detachment in vascular rarefaction. J. Vasc. Res. 51, 247–258 (2014).
Lin, S. L. et al. Targeting endothelium-pericyte cross talk by inhibiting VEGF receptor signaling attenuates kidney microvascular rarefaction and fibrosis. Am. J. Pathol. 178, 911–923 (2011).
Chen, Y.-T. et al. Platelet-derived growth factor receptor signaling activates pericyte–myofibroblast transition in obstructive and post-ischemic kidney fibrosis. Kidney Int. 80, 1170–1181 (2011).
Bijkerk, R. et al. Silencing of microRNA-132 reduces renal fibrosis by selectively inhibiting myofibroblast proliferation. Kidney Int. 89, 1268–1280 (2016).
Leaf, I. A. et al. Pericyte MyD88 and IRAK4 control inflammatory and fibrotic responses to tissue injury. J. Clin. Invest. 127, 321–334 (2017).
Xavier, S. et al. Pericytes and immune cells contribute to complement activation in tubulointerstitial fibrosis. Am. J. Physiol. Renal Physiol. 312, F516–F532 (2017).
Lu, Y., Ye, Y., Yang, Q. & Shi, S. Single-cell RNA-sequence analysis of mouse glomerular mesangial cells uncovers mesangial cell essential genes. Kidney Int. 92, 504–513 (2017).
Hugo, C., Shankland, S. J., Bowen-Pope, D. F., Couser, W. G. & Johnson, R. J. Extraglomerular origin of the mesangial cell after injury. A new role of the juxtaglomerular apparatus. J. Clin. Invest. 100, 786–794 (1997).
Starke, C. et al. Renin lineage cells repopulate the glomerular mesangium after injury. J. Am. Soc. Nephrol. 26, 48–54 (2015).
Baker, A. J. et al. Mesangial cell apoptosis: the major mechanism for resolution of glomerular hypercellularity in experimental mesangial proliferative nephritis. J. Clin. Invest. 94, 2105–2116 (1994).
Villa, L. et al. Late angiotensin II receptor blockade in progressive rat mesangioproliferative glomerulonephritis: new insights into mechanisms. J. Pathol. 229, 672–684 (2013).
Al Hussain, T., Hussein, M. H., Al Mana, H. & Akhtar, M. Pathophysiology of IgA nephropathy. Adv. Anat. Pathol. 24, 56–62 (2017).
Roberts, I. S. D. et al. The Oxford classification of IgA nephropathy: pathology definitions, correlations, and reproducibility. Kidney Int. 76, 546–556 (2009).
Kroeger, P. T. & Wingert, R. A. Using zebrafish to study podocyte genesis during kidney development and regeneration. Genesis 52, 771–792 (2014).
Wingert, R. A. & Davidson, A. J. The zebrafish pronephros: a model to study nephron segmentation. Kidney Int. 73, 1120–1127 (2008).
Diep, C. Q. et al. Identification of adult nephron progenitors capable of kidney regeneration in zebrafish. Nature 470, 95–100 (2011).
Huang, J. et al. A zebrafish model of conditional targeted podocyte ablation and regeneration. Kidney Int. 83, 1193–1200 (2013).
Zhou, W., Boucher, R. C., Bollig, F., Englert, C. & Hildebrandt, F. Characterization of mesonephric development and regeneration using transgenic zebrafish. Am. J. Physiol. Renal Physiol. 299, F1040–F1047 (2010).
Rider, S. A., Mullins, L. J., Verdon, R. F., MacRae, C. A. & Mullins, J. J. Renin expression in developing zebrafish is associated with angiogenesis and requires the Notch pathway and endothelium. Am. J. Physiol. Renal Physiol. 309, F531–F539 (2015).
Santoro, M. M., Pesce, G. & Stainier, D. Y. Characterization of vascular mural cells during zebrafish development. Mech. Dev. 126, 638–649 (2009).
Ando, K. et al. Clarification of mural cell coverage of vascular endothelial cells by live imaging of zebrafish. Development 143, 1328–1339 (2016).
Whitesell, T. R. et al. An α-smooth muscle actin (acta2/αsma) zebrafish transgenic line marking vascular mural cells and visceral smooth muscle cells. PLoS ONE 9, e90590 (2014).
Liang, P. Genomic characterization and expression analysis of the first nonmammalian renin genes from zebrafish and pufferfish. Physiol. Genomics. 16, 314–322 (2004).
Drummond, I. Making a zebrafish kidney: a tale of two tubes. Trends Cell Biol. 13, 357–365 (2003).
Drummond, I. A. et al. Early development of the zebrafish pronephros and analysis of mutations affecting pronephric function. Development 125, 4655–4667 (1998).
Isogai, S., Horiguchi, M. & Weinstein, B. M. The vascular anatomy of the developing zebrafish: an atlas of embryonic and early larval development. Dev. Biol. 230, 278–301 (2001).
Rider, S. A. et al. Zebrafish mesonephric renin cells are functionally conserved and comprise two distinct morphological populations. Am. J. Physiol. Renal Physiol. 312, F778–F790 (2017).
Curado, S. et al. Conditional targeted cell ablation in zebrafish: a new tool for regeneration studies. Dev. Dyn. 236, 1025–1035 (2007).
Zhou, W. & Hildebrandt, F. Inducible podocyte injury and proteinuria in transgenic zebrafish. J. Am. Soc. Nephrol. 23, 1039–1047 (2012).
Bulina, M. E. et al. A genetically encoded photosensitizer. Nat. Biotechnol. 24, 95–99 (2006).
Teh, C. et al. Optogenetic in vivo cell manipulation in KillerRed-expressing zebrafish transgenics. BMC Dev. Biol. 10, 110 (2010).
Buckley, C. et al. Precise spatio-temporal control of rapid optogenetic cell ablation with mem-KillerRed in Zebrafish. Sci. Rep. 7, 5096 (2017).
Kramer-Zucker, A. G., Wiessner, S., Jensen, A. M. & Drummond, I. A. Organization of the pronephric filtration apparatus in zebrafish requires nephrin, podocin and the FERM domain protein mosaic eyes. Dev. Biol. 285, 316–329 (2005).
Zhu, X. et al. Ultrastructural characterization of the pronephric glomerulus development in zebrafish. J. Morphol. 277, 1104–1112 (2016).
Dunn, K. W., Sutton, T. A. & Sandoval, R. M. Live-animal imaging of renal function by multiphoton microscopy. Curr. Protoc. Cytom. https://doi.org/10.1002/0471142956.cy1209s62 (2012).
Mosimann, C. et al. Ubiquitous transgene expression and Cre-based recombination driven by the ubiquitin promoter in zebrafish. Development 138, 169–177 (2011).
Pan, Y. A. et al. Zebrabow: multispectral cell labeling for cell tracing and lineage analysis in zebrafish. Development 140, 2835–2846 (2013).
Weissman, T. A. & Pan, Y. A. Brainbow: new resources and emerging biological applications for multicolor genetic labeling and analysis. Genetics 199, 293–306 (2015).
Rider, S. A., Bruton, F. A., Collins, R. G., Conway, B. R. & Mullins, J. J. The efficacy of puromycin and adriamycin for induction of glomerular failure in larval zebrafish validated by an assay of glomerular permeability dynamics. Zebrafish https://doi.org/10.1089/zeb.2017.1527 (2018).
Armulik, A. et al. Pericytes regulate the blood–brain barrier. Nature 468, 557–561 (2010).
Chen, W. et al. Human myocardial pericytes: multipotent mesodermal precursors exhibiting cardiac specificity. Stem Cells 33, 557–573 (2015).
Guimarães-Camboa, N. et al. Pericytes of multiple organs do not behave as mesenchymal stem cells in vivo. Cell Stem Cell 20, 345–359.e5 (2017).
Tang, W. et al. White fat progenitor cells reside in the adipose vasculature. Science 322, 583–586 (2008).
Zhao, H. et al. Secretion of shh by a neurovascular bundle niche supports mesenchymal stem cell homeostasis in the adult mouse incisor. Cell Stem Cell 14, 160–173 (2014).
Dellavalle, A. et al. Pericytes resident in postnatal skeletal muscle differentiate into muscle fibres and generate satellite cells. Nat. Commun. 2, 499 (2011).
Dickinson, S. C. et al. The Wnt5a receptor, receptor tyrosine kinase-like orphan receptor 2, is a predictive cell surface marker of human mesenchymal stem cells with an enhanced capacity for chondrogenic differentiation. Stem Cells 35, 2280–2291 (2017).
Shen, J. et al. Pericyte antigens in perivascular soft tissue tumors. Int. J. Surg. Pathol. 23, 638–648 (2015).
Wong, L. et al. Reninoma: case report and literature review. J. Hypertens. 26, 368–373 (2008).
Cooke, V. G. et al. Pericyte depletion results in hypoxia-associated epithelial-to-mesenchymal transition and metastasis mediated by met signaling pathway. Cancer Cell 21, 66–81 (2012).
Péault, B. Peril in perishing pericytes. J. Natl Cancer Inst. 107, djv254 (2015).
Bentolila, L. A. et al. Imaging of angiotropism/vascular co-option in a murine model of brain melanoma: implications for melanoma progression along extravascular pathways. Sci. Rep. 6, 23834 (2016).
O’Farrel, F. M. et al. Capillary pericytes mediate coronary no-reflow after myocardial ischaemia. eLife 6, e29280 (2017).
Reviewer information
Nature Reviews Nephrology thanks R. Kramann and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
All authors wrote the manuscript. B.P., I.S. and J.H. made substantial contributions to discussions of the content and reviewed or edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Angiogenesis
-
The formation and remodelling of new blood vessels and capillaries from the growth of existing blood vessels.
- Mesangial cells
-
Specialized perivascular cells of the glomerulus that provide structural support to the capillary tuft.
- Adventitial cells
-
Perivascular mesenchymal cells that are located in the tunica adventitia layer of large arteries and veins and can give rise to mesenchymal stem/stromal cells in culture. Molecularly defined as CD45−CD31−CD146−CD34+.
- Vasa recta
-
Bundles of parallel ascending and descending vessels that run from the cortex to the inner medulla of the kidney and form a countercurrent exchange system for water and solutes.
- Ureteric bud
-
(UB). An outgrowth of the mesonephric (or Wolffian) duct that invades the metanephric mesenchyme and undergoes subsequent rounds of branching to give rise to the collecting ducts of the kidney and to the ureter.
- Cap mesenchyme
-
Metanephric mesenchyme that condenses around the tip of the ureteric bud during kidney development and gives rise to cells of the nephron. The cap mesenchyme is characterized by expression of SIX2 and Cbp/p300-interacting transactivator 1 (CITED1).
- Renin–angiotensin system
-
(RAS). A hormonal system of blood pressure regulation. In response to low blood pressure, juxtaglomerular pericytes secrete renin, which converts circulating angiotensinogen to angiotensin I, leading to a cascade that ultimately increases blood pressure.
- Vasculogenesis
-
The de novo formation of new blood vessels via the differentiation of endothelial progenitor cells.
- Mesenchymal stem/stromal cells
-
(MSCs). Plastic adherent cells that express CD105, CD73 and CD90, do not express endothelial and haematopoietic cell markers, and have the ability to differentiate into osteoblasts, adipocytes and chondroblasts in vitro. Pericytes and adventitial cells are in vivo precursors of culture-derived MSCs, which have potential therapeutic properties.
- Acute kidney injury
-
(AKI). An abrupt loss of kidney function in response to a pathological stimulus. AKI is clinically defined as an increase in serum creatinine and/or blood urea nitrogen levels and is characterized by tubular death. Although typically short term and reversible, AKI may predispose to chronic kidney disease.
- Chronic kidney disease
-
(CKD). Long-term, typically irreversible decline in renal function characterized by glomerular and tubulointerstitial fibrosis.
- Myofibroblast
-
Scar-forming cell present in the glomeruli or interstitium of injured kidney that are characterized by the expression of α-smooth muscle actin.
- Nitroreductase (NTR) system
-
A cell-ablation technique in which bacterial nitroreductase enzyme is transgenically expressed in cells of interest, thus sensitizing them to metronidazole.
- mem-KillerRed
-
A membrane-bound protein that when excited by 520–590 nm light produces reactive oxygen species that are toxic to the cell. This protein can be genetically targeted to cells of interest for user-defined spatial and temporal cell ablation in light-accessible tissue.
- Bessel beam
-
A specialized form of laser beam formed either by passing a beam through an axicon lens or by reflecting it off a spatial light modulator. Bessel beams are resistant to scattering and diffraction and therefore can penetrate further into tissue than other types of laser beams.
Rights and permissions
About this article
Cite this article
Shaw, I., Rider, S., Mullins, J. et al. Pericytes in the renal vasculature: roles in health and disease. Nat Rev Nephrol 14, 521–534 (2018). https://doi.org/10.1038/s41581-018-0032-4
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41581-018-0032-4
This article is cited by
-
Role of perivascular cells in kidney homeostasis, inflammation, repair and fibrosis
Nature Reviews Nephrology (2023)
-
Upregulation of Piezo2 in the mesangial, renin, and perivascular mesenchymal cells of the kidney of Dahl salt-sensitive hypertensive rats and its reversal by esaxerenone
Hypertension Research (2023)
-
Vasculogenesis in kidney organoids upon transplantation
npj Regenerative Medicine (2022)
-
A novel renal perivascular mesenchymal cell subset gives rise to fibroblasts distinct from classic myofibroblasts
Scientific Reports (2022)
-
Versatile subtypes of pericytes and their roles in spinal cord injury repair, bone development and repair
Bone Research (2022)