Abstract
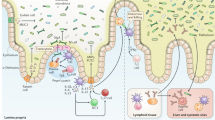
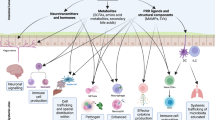
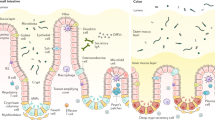
The mucosal lining of the gut has co-evolved with a diverse microbiota over millions of years, leading to the development of specialized mechanisms to actively limit the invasion of pathogens. However, some enteric microorganisms have adapted against these measures, developing ways to hijack or overcome epithelial micro-integrity mechanisms. This breach of the gut barrier not only enables the leakage of host factors out of circulation but can also initiate a cascade of detrimental systemic events as microbiota, pathogens and their affiliated secretions passively leak into extra-intestinal sites. Under normal circumstances, gut damage is rapidly repaired by intestinal stem cells. However, with substantial and deep perturbation to the gut lining and the systemic dissemination of gut contents, we now know that some enteric infections can cause the impairment of host regenerative processes. Although these local and systemic aspects of enteric disease are often studied in isolation, they heavily impact one another. In this Review, by examining the journey of enteric infections from initial establishment to systemic sequelae and how, or if, the host can successfully repair damage, we will tie together these complex interactions to provide a holistic overview of the impact of enteric infections at and beyond the epithelial barrier.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Karmakar, S., Deng, L., He, X. C. & Li, L. Intestinal epithelial regeneration: active versus reserve stem cells and plasticity mechanisms. Am. J. Physiol. Gastrointest. Liver Physiol. 318, G796–G802 (2020).
Barker, N. Adult intestinal stem cells: critical drivers of epithelial homeostasis and regeneration. Nat. Rev. Mol. Cell Biol. 15, 19–33 (2014).
Peck, B. C. E., Shanahan, M. T., Singh, A. P. & Sethupathy, P. Gut microbial influences on the mammalian intestinal stem cell niche. Stem Cell Int. 2017, 5604727 (2017).
Leslie, J. L. et al. Persistence and toxin production by Clostridium difficile within human intestinal organoids result in disruption of epithelial paracellular barrier function. Infect. Immun. 83, 138–145 (2015).
Bonfini, A., Liu, X. & Buchon, N. From pathogens to microbiota: how Drosophila intestinal stem cells react to gut microbes. Dev. Comp. Immunol. 64, 22–38 (2016).
Hoffmann, W. Regeneration of the gastric mucosa and its glands from stem cells. Curr. Med. Chem. 15, 3133–3144 (2008).
Pitsouli, C., Apidianakis, Y. & Perrimon, N. Homeostasis in infected epithelia: stem cells take the lead. Cell Host Microbe 6, 301–307 (2009).
Backert, S., Boehm, M., Wessler, S. & Tegtmeyer, N. Transmigration route of Campylobacter jejuni across polarized intestinal epithelial cells: paracellular, transcellular or both? Cell Commun. Signal. 11, 72 (2013).
Buchon, N., Broderick, N. A., Chakrabarti, S. & Lemaitre, B. Invasive and indigenous microbiota impact intestinal stem cell activity through multiple pathways in Drosophila. Genes Dev. 23, 2333–2344 (2009).
McCormick, B. A., Stocker, B. A., Laux, D. C. & Cohen, P. S. Roles of motility, chemotaxis, and penetration through and growth in intestinal mucus in the ability of an avirulent strain of Salmonella Typhimurium to colonize the large intestine of streptomycin-treated mice. Infect. Immun. 56, 2209–2217 (1988).
Boyle, E. C., Brown, N. F. & Finlay, B. B. Salmonella enterica serovar Typhimurium effectors SopB, SopE, SopE2 and SipA disrupt tight junction structure and function. Cell Microbiol. 8, 1946–1957 (2006).
Prizont, R. & Reed, W. P. Differences in blood group B-specific mucinase activity between virulent and avirulent Shigella flexneri 2a strains. Microb. Pathog. 11, 129–135 (1991).
Silva, A. J., Pham, K. & Benitez, J. A. Haemagglutinin/protease expression and mucin gel penetration in El Tor biotype Vibrio cholerae. Microbiology 149, 1883–1891 (2003).
Hoy, B. et al. Helicobacter pylori HtrA is a new secreted virulence factor that cleaves E-cadherin to disrupt intercellular adhesion. EMBO Rep. 11, 798–804 (2010).
Mileto, S. J. et al. Clostridioides difficile infection damages colonic stem cells via TcdB, impairing epithelial repair and recovery from disease. Proc. Natl Acad. Sci. USA 117, 8064–8073 (2020). This is an important study demonstrating the direct interaction and impairment of colonic stem cells by a specific enteric pathogen.
Stanley, R. A., Ram, S. P., Wilkinson, R. K. & Roberton, A. M. Degradation of pig gastric and colonic mucins by bacteria isolated from the pig colon. Appl. Environ. Microbiol. 51, 1104–1119 (1986).
Lecuit, M. et al. A transgenic model for listeriosis: role of internalin in crossing the intestinal barrier. Science 292, 1722–1725 (2001).
Boehm, M. et al. Rapid paracellular transmigration of Campylobacter jejuni across polarized epithelial cells without affecting TER: role of proteolytic-active HtrA cleaving E-cadherin but not fibronectin. Gut Pathog. 4, 3 (2012).
Wadolkowski, E. A., Laux, D. C. & Cohen, P. S. Colonization of the streptomycin-treated mouse large intestine by a human fecal Escherichia coli strain: role of growth in mucus. Infect. Immun. 56, 1030–1035 (1988).
Hausmann, A. et al. Intestinal epithelial NAIP/NLRC4 restricts systemic dissemination of the adapted pathogen Salmonella Typhimurium due to site-specific bacterial PAMP expression. Mucosal Immunol. 13, 530–544 (2020).
Di Domenico, E. G., Cavallo, I., Pontone, M., Toma, L. & Ensoli, F. Biofilm producing Salmonella Typhi: chronic colonization and development of gallbladder cancer. Int. J. Mol. Sci. 18, 1887 (2017).
Johnson, R. et al. Comparison of Salmonella enterica Serovars Typhi and Typhimurium reveals typhoidal serovar-specific responses to bile. Infect. Immun. 86, e00490-17 (2018).
Zha, L., Garrett, S. & Sun, J. Salmonella Infection in chronic inflammation and gastrointestinal cancer. Diseases 7, 28 (2019).
Carter, G. P. et al. Defining the roles of TcdA and TcdB in localized gastrointestinal disease, systemic organ damage, and the host response during Clostridium difficile infections. mBio 6, e00551 (2015).
Mimuro, H. et al. Helicobacter pylori dampens gut epithelial self-renewal by inhibiting apoptosis, a bacterial strategy to enhance colonization of the stomach. Cell Host Microbe 2, 250–263 (2007).
Costa, A. M., Leite, M., Seruca, R. & Figueiredo, C. Adherens junctions as targets of microorganisms: a focus on Helicobacter pylori. FEBS Lett. 587, 259–265 (2013).
De Filippo, C. et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl Acad. Sci. USA 107, 14691–14696 (2010).
Jakobsson, H. E. et al. Short-term antibiotic treatment has differing long-term impacts on the human throat and gut microbiome. PLoS ONE 5, e9836 (2010).
Tourret, J. et al. Immunosuppressive treatment alters secretion of ileal antimicrobial peptides and gut microbiota, and favors subsequent colonization by uropathogenic Escherichia coli. Transplantation 101, 74–82 (2017).
Jones, T. A., Hernandez, D. Z., Wong, Z. C., Wandler, A. M. & Guillemin, K. The bacterial virulence factor CagA induces microbial dysbiosis that contributes to excessive epithelial cell proliferation in the Drosophila gut. PLoS Pathog. 13, e1006631 (2017).
Ferreyra, J. A. et al. Gut microbiota-produced succinate promotes C. difficile infection after antibiotic treatment or motility disturbance. Cell Host Microbe 16, 770–777 (2014).
Johansson, M. E. V., Sjövall, H. & Hansson, G. C. The gastrointestinal mucus system in health and disease. Nat. Rev. Gastroenterol. Hepatol. 10, 352–361 (2013). A thorough review encompassing the structure and function of mucous within the intestines during health and disease, particularly parasitic and bacterial diseases.
Wheeler, K. M. et al. Mucin glycans attenuate the virulence of Pseudomonas aeruginosa in infection. Nat. Microbiol. 4, 2146–2154 (2019).
Furter, M., Sellin, M. E., Hansson, G. C. & Hardt, W. D. Mucus architecture and near-surface swimming affect distinct Salmonella Typhimurium infection patterns along the murine intestinal tract. Cell Rep. 27, 2665–2678.e3 (2019).
Johansson, M. E. V., Larsson, J. M. H. & Hansson, G. C. The two mucus layers of colon are organized by the MUC2 mucin, whereas the outer layer is a legislator of host–microbial interactions. Proc. Natl Acad. Sci. USA 108, 4659–4665 (2011).
Johansson, M. E. V. et al. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc. Natl Acad. Sci. USA 105, 15064–15069 (2008).
Li, H. et al. The outer mucus layer hosts a distinct intestinal microbial niche. Nat. Commun. 6, 8292 (2015).
Bäckhed, F., Ley, R. E., Sonnenburg, J. L., Peterson, D. A. & Gordon, J. I. Host-bacterial mutualism in the human intestine. Science 307, 1915–1920 (2005).
Engevik, M. A. et al. Human Clostridium difficile infection: altered mucus production and composition. Am. J. Physiol. Gastrointest. Liver Physiol. 308, G510–G524 (2014).
Johansson, M. E. Fast renewal of the distal colonic mucus layers by the surface goblet cells as measured by in vivo labeling of mucin glycoproteins. PLoS ONE 7, e41009 (2012).
Johansson, M. E. V. et al. Normalization of host intestinal mucus layers requires long-term microbial colonization. Cell Host Microbe 18, 582–592 (2015).
Feng, Y. et al. Antibiotics induced intestinal tight junction barrier dysfunction is associated with microbiota dysbiosis, activated NLRP3 inflammasome and autophagy. PLoS ONE 14, e0218384 (2019).
Kim, S. et al. Mucin degrader Akkermansia muciniphila accelerates intestinal stem cell-mediated epithelial development. Gut Microbes 13, 1–20 (2021).
Wrzosek, L. et al. Bacteroides thetaiotaomicron and Faecalibacterium prausnitzii influence the production of mucus glycans and the development of goblet cells in the colonic epithelium of a gnotobiotic model rodent. BMC Biol. 11, 61 (2013).
Valeri, M. et al. Pathogenic E. coli exploits SslE mucinase activity to translocate through the mucosal barrier and get access to host cells. PLoS ONE 10, e0117486 (2015).
Luo, Q. et al. Enterotoxigenic Escherichia coli secretes a highly conserved mucin-degrading metalloprotease to effectively engage intestinal epithelial cells. Infect. Immun. 82, 509–521 (2014).
Wilson, K. H. & Perini, F. Role of competition for nutrients in suppression of Clostridium difficile by the colonic microflora. Infect. Immun. 56, 2610–2614 (1988).
Farquhar, M. G. & Palade, G. E. Junctional complexes in various epithelia. J. Cell Biol. 17, 375–412 (1963).
Drolia, R. & Bhunia, A. K. Crossing the intestinal barrier via Listeria adhesion protein and internalin A. Trends Microbiol. 27, 408–425 (2019).
Drolia, R., Tenguria, S., Durkes, A. C., Turner, J. R. & Bhunia, A. K. Listeria adhesion protein induces intestinal epithelial barrier dysfunction for bacterial translocation. Cell Host Microbe 23, 470–484.e7 (2018).
Fattinger, S. A. et al. Salmonella Typhimurium discreet-invasion of the murine gut absorptive epithelium. PLoS Pathog. 16, e1008503 (2020).
Meza-Segura, M. et al. SepA enhances Shigella invasion of epithelial cells by degrading alpha-1 antitrypsin and producing a neutrophil chemoattractant. mBio 12, e0283321 (2021).
Das, S. et al. Hemochromatosis drives acute lethal intestinal responses to hyperyersiniabactin-producing Yersinia pseudotuberculosis. Proc. Natl Acad. Sci. USA 119, e2110166119 (2022).
Guttman, J. A. & Finlay, B. B. Tight junctions as targets of infectious agents. Biochim. Biophys. Acta 1788, 832–841 (2009). An extensive review highlighting the profound effect pathogen-induced breakdown of tight junctions has on human health and disease.
Grosheva, I. et al. High-throughput screen identifies host and microbiota regulators of intestinal barrier function. Gastroenterology 159, 1807–1823 (2020).
Wine, E., Chan, V. L. & Sherman, P. M. Campylobacter jejuni mediated disruption of polarized epithelial monolayers is cell-type specific, time dependent, and correlates with bacterial invasion. Pediatr. Res. 64, 599–604 (2008).
Kazmierczak, B. I., Mostov, K. & Engel, J. N. Interaction of bacterial pathogens with polarized epithelium. Annu. Rev. Microbiol. 55, 407–435 (2001).
Sears, C. L. Molecular physiology and pathophysiology of tight junctions V. assault of the tight junction by enteric pathogens. Am. J. Physiol. Gastrointest. Liver Physiol. 279, G1129–G1134 (2000).
Hoy, B. et al. Distinct roles of secreted HtrA proteases from gram-negative pathogens in cleaving the junctional protein and tumor suppressor E-cadherin. J. Biol. Chem. 287, 10115–10120 (2012).
Wu, S., Lim, K. C., Huang, J., Saidi, R. F. & Sears, C. L. Bacteroides fragilis enterotoxin cleaves the zonula adherens protein, E-cadherin. Proc. Natl Acad. Sci. USA 95, 14979–14984 (1998).
Steck, N. et al. Enterococcus faecalis metalloprotease compromises epithelial barrier and contributes to intestinal inflammation. Gastroenterology 141, 959–971 (2011). A study delineating the ability of bacterial enzymes to compromise epithelial barrier integrity contributing to chronic intestinal inflammation.
Inoshima, I. et al. A Staphylococcus aureus pore-forming toxin subverts the activity of ADAM10 to cause lethal infection in mice. Nat. Med. 17, 1310–1314 (2011).
Viswanathan, V. K. et al. Comparative analysis of EspF from enteropathogenic and enterohemorrhagic Escherichia coli in alteration of epithelial barrier function. Infect. Immun. 72, 3218–3227 (2004).
Matsuzawa, T., Kuwae, A. & Abe, A. Enteropathogenic Escherichia coli type III effectors EspG and EspG2 alter epithelial paracellular permeability. Infect. Immun. 73, 6283–6289 (2005).
Dean, P. & Kenny, B. Intestinal barrier dysfunction by enteropathogenic Escherichia coli is mediated by two effector molecules and a bacterial surface protein. Mol. Microbiol. 54, 665–675 (2004).
Runswick, S., Mitchell, T., Davies, P., Robinson, C. & Garrod, D. R. Pollen proteolytic enzymes degrade tight junctions. Respirology 12, 834–842 (2007).
Saitoh, Y. et al. Tight junctions. Structural insight into tight junction disassembly by Clostridium perfringens enterotoxin. Science 347, 775–778 (2015).
Chen, M. L., Ge, Z., Fox, J. G. & Schauer, D. B. Disruption of tight junctions and induction of proinflammatory cytokine responses in colonic epithelial cells by Campylobacter jejuni. Infect. Immun. 74, 6581–6589 (2006).
Lobo de Sá, F. D. et al. Resveratrol prevents Campylobacter jejuni-induced leaky gut by restoring occludin and claudin-5 in the paracellular leak pathway. Front. Pharmacol. 12, 640572 (2021).
Simonovic, I., Arpin, M., Koutsouris, A., Falk-Krzesinski, H. J. & Hecht, G. Enteropathogenic Escherichia coli activates ezrin, which participates in disruption of tight junction barrier function. Infect. Immun. 69, 5679–5688 (2001).
Zhang, Q. et al. Enteropathogenic Escherichia coli changes distribution of occludin and ZO-1 in tight junction membrane microdomains in vivo. Microb. Pathog. 48, 28–34 (2010).
Köhler, H. et al. Salmonella enterica serovar Typhimurium regulates intercellular junction proteins and facilitates transepithelial neutrophil and bacterial passage. Am. J. Physiol. Gastrointest. Liver Physiol. 293, G178–G187 (2007).
Sakaguchi, T., Köhler, H., Gu, X., McCormick, B. A. & Reinecker, H. C. Shigella flexneri regulates tight junction-associated proteins in human intestinal epithelial cells. Cell Microbiol. 4, 367–381 (2002).
Vikström, E., Bui, L., Konradsson, P. & Magnusson, K. E. The junctional integrity of epithelial cells is modulated by Pseudomonas aeruginosa quorum sensing molecule through phosphorylation-dependent mechanisms. Exp. Cell Res. 315, 313–326 (2009).
Peralta-Ramírez, J. et al. EspF interacts with nucleation-promoting factors to recruit junctional proteins into pedestals for pedestal maturation and disruption of paracellular permeability. Infect. Immun. 76, 3854–3868 (2008).
Shifflett, D. E., Clayburgh, D. R., Koutsouris, A., Turner, J. R. & Hecht, G. A. Enteropathogenic E. coli disrupts tight junction barrier function and structure in vivo. Lab. Invest. 85, 1308–1324 (2005).
Fasano, A. et al. Zonula occludens toxin modulates tight junctions through protein kinase C-dependent actin reorganization, in vitro. J. Clin. Investig. 96, 710–720 (1995).
Maldonado-Contreras, A. et al. Shigella depends on SepA to destabilize the intestinal epithelial integrity via cofilin activation. Gut Microbes 8, 544–560 (2017).
Wang, F. et al. Interferon-gamma and tumor necrosis factor-alpha synergize to induce intestinal epithelial barrier dysfunction by up-regulating myosin light chain kinase expression. Am. J. Pathol. 166, 409–419 (2005).
Odenwald, M. A. & Turner, J. R. The intestinal epithelial barrier: a therapeutic target? Nat. Rev. Gastroenterol. Hepatol. 14, 9–21 (2017).
Wotzka, S. Y. et al. Escherichia coli limits Salmonella Typhimurium infections after diet shifts and fat-mediated microbiota perturbation in mice. Nat. Microbiol. 4, 2164–2174 (2019).
Sun, Y. et al. Infection-generated electric field in gut epithelium drives bidirectional migration of macrophages. PLoS Biol. 17, e3000044 (2019).
Fasciano, A. C. et al. Yersinia pseudotuberculosis YopE prevents uptake by M cells and instigates M cell extrusion in human ileal enteroid-derived monolayers. Gut Microbes 13, 1988390 (2021).
Zhang, K. et al. Minimal SPI1-T3SS effector requirement for Salmonella enterocyte invasion and intracellular proliferation in vivo. PLoS Pathog. 14, e1006925 (2018).
Crowley, S. M. et al. Intestinal restriction of Salmonella Typhimurium requires caspase-1 and caspase-11 epithelial intrinsic inflammasomes. PLoS Pathog. 16, e1008498 (2020).
Fehon, R. G., McClatchey, A. I. & Bretscher, A. Organizing the cell cortex: the role of ERM proteins. Nat. Rev. Mol. Cell Biol. 11, 276–287 (2010). A thorough review of the structure and function of polarity-determining proteins within the body.
Tapia, R., Kralicek, S. E. & Hecht, G. A. Enteropathogenic Escherichia coli (EPEC) recruitment of PAR polarity protein atypical PKCζ to pedestals and cell-cell contacts precedes disruption of tight junctions in intestinal epithelial cells. Int. J. Mol. Sci. 21, 527 (2020).
Rauch, I. et al. NAIP-NLRC4 inflammasomes coordinate intestinal epithelial cell expulsion with eicosanoid and IL-18 release via activation of caspase-1 and -8. Immunity 46, 649–659 (2017).
Aktories, K., Schwan, C. & Jank, T. Clostridium difficile toxin biology. Annu. Rev. Microbiol. 71, 281–307 (2017).
Robert, A. & Wiels, J. Shiga toxins as antitumor tools. Toxins 13, 690 (2021).
Navarro, M. A., McClane, B. A. & Uzal, F. A. Mechanisms of action and cell death associated with Clostridium perfringens toxins. Toxins 10, 212 (2018).
Chen, P. et al. Structural basis for CSPG4 as a receptor for TcdB and a therapeutic target in Clostridioides difficile infection. Nat. Commun. 12, 3748–3748 (2021).
Holly, M. K. et al. Salmonella enterica infection of murine and human enteroid-derived monolayers elicits differential activation of epithelium-intrinsic inflammasomes. Infect. Immun. 88, e00017-20 (2020).
Balk, R. A. Systemic inflammatory response syndrome (SIRS): where did it come from and is it still relevant today? Virulence 5, 20–26 (2014).
Ho, J. T. K., Chan, G. C. F. & Li, J. C. B. Systemic effects of gut microbiota and its relationship with disease and modulation. BMC Immunol. 16, 21 (2015). A review discussing the relationship between a balanced or dysbiotic gut microbiota and systemic health or disease.
Tao, L. et al. Frizzled proteins are colonic epithelial receptors for C. difficile toxin B. Nature 538, 350–355 (2016).
Dobson, G., Hickey, C. & Trinder, J. Clostridium difficile colitis causing toxic megacolon, severe sepsis and multiple organ dysfunction syndrome. Intensive Care Med. 29, 1030 (2003).
Steele, J. et al. Systemic dissemination of Clostridium difficile toxins A and B is associated with severe, fatal disease in animal models. J. Infect. Dis. 205, 384–391 (2012).
Yu, H. et al. Identification of toxemia in patients with Clostridium difficile infection. PLoS ONE 10, e0124235 (2015).
Sae-Khow, K. et al. Pathogen-associated molecules from gut translocation enhance severity of cecal ligation and puncture sepsis in iron-overload β-thalassemia mice. J. Inflamm. Res. 13, 719–735 (2020).
Thim-Uam, A. et al. Leaky-gut enhanced lupus progression in the Fc gamma receptor-IIb deficient and pristane-induced mouse models of lupus. Sci. Rep. 10, 777 (2020).
Li, J., Moturi, K. R., Wang, L., Zhang, K. & Yu, C. Gut derived-endotoxin contributes to inflammation in severe ischemic acute kidney injury. BMC Nephrol. 20, 16 (2019).
Karmali, M. A. et al. The association between idiopathic hemolytic uremic syndrome and infection by verotoxin-producing Escherichia coli. J. Infect. Dis. 151, 775–782 (1985).
Banatvala, N. et al. The United States National Prospective Hemolytic Uremic Syndrome Study: microbiologic, serologic, clinical, and epidemiologic findings. J. Infect. Dis. 183, 1063–1070 (2001).
Lill, J. K. et al. Tissue-resident macrophages mediate neutrophil recruitment and kidney injury in shiga toxin-induced hemolytic uremic syndrome. Kidney Int. 100, 349–363 (2021).
Karpman, D. et al. The role of lipopolysaccharide and Shiga-like toxin in a mouse model of Escherichia coli O157:H7 infection. J. Infect. Dis. 175, 611–620 (1997).
Szeto, C. C., McIntyre, C. W. & Li, P. K. Circulating bacterial fragments as cardiovascular risk factors in CKD. J. Am. Soc. Nephrol. 29, 1601–1608 (2018).
Awad, M. M. et al. Human plasminogen exacerbates Clostridioides difficile enteric disease and alters the spore surface. Gastroenterology 159, 1431–1443.e6 (2020).
Menzel, K. et al. Cathepsins B, L and D in inflammatory bowel disease macrophages and potential therapeutic effects of cathepsin inhibition in vivo. Clin. Exp. Immunol. 146, 169–180 (2006).
Tarlton, J. F. et al. The role of up-regulated serine proteases and matrix metalloproteinases in the pathogenesis of a murine model of colitis. Am. J. Pathol. 157, 1927–1935 (2000).
Palmieri, V. et al. Interleukin-33 signaling exacerbates experimental infectious colitis by enhancing gut permeability and inhibiting protective Th17 immunity. Mucosal Immunol. 14, 923–936 (2021).
Cebra, J. J. Influences of microbiota on intestinal immune system development. Am. J. Clin. Nutr. 69, 1046s–1051s (1999).
Vĕtvicka, V., Tlaskalová-Hogenová, H. & Stĕpánková, R. Effects of microflora antigens on lymphocyte migration patterns in germfree and conventional rats. Folia Biol. 29, 412–418 (1983).
Kamada, N. et al. Regulated virulence controls the ability of a pathogen to compete with the gut microbiota. Science 336, 1325–1329 (2012).
Rea, M. C. et al. Bioavailability of the anti-clostridial bacteriocin thuricin CD in gastrointestinal tract. Microbiology 160, 439–445 (2014).
Cash, H. L., Whitham, C. V., Behrendt, C. L. & Hooper, L. V. Symbiotic bacteria direct expression of an intestinal bactericidal lectin. Science 313, 1126–1130 (2006).
Lu, X., Xie, S., Ye, L., Zhu, L. & Yu, Q. Lactobacillus protects against S. Typhimurium-induced intestinal inflammation by determining the fate of epithelial proliferation and differentiation. Mol. Nutr. Food Res. 64, e1900655 (2020). An important study demonstrating the links between microbiota, enteric pathogens, host immune responses, and colonocyte proliferation and differentiation during enteric disease.
Velazquez, E. M. et al. Endogenous Enterobacteriaceae underlie variation in susceptibility to Salmonella infection. Nat. Microbiol. 4, 1057–1064 (2019).
Drolia, R. et al. Receptor-targeted engineered probiotics mitigate lethal Listeria infection. Nat. Commun. 11, 6344 (2020).
Taha-Abdelaziz, K. et al. In vitro assessment of immunomodulatory and anti-Campylobacter activities of probiotic lactobacilli. Sci. Rep. 9, 17903 (2019).
Nattramilarasu, P. K., Lobo de Sá, F. D., Schulzke, J. D. & Bücker, R. Immune-mediated aggravation of the Campylobacter concisus-induced epithelial barrier dysfunction. Int. J. Mol. Sci. 22, 2043 (2021).
Sakaue, M. et al. Type A fulminant Clostridium perfringens sepsis indicated RBC/Hb discrepancy; a case report. BMC Infect. Dis. 19, 719 (2019).
Shibata, W. et al. Helicobacter-induced gastric inflammation alters the properties of gastric tissue stem/progenitor cells. BMC Gastroenterol. 17, 145 (2017). This study establishes the link between enteric bacterial pathogens, the inflammation and host immune responses they induce and, in turn, the effects they can elicit on stem or progenitor cells during infection.
Beumer, J. & Clevers, H. Regulation and plasticity of intestinal stem cells during homeostasis and regeneration. Development 143, 3639–3649 (2016). A review encompassing the stem cell populations and regeneration mechanisms within the intestines during health and repair from injury.
van der Wath, R. C., Gardiner, B. S., Burgess, A. W. & Smith, D. W. Cell organisation in the colonic crypt: a theoretical comparison of the pedigree and niche concepts. PLoS ONE 8, e73204 (2013).
Hua, T. et al. A reserve stem cell population in small intestine renders Lgr5-positive cells dispensable. Nature 478, 255–259 (2011).
Liu, X., Lu, R., Wu, S. & Sun, J. Salmonella regulation of intestinal stem cells through the Wnt/beta-catenin pathway. FEBS Lett. 584, 911–916 (2010).
Huan, Y. W. et al. Lawsonia intracellularis exploits beta-catenin/Wnt and Notch signalling pathways during infection of intestinal crypt to alter cell homeostasis and promote cell proliferation. PLoS ONE 12, e0173782 (2017).
Flanagan, D. J. et al. Frizzled7 functions as a wnt receptor in intestinal epithelial Lgr5+ stem cells. Stem Cell Rep. 4, 759–767 (2015).
Holmberg, J. et al. EphB receptors coordinate migration and proliferation in the intestinal stem cell niche. Cell 125, 1151–1163 (2006).
Gregorieff, A. et al. Expression pattern of wnt signaling components in the adult intestine. Gastroenterology 129, 626–638 (2005).
Gonzalez, L. M. et al. Preservation of reserve intestinal epithelial stem cells following severe ischemic injury. Am. J. Physiol. Gastrointest. Liver Physiol. 316, G482–G494 (2019).
Karin, M. & Clevers, H. Reparative inflammation takes charge of tissue regeneration. Nature 529, 307–315 (2016).
Potten, C. S., Owen, G. & Booth, D. Intestinal stem cells protect their genome by selective segregation of template DNA strands. J. Cell Sci. 115, 2381–2388 (2002).
Merlos-Suárez, A. et al. The intestinal stem cell signature identifies colorectal cancer stem cells and predicts disease relapse. Cell Stem Cell 8, 511–524 (2011).
Bankaitis, E. D., Ha, A., Kuo, C. J. & Magness, S. T. Reserve stem cells in intestinal homeostasis and injury. Gastroenterology 155, 1348–1361 (2018).
Chatzivasileiou, K., Kriebel, K., Steinhoff, G., Kreikemeyer, B. & Lang, H. Do oral bacteria alter the regenerative potential of stem cells? A concise review. J. Cell Mol. Med. 19, 2067–2074 (2015).
O’Rourke, F. & Kempf, V. A. J. Interaction of bacteria and stem cells in health and disease. FEMS Microbiol. Rev. 43, 162–180 (2019). A review presenting current knowledge on the diverse interactions between bacteria and stem cells throughout the body in regulating stem cell renewal and homeostasis as well as targets for bacterial pathogenicity strategies.
Sellin, J. H., Wang, Y., Singh, P. & Umar, S. beta-Catenin stabilization imparts crypt progenitor phenotype to hyperproliferating colonic epithelia. Exp. Cell Res. 315, 97–109 (2009).
van der Post, S., Birchenough, G. M. H. & Held, J. M. NOX1-dependent redox signaling potentiates colonic stem cell proliferation to adapt to the intestinal microbiota by linking EGFR and TLR activation. Cell Rep. 35, 108949 (2021).
Lee, Y. S. et al. Microbiota-derived lactate accelerates intestinal stem-cell-mediated epithelial development. Cell Host Microbe 24, 833–846.e6 (2018).
Reedy, A. R., Luo, L., Neish, A. S. & Jones, R. M. Commensal microbiota-induced redox signaling activates proliferative signals in the intestinal stem cell microenvironment. Development 146, dev171520 (2019).
Sellin, M. E. et al. Epithelium-intrinsic NAIP/NLRC4 inflammasome drives infected enterocyte expulsion to restrict Salmonella replication in the intestinal mucosa. Cell Host Microbe 16, 237–248 (2014).
Fast, D., Duggal, A. & Foley, E. Monoassociation with Lactobacillus plantarum disrupts intestinal homeostasis in adult Drosophila melanogaster. mBio 9, e01114-18 (2018).
Kuo, W. T. et al. the tight junction protein ZO-1 is dispensable for barrier function but critical for effective mucosal repair. Gastroenterology 161, 1924–1939 (2021).
Samba-Louaka, A., Nougayrède, J. P., Watrin, C., Oswald, E. & Taieb, F. The enteropathogenic Escherichia coli effector Cif induces delayed apoptosis in epithelial cells. Infect. Immun. 77, 5471–5477 (2009).
Iwai, H. et al. A bacterial effector targets Mad2L2, an APC inhibitor, to modulate host cell cycling. Cell 130, 611–623 (2007).
Glotfelty, L. G. et al. Enteropathogenic E. coli effectors EspG1/G2 disrupt microtubules, contribute to tight junction perturbation and inhibit restoration. Cell Microbiol. 16, 1767–1783 (2014).
Pearson, J. S. et al. A type III effector antagonizes death receptor signalling during bacterial gut infection. Nature 501, 247–251 (2013).
Li, S. et al. Pathogen blocks host death receptor signalling by arginine GlcNAcylation of death domains. Nature 501, 242–246 (2013).
Parigi, S. M. et al. The spatial transcriptomic landscape of the healing mouse intestine following damage. Nat. Commun. 13, 828 (2022). This study utilizes an exciting new technology that combines spatial biology with transcriptomics to assess intestinal repair in an approach dubbed by Nature Methods as ‘method of the year’ for 2020.
Leblond, F., Davis, S. C., Valdés, P. A. & Pogue, B. W. Pre-clinical whole-body fluorescence imaging: review of instruments, methods and applications. J. Photochem. Photobiol. B 98, 77–94 (2010).
Engel, R. M. et al. Patient-derived colorectal cancer organoids upregulate revival stem cell marker genes following chemotherapeutic treatment. J. Clin. Med. 9, 128 (2020).
Nusse, Y. M. et al. Parasitic helminths induce fetal-like reversion in the intestinal stem cell niche. Nature 559, 109–113 (2018). An important study demonstrating the ability of parasitic helminths, and in turn microorganisms, to affect intestinal stem cell function and microenvironment for their own gain.
Zhang, J., Liang, Y., Bradford, W. H. & Sheikh, F. Desmosomes: emerging pathways and non-canonical functions in cardiac arrhythmias and disease. Biophys. Rev. 13, 697–706 (2021).
Tsukita, S., Furuse, M. & Itoh, M. Multifunctional strands in tight junctions. Nat. Rev. Mol. Cell Biol. 2, 285–293 (2001).
Nitta, T. et al. Size-selective loosening of the blood-brain barrier in claudin-5-deficient mice. J. Cell Biol. 161, 653–660 (2003).
Balda, M. S. & Matter, K. Tight junctions at a glance. J. Cell Sci. 121, 3677–3682 (2008).
Guillemot, L., Paschoud, S., Pulimeno, P., Foglia, A. & Citi, S. The cytoplasmic plaque of tight junctions: a scaffolding and signalling center. Biochim. Biophys. Acta Biomembr. 1778, 601–613 (2008).
Krause, G. et al. Structure and function of claudins. Biochim. Biophys. Acta Biomembr. 1778, 631–645 (2008).
Ayyaz, A. et al. Single-cell transcriptomes of the regenerating intestine reveal a revival stem cell. Nature 569, 121–125 (2019). A pioneering study defining a unique stem cell population that is activated by damage to regenerate the injured intestinal epithelium separate to homeostatic intestinal stem cell populations.
Bjerknes, M. & Cheng, H. Clonal analysis of mouse intestinal epithelial progenitors. Gastroenterology 116, 7–14 (1999).
Yan, K. S. et al. The intestinal stem cell markers Bmi1 and Lgr5 identify two functionally distinct populations. Proc. Natl Acad. Sci. USA 109, 466–471 (2012).
Harnack, C. et al. R-spondin 3 promotes stem cell recovery and epithelial regeneration in the colon. Nat. Commun. 10, 4368 (2019).
Davidson, L. A. et al. Alteration of colonic stem cell gene signatures during the regenerative response to injury. Biochim. Biophys. Acta 1822, 1600–1607 (2012).
Acknowledgements
The authors thank the referees for their helpful comments on this work. The authors also thank H. Abud for stimulating discussions about intestinal stem cells. The authors would also like to acknowledge that, due to space limitations, many important studies and citations could not be included.
Author information
Authors and Affiliations
Contributions
A.P.R., S.J.M. and D.L. researched data for the article. All authors substantially contributed to the discussion of content. A.P.R. wrote the manuscript. S.J.M. and D.L. reviewed or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Microbiology thanks Arun Bhunia, Mikael Sellin and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Goblet cells
-
Intestinal epithelial cells with the primary function to synthesize and secrete mucous.
- Bacteraemia
-
An infection of the bloodstream.
- Sepsis
-
An overactivation of the immune response following infection that causes inflammation throughout the body and can be life threatening.
- Haemolytic uraemic syndrome
-
A syndrome most commonly caused by infection, which leads to kidney damage, blood cell lysis and retention of kidney filtrates.
- Paneth cells
-
Specialized epithelial cells located at the small intestinal crypt base that secrete stem cell-supporting factors.
- Stem cell niche
-
Specific microenvironment at the base of the intestinal crypt that promotes an undifferentiated, self-renewable state.
Rights and permissions
About this article
Cite this article
Rogers, A.P., Mileto, S.J. & Lyras, D. Impact of enteric bacterial infections at and beyond the epithelial barrier. Nat Rev Microbiol 21, 260–274 (2023). https://doi.org/10.1038/s41579-022-00794-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41579-022-00794-x
This article is cited by
-
The interaction of inflammasomes and gut microbiota: novel therapeutic insights
Cell Communication and Signaling (2024)
-
Claudin-7 is essential for the maintenance of colonic stem cell homoeostasis via the modulation of Wnt/Notch signalling
Cell Death & Disease (2024)
-
Cleavage of cell junction proteins as a host invasion strategy in leptospirosis
Applied Microbiology and Biotechnology (2024)
-
Balancing Act of the Intestinal Antimicrobial Proteins on Gut Microbiota and Health
Journal of Microbiology (2024)
-
Multistrain Probiotics with Fructooligosaccharides Improve Middle Cerebral Artery Occlusion–Driven Neurological Deficits by Revamping Microbiota-Gut-Brain Axis
Probiotics and Antimicrobial Proteins (2023)