Abstract
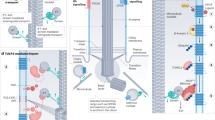
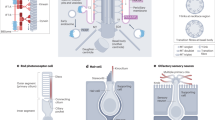
The primary cilium is a hair-like surface-exposed organelle of the eukaryotic cell that decodes a variety of signals — such as odorants, light and Hedgehog morphogens — by altering the local concentrations and activities of signalling proteins. Signalling within the cilium is conveyed through a diverse array of second messengers, including conventional signalling molecules (such as cAMP) and some unusual intermediates (such as sterols). Diffusion barriers at the ciliary base establish the unique composition of this signalling compartment, and cilia adapt their proteome to signalling demands through regulated protein trafficking. Much progress has been made on the molecular understanding of regulated ciliary trafficking, which encompasses not only exchanges between the cilium and the rest of the cell but also the shedding of signalling factors into extracellular vesicles.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Reiter, J. F. & Leroux, M. R. Genes and molecular pathways underpinning ciliopathies. Nat. Rev. Mol. Cell. Biol. 18, 533–547 (2017).
de Robertis, E. Electron microscope observations on the submicroscopic organization of the retinal rods. J. Biophys. Biochem. Cytol. 2, 319–330 (1956).
Bloom, G. Studies on the olfactory epithelium of the frog and the toad with the aid of light and electron microscopy. Z. Zellforsch. Mikrosk. Anat. 41, 89–100 (1954).
Huangfu, D. et al. Hedgehog signalling in the mouse requires intraflagellar transport proteins. Nature 426, 83–87 (2003).
Gerdes, J. M. et al. Disruption of the basal body compromises proteasomal function and perturbs intracellular Wnt response. Nat. Genet. 39, 1350–1360 (2007).
Ezratty, E. J. et al. A role for the primary cilium in Notch signaling and epidermal differentiation during skin development. Cell 145, 1129–1141 (2011).
Ross, A. J. et al. Disruption of Bardet-Biedl syndrome ciliary proteins perturbs planar cell polarity in vertebrates. Nat. Genet. 37, 1135–1140 (2005).
Schneider, L. et al. PDGFRαα signaling is regulated through the primary cilium in fibroblasts. Curr. Biol. 15, 1861–1866 (2005).
Yeh, C. et al. IGF-1 activates a cilium-localized noncanonical Gβγ signaling pathway that regulates cell-cycle progression. Dev. Cell 26, 358–368 (2013).
Schou, K. B., Pedersen, L. B. & Christensen, S. T. Ins and outs of GPCR signaling in primary cilia. EMBO Rep. 16, 1099–1113 (2015).
Clement, C. A. et al. TGF-β signaling is associated with endocytosis at the pocket region of the primary cilium. Cell Rep. 3, 1806–1814 (2013).
Siljee, J. E. et al. Subcellular localization of MC4R with ADCY3 at neuronal primary cilia underlies a common pathway for genetic predisposition to obesity. Nat. Genet. 50, 180–185 (2018).
Bloodgood, R. A. Sensory reception is an attribute of both primary cilia and motile cilia. J. Cell Sci. 123, 505–509 (2010).
Wachten, D., Jikeli, J. F. & Kaupp, U. B. Sperm sensory signaling. Cold Spring Harb. Perspect. Biol. 9, a028225 (2017).
Calvert, P. D., Schiesser, W. E. & Pugh, E. N. Diffusion of a soluble protein, photoactivatable GFP, through a sensory cilium. J. Gen. Physiol. 135, 173–196 (2010).
Bhandawat, V., Reisert, J. & Yau, K.-W. Signaling by olfactory receptor neurons near threshold. Proc. Natl Acad. Sci. USA 107, 18682–18687 (2010).
Corbit, K. C. et al. Vertebrate Smoothened functions at the primary cilium. Nature 437, 1018–1021 (2005).
Haycraft, C. J. et al. Gli2 and Gli3 localize to cilia and require the intraflagellar transport protein polaris for processing and function. PLOS Genet. 1, e53 (2005).
May, S. R. et al. Loss of the retrograde motor for IFT disrupts localization of Smo to cilia and prevents the expression of both activator and repressor functions of Gli. Dev. Biol. 287, 378–389 (2005).
Belzile, O., Hernandez-Lara, C. I., Wang, Q. & Snell, W. J. Regulated membrane protein entry into flagella is facilitated by cytoplasmic microtubules and does not require IFT. Curr. Biol. 23, 1460–1465 (2013).
Rohatgi, R., Milenkovic, L. & Scott, M. P. Patched1 regulates hedgehog signaling at the primary cilium. Science 317, 372–376 (2007).
Bangs, F. & Anderson, K. V. Primary cilia and mammalian Hedgehog signaling. Cold Spring Harb. Perspect. Biol. 9, a028175 (2017).
Pablo, J. L., DeCaen, P. G. & Clapham, D. E. Progress in ciliary ion channel physiology. J. Gen. Physiol. 149, 37–47 (2017).
Johnson, J.-L. F. & Leroux, M. R. cAMP and cGMP signaling: sensory systems with prokaryotic roots adopted by eukaryotic cilia. Trends Cell Biol. 20, 435–444 (2010).
Garcia, G., Raleigh, D. R. & Reiter, J. F. How the ciliary membrane is organized inside-out to communicate outside-in. Curr. Biol. 28, R421–R434 (2018).
Luchetti, G. et al. Cholesterol activates the G-protein coupled receptor Smoothened to promote Hedgehog signaling. eLife 5, e20304 (2016).
Huang, P. et al. Cellular cholesterol directly activates Smoothened in Hedgehog signaling. Cell 166, 1176–1187 (2016).
Raleigh, D. R. et al. Cilia-associated oxysterols activate Smoothened. Mol. Cell 72, 316–327 (2018). This paper identifies the oxysterols 24,25-epoxycholesterol and 7β,27-dihydroxycholesterol as ciliary lipids that bind and activate Smoothened.
Nager, A. R. et al. An actin network dispatches ciliary GPCRs into extracellular vesicles to modulate signaling. Cell 168, 252–263 (2017). This paper uncovers signal-dependent ectocytosis as a means for ciliary exit of activated GPCRs in mammalian cells; see also Cao et al. (2015). It also discovers a role for actin in ectosome scission; see also Phua et al. (2017).
Phua, S. C. et al. Dynamic remodeling of membrane composition drives cell cycle through primary cilia excision. Cell 168, 264–279 (2017). This paper presents the discovery of a role for actin in ectosome release; see also Nager et al. (2017). It also suggests that cilia shorten by shedding ectosomes; see also Long et al. (2016).
Long, H. et al. Comparative analysis of ciliary membranes and ectosomes. Curr. Biol. 26, 3327–3335 (2016). This study demonstrates that cilia shorten, at least in part, by shedding ectosomes; see also Phua et al. (2017). It also suggests a role for ESCRT proteins ALG2 and VPS4 in ciliary ectocytosis.
Fisch, C. & Dupuis-Williams, P. Ultrastructure of cilia and flagella — back to the future! Biol. Cell 103, 249–270 (2011).
Garcia-Gonzalo, F. R. & Reiter, J. F. Open sesame: how transition fibers and the transition zone control ciliary composition. Cold Spring Harb. Perspect. Biol. 9, a028134 (2017).
Gonçalves, J. & Pelletier, L. The ciliary transition zone: finding the pieces and assembling the gate. Mol. Cells 40, 243–253 (2017).
Breslow, D., Koslover, E. F., Seydel, F., Spakowitz, A. J. & Nachury, M. V. An in vitro assay for entry into cilia reveals unique properties of the soluble diffusion barrier. J. Cell Biol. 203, 129–147 (2013).
Cevik, S. et al. Active transport and diffusion barriers restrict Joubert Syndrome-associated ARL13B/ARL-13 to an Inv-like ciliary membrane subdomain. PLOS Genet. 9, e1003977 (2013).
Chih, B. et al. A ciliopathy complex at the transition zone protects the cilia as a privileged membrane domain. Nat. Cell Biol. 14, 61–72 (2011).
Hu, Q. et al. A septin diffusion barrier at the base of the primary cilium maintains ciliary membrane protein distribution. Science 329, 436–439 (2010).
Kee, H. L. et al. A size-exclusion permeability barrier and nucleoporins characterize a ciliary pore complex that regulates transport into cilia. Nat. Cell Biol. 14, 431–437 (2012).
Lin, Y.-C. et al. Chemically inducible diffusion trap at cilia reveals molecular sieve-like barrier. Nat. Chem. Biol. 9, 437–443 (2013).
Yang, T. T. et al. Super-resolution architecture of mammalian centriole distal appendages reveals distinct blade and matrix functional components. Nat. Commun. 9, 2023 (2018). This study establishes the existence of the DAM and suggests that the DAM functions as a barrier for ciliary membrane proteins.
Ye, F., Nager, A. R. & Nachury, M. V. BBSome trains remove activated GPCRs from cilia by enabling passage through the transition zone. J. Cell Biol. 217, 1847–1868 (2018). Using single-molecule tracking, this study directly demonstrates that the transition zone is breached by exiting GPCRs in a BBSome-dependent manner.
Najafi, M., Maza, N. A. & Calvert, P. D. Steric volume exclusion sets soluble protein concentrations in photoreceptor sensory cilia. Proc. Natl Acad. Sci. USA 109, 203–208 (2012).
Lambacher, N. J. et al. TMEM107 recruits ciliopathy proteins to subdomains of the ciliary transition zone and causes Joubert syndrome. Nat. Cell Biol. 18, 122–131 (2016).
Takao, D., Wang, L., Boss, A. & Verhey, K. J. Protein interaction analysis provides a map of the spatial and temporal organization of the ciliary gating zone. Curr. Biol. 27, 2296–2306 (2017).
Craige, B. et al. CEP290 tethers flagellar transition zone microtubules to the membrane and regulates flagellar protein content. J. Cell Biol. 190, 927–940 (2010).
Awata, J. et al. NPHP4 controls ciliary trafficking of membrane proteins and large soluble proteins at the transition zone. J. Cell Sci. 127, 4714–4727 (2014).
Scheidel, N. & Blacque, O. E. Intraflagellar transport complex A genes differentially regulate cilium formation and transition zone gating. Curr. Biol. 28, 3279–3287 (2018).
Vuolo, L., Stevenson, N. L., Heesom, K. J. & Stephens, D. J. Dynein-2 intermediate chains play crucial but distinct roles in primary cilia formation and function. eLife 7, e39655 (2018).
Jensen, V. L. et al. Role for intraflagellar transport in building a functional transition zone. EMBO Rep. 19, e45862 (2018).
Dharmat, R. et al. SPATA7 maintains a novel photoreceptor-specific zone in the distal connecting cilium. J. Cell Biol. 217, 2851–2865 (2018).
Wiegering, A. et al. Cell type-specific regulation of ciliary transition zone assembly in vertebrates. EMBO J. 37, e97791 (2018).
Jana, S. C. et al. Differential regulation of transition zone and centriole proteins contributes to ciliary base diversity. Nat. Cell Biol. 20, 928–941 (2018).
Lewis, W. R. et al. Mks6 mutations reveal tissue- and cell type-specific roles for the cilia transition zone. FASEB J. 33, 1440–1455 (2018).
Jensen, V. L. et al. Formation of the transition zone by Mks5/Rpgrip1L establishes a ciliary zone of exclusion (CIZE) that compartmentalises ciliary signalling proteins and controls PIP2 ciliary abundance. EMBO J. 34, 2537–2556 (2015).
Williams, C. L. et al. MKS and NPHP modules cooperate to establish basal body/transition zone membrane associations and ciliary gate function during ciliogenesis. J. Cell Biol. 192, 1023–1041 (2011).
Yee, L. E. et al. Conserved genetic interactions between ciliopathy complexes cooperatively support ciliogenesis and ciliary signaling. PLOS Genet. 11, e1005627 (2015).
Roberson, E. C. et al. TMEM231, mutated in orofaciodigital and Meckel syndromes, organizes the ciliary transition zone. J. Cell Biol. 209, 129–142 (2015).
Milenkovic, L. et al. Single-molecule imaging of Hedgehog pathway protein Smoothened in primary cilia reveals binding events regulated by Patched1. Proc. Natl Acad. Sci. USA 112, 8320–8325 (2015).
Shi, X. et al. Super-resolution microscopy reveals that disruption of ciliary transition-zone architecture causes Joubert syndrome. Nat. Cell Biol. 19, 1178–1188 (2017).
Tanos, B. E. et al. Centriole distal appendages promote membrane docking, leading to cilia initiation. Genes Dev. 27, 163–168 (2013).
Li, W. et al. Centriole translocation and degeneration during ciliogenesis in Caenorhabditis elegans neurons. EMBO J. 36, 2553–2566 (2017).
Nechipurenko, I. V., Berciu, C., Sengupta, P. & Nicastro, D. Centriolar remodeling underlies basal body maturation during ciliogenesis in Caenorhabditis elegans. eLife 6, e25686 (2017).
Serwas, D., Su, T. Y., Roessler, M., Wang, S. & Dammermann, A. Centrioles initiate cilia assembly but are dispensable for maturation and maintenance in C. elegans. J. Cell Biol. 216, 1659–1671 (2017).
Sugimoto, M. et al. The keratin-binding protein Albatross regulates polarization of epithelial cells. J. Cell Biol. 183, 19–28 (2008).
Sang, L. et al. Mapping the NPHP-JBTS-MKS protein network reveals ciliopathy disease genes and pathways. Cell 145, 513–528 (2011).
Delous, M. et al. Nephrocystin-1 and nephrocystin-4 are required for epithelial morphogenesis and associate with PALS1/PATJ and Par6. Hum. Mol. Genet. 18, 4711–4723 (2009).
Wei, Q. et al. The hydrolethalus syndrome protein HYLS-1 regulates formation of the ciliary gate. Nat. Commun. 7, 12437 (2016).
DeCaen, P. G., Delling, M., Vien, T. N. & Clapham, D. E. Direct recording and molecular identification of the calcium channel of primary cilia. Nature 504, 315–318 (2013).
Delling, M., DeCaen, P. G., Doerner, J. F., Febvay, S. & Clapham, D. E. Primary cilia are specialized calcium signalling organelles. Nature 504, 311–314 (2013).
Borisy, F. F. et al. Calcium/calmodulin-activated phosphodiesterase expressed in olfactory receptor neurons. J. Neurosci. 12, 915–923 (1992).
Mukherjee, S. et al. A novel biosensor to study cAMP dynamics in cilia and flagella. eLife 5, e14052 (2016).
Moore, B. S. et al. Cilia have high cAMP levels that are inhibited by Sonic Hedgehog-regulated calcium dynamics. Proc. Natl Acad. Sci. USA 113, 13069–13074 (2016).
Grarup, N. et al. Loss-of-function variants in ADCY3 increase risk of obesity and type 2 diabetes. Nat. Genet. 50, 172–174 (2018).
Saeed, S. et al. Loss-of-function mutations in ADCY3 cause monogenic severe obesity. Nat. Genet. 50, 175–179 (2018).
Rees, S. et al. Adenylyl cyclase 6 deficiency ameliorates polycystic kidney disease. J. Am. Soc. Nephrol. 25, 232–237 (2014).
Wang, Q. et al. Adenylyl cyclase 5 deficiency reduces renal cyclic AMP and cyst growth in an orthologous mouse model of polycystic kidney disease. Kidney Int. 93, 403–415 (2018).
Breslow, D. K. et al. A CRISPR-based screen for Hedgehog signaling provides insights into ciliary function and ciliopathies. Nat. Genet. 50, 460–471 (2018).
Pusapati, G. V. et al. CRISPR screens uncover genes that regulate target cell sensitivity to the morphogen Sonic Hedgehog. Dev. Cell 44, 113–129 (2018).
Vuolo, L., Herrera, A., Torroba, B., Menendez, A. & Pons, S. Ciliary adenylyl cyclases control the Hedgehog pathway. J. Cell Sci. 128, 2928–2937 (2015).
Kwon, R. Y., Temiyasathit, S., Tummala, P., Quah, C. C. & Jacobs, C. R. Primary cilium-dependent mechanosensing is mediated by adenylyl cyclase 6 and cyclic AMP in bone cells. FASEB J. 24, 2859–2868 (2010).
Sadana, R. & Dessauer, C. W. Physiological roles for G protein-regulated adenylyl cyclase isoforms: insights from knockout and overexpression studies. Neurosignals 17, 5–22 (2009).
Hilgendorf, K. I., Johnson, C. T. & Jackson, P. K. The primary cilium as a cellular receiver: organizing ciliary GPCR signaling. Curr. Opin. Cell Biol. 39, 84–92 (2016).
Pedersen, L. B., Geimer, S., Sloboda, R. D. & Rosenbaum, J. L. The microtubule plus end-tracking protein EB1 is localized to the flagellar tip and basal bodies in Chlamydomonas reinhardtii. Curr. Biol. 13, 1969–1974 (2003).
Harris, J. A., Liu, Y., Yang, P., Kner, P. & Lechtreck, K. F. Single-particle imaging reveals intraflagellar transport-independent transport and accumulation of EB1 in Chlamydomonas flagella. Mol. Biol. Cell 27, 295–307 (2016).
Chávez, M. et al. Modulation of ciliary phosphoinositide content regulates trafficking and Sonic Hedgehog signaling output. Dev. Cell 34, 338–350 (2015).
Garcia-Gonzalo, F. R. et al. Phosphoinositides regulate ciliary protein trafficking to modulate Hedgehog signaling. Dev. Cell 34, 400–409 (2015). Chavez et al. (2015) and Garcia-Gonzalo et al. (2015) demonstrate that the lipid PtdIns(4)P is enriched in the ciliary membrane thanks to the phosphatase INPP5E, which converts PtdIns(4,5)P 2 into PtdIns(4)P.
Poo, M. & Cone, R. A. Lateral diffusion of rhodopsin in the photoreceptor membrane. Nature 247, 438–441 (1974).
Aveldaño, M. I. & Bazán, N. G. Molecular species of phosphatidylcholine, -ethanolamine, -serine, and -inositol in microsomal and photoreceptor membranes of bovine retina. J. Lipid Res. 24, 620–627 (1983).
Boesze-Battaglia, K. & Schimmel, R. Cell membrane lipid composition and distribution: implications for cell function and lessons learned from photoreceptors and platelets. J. Exp. Biol. 200, 2927–2936 (1997).
Andrews, D. & Nelson, D. L. Biochemical studies of the excitable membrane of Paramecium tetraurelia. II. Phospholipids of ciliary and other membranes. Biochim. Biophys. Acta 550, 174–187 (1979).
Chailley, B. & Boisvieux-Ulrich, E. Detection of plasma membrane cholesterol by filipin during microvillogenesis and ciliogenesis in quail oviduct. J. Histochem. Cytochem. 33, 1–10 (1985).
Lobasso, S. et al. Lipidomic analysis of porcine olfactory epithelial membranes and cilia. Lipids 45, 593–602 (2010).
Montesano, R. Inhomogeneous distribution of filipin-sterol complexes in the ciliary membrane of rat tracheal epithelium. Am. J. Anat. 156, 139–145 (1979).
Morris, R. J. & Bone, Q. Metazoan lipids: an unusual association of saturated sterols with relatively saturated fatty acids in the cilia of Ciona intestinalis. Lipids 18, 900–901 (1983).
Morris, R. J. & Bone, Q. Highly saturated lipid composition of ctenophore cilia: possible indication of low membrane permeability. Lipids 20, 933–935 (1985).
Souto-Padrón, T. & de Souza, W. Freeze-fracture localization of filipin-cholesterol complexes in the plasma membrane of Trypanosoma cruzi. J. Parasitol. 69, 129–137 (1983).
Tyler, K. M. et al. Flagellar membrane localization via association with lipid rafts. J. Cell Sci. 122, 859–866 (2009).
Emmer, B. T., Maric, D. & Engman, D. M. Molecular mechanisms of protein and lipid targeting to ciliary membranes. J. Cell Sci. 123, 529–536 (2010).
Nachury, M. V., Seeley, E. S. & Jin, H. Trafficking to the ciliary membrane: how to get across the periciliary diffusion barrier? Annu. Rev. Cell Dev. Biol. 26, 59–87 (2010).
Serricchio, M. et al. Flagellar membranes are rich in raft-forming phospholipids. Biol. Open 4, 1143–1153 (2015).
Nguyen, P. A. T., Liou, W., Hall, D. H. & Leroux, M. R. Ciliopathy proteins establish a bipartite signaling compartment in a C. elegans thermosensory neuron. J. Cell Sci. 127, 5317–5330 (2014).
Schou, K. B. et al. KIF13B establishes a CAV1-enriched microdomain at the ciliary transition zone to promote Sonic hedgehog signalling. Nat. Commun. 8, 14177 (2017). This study demonstrates cholesterol-dependent localization of the cholesterol-binding protein caveolin to the transition zone, which suggests a specialized lipid organization at the transition zone.
Kamiya, R. & Witman, G. B. Submicromolar levels of calcium control the balance of beating between the two flagella in demembranated models of Chlamydomonas. J. Cell Biol. 98, 97–107 (1984).
Lechtreck, K. F. IFT–cargo interactions and protein transport in cilia. Trends Biochem. Sci. 40, 765–778 (2015).
Nachury, M. V. The molecular machines that traffic signaling receptors into and out of cilia. Curr. Opin. Cell Biol. 51, 124–131 (2018).
Nakayama, K. & Katoh, Y. Ciliary protein trafficking mediated by IFT and BBSome complexes with the aid of kinesin-2 and dynein-2 motors. J. Biochem. 163, 155–164 (2018).
Bhogaraju, S. et al. Molecular basis of tubulin transport within the cilium by IFT74 and IFT81. Science 341, 1009–1012 (2013).
Craft, J. M., Harris, J. A., Hyman, S., Kner, P. & Lechtreck, K. F. Tubulin transport by IFT is upregulated during ciliary growth by a cilium-autonomous mechanism. J. Cell Biol. 208, 223–237 (2015).
Kubo, T. et al. Together, the IFT81 and IFT74 N-termini form the main module for intraflagellar transport of tubulin. J. Cell Sci. 129, 2106–2119 (2016).
Taschner, M., Mourão, A., Awasthi, M., Basquin, J. & Lorentzen, E. Structural basis of outer dynein arm intraflagellar transport by the transport adaptor protein ODA16 and the intraflagellar transport protein IFT46. J. Biol. Chem. 292, 7462–7473 (2017).
Hou, Y., Witman, G. B. & Marshall, W. The N-terminus of IFT46 mediates intraflagellar transport of outer arm dynein and its cargo-adaptor ODA16. Mol. Biol. Cell 28, 2420–2433 (2017).
Hunter, E. L. et al. The IDA3 adapter, required for intraflagellar transport of I1 dynein, is regulated by ciliary length. Mol. Biol. Cell 29, 886–896 (2018).
Ye, F. et al. Single molecule imaging reveals a major role for diffusion in the exploration of ciliary space by signaling receptors. eLife 2, e00654 (2013).
Monis, W. J., Faundez, V. & Pazour, G. J. BLOC-1 is required for selective membrane protein trafficking from endosomes to primary cilia. J. Cell Biol. 216, 2131–2150 (2017).
Morthorst, S. K., Christensen, S. T. & Pedersen, L. B. Regulation of ciliary membrane protein trafficking and signalling by kinesin motor proteins. FEBS J. 285, 4535–4564 (2018).
Blacque, O. E., Scheidel, N. & Kuhns, S. Rab GTPases in cilium formation and function. Small GTPases 9, 76–94 (2018).
Brown, J. M., Cochran, D. A., Craige, B., Kubo, T. & Witman, G. B. Assembly of IFT trains at the ciliary base depends on IFT74. Curr. Biol. 25, 1583–1593 (2015).
Deane, J. A., Cole, D. G., Seeley, E. S., Diener, D. R. & Rosenbaum, J. L. Localization of intraflagellar transport protein IFT52 identifies basal body transitional fibers as the docking site for IFT particles. Curr. Biol. 11, 1586–1590 (2001).
Wingfield, J. L. et al. IFT trains in different stages of assembly queue at the ciliary base for consecutive release into the cilium. eLife 6, e26609 (2017).
Huang, K., Diener, D. R., Mitchell, A. & Rosenbaum, J. L. Function and dynamics of PKD2 in Chlamydomonas reinhardtii flagella. J. Cell Biol. 179, 501–514 (2007).
Brear, A. G., Yoon, J., Wojtyniak, M. & Sengupta, P. Diverse cell type-specific mechanisms localize G protein-coupled receptors to Caenorhabditis elegans sensory cilia. Genetics 197, 667–684 (2014).
Bae, Y. K. et al. General and cell-type specific mechanisms target TRPP2/PKD-2 to cilia. Development 133, 3859–3870 (2006).
Dwyer, N. D., Adler, C. E., Crump, J. G., L’Etoile, N. D. & Bargmann, C. I. Polarized dendritic transport and the AP-1 mu1 clathrin adaptor UNC-101 localize odorant receptors to olfactory cilia. Neuron 31, 277–287 (2001).
Eguether, T., Cordelieres, F. P. & Pazour, G. J. Intraflagellar transport is deeply integrated in hedgehog signaling. Mol. Biol. Cell 29, 1178–1189 (2018).
Liu, P. & Lechtreck, K. F. The Bardet–Biedl syndrome protein complex is an adapter expanding the cargo range of intraflagellar transport trains for ciliary export. Proc. Natl Acad. Sci. USA 115, E934–E943 (2018).
Badgandi, H. B., Hwang, S., Shimada, I. S., Loriot, E. & Mukhopadhyay, S. Tubby family proteins are adapters for ciliary trafficking of integral membrane proteins. J. Cell Biol. 216, 743–760 (2017).
Fu, W., Wang, L., Kim, S., Li, J. & Dynlacht, B. D. Role for the IFT-A complex in selective transport to the primary cilium. Cell Rep. 17, 1505–1517 (2016).
Hirano, T., Katoh, Y. & Nakayama, K. Intraflagellar transport-A complex mediates ciliary entry and retrograde trafficking of ciliary G protein-coupled receptors. Mol. Biol. Cell 28, 429–439 (2017).
Mukhopadhyay, S. et al. TULP3 bridges the IFT-A complex and membrane phosphoinositides to promote trafficking of G protein-coupled receptors into primary cilia. Genes Dev. 24, 2180–2193 (2010).
Picariello, T. et al. A global analysis of IFT-A function reveals specialization for transport of membrane-associated proteins into cilia. J. Cell Sci. 132, jcs.220749 (2019).
Sun, X. et al. Tubby is required for trafficking G protein-coupled receptors to neuronal cilia. Cilia 1, 21 (2012).
Oswald, F., Prevo, B., Acar, S. & Peterman, E. J. G. Interplay between ciliary ultrastructure and IFT-train dynamics revealed by single-molecule super-resolution imaging. Cell Rep. 25, 224–235 (2018).
Yang, T., Tran, M. N. T., Chong, W. M., Huang, C.-E. & Liao, J.-C. Single-particle tracking localization microscopy reveals nonaxonemal dynamics of intraflagellar transport proteins at the base of mammalian primary cilia. Mol. Biol. Cell 30, 828–837 (2019).
Kanie, T. et al. The CEP19-RABL2 GTPase complex binds IFT-B to initiate intraflagellar transport at the ciliary base. Dev. Cell 42, 22–36 (2017).
Nishijima, Y. et al. RABL2 interacts with the intraflagellar transport-B complex and CEP19 and participates in ciliary assembly. Mol. Biol. Cell 28, 1652–1666 (2017).
Park, J. et al. dTULP, the Drosophila melanogaster homolog of Tubby, regulates transient receptor potential channel localization in cilia. PLOS Genet. 9, e1003814 (2013).
Park, J. et al. Ciliary phosphoinositide regulates ciliary protein trafficking in Drosophila. Cell Rep. 13, 2808–2816 (2015).
Ismail, S. A. et al. Arl2-GTP and Arl3-GTP regulate a GDI-like transport system for farnesylated cargo. Nat. Chem. Biol. 7, 942–949 (2011).
Ismail, S. A. et al. Structural basis for Arl3-specific release of myristoylated ciliary cargo from UNC119. EMBO J. 31, 4085–4094 (2012).
Wright, K. J. et al. An ARL3-UNC119-RP2 GTPase cycle targets myristoylated NPHP3 to the primary cilium. Genes Dev. 25, 2347–2360 (2011).
Zhang, H. et al. UNC119 is required for G protein trafficking in sensory neurons. Nat. Neurosci. 14, 874–880 (2011). Ismail et al. (2011), Ismail et al. (2012), Wright et al. (2011) and Zhang, H. et al. ( 2011) describe the myristoyl shuttle UNC119 and the farnesyl shuttle PDE6δ and the release of their cargoes by ARL3–GTP inside cilia as a cogent mechanism of ciliary import of lipidated proteins.
Jensen, V. L. & Leroux, M. R. Gates for soluble and membrane proteins, and two trafficking systems (IFT and LIFT), establish a dynamic ciliary signaling compartment. Curr. Opin. Cell Biol. 47, 83–91 (2017).
Stephen, L. A. & Ismail, S. Shuttling and sorting lipid-modified cargo into the cilia. Biochem. Soc. Trans. 44, 1273–1280 (2016).
Evans, R. J. et al. The retinitis pigmentosa protein RP2 links pericentriolar vesicle transport between the Golgi and the primary cilium. Hum. Mol. Genet. 19, 1358–1367 (2010).
Blacque, O. E. et al. Functional genomics of the cilium, a sensory organelle. Curr. Biol. 15, 935–941 (2005).
Stephan, A., Vaughan, S., Shaw, M. K., Gull, K. & McKean, P. G. An essential quality control mechanism at the eukaryotic Basal body prior to intraflagellar transport. Traffic 8, 1323–1330 (2007).
Gotthardt, K. et al. A G-protein activation cascade from Arl13B to Arl3 and implications for ciliary targeting of lipidated proteins. eLife 4, e11859 (2015).
Ivanova, A. A. et al. Biochemical characterization of purified mammalian ARL13B protein indicates that it is an atypical GTPase and ARL3 guanine nucleotide exchange factor (GEF). J. Biol. Chem. 292, 11091–11108 (2017).
Zhang, Q. et al. GTP-binding of ARL-3 is activated by ARL-13 as a GEF and stabilized by UNC-119. Sci. Rep. 6, 24534 (2016).
Fansa, E. K., Kösling, S. K., Zent, E., Wittinghofer, A. & Ismail, S. PDE6δ-mediated sorting of INPP5E into the cilium is determined by cargo-carrier affinity. Nat. Commun. 7, 11366 (2016).
Hurd, T. et al. The retinitis pigmentosa protein RP2 interacts with polycystin 2 and regulates cilia-mediated vertebrate development. Hum. Mol. Genet. 19, 4330–4344 (2010).
Humbert, M. C. et al. ARL13B, PDE6D, and CEP164 form a functional network for INPP5E ciliary targeting. Proc. Natl Acad. Sci. USA 109, 19691–19696 (2012).
Revenkova, E., Liu, Q., Gusella, G. L. & Iomini, C. The Joubert syndrome protein ARL13B binds tubulin to maintain uniform distribution of proteins along the ciliary membrane. J. Cell Sci. 131, jcs212324 (2018).
Nozaki, S. et al. Regulation of ciliary retrograde protein trafficking by the Joubert syndrome proteins ARL13B and INPP5E. J. Cell Sci. 130, 563–576 (2017).
Liem, K. F. et al. The IFT-A complex regulates Shh signaling through cilia structure and membrane protein trafficking. J. Cell Biol. 197, 789–800 (2012).
Roy, K. et al. Palmitoylation of the ciliary GTPase ARL13b is necessary for its stability and its role in cilia formation. J. Biol. Chem. 292, 17703–17717 (2017).
Cevik, S. et al. Joubert syndrome Arl13b functions at ciliary membranes and stabilizes protein transport in Caenorhabditis elegans. J. Cell Biol. 188, 953–969 (2010).
Li, Y., Wei, Q., Zhang, Y., Ling, K. & Hu, J. The small GTPases ARL-13 and ARL-3 coordinate intraflagellar transport and ciliogenesis. J. Cell Biol. 189, 1039–1051 (2010).
Mick, D. U. et al. Proteomics of primary cilia by proximity labeling. Dev. Cell 35, 497–512 (2015).
Viau, A. et al. Cilia-localized LKB1 regulates chemokine signaling, macrophage recruitment, and tissue homeostasis in the kidney. EMBO J. 37, e98615 (2018).
Emmer, B. T. et al. Identification of a palmitoyl acyltransferase required for protein sorting to the flagellar membrane. J. Cell Sci. 122, 867–874 (2009).
Follit, J. A., Li, L., Vucica, Y. & Pazour, G. J. The cytoplasmic tail of fibrocystin contains a ciliary targeting sequence. J. Cell Biol. 188, 21–28 (2010).
Goodwin, J. S. et al. Depalmitoylated Ras traffics to and from the Golgi complex via a nonvesicular pathway. J. Cell Biol. 170, 261–272 (2005).
Rocks, O. et al. An acylation cycle regulates localization and activity of palmitoylated Ras isoforms. Science 307, 1746–1752 (2005).
Huang, K., Diener, D. R. & Rosenbaum, J. L. The ubiquitin conjugation system is involved in the disassembly of cilia and flagella. J. Cell Biol. 186, 601–613 (2009).
Green, J. A. et al. Recruitment of β-arrestin into neuronal cilia modulates somatostatin receptor subtype 3 ciliary localization. Mol. Cell. Biol. 36, 223–235 (2015).
Pal, K. et al. Smoothened determines β-arrestin-mediated removal of the G protein-coupled receptor Gpr161 from the primary cilium. J. Cell Biol. 212, 861–875 (2016).
Datta, P. et al. Accumulation of non-outer segment proteins in the outer segment underlies photoreceptor degeneration in Bardet-Biedl syndrome. Proc. Natl Acad. Sci. USA 112, E4400–E4409 (2015).
Lechtreck, K. F. et al. The Chlamydomonas reinhardtii BBSome is an IFT cargo required for export of specific signaling proteins from flagella. J. Cell Biol. 187, 1117–1132 (2009).
Lechtreck, K. F. et al. Cycling of the signaling protein phospholipase D through cilia requires the BBSome only for the export phase. J. Cell Biol. 201, 249–261 (2013).
Eguether, T. et al. IFT27 links the BBSome to IFT for maintenance of the ciliary signaling compartment. Dev. Cell 31, 279–290 (2014).
Liew, G. M. et al. The intraflagellar transport protein IFT27 promotes BBSome exit from cilia through the GTPase ARL6/BBS3. Dev. Cell 31, 265–278 (2014).
Jin, H. et al. The conserved Bardet-Biedl syndrome proteins assemble a coat that traffics membrane proteins to cilia. Cell 141, 1208–1219 (2010).
Klink, B. U. et al. A recombinant BBSome core complex and how it interacts with ciliary cargo. eLife 6, e27434 (2017).
Baker, S. A. et al. The outer segment serves as a default destination for the trafficking of membrane proteins in photoreceptors. J. Cell Biol. 183, 485–498 (2008).
Ocbina, P. J. R. & Anderson, K. V. Intraflagellar transport, cilia, and mammalian Hedgehog signaling: analysis in mouse embryonic fibroblasts. Dev. Dyn. 237, 2030–2038 (2008).
Zhang, Q. et al. Bardet-Biedl syndrome 3 (Bbs3) knockout mouse model reveals common BBS-associated phenotypes and Bbs3 unique phenotypes. Proc. Natl Acad. Sci. USA 108, 20678–20683 (2011).
Trimble, W. S. & Grinstein, S. Barriers to the free diffusion of proteins and lipids in the plasma membrane. J. Cell Biol. 208, 259–271 (2015).
Schmidt, H. B. & Görlich, D. Transport selectivity of nuclear pores, phase separation, and membraneless organelles. Trends Biochem. Sci. 41, 46–61 (2016).
Papermaster, D. S., Schneider, B. G. & Besharse, J. C. Vesicular transport of newly synthesized opsin from the Golgi apparatus toward the rod outer segment. Ultrastructural immunocytochemical and autoradiographic evidence in Xenopus retinas. Invest. Ophthalmol. Vis. Sci. 26, 1386–1404 (1985).
Pedersen, L. B., Mogensen, J. B. & Christensen, S. T. Endocytic control of cellular signaling at the primary cilium. Trends Biochem. Sci. 41, 784–797 (2016).
Benmerah, A. The ciliary pocket. Curr. Opin. Cell Biol. 25, 78–84 (2013).
Langousis, G. et al. Loss of the BBSome perturbs endocytic trafficking and disrupts virulence of Trypanosoma brucei. Proc. Natl Acad. Sci. USA 113, 632–637 (2016).
Lobingier, B. T. & von Zastrow, M. When trafficking and signaling mix: how subcellular location shapes G protein-coupled receptor activation of heterotrimeric G proteins. Traffic 20, 130–136 (2019).
Pan, B. T., Teng, K., Wu, C., Adam, M. & Johnstone, R. M. Electron microscopic evidence for externalization of the transferrin receptor in vesicular form in sheep reticulocytes. J. Cell Biol. 101, 942–948 (1985).
Von Bartheld, C. S. & Altick, A. L. Multivesicular bodies in neurons: distribution, protein content, and trafficking functions. Prog. Neurobiol. 93, 313–340 (2011).
Besharse, J. C., Hollyfield, J. G. & Rayborn, M. E. Turnover of rod photoreceptor outer segments. II. Membrane addition and loss in relationship to light. J. Cell Biol. 75, 507–527 (1977).
Young, R. W. & Bok, D. Participation of the retinal pigment epithelium in the rod outer segment renewal process. J. Cell Biol. 42, 392–403 (1969).
Salinas, R. Y. et al. Photoreceptor discs form through peripherin-dependent suppression of ciliary ectosome release. J. Cell Biol. 216, 1489–1499 (2017). This paper provides an in vivo demonstration of a role for ciliary ectocytosis in disposing of excess membranes. Ectocytosis must hence be suppressed for the formation of fresh discs in photoreceptor outer segments.
Cao, M. et al. Uni-directional ciliary membrane protein trafficking by a cytoplasmic retrograde IFT motor and ciliary ectosome shedding. eLife 4, e05242 (2015). This paper uncovers signal-dependent ectocytosis as a means for ciliary exit of activated adhesion molecules in C. reinhardtii ; see also Nager et al. (2017).
Loktev, A. V. & Jackson, P. K. Neuropeptide Y family receptors traffic via the Bardet-Biedl syndrome pathway to signal in neuronal primary cilia. Cell Rep. 5, 1316–1329 (2013).
Marion, S., Oakley, R. H., Kim, K.-M., Caron, M. G. & Barak, L. S. A beta-arrestin binding determinant common to the second intracellular loops of rhodopsin family G protein-coupled receptors. J. Biol. Chem. 281, 2932–2938 (2006).
van Dam, T. J. P. et al. Evolution of modular intraflagellar transport from a coatomer-like progenitor. Proc. Natl Acad. Sci. USA 110, 6943–6948 (2013).
Shida, T., Cueva, J. G., Xu, Z., Goodman, M. B. & Nachury, M. V. The major alpha-tubulin K40 acetyltransferase alphaTAT1 promotes rapid ciliogenesis and efficient mechanosensation. Proc. Natl Acad. Sci. USA 107, 21517–21522 (2010).
Dentler, W. A. Role for the membrane in regulating Chlamydomonas flagellar length. PLOS ONE 8, e53366 (2013).
Rajagopalan, V., Subramanian, A., Wilkes, D. E., Pennock, D. G. & Asai, D. J. Dynein-2 affects the regulation of ciliary length but is not required for ciliogenesis in Tetrahymena thermophila. Mol. Biol. Cell 20, 708–720 (2009).
Hao, L., Efimenko, E., Swoboda, P. & Scholey, J. M. The retrograde IFT machinery of C. elegans cilia: two IFT dynein complexes? PLOS ONE 6, e20995 (2011).
Ocbina, P. J. R., Eggenschwiler, J. T., Moskowitz, I. & Anderson, K. V. Complex interactions between genes controlling trafficking in primary cilia. Nat. Genet. 43, 547–553 (2011).
Das, R. M. & Storey, K. G. Apical abscission alters cell polarity and dismantles the primary cilium during neurogenesis. Science 343, 200–204 (2014).
Mirvis, M., Siemers, K. A., Nelson, W. J. & Stearns, T. Heterogeneous dynamics and mechanisms of primary cilium disassembly in mammalian cells. Preprint at bioRxiv https://www.biorxiv.org/content/10.1101/433144v2 (2018).
Niel, Gvan, D’Angelo, G. & Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 19, 213–228 (2018).
Wood, C. R. & Rosenbaum, J. L. Ciliary ectosomes: transmissions from the cell’s antenna. Trends Cell Biol. 25, 276–285 (2015).
Wood, C. R., Huang, K., Diener, D. R. & Rosenbaum, J. L. The cilium secretes bioactive ectosomes. Curr. Biol. 23, 906–911 (2013).
Szempruch, A. J. et al. Extracellular vesicles from Trypanosoma brucei mediate virulence factor transfer and cause host anemia. Cell 164, 246–257 (2016).
Wang, J. et al. C. elegans ciliated sensory neurons release extracellular vesicles that function in animal communication. Curr. Biol. 24, 519–525 (2014).
Hurley, J. H. ESCRTs are everywhere. EMBO J. 34, 2398–2407 (2015).
Diener, D. R., Lupetti, P. & Rosenbaum, J. L. Proteomic analysis of isolated ciliary transition zones reveals the presence of ESCRT proteins. Curr. Biol. 25, 379–384 (2015).
Mierzwa, B. & Gerlich, D. W. Cytokinetic abscission: molecular mechanisms and temporal control. Dev. Cell 31, 525–538 (2014).
Chiaruttini, N. et al. Relaxation of loaded ESCRT-III spiral springs drives membrane deformation. Cell 163, 866–879 (2015).
Wollert, T., Wunder, C., Lippincott-Schwartz, J. & Hurley, J. H. Membrane scission by the ESCRT-III complex. Nature 458, 172–177 (2009).
Liu, A. P. & Fletcher, D. A. Actin polymerization serves as a membrane domain switch in model lipid bilayers. Biophys. J. 91, 4064–4070 (2006).
Römer, W. et al. Actin dynamics drive membrane reorganization and scission in clathrin-independent endocytosis. Cell 140, 540–553 (2010).
Allain, J.-M., Storm, C., Roux, A., Amar, M. B. & Joanny, J.-F. Fission of a multiphase membrane tube. Phys. Rev. Lett. 93, 158104 (2004).
Ma, M., Gallagher, A.-R. & Somlo, S. Ciliary mechanisms of cyst formation in polycystic kidney disease. Cold Spring Harb. Perspect. Biol. 9, a028209 (2017).
Pazour, G. J. et al. Polycystin-2 localizes to kidney cilia and the ciliary level is elevated in orpk mice with polycystic kidney disease. Curr. Biol. 12, R378–R380 (2002).
Yoder, B. K., Hou, X. & Guay-Woodford, L. M. The polycystic kidney disease proteins, polycystin-1, polycystin-2, polaris, and cystin, are co-localized in renal cilia. J. Am. Soc. Nephrol. 13, 2508–2516 (2002).
Kleene, S. J. & Kleene, N. K. The native TRPP2-dependent channel of murine renal primary cilia. Am. J. Physiol. Renal Physiol. 312, F96–F108 (2017).
Liu, X. et al. Polycystin-2 is an essential ion channel subunit in the primary cilium of the renal collecting duct epithelium. eLife 7, e33183 (2018).
Nauli, S. M. et al. Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells. Nat. Genet. 33, 129–137 (2003).
Delling, M. et al. Primary cilia are not calcium-responsive mechanosensors. Nature 531, 656–660 (2016).
Pazour, G. J. et al. Chlamydomonas IFT88 and its mouse homologue, polycystic kidney disease gene Tg737, are required for assembly of cilia and flagella. J. Cell Biol. 151, 709–718 (2000).
Braun, D. A. & Hildebrandt, F. Ciliopathies. Cold Spring Harb. Perspect. Biol. 9, a028191 (2017).
Ma, M., Tian, X., Igarashi, P., Pazour, G. J. & Somlo, S. Loss of cilia suppresses cyst growth in genetic models of autosomal dominant polycystic kidney disease. Nat. Genet. 45, 1004–1012 (2013).
Riobo, N. A. in The Smoothened Receptor in Cancer and Regenerative Medicine (ed. Ruat, M.) 13–42 (Springer, 2015).
Liu, Y. C. et al. The PPFIA1-PP2A protein complex promotes trafficking of Kif7 to the ciliary tip and Hedgehog signaling. Sci. Signal. 7, ra117 (2014).
Cheung, H. O.-L. et al. The kinesin protein Kif7 is a critical regulator of Gli transcription factors in mammalian hedgehog signaling. Sci. Signal. 2, ra29 (2009).
Endoh-Yamagami, S. et al. The mammalian Cos2 homolog Kif7 plays an essential role in modulating Hh signal transduction during development. Curr. Biol. 19, 1320–1326 (2009).
He, M. et al. The kinesin-4 protein Kif7 regulates mammalian Hedgehog signalling by organizing the cilium tip compartment. Nat. Cell Biol. 16, 663–672 (2014).
Bidet, M. et al. The hedgehog receptor Patched is involved in cholesterol transport. PLOS ONE 6, e23834 (2011).
Fiorini, L. et al. Natural paniceins from mediterranean sponge inhibit the multidrug resistance activity of Patched and increase chemotherapy efficiency on melanoma cells. Oncotarget 6, 22282–22297 (2015).
Zhang, Y. et al. Structural basis for cholesterol transport-like activity of the Hedgehog receptor Patched. Cell 175, 1352–1364 (2018).
Gong, X. et al. Structural basis for the recognition of Sonic Hedgehog by human Patched1. Science 361, eaas8935 (2018).
Qi, X., Schmiege, P., Coutavas, E., Wang, J. & Li, X. Structures of human Patched and its complex with native palmitoylated sonic hedgehog. Nature 560, 128–132 (2018).
Qi, X., Schmiege, P., Coutavas, E. & Li, X. Two Patched molecules engage distinct sites on Hedgehog yielding a signaling-competent complex. Science 362, eaas8843 (2018). Zhang et al. (2018), Gong et al. (2018), Qi et al. ( Nature , 2018) and Qi et al. (Science , 2018) present structures of Hedgehog receptor Patched 1 that uncover a channel in Patched 1 through which cholesterol may be transported from the inner leaflet to the extracellular domain. Zhang et al. (2018) also provide direct evidence that P atched 1 transports cholesterol from the inner to the outer leaflet of the membrane.
Huang, P. et al. Structural basis of Smoothened activation in Hedgehog signaling. Cell 174, 312–324 (2018). The structure of activated Smoothened, as presented in this paper, reveals a channel through which cholesterol may travel from the inner leaflet to the cysteine-rich extracellular domain, thereby promoting receptor activation.
Cole, D. G. et al. Chlamydomonas kinesin-II-dependent intraflagellar transport (IFT): IFT particles contain proteins required for ciliary assembly in Caenorhabditis elegans sensory neurons. J. Cell Biol. 141, 993–1008 (1998).
Nachury, M. V. et al. A core complex of BBS proteins cooperates with the GTPase Rab8 to promote ciliary membrane biogenesis. Cell 129, 1201–1213 (2007).
Wingfield, J. L., Lechtreck, K.-F. & Lorentzen, E. Trafficking of ciliary membrane proteins by the intraflagellar transport/BBSome machinery. Essays Biochem. 62, 753–763 (2018).
Funabashi, T., Katoh, Y., Okazaki, M., Sugawa, M. & Nakayama, K. Interaction of heterotrimeric kinesin-II with IFT-B-connecting tetramer is crucial for ciliogenesis. J. Cell Biol. 217, 2867–2876 (2018).
Jordan, M. A., Diener, D. R., Stepanek, L. & Pigino, G. The cryo-EM structure of intraflagellar transport trains reveals how dynein is inactivated to ensure unidirectional anterograde movement in cilia. Nat. Cell Biol. 20, 1250–1255 (2018).
Chien, A. et al. Dynamics of the IFT machinery at the ciliary tip. eLife 6, e28606 (2017).
Mijalkovic, J., van Krugten, J., Oswald, F., Acar, S. & Peterman, E. J. G. Single-molecule turnarounds of intraflagellar transport at the C. elegans ciliary tip. Cell Rep. 25, 1701–1707 (2018).
Stepanek, L. & Pigino, G. Microtubule doublets are double-track railways for intraflagellar transport trains. Science 352, 721–724 (2016).
Chaya, T., Omori, Y., Kuwahara, R. & Furukawa, T. ICK is essential for cell type-specific ciliogenesis and the regulation of ciliary transport. EMBO J. 33, 1227–1242 (2014).
Berman, S. A., Wilson, N. F., Haas, N. A., Lefebvre, P. A. A novel MAP kinase regulates flagellar length in Chlamydomonas. Curr. Biol. 13, 1145–1149 (2003).
Burghoorn, J. et al. Mutation of the MAP kinase DYF-5 affects docking and undocking of kinesin-2 motors and reduces their speed in the cilia of Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 104, 7157 (2007).
Bengs, F., Scholz, A., Kuhn, D. & Wiese, M. LmxMPK9, a mitogen-activated protein kinase homologue affects flagellar length in Leishmania mexicana: MAP kinase and flagellar length. Mol. Microbiol. 55, 1606–1615 (2005).
Acknowledgements
The authors thank B. Snell, G. Witman, J. Reiter, M. Delling, D. Breslow, I. Ojeda Naharros and S. Shinde for comments on the manuscript and A. Nager for help with drafting the manuscript. They apologize to their colleagues whose publications they could not cite owing to length restrictions. Research in the Nachury laboratory is supported by NIGMS grant GM089933, a Stein Innovation Award from Research to Prevent Blindness and, in part, by NEI core grant EY002162 and by an RPB Unrestricted Grant. Research in the Mick laboratory is supported by the Deutsche Forschungsgemeinschaft (DFG) SFB894/TPA-22.
Reviewer information
Nature Reviews Molecular Cell Biology thanks L. B. Pedersen and other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- G protein-coupled receptors
-
(GPCRs). Membrane proteins with seven transmembrane domains that sense various external signals (such as drugs and odorants) and relay them into the cell through heterotrimeric G proteins.
- Anorexigenic
-
Refers to an effect related to the loss of appetite (anorexia), thus resulting in lower food consumption and weight loss.
- Photoreceptors
-
Sensory neurons that detect light using rhodopsin localized inside a specialized cilium.
- Rhodopsin
-
A photosensory G protein-coupled receptor (GPCR) that transduces light using a tightly bound molecule of retinal.
- Bardet–Biedl syndrome
-
(BBS). A ciliopathy (with mutations in 21 different genes identified to date) characterized by retinal degeneration, obesity, kidney abnormalities and polydactyly.
- Phosphoinositides
-
Hallmark lipids that mark the cytoplasmic leaflets of cellular membranes.
- Basal body
-
A specialized mother centriole that is a template for the ciliary axoneme.
- Axoneme
-
The microtubule core of cilia composed of nine microtubule doublets.
- Transition fibres
-
Identical to the distal appendages of mother centrioles in unciliated cells, transition fibres physically connect the basal body to the membrane in ciliated cells.
- Y-links
-
Y-shaped electron-dense structures that connect the ciliary axoneme (bottom of the Y) to the membrane (top of the Y) at the transition zone.
- Tight junctions
-
The junctions between adjacent epithelial cells that seal the epithelium and function as a barrier for lipid and integral membrane protein diffusion between the apical and basolateral membranes of cells.
- Heterotrimeric G protein α subunit
-
The GTPase subunit of heterotrimeric G proteins. It becomes loaded with GTP after encountering an active G protein-coupled receptor (GPCR) and modulates downstream activities, such as adenylyl cyclases. Gαs–GTP stimulates adenylyl cyclases, while Gαi–GTP inhibits them.
- GLI transcription factors
-
The GLI1 transcription activator was first identified in glioblastoma (a Hedgehog-driven tumour). The paralogues GLI2 and GLI3 are processed into transcription activators or repressors depending upon regulatory inputs from the Hedgehog pathway.
- Sphingolipids
-
Lipids formed by a backbone that consists of a sphingosine N-acylated by a fatty acid. Named after the Sphinx because of their once-enigmatic functions.
- Oxysterols
-
Products of cholesterol oxidation with hydroxyl, carbonyl or epoxide groups. Their best-established roles are in regulation of cholesterol metabolism.
- Condensed membrane microdomains
-
Tightly packed and viscous regions of the membrane that can form through clustering of lipid rafts.
- Liquid-ordered lipid phase
-
Biological membranes are two-dimensional liquids that can phase separate into liquid-ordered (for example, rafts) and liquid-disordered (for example, fluid mosaic) phases.
- Raft
-
A relatively ordered membrane domain formed by interactions between proteins and lipids. Rafts typically present as cholesterol and sphingolipidenriched membrane nanodomains <200 nm in size that are thought to facilitate interactions between signalling molecules.
- BBSome
-
A complex of eight Bardet–Biedl syndrome proteins that removes proteins from cilia.
- IFT trains
-
High-order oligomers of intraflagellar transport (IFT) complexes that transport cargo along the axoneme.
- Polycystic kidney and hepatic disease 1
-
(PKHD1). A large single-pass membrane protein of unknown function mutated in autosomal recessive polycystic kidney disease.
- Ciliary targeting signal
-
(CTS). In the strictest sense, a short stretch of amino acids that is necessary and sufficient for targeting a protein to cilia. Some CTSs (for example, in somatostatin receptor 3) are sufficient but not necessary for ciliary targeting.
- Somatostatin receptor 3
-
(SSTR3). A ciliary G protein-coupled receptor (GPCR) expressed in hippocampal neurons and required for novelty recognition. It couples to Gαi.
- Melanocortin concentrating hormone receptor 1
-
(MCHR1). A ciliary G protein-coupled receptor (GPCR) that has a modest role in body weight homeostasis.
- ARL3
-
A small GTPase of the ARF-like family, most members of which regulate vesicular trafficking.
- β-Arrestin 2
-
Recognizes activated G protein-coupled receptors (GPCRs), blocks GPCR-to-Gα communication and promotes GPCR endocytosis, thereby typically driving termination of GPCR signalling.
- Axon initial segment
-
In neurons, the first few micrometres of the axon. It is an area rich in actin that functionally separates the axonal plasma membrane from the soma plasma membrane.
- Karyopherins
-
Nuclear transport receptors that ferry proteins across the nuclear pore complex.
- Ectosomes
-
Extracellular vesicles that directly bud from the limiting membrane of the cell. Also termed microvesicles.
- Neuropeptide Y receptor type 2
-
(NPY2R). A Gαs-coupled G protein-coupled receptor (GPCR) that regulates appetite under stress conditions.
- Exosomes
-
Extracellular vesicles that originate as vesicles inside the lumen of the multivesicular body and become extracellular upon fusion of the multivesicular body with the plasma membrane.
- Endosomal sorting complex required for transport
-
(ESCRT). A cascade of protein complexes (0, I, II and III in order of action) discovered initially in yeast as factors involved in budding transmembrane proteins from the endosomal membrane into intraluminal vesicles. ESCRTs have since been demonstrated to be necessary for many cellular processes involving a bud-like topology.
- Line tension
-
The 2D version of surface tension, which reflects the energy minimization of a system in which an energetically costly discontinuity exists between two neighbouring phases.
Rights and permissions
About this article
Cite this article
Nachury, M.V., Mick, D.U. Establishing and regulating the composition of cilia for signal transduction. Nat Rev Mol Cell Biol 20, 389–405 (2019). https://doi.org/10.1038/s41580-019-0116-4
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41580-019-0116-4
This article is cited by
-
Landscapes of gut bacterial and fecal metabolic signatures and their relationship in severe preeclampsia
Journal of Translational Medicine (2024)
-
Regulation of ciliary homeostasis by intraflagellar transport-independent kinesins
Cell Death & Disease (2024)
-
Emerging mechanistic understanding of cilia function in cellular signalling
Nature Reviews Molecular Cell Biology (2024)
-
The exocyst complex and intracellular vesicles mediate soluble protein trafficking to the primary cilium
Communications Biology (2024)
-
XIAP-mediated degradation of IFT88 disrupts HSC cilia to stimulate HSC activation and liver fibrosis
EMBO Reports (2024)