Abstract
Virtually all plants and animals, including humans, are home to symbiotic microorganisms. Symbiotic interactions can be neutral, harmful or have beneficial effects on the host organism. However, growing evidence suggests that microbial symbionts can evolve rapidly, resulting in drastic transitions along the parasite–mutualist continuum. In this Review, we integrate theoretical and empirical findings to discuss the mechanisms underpinning these evolutionary shifts, as well as the ecological drivers and why some host–microorganism interactions may be stuck at the end of the continuum. In addition to having biomedical consequences, understanding the dynamic life of microorganisms reveals how symbioses can shape an organism’s biology and the entire community, particularly in a changing world.
Similar content being viewed by others
Introduction
Parasitic and mutualistic microbial symbioses exist widely in nature. These interactions occur when microorganisms (that is, bacteria, fungi and viruses) take up residence in or on animals or plants, and cause damage or confer benefits to the host. Parasitic microorganisms (including pathogens) can exploit the host, and in doing so, cause harm. The term mutualist classically refers to any organism in a mutually beneficial relationship with another. However, the assumed benefits are rarely empirically tested for the symbiont1. There is thus an emerging awareness that many putative mutualisms may even be hosts exploiting symbionts2,3,4, in an interaction referred to as inverted parasitism5.
The continuum
The designation of entities as ‘parasite’ or ‘mutualist’ implies a simple binary system whereby species incur positive or negative impacts on fitness during interactions. However, these terms represent ends of a continuum along which an interaction between a host and symbiont can shift. These transitions occur as the relative benefits and costs to each species in the relationship strengthen or weaken (Fig. 1) across ecological or evolutionary time. Transitions can be driven by changes in the environment and ecology of the interacting species or communities. At the centre of the continuum sit commensals, which benefit from the interaction with hosts, but do not cause a detectable cost6.
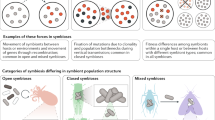
Examples from nature of microorganisms transitioning from free-living to host-associated lifestyles include the evolution of parasitic species in the Bacillus cereus group (for example, the causative agent of anthrax) from soil-dwelling ancestors237 (part a), and environmental Pantoea bacteria evolving obligate mutualistic roles in stink bug growth and development16 (part b). Examples involving transitions along the continuum are the widespread plant parasite Pseudomonas syringae likely evolving from mutualistic ancestors, driven by horizontal gene transfer (HGT) of type III secretion systems29,79 (part c), and entomopathogens taking over the metabolic role of an ancient and degraded endosymbiont in cicadas165 (part d). Image credits: part a (right) Getty images Smith Collection/Gado.Contributor; part b is adapted from ref.238, CC BY 4.0 (https://creativecommons.org/licenses/by/4.0/); part c (left), image courtesey of Gerald Holmes; part c (right) is adapted from ref.239, CC BY 4.0 (https://creativecommons.org/licenses/by/4.0/); part d (left), image courtesey of Yu Matsuura; part d (right) adapted with permission from ref.165, PNAS.
The concept of the parasite–mutualist continuum dates back several decades. An early discussion by Ewald7 focused on the fundamental role of transmission route in driving evolutionary transitions between parasitism and mutualism in symbiotic associations. The conditionality of symbiotic interactions was later highlighted by Bronstein8. She reviewed evidence that the costs and benefits of interspecific interactions vary greatly with ecological context, and thus the outcome of a symbiosis can change throughout an organism’s lifetime.
Evolution of microorganisms into parasites or mutualists
Microorganisms can rapidly adapt to new environments. Short generation times, large population sizes and high mutation rates combined with genome flexibility all facilitate accelerated microbial evolution9. Furthermore, their capacity for plastic responses10,11,12 and the dynamic nature of the communities that microorganisms are nested and interact within13,14 provide further routes for changing costs and benefits of association with hosts.
Free-living environmental microorganisms, which do not associate with hosts, were the progenitors for all symbiont diversity observed today15. Free-living microorganisms can evolve to be parasites or mutualists16,17,18,19,20,21. A new host-associated lifestyle often remains facultative for the microorganism22,23, but in some cases the microorganism evolves an obligate dependency on the host24,25. Transitions from free-living to host association are sometimes facilitated by horizontal transfer of genes, often encoding traits that facilitate immediate exploitation of, or benefit to, hosts (for example, immune evasion, toxin production, nitrogen fixation and bioluminescence)15,26. Once associated with a host, symbiotic interactions can shift along the continuum (Fig. 1). For instance, parasites can evolve to be less antagonistic to hosts. Reduced antagonism is thought to be favoured if alternative hosts are rarely available or if transmission of the parasite is enhanced by increases in host fitness27,28. Molecular phylogenetics corroborates this trajectory, showing that parasites have frequently served as progenitors for the independent descent of symbionts that now exhibit mutualistic traits15,29. In this context, microorganisms shifting into novel host taxa is an important process, often forging novel associations on the continuum30,31. Transitions can also occur if a parasite’s own selfish traits benefit a host as a by-product27, or by hosts rewarding32 or capturing33 symbiont genotypes that confer benefits. Conversely, mutualisms can break down into parasitisms. This breakdown can occur owing to the spread of cheater symbionts, which exploit the benefits of host association without paying the cost of returning a benefit27,34. However, shifts from mutualism to parasitism appear rare in nature15,29,35. More frequently, symbionts leave the host-association continuum by reverting to free-living environmental lifestyles, as demonstrated by Actinobacteria abandoning ant hosts15,36.
In this Review, we discuss the evolutionary transitions of host–microorganism symbioses along the parasite–mutualist continuum, the mechanisms underlying evolutionary changes, the selective pressures involved and common empirical approaches for studying them (Box 1). We also briefly discuss context-dependent transitions and the consequences faced by microorganisms when their symbioses are constrained to the extreme ends of the continuum. Moreover, we focus the Review on eukaryotic host–microorganism symbioses; however, we note that microbial interactions with mobile genetic elements (MGEs) can be analogous to symbioses (Box 2) given the ability of these elements to confer beneficial traits and cause harm to bacterial hosts37,38.
Mechanisms of evolution along the continuum
The gradual emergence of microbial mutualists from parasitic ancestors15,29,31,39 and the rapid leaps in symbiont phenotypes observed in real time40,41,42,43,44 provide fascinating insights into the proliferation of microbial symbiotic diversity. The genetic changes involved in microbial evolution are key contributors to the formation of mutualisms and parasitisms and their transitions along the symbiotic continuum. Mechanisms that result in these changes include, for example, selection on existing genetic variation45,46, de novo mutations40,43,47,48,49 and genome rearrangements50,51,52. Genome rearrangements include inversions, duplications, translocations and gene loss50,53,54 (for further discussion of gene loss, see later section, Stuck at the end of the line). Horizontal gene transfer (HGT) events, whereby genetic material moves between organisms in a manner other than vertically, are also important factors in microbial evolutionary transitions42,55,56,57,58. These events often involve MGEs — such as plasmids, transposons, insertion elements and phages — coding for traits that are beneficial or harmful to hosts during their interaction.
Shifts between microbial parasitism and mutualism can involve selection on existing variation. Through experimental evolution of the bacterial symbiont Parachlamydia acanthamoebae and its protist host Acanthamoeba sp., one study46 observed an evolutionary shift of the microbial symbiont towards parasitism under horizontal transmission conditions. The molecular basis of this transition was a pronounced increase in the frequency of specific genetic variants within the original symbiont population, alongside marked changes in the expression of machinery necessary for manipulating host cells, such as the type III secretion system (T3SS).
Selection on de novo mutations in bacterial populations has also been detected in evolution experiments, resulting in movement along the continuum. In these cases, experiments are started by propagating a single clone in hosts. In one study40, a clonal population of Enterococcus faecalis was introduced into nematode host populations, and mutations that arose favoured enhanced production of reactive oxygen species. This phenotype allowed E. faecalis to become highly beneficial to hosts, as production of these antimicrobials suppressed infection by Staphylococcus aureus. A similar direction of travel, but from parasite to commensal, has been observed in nematode host populations by evolving Pseudomonas aeruginosa from a single clone49. Conversely, within the guts of old mice, mutations arising in clones of commensal Escherichia coli may have resulted in evolution towards pathogenicity59. In comparison with evolution within young mice, mutational targets linked to stress-related functions and associated with virulence were under strong selection in the inflamed guts of older mice. Mutation might have a prominent role in transitions when symbionts have a low initial diversity upon colonization. This situation could occur naturally when symbionts have a low infectious dose or when transmission causes population bottlenecks (see section on Transmission below).
Wide-ranging genetic changes — HGT, gene loss and genome rearrangements — have had a profound role in Yersinia pestis becoming more virulent and adapting to new host species50,60,61. Y. pestis is the causative agent of plague in mammalian and arthropod hosts. It is thought to have diverged from its less harmful ancestor Yersinia pseudotuberculosis 1,500–55,000 years ago62,63. Sequencing of isolates of the two species revealed that both HGT and insertion sequence-mediated genome rearrangements and deletions facilitated Y. pestis evolution50,60,61. The bacterium acquired two plasmids, namely pMT1 and pPCP1, making it more virulent compared with its Y. pseudotuberculosis ancestor. The former plasmid carries the ymt gene encoding Yersinia murine toxin, required for the colonization of the flea host64,65, and the capsular antigen fraction 1, which inhibits phagocytosis65,66. These acquisitions contributed to the evolution of Y. pestis towards greater virulence. Adaptation of the parasite to new hosts was mediated by genome rearrangements, particularly via insertion sequences and gene loss. Gene loss was crucial in reducing the toxicity of Y. pestis to the flea vector, allowing biofilm to develop in the flea foregut67. Gene disruption by insertion sequences, in combination with deletion events, point mutations and frameshifts, further created an extensive number of pseudogenes within the Y. pestis genome50,60,61. Altogether, these genetic changes facilitated a shift in lifestyle, from a less harmful mammalian enteropathogen to systemic pathogen of both mammalian and arthropod hosts.
Infection by various phages (mostly lytic, λ-like phages) along with other MGEs facilitated the divergence of the highly pathogenic enterohaemorrhagic E. coli strain O157 Sakai from its ancestor. The commensal E. coli strain K12 is also descended from this common ancestor68. In strain O157 Sakai, prophages and prophage-like elements encode a variety of virulence-related genes — adhesins, tellurite resistance genes and urease — contributing to the acquisition of virulence factors that have determined this bacterium’s trajectory towards increased virulence in humans. One of these elements also encodes the major virulence factor, the locus of enterocyte effacement (LEE), which is responsible for bacterial attachment followed by development of the disease-causing effacing lesions in the intestine69. Lambda-like phages on the Sakai chromosome also encode the destructive Shiga toxin, as well as proteins involved in serum resistance and cell adhesion. Having become integral to the organism’s virulence in this way, the prophages themselves have transitioned from parasitic to mutualistic elements within the O157 Sakai genome (for further discussion of MGEs as symbionts, see Box 2).
How commonly do shifts across the continuum occur owing to de novo mutation or machinery acquired by HGT? Host environments with complex, often open, microbial communities, such as the mammalian gut, might generate more extensive opportunities for HGT70,71,72. For example, phage-driven HGT from the resident community can dictate the evolution of invading strains73 and instigate change more rapidly than is achievable by mutation accumulation74. HGT has had a considerable role in major evolutionary transitions of living organisms; it is increasingly confirmed as a dominant force in the evolution of host–symbiont associations20,29,54,58,65,75,76,77,78,79,80. Yet, for symbionts nested within simple microbial communities (for example, intracellular environments), scarce opportunities for HGT may mean de novo mutation is more likely to underpin shifts along the continuum. Studies reporting selection on de novo mutation during transitions40,49,59 highlight the power of this genetic means to generate remarkable change on the continuum. These experiments typically involve a small number of microbial species and/or low levels of initial genetic diversity upon colonization. When incorporating a host background with an ecologically relevant microbiota, HGT might be more dominant.
Drivers of evolution along the continuum
Ecological sources of selection can drive microbial symbiont evolution towards increasing host benefits (Table 1) or harm (Table 2). Shifts occur across generations as microbial symbionts adapt to life in a new host species, encounter different transmission opportunities and face hosts that reciprocally evolve in response. The presence or absence of additional interacting species in the community can also drive evolutionary change in a host–symbiont relationship owing to changing distribution of net benefits and costs across the community. Essentially, given a strong source of selection, genetic change can occur within just a handful of microbial generations. These transitions are often investigated using experimental evolution or over macro-evolutionary timescales via phylogenetic comparisons (Box 1).
Novel hosts
Microorganisms frequently encounter novel host environments. They can jump across species boundaries or colonize hosts from pools of free-living environmental microorganisms. Novel infections can generate new diversity on the symbiosis continuum through divergence and speciation81. High-profile cases of host shifts, such as the recent SARS-CoV-2 pandemic82, highlight the potential for investigating evolutionary changes in virulence upon emergence83,84,85. New associations are often maladaptive for both host and parasite86, and associations can move unpredictably on the continuum or burn out. This trajectory has been observed in emergences of avian influenza virus, where case fatality rates can be high but human-to-human transmission is low87.
Shifts between host species, possibly driven by HGT of virulence-associated genes, appear to have been important in the emergence of the Q fever parasite, Coxiella burnetii30,88. This proposed mutualist-to-parasite transition is a complex case for which the full evolutionary story remains unknown. However, phylogenetic analysis suggests that this highly infectious bacterium recently emerged from a clade of vertically transmitted mutualistic endosymbionts of ticks30. C. burnetii may have evolved mechanisms to infect vertebrate cells, persist in the environment and be airborne-transmitted. These traits are unlikely to be found in the arthropod-restricted ancestors30. Ticks feeding on vertebrates likely provided the ecological bridge. Similar transitions occurred within Sodalis-allied symbionts, a group of host-restricted bacteria common to insects including the tsetse fly vector. A free-living Sodalis sp. was isolated after a person suffered a wound from a tree branch, and this serendipitous finding provided evidence that symbiont lineages emerged from environmental ancestors31. Early vectoring of these environmental strains by insects was likely pivotal in the evolution of the beneficial, heritable Sodalis endosymbionts observed today.
Novel species interactions can drive rapid innovation. This might particularly be the case if a microorganism bears characteristics that can provide instant benefits. Microorganisms encoding functions of light generation, photosynthesis, nitrogen fixation or antimicrobials may provide such rapid benefits15. These characteristics may be remodelled (or act as pre-adaptations) for transitions in symbiosis15. Such repurposing may have occurred in the antifungal-producing Burkholderia symbionts associated with Lagriinae beetles. Burkholderia symbionts appear to have transitioned from a plant parasite to insect mutualist. In this context, secondary metabolites previously used as virulence factors against plants may have been repurposed for antifungal defence on beetle eggs89. Additional evidence comes from marine hosts, including within the bulbs of anglerfish and the Vibrio fischeri-filled light organs of bobtail squid. These hosts benefit from these bioluminescent bacteria to lure prey and avoid predation, respectively, and the symbionts often retain the capacity to live freely, or persist in the environment22,51.
Transmission opportunities
Transmission mode has been considered to predict the direction of a symbiont’s evolution on the continuum. When horizontally transmitted symbionts can move between unrelated host individuals, the fitness interests between species are uncoupled, a scenario thought to favour parasitism7. The degree of harm caused to hosts from infection is often framed by the virulence–transmission trade-off90,91. The relationship assumes that virulence — the reduction in host fitness caused by parasite infection — is costly to the parasite as host resources are needed for replication92. The cost of harming the host too much or too soon from replication might result in less transmission. Thus, it is predicted that transmission should be highest at intermediate virulence, which balances the costs of within‐host replication and infectious period length90. This model is particularly pertinent for symbionts that rely on a mobile host for transmission (for example, socially transmitted microorganisms). Those that do not (for example, vector- and water-borne microorganisms) can bypass trade-offs between virulence and transmission91. This conventional model goes some way to hypothesizing on general patterns of virulence, yet several extensions and alternatives have been suggested93,94,95.
It has been suggested that mutualists may evolve from parasitic ancestors when the frequency of horizontal transmission routes is reduced or lost7. If vertical transmission is the remaining dominant mode of transmission then the fitness of host and symbiont can become tightly coupled, reducing the arena for evolutionary conflict and thereby favouring selection for mutual benefit7,90,96. Mutualisms involving symbiont inheritance are predicted to be stable on the continuum and unlikely to revert to parasitism15,97. But exclusively vertical transmission can endanger associations via genetic bottlenecking (see section on Stuck at the end of the line). Clearly, becoming inherited is not the sole route by which bacterial mutualists evolve. Comparative analysis has found no evidence for vertical transmission preceding the origin of mutualism15. Many mutualisms involve horizontal transmission such as conjugative plasmids in bacterial populations98 and the vast networks of mycorrhizae that improve plant productivity99,100. In particular, evolution of defensive traits in symbionts are proposed to be facilitated by the genetic diversity and selection for innovation promoted by horizontal transmission101. Many horizontally transmitted microbial symbionts are obligate for host fitness16,22,102, but many can be facultative24 and confer costs in different environments.
Conversely, not all inherited microorganisms become mutualists103. Wolbachia, Spiroplasma and Arsenophonus species are common inherited parasites that manipulate host reproduction, maximizing resource allocation to the transmitting host sex (females) by feminizing hosts or killing their sons104. However, theory suggests that the spread of such reproductive parasites will be enhanced by the evolution of traits that benefit hosts105. A beneficial trait (that is, defence) may even interact with a parasitic trait (that is, reproductive manipulation) to completely exclude a natural enemy105. Indeed, cryptic benefits are now found in several systems106,107, and there is evidence that some reproductive parasites may need to also transmit horizontally just to persist108.
Transmission as a determinant of the location of a symbiosis along the continuum is complex. There are numerous exceptions to classical theory. Nonetheless, experimental manipulation of transmission modes finds general support for the theory that horizontal transmission can select for parasitism and vertical transmission for reduced antagonism (Table 1; Table 2). In a symbiosis between a jellyfish and the alga Symbiodinium microadriaticum, cooperative traits, including growth enhancement, were selected when transmission was restricted to heritable routes109. Such cooperative traits are fundamental for stable mutualisms, protecting against transitions to parasitism or abandonment events. In the reverse experiment, restriction of the alga to horizontal transmission selected for faster proliferation and dispersal (traits associated with parasitism), and declines in host fitness were detected109. Such findings are mirrored across terrestrial systems46,110,111. The common pill bug hosts a Wolbachia strain (wVulC) that feminizes genetic males112. Blocking the typical vertical route, and mimicking horizontal transmission, saw systemic increases in Wolbachia (wVulC) density and a drastic transition from a benign partner to a highly virulent one110.
The community
The drivers of transitions along the parasite–mutualist continuum can be complex and stem from the ecological and evolutionary movements of many different players. Defensive symbiosis113,114, whereby there are at least three interacting species (host, defensive symbiont and an attacking enemy) is particularly dynamic along the continuum in response to community composition changes. The absence of the symbiont or enemy can have evolutionary consequences for other species in the community, even without direct interactions115,116. Co-infections in hosts can also influence transitions in the symbiosis by providing new phenotypes via HGT of genetic material (for example, symbiosis islands, plasmids and phages)78,80,114,117.
The impact of community complexity is demonstrated by the bacterium Hamiltonella defensa and its lysogenic phage, APSE. This association protects host aphids against parasitoid wasps118,119 (Fig. 2). In this context, the fitness benefit afforded to the aphid host is contingent on parasitoid presence — in its absence, H. defensa with APSE phage is costly to the aphid120. The mechanism of protection (toxin production) hinges on the initial lateral transfer of phage from a co-infecting symbiont117,121. Subsequent loss of the phage can move the interaction between H. defensa and aphids back towards parasitism122. Theory105,116, experimental evolution40 and field studies123 have captured how microorganisms, even parasitic ones, can evolve rapidly to protect their hosts when collectively threatened, often crossing the parasite–mutualist continuum in the process. In Caenorhabditis elegans nematodes, a mildly parasitic gut bacterium was shown to evolve an enhanced ability to protect against infection by a more virulent parasite40. In the parasite’s absence, the gut bacterium did not emerge as a microbial line of host defence.
Defensive symbioses involve multiple species, including a host (H) and defensive microorganism (DM) that protects against an attacking parasite (P)113. Often, hidden players exist within a DM, such as mobile genetic elements (MGEs; for example phages, plasmids and transposable elements) that encode factors involved in the protective function of the DM. In this community, the evolutionary and ecological moves (examples denoted by arrows) of each player can affect the relative position of another on the parasite–mutualist continuum. Players may move, resulting in an overall beneficial (net+), detrimental (net–) or negligible (net 0) effect on host fitness. For example, if a MGE encodes key protective functions, then its loss (move 2) will shift the DM’s position towards parasitism (all cost and no benefit to host). Meanwhile, the costs of P to H will increase now that H is no longer protected by the DM and its MGE. Transitions here can also alter the coevolutionary patterns and processes between players and species.
Additional symbionts, with previously unknown effects, are increasingly being identified even in iconic ‘two-player’ symbioses, such as corals124 and lichens125,126. It is thus not surprising that the complexity of a host’s whole microbiota (which often includes a diverse repertoire of bacteria, fungi and viruses) can interact to produce new outcomes for individual strains, species and the community as a whole. Members of the microbiota compete and cooperate in a myriad of ways127, influencing the virulence of one another via processes such as the suppression of public goods128 or the facilitation of biofilm formation129 and epithelial translocation130. The passage of Candida albicans in mice lacking gut microbiota has highlighted the role of communities in determining fate on the parasite–mutualist continuum. In the absence of a gut microbiota, C. albicans mutants emerge that are defective in hyphal formation, no longer requiring it for competition against other microbiota members. When compared with the wild-type ancestor that coexists with a microbiota, these C. albicans mutants are less virulent and protect their hosts against Aspergillus fumigatus infection in a manner independent of host adaptive immunity43. This transition from pathobiont to conditional mutualist in this context appears to hinge on the absence of competing microorganisms. However, given a gradient of increasing microbiome diversity, it would be valuable to understand when the selective advantage of the transition disappears. Other recent work, in microbiota-free mice, noted that when E. coli is a lone colonizer of the gut, it is consistently selected to increase metabolism of amino acids serine and threonine. A small increase in microbiome diversity (the addition of a single competing species) alters the evolutionary trajectory of E. coli substantially, instead favouring mutations associated with anaerobic metabolism131. This outcome suggests that bacteria may have low fidelity in metabolic function even within a single host generation132. Such a finding suggests host–microbial symbioses may not adhere to the idea of the ‘holobiont’ being a cohesive unit of selection133. This idea relies on high fidelity between partners134, which may easily be disrupted by changes to the surrounding microbial community.
If we can selectively drive the evolution of microorganisms and their communities, applications may improve on the already promising use of faecal microbiota transplants in medicine135, symbiont-mediated vector control136,137 and the manipulation of crop parasites42. There is, however, a pressing need to understand the long-term response of microbial communities to the engineering of symbionts. Recently, theoretical models have treated virulence as a cost shared by all symbionts coexisting in a host138,139. These models find that defence by a symbiont often drives reduced virulence across the microbial community (including in attacking parasites), an outcome dependent on the cost of defence being low and the shared cost of virulence also being low139. However, defensive microorganisms may also select for resistance mechanisms (for example, toxin production and inflammatory stimulation) in the parasites they protect against, causing collateral damage to hosts and driving increased parasite virulence140. This is akin to established predictions for co-infecting parasite species, whereby competition selects for increased virulence141,142,143. Promisingly though, and in line with some theory138,139, selection for reduced parasite virulence has been revealed in response to microorganism-mediated protection144. Others also report long-term efficacy of protection mechanisms despite an evolving pathosphere145.
Host control
Beyond microbial symbiont evolution, hosts can affect the position of the symbiosis on the continuum146. Hosts can be resistant (that is, reducing symbiont colonization) and tolerant (that is, coping with symbiont-associated damage without limiting colonization)147, which reduces any negative impacts of the host–symbiont interaction. Evolving control mechanisms (for example, sanctions and rewards, and microbiome modulators)146,148, or acquiring symbiotic function from an alternative source (for example, symbiont switching and HGT)100 can also limit or cause a change in the position of the interaction along the continuum.
Resistance to symbiont infection is observed ubiquitously across evolving host–parasite associations149,150. Mutations associated with membrane transporters in the bacterium Actinomyces odontolyticus coincided with a reduction in the negative effects of its ectoparasite (Nanosynbacter lyticus)151, perhaps indicating an adaptive host response to block resources to the ectoparasite or prevent its attachment151. As host resistance and tolerance strategies can affect parasitic symbiont fitness, they can counter-adapt152,153. This process may lead to a repeated back and forth along the continuum.
Hosts can also have key roles in restraining symbiont-driven shifts along the continuum. They may act to prevent the emergence of cheating symbionts, which exploit the benefits of host association without paying the cost of returning a benefit27,34. Alternatively, hosts may maintain the association at a position optimal for their own fitness. Sanction and reward strategies, spatial segregation of symbionts and partner choice mechanisms have evolved to promote and maintain cooperation27,154,155. For instance, legumes may sanction defective nitrogen-fixing bacteria by blocking resources to the respective root nodule32,154, and plants reward helpful mycorrhizal fungi with extra carbohydrate156. These mechanisms protect the host from investing in symbionts with net costs and avoid trajectories towards antagonism.
There is mounting theoretical and empirical evidence that many putative mutualisms may actually be a product of hosts exploiting symbionts2,3,4,33. Interactions can benefit the host, but with no reciprocity to the symbiont whose fitness is markedly reduced within the walls of host confinement1. These may be viewed as cases of inverted parasitism5. The host is the parasite of its smaller guest. This phenomenon is exemplified by zooxanthellae in which replication rates are severely compromised by host association4, rising from 3 days outside of coral hosts157 to around 70 days within158. Another example comes from Paramecium bursaria and photosynthetic Chlorella symbionts. Chlorella species provide fixed carbon in return for organic nitrogen, but the host tightly controls symbiont density in response to light conditions, ensuring the best nutrient trade for itself159. Control of the symbiont potentially occurs via digestion of Chlorella cells160. The host may win twofold, paying the workforce only when required and acquiring nutrition via digestion of surplus symbionts. The growth rate for Chlorella remains consistently better outside the host159, but inside, this symbiont avoids algal competitors161 and may be protected against its own parasites162. Research on exploitation by hosts is in its infancy, with the greatest evidence coming from interactions with photosynthetic symbionts4,159,163. Many questions remain, including the ubiquity of the phenomenon and whether some classes of symbiont are more vulnerable to exploitation than others.
Although considered relatively rare over evolutionary time, hosts may also eschew parasitic164 and mutualistic associations100. Fleeing the infectious environment is one strategy. Spatiotemporal escape by asexual rotifers prevents them interacting with fungal parasites consistently over evolutionary time. By drying up and blowing away in the wind, these animals are protected from infection, which allows them to maintain their asexual reproductive strategy164. Mutualistic associations can be abandoned via the recruitment of new symbionts100. As the Hodgkinia endosymbionts of cicadas teetered on the edge of genomic collapse, Ophiocordyceps fungi (commonly parasites) began to take over the essential roles in amino acid synthesis for the host165. Abandonment can also occur via exploitation of an alternative resource100. For example, the evolution of carnivory in plants led to several plant species deserting arbuscular mycorrhizal fungal symbionts, as the plant now gains nutrients directly from prey100. These cases chime with a growing debate over whether hosts can have the upper hand in symbioses, despite generally being the species that evolves more slowly (known as the Red King effect166,167), exploiting and imprisoning their microorganisms to gain disproportionate control and benefit2,3,4,33,159,168.
Context-dependent shifts
The outcome of many microbial interactions with hosts are context dependent14. Both facultative and obligate symbioses can make shifts along the parasite–mutualist continuum that do not involve evolution, often occurring within a generation and driven by ecological change or opportunity (Table 3). Abiotic factors such as temperature169, resource availability170, environmental toxicity171 and the biotic composition of the surrounding community119 or host ontogeny172,173 can all affect the distribution of costs and benefits incurred by the host and microbial symbiont. The position on the continuum can also change if the microbial symbiont becomes infected with its own symbionts (for example, phages and mycoviruses)42,122. Here, we focus on short-term disruptions to host–symbiont associations, but note that sustained alterations to context will feed back to evolutionary change for the interacting species.
Generally, theory predicts that nutrient-limited environments, or other harsh environments, can foster beneficial interactions between compatible players27,174 via mechanisms such as cross-protection and cross-feeding. This outcome has been substantiated by empirical work175,176,177. For symbionts that have nutritional roles (for example, vitamin synthesis and nitrogen fixation), abundant resources can substantially undermine the net benefit gained by the host. The provisioning of mineral nitrogen from fertilizer erases the benefit Bradyrhizobium symbionts provide to legume hosts (Lotus strigosusas) as this acquisition route is less energetically costly for the legume than its symbiont-fixed equivalent178. Some hosts evade context-dependent costs by divesting themselves of associations when ecological conditions change, such as the phytoplankton that abandon their nitrogen-fixing cyanobacteria when environmental nitrogen is abundant179. For host–parasite systems, there is no evidence for a one-way effect of nutrient availability to hosts on the harm caused by infection180. One study180 suggested that the level of parasite virulence in a given environment is likely the result of a balance between the effect of host nutrition on the immune system and on parasite resources.
Temperature can affect symbiont phenotypes181,182, which directly impact symbiont virulence or benefit, such as the regulation of toxin production183 or molecules required for nutrient scavenging184. Some obligate mutualists can constitute thermally ‘weak links’ for hosts, becoming non-functional or even lost from hosts outside adapted temperature ranges, which can have catastrophic consequences for host fitness185,186. Interactions can occur between abiotic and biotic factors. For instance, a 5 °C increase in temperature diminishes H. defensa-mediated defence against parasitoids187,188. This temperature-dependent reduction in defence may be ameliorated if co-infection with an additional bacterium, known as pea aphid X-type symbiont, occurs187.
In other cases, community composition alone can temporarily cause transitions. Defensive symbioses present a clear demonstration of community context-dependent shifts, whereby benefits to the host are contingent on the presence of an enemy species113,114. In the absence of the enemy, the host pays the cost with no detectable benefit, and the association moves towards one that is parasitic114,189. Infection of a symbiont with its own symbionts (that is, hyperparasitism190) can also generate transitions. Recent work found that the devastating effects of a fungal parasite on rapeseed crop are significantly reduced if the fungus becomes infected with mycovirus SsHADV-1 (ref.42). The presence of the mycovirus appeared to affect the expression of a suite of both fungal and crop genes, including those encoding plant cell-wall-degrading enzymes and crop signalling pathways42.
Pathobionts provide an excellent example of context-dependent transitions from neutral to harmful agents191. In a host with a functional immune system and healthy microbiota, pathobionts can exist as commensals191,192,193. Pathobionts are well adapted to proliferate beyond their normal niche. During dysbiosis (for example, compromised immunity, disruption of the microbiota or introduction of medical devices such as catheters or surgical implants) pathobionts can cause disease in a wide variety of forms, from minor infections to more serious chronic or invasive disease194. This ability to transition from harmless to harmful in different contexts makes pathobionts hard to place on the continuum. They are neither consistent parasites nor consistent commensals, with the state of the host generally determining their transition from one to the other.
Stuck at the end of the line
At either end of the continuum lie the extremes of host-killing (or castration) and mutual dependence. What maintains an association here, and what is its future?
The ability to shift along the continuum for some parasitic microorganisms could depend on transmission route. Some infectious agents may stay hypervirulent owing to a high degree of environmental transmission or a lack of reliance on hosts to transmit and propagate. The ‘curse of the Pharoah’ hypothesis195 posits that microorganisms able to ‘sit and wait’ in the environment can be perpetual killers, whereas others suggest that traits that enable persistence in the environment will be traded off with virulence196. There may also be constraints of the parasitic life cycle that prevent a transition. Microbial parasites that must lyse host cells to transmit (for example, lytic phages and Plasmodium species in mammals) or steal resources in a way that castrates the host (Pasteuria bacterial parasite infecting Daphnia magna197) are systems in which transitions away from antagonism are unlikely.
At the opposing end of the continuum lie inherited, obligate endosymbionts, which often have nutritional roles. Although many of these associations are ancient and bestow mutual benefits, they can be risky, particularly for the endosymbiont3,53,198. The genomes of these symbionts can gradually decay as transmission bottlenecks allow deleterious mutations to become fixed by genetic drift, and mutational bias towards deletions removes genes199,200,201,202. Genomic decay can lead to extinction, unless heightened genetic and cellular support is provided by the host203 or other symbionts78,204,205. For example, leafhoppers show gene expression patterns that appear tailored to the deficiencies of each of their endosymbionts’ highly degraded genomes203. In rare cases, symbionts may transition to organelle status206, notoriously achieved by mitochondria and plastids, but this does not guarantee shelter against further gene loss or extinction207,208. Hosts may also avoid extinction alongside an endosymbiont by exploiting alternative nutritional resources or gaining new symbionts158,159.
Conclusions and future perspectives
Plants and animals, including humans, are colonized by innumerable microorganisms. This observation has sparked a revolution in studying the impacts of those microorganisms on host biology and health. Many more examples of microbial evolution causing transitions across the parasite–mutualist continuum will emerge through further research using experimental evolution and investigating the microbiome in an evolutionary context. The potential evolution of species in the human microbiome from good to bad209,210, and the degree to which beneficial interactions could be upset by microbiome perturbation211, are of clinical relevance for individuals vulnerable to infectious disease. In the future, such individuals may benefit from engineering of the microbiome or symbiont communities, via either direct genetic modifications to key transition loci in microbiome members, or exposure to selection sources with known outcomes. This approach has recently been achieved for honeybees, with the genetic modification of a core gut bacterium improving resistance to viral infection212. These are exciting applications, but we must strive to understand the evolutionary consequences for the parasites targeted too.
More fundamentally, understanding causes of transitions will provide insight into the dynamics of how an organism’s biology and its community are shaped by microbial inhabitants. The ecological and evolutionary transitions of other species, as well as environmental change, can alter the scope for conflict in symbioses involving microorganisms. Interest has grown in thinking of host–microorganism symbioses as holobionts with highly aligned selective interests134. Many associations may be also viewed in an ecological community context13,146 in which constant shifts occur back and forth on the parasite–mutualist continuum. The degree to which the host and symbiont, or both, have control over those shifts remains relatively unexplored. Research in the field has focused on the propensity of symbionts to invade unwilling hosts or cheat reciprocal arrangements. Yet an exciting new avenue is emerging, one that is exposing hosts as exploiters and imprisoners of microorganisms33,198. The extent to which microorganisms are able to evolve to counter or take advantage of that exploitation is also unclear.
Moreover, environmental changes have the potential to substantially alter selection in symbiotic interactions213. In addition to altering established symbioses, marked changes to abiotic variables can also move the boundaries of environmental constraint, fostering the evolution of new interactions on the continuum that were previously impossible or profitless. How will the collectively growing impact of humans affect the stability of beneficial associations and the emergence of parasites globally (for example, see refs214,215)? This question is particularly timely given the COVID-19 pandemic. Undoubtedly, as environmental perturbations increase in magnitude and frequency, and as the use of antimicrobials grows, understanding the effects on the real-time evolution of host–symbiont interactions will become more and more valuable.
References
Garcia, J. R. & Gerardo, N. M. The symbiont side of symbiosis: do microbes really benefit? Front. Microbiol. 5, 510 (2014).
Law, R. & Dieckmann, U. Symbiosis through exploitation and the merger of lineages in evolution. Proc. Biol. Sci. 265, 1245–1253 (1998).
Keeling, P. J. & McCutcheon, J. P. Endosymbiosis: the feeling is not mutual. J. Theor. Biol. 434, 75–79 (2017).
Wooldridge, S. A. Is the coral-algae symbiosis really ‘mutually beneficial’ for the partners? BioEssays 32, 615–625 (2010).
Mushegian, A. A. & Ebert, D. Rethinking ‘mutualism’ in diverse host-symbiont communities. BioEssays 38, 100–108 (2016).
Mathis, K. A. & Bronstein, J. L. Our current understanding of commensalism. Ann. Rev. Ecol. Evol. Syst. 51, 167–189 (2020).
Ewald, P. W. Transmission modes and evolution of the parasitism-mutualism continuum. Ann. N. Y. Acad. Sci. 503, 295–306 (1987).
Bronstein, J. L. Conditional outcomes in mutualistic interactions. Trends Ecol. Evol. 9, 214–217 (1994).
Schu, M. G. & Schrallhammer, M. Cultivation conditions can cause a shift from mutualistic to parasitic behavior in the symbiosis between Paramecium and its bacterial symbiont Caedibacter taeniospiralis. Curr. Microbiol. 75, 1099–1102 (2018).
Osman, E. O. et al. Coral microbiome composition along the northern Red Sea suggests high plasticity of bacterial and specificity of endosymbiotic dinoflagellate communities. Microbiome 8, 8 (2020).
Kümmerli, R., Jiricny, N., Clarke, L. S., West, S. A. & Griffin, A. S. Phenotypic plasticity of a cooperative behaviour in bacteria. J. Evol. Biol. 22, 589–598 (2009).
Kumamoto, C. A. Niche-specific gene expression during C. albicans infection. Curr. Opin. Microbiol. 11, 325–330 (2008).
Thrall, P. H., Hochberg, M. E., Burdon, J. J. & Bever, J. D. Coevolution of symbiotic mutualists and parasites in a community context. Trends Ecol. Evol. 22, 120–126 (2007).
Chamberlain, S. A., Bronstein, J. L. & Rudgers, J. A. How context dependent are species interactions? Ecol. Lett. 17, 881–890 (2014).
Sachs, J. L., Skophammer, R. G. & Regus, J. U. Evolutionary transitions in bacterial symbiosis. Proc. Natl Acad. Sci. USA 108 (Suppl. 2), 10800–10807 (2011).
Hosokawa, T. et al. Obligate bacterial mutualists evolving from environmental bacteria in natural insect populations. Nat. Microbiol. 1, 1–7 (2016).
Gupta, A. & Nair, S. Dynamics of insect–microbiome interaction influence host and microbial symbiont. Front. Microbiol. 11, 1357 (2020).
Lutzoni, F. & Pagel, M. Accelerated evolution as a consequence of transitions to mutualism. Proc. Natl Acad. Sci. USA 94, 11422–11427 (1997).
Kaltenpoth, M. et al. Partner choice and fidelity stabilize coevolution in a Cretaceous-age defensive symbiosis. Proc. Natl Acad. Sci. USA 111, 6359–6364 (2014).
Manzano-Marı́n, A. et al. Serial horizontal transfer of vitamin-biosynthetic genes enables the establishment of new nutritional symbionts in aphids’ di-symbiotic systems. ISME J. 14, 259–273 (2020).
Miyauchi, S. et al. Large-scale genome sequencing of mycorrhizal fungi provides insights into the early evolution of symbiotic traits. Nat. Commun. 11, 5125 (2020).
McFall-Ngai, M. J. The importance of microbes in animal development: lessons from the squid-Vibrio symbiosis. Annu. Rev. Microbiol. 68, 177–194 (2014).
Brown, S. P., Cornforth, D. M. & Mideo, N. Evolution of virulence in opportunistic pathogens: generalism, plasticity, and control. Trends Microbiol. 20, 336–342 (2012).
Fisher, R. M., Henry, L. M., Cornwallis, C. K., Kiers, E. T. & West, S. A. The evolution of host-symbiont dependence. Nat. Commun. 8, 15973 (2017).
McDowell, J. M. Genomes of obligate plant pathogens reveal adaptations for obligate parasitism. Proc. Natl Acad. Sci. USA 108, 8921–8922 (2011).
Wilson, B. A. & Salyers, A. A. Is the evolution of bacterial pathogens an out-of-body experience? Trends Microbiol. 11, 347–350 (2003).
Sachs, J. L., Mueller, U. G., Wilcox, T. P. & Bull, J. J. The evolution of cooperation. Q. Rev. Biol. 79, 135–160 (2004).
Bull, J. J. & Rice, W. R. Distinguishing mechanisms for the evolution of co-operation. J. Theor. Biol. 149, 63–74 (1991).
Sachs, J. L., Skophammer, R. G., Bansal, N. & Stajich, J. E. Evolutionary origins and diversification of proteobacterial mutualists. Proc. Biol. Sci. 281, 20132146 (2014).
Duron, O. et al. The recent evolution of a maternally-inherited endosymbiont of ticks led to the emergence of the Q fever pathogen, Coxiella burnetii. PLoS Pathog. 11, e1004892 (2015).
Clayton, A. L. et al. A novel human-infection-derived bacterium provides insights into the evolutionary origins of mutualistic insect–bacterial symbioses. PLoS Genet. 8, e1002990 (2012).
West, S. A., Kiers, E. T., Simms, E. L. & Denison, R. F. Sanctions and mutualism stability: why do rhizobia fix nitrogen? Proc. Biol. Sci. 269, 685–694 (2002).
Sørensen, M. E. S. et al. The role of exploitation in the establishment of mutualistic microbial symbioses. FEMS Microbiol. Lett. 366, fnz148 (2019).
Trivers, R. L. The evolution of reciprocal altruism. Q. Rev. Biol. 46, 35–57 (1971).
Frederickson, M. E. Mutualisms are not on the verge of breakdown. Trends Ecol. Evol. 32, 727–734 (2017).
Mueller, U. G., Ishak, H., Lee, J. C., Sen, R. & Gutell, R. R. Placement of attine ant-associated Pseudonocardia in a global Pseudonocardia phylogeny (Pseudonocardiaceae, Actinomycetales): a test of two symbiont-association models. Antonie Van Leeuwenhoek 98, 195–212 (2010).
Dietel, A.-K., Kaltenpoth, M. & Kost, C. Convergent evolution in intracellular elements: plasmids as model endosymbionts. Trends Microbiol. 26, 755–768 (2018).
Hurst, G. D. D. Extended genomes: symbiosis and evolution. Interface Focus. 7, 20170001 (2017).
Melnyk, R. A., Hossain, S. S. & Haney, C. H. Convergent gain and loss of genomic islands drive lifestyle changes in plant-associated Pseudomonas. ISME J. 13, 1575–1588 (2019).
King, K. C. et al. Rapid evolution of microbe-mediated protection against pathogens in a worm host. ISME J. 10, 1915–1924 (2016).
Shapiro, J. W. & Turner, P. E. Evolution of mutualism from parasitism in experimental virus populations. Evolution 72, 707–712 (2018).
Zhang, H. et al. A 2-kb mycovirus converts a pathogenic fungus into a beneficial endophyte for brassica protection and yield enhancement. Mol. Plant. 13, 1420–1433 (2020).
Tso, G. H. W. et al. Experimental evolution of a fungal pathogen into a gut symbiont. Science 362, 589–595 (2018).
Harrison, E., Guymer, D., Spiers, A. J., Paterson, S. & Brockhurst, M. A. Parallel compensatory evolution stabilizes plasmids across the parasitism-mutualism continuum. Curr. Biol. 25, 2034–2039 (2015).
Porter, S. S., Faber-Hammond, J., Montoya, A. P., Friesen, M. L. & Sackos, C. Dynamic genomic architecture of mutualistic cooperation in a wild population of Mesorhizobium. ISME J. 13, 301–315 (2019).
Herrera, P. et al. Molecular causes of an evolutionary shift along the parasitism–mutualism continuum in a bacterial symbiont. Proc. Natl Acad. Sci. USA 117, 21658–21666 (2020).
Li, E. et al. Rapid evolution of bacterial mutualism in the plant rhizosphere. Preprint at bioRxiv https://doi.org/10.1101/2020.12.07.414607 (2020).
Pankey, M. S. et al. Host-selected mutations converging on a global regulator drive an adaptive leap towards symbiosis in bacteria. eLife 6, e24414 (2017).
Jansen, G. et al. Evolutionary transition from pathogenicity to commensalism: global regulator mutations mediate fitness gains through virulence attenuation. Mol. Biol. Evol. 32, 2883–2896 (2015).
Chain, P. S. G. et al. Insights into the evolution of Yersinia pestis through whole-genome comparison with Yersinia pseudotuberculosis. Proc. Natl Acad. Sci. USA 101, 13826–13831 (2004).
Hendry, T. A. et al. Ongoing transposon-mediated genome reduction in the luminous bacterial symbionts of deep-sea ceratioid anglerfishes. mBio 9, e01033-18 (2018).
Nygaard, S. et al. Reciprocal genomic evolution in the ant–fungus agricultural symbiosis. Nat. Commun. 7, 12233 (2016).
Bennett, G. M. & Moran, N. A. Heritable symbiosis: the advantages and perils of an evolutionary rabbit hole. Proc. Natl Acad. Sci. USA 112, 10169–10176 (2015).
Gluck-Thaler, E. et al. Repeated gain and loss of a single gene modulates the evolution of vascular pathogen lifestyles. bioRxiv https://doi.org/10.1101/2020.04.24.058529 (2020).
Arredondo-Alonso, S. et al. Plasmids shaped the recent emergence of the major nosocomial pathogen Enterococcus faecium. mBio 11, e03284-19 (2020).
Driscoll, T. P. et al. Evolution of Wolbachia mutualism and reproductive parasitism: insight from two novel strains that co-infect cat fleas. Preprint at bioRxiv https://doi.org/10.1101/2020.06.01.128066 (2020).
Frantzeskakis, L. et al. Signatures of host specialization and a recent transposable element burst in the dynamic one-speed genome of the fungal barley powdery mildew pathogen. BMC Genomics 19, 381 (2018).
Savory, E. A. et al. Evolutionary transitions between beneficial and phytopathogenic Rhodococcus challenge disease management. eLife 6, e30925 (2017).
Barreto, H. C., Sousa, A. & Gordo, I. The landscape of adaptive evolution of a gut commensal bacteria in aging mice. Curr. Biol. 30, 1102–1109.e5 (2020).
Parkhill, J. et al. Genome sequence of Yersinia pestis, the causative agent of plague. Nature 413, 523–527 (2001).
Deng, W. et al. Genome sequence of Yersinia pestis KIM. J. Bacteriol. 184, 4601–4611 (2002).
Achtman, M. et al. Yersinia pestis, the cause of plague, is a recently emerged clone of Yersinia pseudotuberculosis. Proc. Natl Acad. Sci. USA 96, 14043–14048 (1999).
Rasmussen, S. et al. Early divergent strains of Yersinia pestis in Eurasia 5,000 years ago. Cell 163, 571–582 (2015).
Hinnebusch, B. J. et al. Role of Yersinia murine toxin in survival of Yersinia pestis in the midgut of the flea vector. Science 296, 733–735 (2002).
Lindler, L. E., Plano, G. V., Burland, V., Mayhew, G. F. & Blattner, F. R. Complete DNA sequence and detailed analysis of the Yersinia pestis KIM5 plasmid encoding murine toxin and capsular antigen. Infect. Immun. 66, 5731–5742 (1998).
Du, Y., Rosqvist, R. & Forsberg, Å. Role of fraction 1 antigen of Yersinia pestis in inhibition of phagocytosis. Infect. Immun. 70, 1453–1460 (2002).
Sun, Y.-C., Jarrett, C. O., Bosio, C. F. & Hinnebusch, B. J. Retracing the evolutionary path that led to flea-borne transmission of Yersinia pestis. Cell Host Microbe 15, 578–586 (2014).
Ohnishi, M., Kurokawa, K. & Hayashi, T. Diversification of Escherichia coli genomes: are bacteriophages the major contributors? Trends Microbiol. 9, 481–485 (2001).
Franzin, F. M. & Sircili, M. P. Locus of enterocyte effacement: a pathogenicity island involved in the virulence of enteropathogenic and enterohemorragic Escherichia coli subjected to a complex network of gene regulation. Biomed. Res. Int. 2015, 534738 (2015).
Brito, I. L. et al. Mobile genes in the human microbiome are structured from global to individual scales. Nature 535, 435–439 (2016).
Broaders, E., O’Brien, C., Gahan, C. G. M. & Marchesi, J. R. Evidence for plasmid-mediated salt tolerance in the human gut microbiome and potential mechanisms. FEMS Microbiol. Ecol. 92, fiw019 (2016).
McCarthy, A. J. et al. Extensive horizontal gene transfer during Staphylococcus aureus co-colonization in vivo. Genome Biol. Evol. 6, 2697–2708 (2014).
Frazão, N., Sousa, A., Lässig, M. & Gordo, I. Horizontal gene transfer overrides mutation in Escherichia coli colonizing the mammalian gut. Proc. Natl Acad. Sci. USA 116, 17906–17915 (2019).
Niehus, R., Mitri, S., Fletcher, A. G. & Foster, K. R. Migration and horizontal gene transfer divide microbial genomes into multiple niches. Nat. Commun. 6, 8924 (2015).
Koonin, E. V. Horizontal gene transfer: essentiality and evolvability in prokaryotes, and roles in evolutionary transitions. F1000Res https://doi.org/10.12688/f1000research.8737.1 (2016).
Nowack, E. C. M. et al. Gene transfers from diverse bacteria compensate for reductive genome evolution in the chromatophore of Paulinella chromatophora. Proc. Natl Acad. Sci. USA 113, 12214–12219 (2016).
Bordenstein, S. R. & Bordenstein, S. R. Eukaryotic association module in phage WO genomes from Wolbachia. Nat. Commun. 7, 13155 (2016).
Waterworth, S. C. et al. Horizontal gene transfer to a defensive symbiont with a reduced genome in a multipartite beetle microbiome. mBio 11, e02430-19 (2020).
Ma, W., Dong, F. F. T., Stavrinides, J. & Guttman, D. S. Type III effector diversification via both pathoadaptation and horizontal transfer in response to a coevolutionary arms race. PLoS Genet. 2, e209 (2006).
Nikoh, N. et al. Evolutionary origin of insect–Wolbachia nutritional mutualism. Proc. Natl Acad. Sci. USA 111, 10257–10262 (2014).
Sheppard, S. K., Guttman, D. S. & Fitzgerald, J. R. Population genomics of bacterial host adaptation. Nat. Rev. Genet. 19, 549–565 (2018).
Day, T., Gandon, S., Lion, S. & Otto, S. P. On the evolutionary epidemiology of SARS-CoV-2. Curr. Biol. 30, R849–R857 (2020).
Tardy, L., Giraudeau, M., Hill, G. E., McGraw, K. J. & Bonneaud, C. Contrasting evolution of virulence and replication rate in an emerging bacterial pathogen. Proc. Natl Acad. Sci. USA 116, 16927–16932 (2019).
Alves, J. M. et al. Parallel adaptation of rabbit populations to myxoma virus. Science 363, 1319–1326 (2019).
Kerr, P. J. Myxomatosis in Australia and Europe: a model for emerging infectious diseases. Antivir. Res. 93, 387–415 (2012).
Longdon, B. et al. The causes and consequences of changes in virulence following pathogen host shifts. PLoS Pathog. 11, e1004728 (2015).
van Boven, M. et al. Detecting emerging transmissibility of avian influenza virus in human households. PLoS Comput. Biol. 3, e145 (2007).
Moses, A. S., Millar, J. A., Bonazzi, M., Beare, P. A. & Raghavan, R. Horizontally acquired biosynthesis genes boost Coxiella burnetii’s physiology. Front. Cell Infect. Microbiol. 7, 174 (2017).
Flórez, L. V. et al. Antibiotic-producing symbionts dynamically transition between plant pathogenicity and insect-defensive mutualism. Nat. Commun. 8, 1–9 (2017).
Anderson, R. M. & May, R. M. Coevolution of hosts and parasites. Parasitology 85, 411–426 (1982).
Ewald, P. W. Host-parasite relations, vectors, and the evolution of disease severity. Annu. Rev. Ecol. Syst. 14, 465–485 (1983).
Bull, J. J. Perspective: Virulence. Evolution 48, 1423–1437 (1994).
Rafaluk, C., Jansen, G., Schulenburg, H. & Joop, G. When experimental selection for virulence leads to loss of virulence. Trends Parasitol. 31, 426–434 (2015).
Alizon, S. & Van Baalen, M. Transmission-virulence trade-offs in vector-borne diseases. Theor. Popul. Biol. 74, 6–15 (2008).
Cressler, C. E., McLeod, D. V., Rozins, C., Hoogen, J. V. D. & Day, T. The adaptive evolution of virulence: a review of theoretical predictions and empirical tests. Parasitology 143, 915–930 (2016).
Axelrod, R. & Hamilton, W. D. The evolution of cooperation. Science 211, 1390–1396 (1981).
Yamamura, N. Vertical transmission and evolution of mutualism from parasitism. Theor. Popul. Biol. 44, 95–109 (1993).
Hall, J. P. J., Brockhurst, M. A., Dytham, C. & Harrison, E. The evolution of plasmid stability: are infectious transmission and compensatory evolution competing evolutionary trajectories? Plasmid 91, 90–95 (2017).
Kiers, E. T. & Denison, R. F. Sanctions, cooperation, and the stability of plant-rhizosphere mutualisms. Annu. Rev. Ecol. Evol. Syst. 39, 215–236 (2008).
Werner, G. D. A. et al. Symbiont switching and alternative resource acquisition strategies drive mutualism breakdown. Proc. Natl Acad. Sci. USA 115, 5229–5234 (2018).
Herre, E. A. et al. The evolution of mutualisms: exploring the paths between conflict and cooperation. Trends Ecol. Evol. 14, 49–53 (1999).
Nussbaumer, A. D., Fisher, C. R. & Bright, M. Horizontal endosymbiont transmission in hydrothermal vent tubeworms. Nature 441, 345–348 (2006).
Dusi, E., Krenek, S., Petzoldt, T., Kaltz, O. & Berendonk, T. U. When enemies do not become friends: experimental evolution of heat-stress adaptation in a vertically transmitted parasite. Preprint at bioRxiv https://doi.org/10.1101/2020.01.23.917773 (2020).
Engelstädter, J. & Hurst, G. D. D. The ecology and evolution of microbes that manipulate host reproduction. Annu. Rev. Ecol. Evol. Syst. 40, 127–149 (2009).
Fenton, A., Johnson, K. N., Brownlie, J. C. & Hurst, G. D. D. Solving the Wolbachia paradox: modeling the tripartite interaction between host, Wolbachia, and a natural enemy. Am. Nat. 178, 333–342 (2011).
Zug, R. & Hammerstein, P. Evolution of reproductive parasites with direct fitness benefits. Heredity 120, 266–281 (2018).
Drew, G. C., Frost, C. L. & Hurst, G. D. Reproductive parasitism and positive fitness effects of heritable microbes. in eLS https://onlinelibrary.wiley.com/doi/abs/10.1002/9780470015902.a0028327 (2019).
Parratt, S. R. et al. Superparasitism drives heritable symbiont epidemiology and host sex ratio in a wasp. PLoS Pathog. 12, e1005629 (2016).
Sachs, J. L. & Wilcox, T. P. A shift to parasitism in the jellyfish symbiont Symbiodinium microadriaticum. Proc. Biol. Sci. 273, 425–429 (2006).
Le Clec’h, W., Dittmer, J., Raimond, M., Bouchon, D. & Sicard, M. Phenotypic shift in Wolbachia virulence towards its native host across serial horizontal passages. Proc. Biol. Sci. 284, 20171076 (2017).
Stewart, A. D., Logsdon, J. M. & Kelley, S. E. An empirical study of the evolution of virulence under both horizontal and vertical transmission. Evolution 59, 730–739 (2005).
Rigaud, T., Souty-Grosset, C., Raimond, R., Mocquard, J.-P. & Juchault, P. Feminizing endocytobiosis in the terrestrial crustacean Armadilidium vulgare Latr. (isopoda) - recent acquisitions. Cell Res. 15, 259–273 (1991).
King, K. C. Defensive symbionts. Curr. Biol. 29, R78–R80 (2019).
Flórez, L. V., Biedermann, P. H. W., Engl, T. & Kaltenpoth, M. Defensive symbioses of animals with prokaryotic and eukaryotic microorganisms. Nat. Prod. Rep. 32, 904–936 (2015).
Couret, J., Huynh-Griffin, L., Antolic-Soban, I., Acevedo-Gonzalez, T. S. & Gerardo, N. M. Even obligate symbioses show signs of ecological contingency: impacts of symbiosis for an invasive stinkbug are mediated by host plant context. Ecol. Evol. 9, 9087–9099 (2019).
Ashby, B. & King, K. Friendly foes: the evolution of host protection by a parasite. Evol. Lett. 1, 211–221 (2017).
Duron, O. Arsenophonus insect symbionts are commonly infected with APSE, a bacteriophage involved in protective symbiosis. FEMS Microbiol. Ecol. 90, 184–194 (2014).
Ferrari, J., Darby, A. C., Daniell, T. J., Godfray, H. C. J. & Douglas, A. E. Linking the bacterial community in pea aphids with host-plant use and natural enemy resistance. Ecol. Entomol. 29, 60–65 (2004).
Oliver, K. M., Russell, J. A., Moran, N. A. & Hunter, M. S. Facultative bacterial symbionts in aphids confer resistance to parasitic wasps. Proc. Natl Acad. Sci. USA 100, 1803–1807 (2003).
Polin, S., Simon, J.-C. & Outreman, Y. An ecological cost associated with protective symbionts of aphids. Ecol. Evol. 4, 826–830 (2014).
Degnan, P. H., Yu, Y., Sisneros, N., Wing, R. A. & Moran, N. A. Hamiltonella defensa, genome evolution of protective bacterial endosymbiont from pathogenic ancestors. Proc. Natl Acad. Sci. USA 106, 9063–9068 (2009).
Weldon, S. R., Strand, M. R. & Oliver, K. M. Phage loss and the breakdown of a defensive symbiosis in aphids. Proc. Biol. Sci. 280, 20122103 (2013).
Weeks, A. R., Turelli, M., Harcombe, W. R., Reynolds, K. T. & Hoffmann, A. A. From parasite to mutualist: rapid evolution of Wolbachia in natural populations of Drosophila. PLoS Biol. 5, e114 (2007).
Kwong, W. K., del Campo, J., Mathur, V., Vermeij, M. J. A. & Keeling, P. J. A widespread coral-infecting apicomplexan with chlorophyll biosynthesis genes. Nature 568, 103–107 (2019).
Tuovinen, V. et al. Two basidiomycete fungi in the cortex of wolf lichens. Curr. Biol. 29, 476–483.e5 (2019).
Spribille, T. et al. Basidiomycete yeasts in the cortex of ascomycete macrolichens. Science 353, 488–492 (2016).
Coyte, K. Z. & Rakoff-Nahoum, S. Understanding competition and cooperation within the mammalian gut microbiome. Curr. Biol. 29, R538–R544 (2019).
Lopez-Medina, E. et al. Candida albicans inhibits Pseudomonas aeruginosa virulence through suppression of pyochelin and pyoverdine biosynthesis. PLoS Pathog. 11, e1005129 (2015).
Harriott, M. M. & Noverr, M. C. Candida albicans and Staphylococcus aureus form polymicrobial biofilms: effects on antimicrobial resistance. Antimicrob. Agents Chemother. 53, 3914–3922 (2009).
Diebel, L. N., Liberati, D. M., Diglio, C. A., Dulchavsky, S. A. & Brown, W. J. Synergistic effects of Candida and Escherichia coli on gut barrier function. J. Trauma. Acute Care Surg. 47, 1045 (1999).
Barroso-Batista, J. et al. Specific eco-evolutionary contexts in the mouse gut reveal Escherichia coli metabolic versatility. Curr. Biol. 30, 1049–1062.e7 (2020).
King, K. C., Stevens, E. & Drew, G. C. Microbiome: evolution in a world of interaction. Curr. Biol. 30, R265–R267 (2020).
Zilber-Rosenberg, I. & Rosenberg, E. Role of microorganisms in the evolution of animals and plants: the hologenome theory of evolution. FEMS Microbiol. Rev. 32, 723–735 (2008).
Douglas, A. E. & Werren, J. H. Holes in the hologenome: why host-microbe symbioses are not holobionts. mBio 7, e02099 (2016).
Bakken, J. S. et al. Treating Clostridium difficile infection with fecal microbiota transplantation. Clin. Gastroenterol. Hepatol. 9, 1044–1049 (2011).
Bourtzis, K. et al. Harnessing mosquito–Wolbachia symbiosis for vector and disease control. Acta Tropica 132, S150–S163 (2014).
O’Neill, S. L. in Dengue and Zika: Control and Antiviral Treatment Strategies (eds Hilgenfeld, R. & Vasudevan, S. G.) 355–360 (Springer, 2018).
Nelson, P. G. & May, G. Coevolution between mutualists and parasites in symbiotic communities may lead to the evolution of lower virulence. Am. Nat. 190, 803–817 (2017).
Nelson, P. & May, G. Defensive symbiosis and the evolution of virulence. Am. Nat. 196, 333–343 (2020).
Ford, S. A. & King, K. C. Harnessing the power of defensive microbes: evolutionary implications in nature and disease control. PLoS Pathog. 12, e1005465 (2016).
Nowak, M. A. & May, R. M. Superinfection and the evolution of parasite virulence. Proc. Biol. Sci. 255, 81–89 (1994).
Alizon, S., de Roode, J. C. & Michalakis, Y. Multiple infections and the evolution of virulence. Ecol. Lett. 16, 556–567 (2013).
Frank, S. A. Host–symbiont conflict over the mixing of symbiotic lineages. Proc. Biol. Sci. 263, 339–344 (1996).
Ford, S. A., Kao, D., Williams, D. & King, K. C. Microbe-mediated host defence drives the evolution of reduced pathogen virulence. Nat. Commun. 7, 1–9 (2016).
Engl, T. et al. Evolutionary stability of antibiotic protection in a defensive symbiosis. Proc. Natl Acad. Sci. USA 115, E2020–E2029 (2018).
Foster, K. R., Schluter, J., Coyte, K. Z. & Rakoff-Nahoum, S. The evolution of the host microbiome as an ecosystem on a leash. Nature 548, 43–51 (2017).
Schneider, D. S. & Ayres, J. S. Two ways to survive infection: what resistance and tolerance can teach us about treating infectious diseases. Nat. Rev. Immunol. 8, 889–895 (2008).
Voges, M. J. E. E. E., Bai, Y., Schulze-Lefert, P. & Sattely, E. S. Plant-derived coumarins shape the composition of an Arabidopsis synthetic root microbiome. Proc. Natl Acad. Sci. USA 116, 12558–12565 (2019).
Gandon, S. & Michalakis, Y. Evolution of parasite virulence against qualitative or quantitative host resistance. Proc. Biol. Sci. 267, 985–990 (2000).
Best, A., White, A. & Boots, M. The coevolutionary implications of host tolerance. Evolution 68, 1426–1435 (2014).
Bor, B. et al. Rapid evolution of decreased host susceptibility drives a stable relationship between ultrasmall parasite TM7x and its bacterial host. Proc. Natl Acad. Sci. USA 115, 12277–12282 (2018).
Schulte, R. D., Makus, C., Hasert, B., Michiels, N. K. & Schulenburg, H. Multiple reciprocal adaptations and rapid genetic change upon experimental coevolution of an animal host and its microbial parasite. Proc. Natl Acad. Sci. USA 107, 7359–7364 (2010).
Kerr, P. J. et al. Next step in the ongoing arms race between myxoma virus and wild rabbits in Australia is a novel disease phenotype. Proc. Natl Acad. Sci. USA 114, 9397–9402 (2017).
Kiers, E. T., Rousseau, R. A., West, S. A. & Denison, R. F. Host sanctions and the legume–rhizobium mutualism. Nature 425, 78–81 (2003).
Frederickson, M. E. Rethinking mutualism stability: cheaters and the evolution of sanctions. Q. Rev. Biol. 88, 269–295 (2013).
Kiers, E. T. et al. Reciprocal rewards stabilize cooperation in the mycorrhizal symbiosis. Science 333, 880–882 (2011).
Fitt, W. K. & Trench, R. K. The relation of diel patterns of cell division to diel patterns of motility in the symbiotic dinoflagellate Symbiodinium microadriaticum Freudenthal in culture. N. Phytol. 94, 421–432 (1983).
Wilkerson, F. P., Kobayashi, D. & Muscatine, L. Mitotic index and size of symbiotic algae in Caribbean reef corals. Coral Reefs 7, 29–36 (1988).
Lowe, C. D., Minter, E. J., Cameron, D. D. & Brockhurst, M. A. Shining a light on exploitative host control in a photosynthetic endosymbiosis. Curr. Biol. 26, 207–211 (2016).
Kodama, Y. & Fujishima, M. Symbiotic Chlorella variabilis incubated under constant dark conditions for 24 hours loses the ability to avoid digestion by host lysosomal enzymes in digestive vacuoles of host ciliate Paramecium bursaria. FEMS Microbiol. Ecol. 90, 946–955 (2014).
Iwai, S., Fujita, K., Takanishi, Y. & Fukushi, K. Photosynthetic endosymbionts benefit from host’s phagotrophy, including predation on potential competitors. Curr. Biol. 29, 3114–3119.e3 (2019).
Reisser, W. et al. Viruses distinguish symbiotic Chlorella spp. of Paramecium bursaria. Endocytobiosis Cell Res. 7, 245–251 (1991).
Ahmadjian, V. The lichen symbiosis. Ann. Botany 75, 101–102 (1993).
Wilson, C. G. & Sherman, P. W. Anciently asexual bdelloid rotifers escape lethal fungal parasites by drying up and blowing away. Science 327, 574–576 (2010).
Matsuura, Y. et al. Recurrent symbiont recruitment from fungal parasites in cicadas. Proc. Natl Acad. Sci. USA 115, E5970–E5979 (2018).
Bergstrom, C. T. & Lachmann, M. The Red King effect: when the slowest runner wins the coevolutionary race. Proc. Natl Acad. Sci. USA 100, 593–598 (2003).
Veller, C., Hayward, L. K., Hilbe, C. & Nowak, M. A. The Red Queen and King in finite populations. Proc. Natl Acad. Sci. USA 114, E5396–E5405 (2017).
Vigneron, A. et al. Insects recycle endosymbionts when the benefit is over. Curr. Biol. 24, 2267–2273 (2014).
Baker, D. M., Freeman, C. J., Wong, J. C. Y., Fogel, M. L. & Knowlton, N. Climate change promotes parasitism in a coral symbiosis. ISME J. 12, 921–930 (2018).
Hom, E. F. Y. & Murray, A. W. Niche engineering demonstrates a latent capacity for fungal-algal mutualism. Science 345, 94–98 (2014).
Hall, J. P. J. et al. Environmentally co-occurring mercury resistance plasmids are genetically and phenotypically diverse and confer variable context-dependent fitness effects. Env. Microbiol. 17, 5008–5022 (2015).
Banaszak, A. T., García Ramos, M. & Goulet, T. L. The symbiosis between the gastropod Strombus gigas and the dinoflagellate Symbiodinium: an ontogenic journey from mutualism to parasitism. J. Exp. Mar. Biol. Ecol. 449, 358–365 (2013).
Nakazawa, T. & Katayama, N. Stage-specific parasitism by a mutualistic partner can increase the host abundance. Front. Ecol. Evol. https://doi.org/10.3389/fevo.2020.602675 (2020).
Wintermute, E. H. & Silver, P. A. Emergent cooperation in microbial metabolism. Mol. Syst. Biol. 6, 407 (2010).
Yurtsev, E. A., Conwill, A. & Gore, J. Oscillatory dynamics in a bacterial cross-protection mutualism. Proc. Natl Acad. Sci. USA 113, 6236–6241 (2016).
Hoek, T. A. et al. Resource availability modulates the cooperative and competitive nature of a microbial cross-feeding mutualism. PLoS Biol. 14, e1002540 (2016).
Hillesland, K. L. & Stahl, D. A. Rapid evolution of stability and productivity at the origin of a microbial mutualism. Proc. Natl Acad. Sci. USA 107, 2124–2129 (2010).
Regus, J. U., Gano, K. A., Hollowell, A. C., Sofish, V. & Sachs, J. L. Lotus hosts delimit the mutualism–parasitism continuum of Bradyrhizobium. J. Evol. Biol. 28, 447–456 (2015).
Hay, M. E. et al. Mutualisms and aquatic community structure: the enemy of my enemy is my friend. Annu. Rev. Ecol. Evol. Syst. 35, 175–197 (2004).
Pike, V. L., Lythgoe, K. A. & King, K. C. On the diverse and opposing effects of nutrition on pathogen virulence. Proc. Biol. Sci. 286, 20191220 (2019).
Corbin, C., Heyworth, E. R., Ferrari, J. & Hurst, G. D. D. Heritable symbionts in a world of varying temperature. Heredity 118, 10–20 (2017).
Thomas, M. B. & Blanford, S. Thermal biology in insect-parasite interactions. Trends Ecol. Evol. 18, 344–350 (2003).
Delor, I. & Cornelis, G. R. Role of Yersinia enterocolitica Yst toxin in experimental infection of young rabbits. Infect. Immun. 60, 4269–4277 (1992).
Kouse, A. B., Righetti, F., Kortmann, J., Narberhaus, F. & Murphy, E. R. RNA-mediated thermoregulation of iron-acquisition genes in Shigella dysenteriae and pathogenic Escherichia coli. PLoS ONE 8, e63781 (2013).
Kishimoto, M., Baird, A. H., Maruyama, S., Minagawa, J. & Takahashi, S. Loss of symbiont infectivity following thermal stress can be a factor limiting recovery from bleaching in cnidarians. ISME J. 14, 3149–3152 (2020).
Zhang, B., Leonard, S. P., Li, Y. & Moran, N. A. Obligate bacterial endosymbionts limit thermal tolerance of insect host species. Proc. Natl Acad. Sci. USA 116, 24712–24718 (2019).
Guay, J.-F., Boudreault, S., Michaud, D. & Cloutier, C. Impact of environmental stress on aphid clonal resistance to parasitoids: role of Hamiltonella defensa bacterial symbiosis in association with a new facultative symbiont of the pea aphid. J. Insect Physiol. 55, 919–926 (2009).
Bensadia, F., Boudreault, S., Guay, J.-F., Michaud, D. & Cloutier, C. Aphid clonal resistance to a parasitoid fails under heat stress. J. Insect Physiol. 52, 146–157 (2006).
Vorburger, C. & Gouskov, A. Only helpful when required: a longevity cost of harbouring defensive symbionts. J. Evol. Biol. 24, 1611–1617 (2011).
Parratt, S. R. & Laine, A.-L. The role of hyperparasitism in microbial pathogen ecology and evolution. ISME J. 10, 1815–1822 (2016).
Kamada, N., Chen, G. Y., Inohara, N. & Núñez, G. Control of pathogens and pathobionts by the gut microbiota. Nat. Immunol. 14, 685–690 (2013).
Hajishengallis, G. & Lamont, R. J. Dancing with the stars: how choreographed bacterial interactions dictate nososymbiocity and give rise to keystone pathogens, accessory pathogens, and pathobionts. Trends Microbiol. 24, 477–489 (2016).
Neville, B. A., d’Enfert, C. & Bougnoux, M.-E. Candida albicans commensalism in the gastrointestinal tract. FEMS Yeast Res. 15, fov081 (2015).
Chow, J., Tang, H. & Mazmanian, S. K. Pathobionts of the gastrointestinal microbiota and inflammatory disease. Curr. Opin. Immunol. 23, 473–480 (2011).
Bonhoeffer, S., Lenski, R. E. & Ebert, D. The curse of the pharaoh: the evolution of virulence in pathogens with long living propagules. Proc. Biol. Sci. 263, 715–721 (1996).
Rafaluk-Mohr, C. The relationship between parasite virulence and environmental persistence: a meta-analysis. Parasitology 146, 897–902 (2019).
Ebert, D., Joachim Carius, H., Little, T. & Decaestecker, E. The evolution of virulence when parasites cause host castration and gigantism. Am. Nat. 164, S19–S32 (2004).
McCutcheon, J. P., Boyd, B. M. & Dale, C. The life of an insect endosymbiont from the cradle to the grave. Curr. Biol. 29, R485–R495 (2019).
Moran, N. A. Accelerated evolution and Muller’s rachet in endosymbiotic bacteria. Proc. Natl Acad. Sci. USA 93, 2873–2878 (1996).
Moran, N. A., McCutcheon, J. P. & Nakabachi, A. Genomics and evolution of heritable bacterial symbionts. Annu. Rev. Genet. 42, 165–190 (2008).
Wernegreen, J. J. Reduced selective constraint in endosymbionts: elevation in radical amino acid replacements occurs genome-wide. PLoS ONE 6, e28905 (2011).
Wernegreen, J. J. Genome evolution in bacterial endosymbionts of insects. Nat. Rev. Genet. 3, 850–861 (2002).
Mao, M., Yang, X. & Bennett, G. M. Evolution of host support for two ancient bacterial symbionts with differentially degraded genomes in a leafhopper host. Proc. Natl Acad. Sci. USA 115, E11691–E11700 (2018).
Husnik, F. et al. Horizontal gene transfer from diverse bacteria to an insect genome enables a tripartite nested mealybug symbiosis. Cell 153, 1567–1578 (2013).
Łukasik, P. et al. Multiple origins of interdependent endosymbiotic complexes in a genus of cicadas. Proc. Natl Acad. Sci. USA 115, E226–E235 (2018).
Keeling, P. J., McCutcheon, J. P. & Doolittle, W. F. Symbiosis becoming permanent: survival of the luckiest. Proc. Natl Acad. Sci. USA 112, 10101–10103 (2015).
Karnkowska, A. et al. A eukaryote without a mitochondrial organelle. Curr. Biol. 26, 1274–1284 (2016).
John, U. et al. An aerobic eukaryotic parasite with functional mitochondria that likely lacks a mitochondrial genome. Sci. Adv. 5, eaav1110 (2019).
Venkova, T., Yeo, C. C. & Espinosa, M. Editorial: The good, the bad, and the ugly: multiple roles of bacteria in human life. Front. Microbiol. 9, 1702 (2018).
Cirstea, M., Radisavljevic, N. & Finlay, B. B. Good bug, bad bug: breaking through microbial stereotypes. Cell Host Microbe 23, 10–13 (2018).
Durack, J. & Lynch, S. V. The gut microbiome: relationships with disease and opportunities for therapy. J. Exp. Med. 216, 20–40 (2019).
Leonard, S. P. et al. Engineered symbionts activate honey bee immunity and limit pathogens. Science 367, 573–576 (2020).
Wolinska, J. & King, K. C. Environment can alter selection in host–parasite interactions. Trends Parasitol. 25, 236–244 (2009).
Kiers, E. T., Palmer, T. M., Ives, A. R., Bruno, J. F. & Bronstein, J. L. Mutualisms in a changing world: an evolutionary perspective. Ecol. Lett. 13, 1459–1474 (2010).
Lafferty, K. D. The ecology of climate change and infectious diseases. Ecology 90, 888–900 (2009).
Magalon, H., Nidelet, T., Martin, G. & Kaltz, O. Host growth conditions influence experimental evolution of life history and virulence of a parasite with vertical and horizontal transmission. Evolution 64, 2126–2138 (2010).
Bull, J. J., Molineux, I. J. & Rice, W. R. Selection of benevolence in a host-parasite system. Evolution 45, 875–882 (1991).
Gibson, A. K. et al. The evolution of reduced antagonism—a role for host–parasite coevolution. Evolution 69, 2820–2830 (2015).
Kubinak, J. L. & Potts, W. K. Host resistance influences patterns of experimental viral adaptation and virulence evolution. Virulence 4, 410–418 (2013).
Matthews, A. C., Mikonranta, L. & Raymond, B. Shifts along the parasite–mutualist continuum are opposed by fundamental trade-offs. Proc. Biol. Sci. 286, 20190236 (2019).
Marchetti, M. et al. Experimental evolution of a plant pathogen into a legume symbiont. PLoS Biol. 8, e1000280 (2010).
Ruby, E. G. et al. Complete genome sequence of Vibrio fischeri: a symbiotic bacterium with pathogenic congeners. Proc. Biol. Sci. 102, 3004–3009 (2005).
Jeon, K. W. Genetic and physiological interactions in the amoeba-bacteria symbiosis. J. Eukaryot. Microbiol. 51, 502–508 (2004).
Wang, X. et al. Cryptic prophages help bacteria cope with adverse environments. Nat. Commun. 1, 1–9 (2010).
Bull, J. J. & Molineux, I. J. Molecular genetics of adaptation in an experimental model of cooperation. Evolution 46, 882–895 (1992).
Kikuchi, Y., Hosokawa, T. & Fukatsu, T. An ancient but promiscuous host-symbiont association between Burkholderia gut symbionts and their heteropteran hosts. ISME J. 5, 446–460 (2011).
Kikuchi, Y., Hosokawa, T. & Fukatsu, T. Insect-microbe mutualism without vertical transmission: a stinkbug acquires a beneficial gut symbiont from the environment every generation. Appl. Env. Microbiol. 73, 4308–4316 (2007).
Shapiro, J. W., Williams, E. S. C. P. & Turner, P. E. Evolution of parasitism and mutualism between filamentous phage M13 and Escherichia coli. PeerJ 4, e2060 (2016).
Porter, S. S. & Simms, E. L. Selection for cheating across disparate environments in the legume-rhizobium mutualism. Ecol. Lett. 17, 1121–1129 (2014).
Weese, D. J., Heath, K. D., Dentinger, B. T. M. & Lau, J. A. Long-term nitrogen addition causes the evolution of less-cooperative mutualists. Evolution 69, 631–642 (2015).
Slater, S. C. et al. Genome sequences of three Agrobacterium biovars help elucidate the evolution of multichromosome genomes in bacteria. J. Bacteriol. 191, 2501–2511 (2009).
Proença, J. T., Barral, D. C. & Gordo, I. Commensal-to-pathogen transition: one-single transposon insertion results in two pathoadaptive traits in Escherichia coli–macrophage interaction. Sci. Rep. 7, 4504 (2017).
Hu, G. et al. Microevolution during serial mouse passage demonstrates FRE3 as a virulence adaptation gene in Cryptococcus neoformans. mBio 5, e00941-14 (2014).
Chrostek, E. et al. Wolbachia variants induce differential protection to viruses in Drosophila melanogaster: a phenotypic and phylogenomic analysis. PLoS Genet. 9, e1003896 (2013).
Sicard, M. et al. When mutualists are pathogens: an experimental study of the symbioses between Steinernema (entomopathogenic nematodes) and Xenorhabdus (bacteria). J. Evol. Biol. 17, 985–993 (2004).
Margulis, L. Words as battle cries: symbiogenesis and the new field of endocytobiology. BioScience 40, 673–677 (1990).
Didelot, X., Barker, M., Falush, D. & Priest, F. G. Evolution of pathogenicity in the Bacillus cereus group. Syst. Appl. Microbiol. 32, 81–90 (2009).
Oishi, S., Moriyama, M., Koga, R. & Fukatsu, T. Morphogenesis and development of midgut symbiotic organ of the stinkbug Plautia stali (Hemiptera: Pentatomidae). Zool. Lett. 5, 16 (2019).
Kang, Y. et al. HopW1 from Pseudomonas syringae disrupts the actin cytoskeleton to promote virulence in Arabidopsis. PLoS Pathog. 10, e1004232 (2014).
Joy, J. B., Liang, R. H., McCloskey, R. M., Nguyen, T. & Poon, A. F. Y. Ancestral reconstruction. PLoS Comput. Biol. 12, e1004763 (2016).
Rafaluk-Mohr, C., Ashby, B., Dahan, D. A. & King, K. C. Mutual fitness benefits arise during coevolution in a nematode-defensive microbe model. Evol. Lett. 2, 246–256 (2018).
Ford, S. A., Williams, D., Paterson, S. & King, K. C. Co-evolutionary dynamics between a defensive microbe and a pathogen driven by fluctuating selection. Mol. Ecol. 26, 1778–1789 (2017).
Hall, A. R., Ashby, B., Bascompte, J. & King, K. C. Measuring coevolutionary dynamics in species-rich communities. Trends Ecol. Evol. 35, 539–550 (2020).
Betts, A., Rafaluk, C. & King, K. C. Host and parasite evolution in a tangled bank. Trends Parasitol. 32, 863–873 (2016).
Partridge, S. R., Kwong, S. M., Firth, N. & Jensen, S. O. Mobile genetic elements associated with antimicrobial resistance. Clin. Microbiol. Rev. 31, e00088-17 (2018).
Unterholzner, S. J., Poppenberger, B. & Rozhon, W. Toxin-antitoxin systems: biology, identification, and application. Mob. Genet. Elem. 3, e26219 (2013).
Croucher, N. J. et al. Rapid pneumococcal evolution in response to clinical interventions. Science 331, 430–434 (2011).
Wu, M. et al. Phylogenomics of the reproductive parasite wolbachia pipientis wMel: a streamlined genome overrun by mobile genetic elements. PLoS Biol. 2, E69 (2004).
Frost, C. L. et al. The hypercomplex genome of an insect reproductive parasite highlights the importance of lateral gene transfer in symbiont biology. mBio 11, e02590-19 (2020).
Bamford, D. H. Do viruses form lineages across different domains of life? Res. Microbiol. 154, 231–236 (2003).
Casjens, S. et al. A bacterial genome in flux: the twelve linear and nine circular extrachromosomal DNAs in an infectious isolate of the Lyme disease spirochete Borrelia burgdorferi. Mol. Microbiol. 35, 490–516 (2000).
Casjens, S. Prophages and bacterial genomics: what have we learned so far? Mol. Microbiol. 49, 277–300 (2003).
Acknowledgements
This work was funded by a European Starter Grant (COEVOPRO 802242) to K.C.K. The authors are grateful to four reviewers for their comments.
Author information
Authors and Affiliations
Contributions
All authors researched data for the article, contributed substantially to discussion of the content, wrote the article and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Inverted parasitism
-
An interaction whereby the classically viewed host exploits its smaller symbiont, implementing a fitness cost to the symbiont.
- Parasitism
-
An antagonistic symbiotic relationship in which one species is harmed, while the other benefits.
- Mutualism
-
A symbiotic relationship in which both interacting species benefit, or are perceived to benefit. Benefit is often only confirmed empirically for the host.
- Symbiosis
-
An association between two dissimilar organisms that have some degree of physical association, which is potentially long lasting, regardless of the implications for the fitness of either organism.
- Free-living
-
A microbial lifestyle not dependent on association with a host for long-term survival and replication; this is the ancestral state of all symbionts.
- Mobile genetic elements
-
(MGEs). Sequences of genetic material that can be exchanged between chromosomes or organisms via either their own mobilizing machinery or that of their host. Examples include transposable elements, plasmids and phages.
- Horizontal gene transfer
-
(HGT). The movement of genetic material between organisms that does not flow from parent to offspring.
- Virulence
-
The damage caused to the host due to infection by a parasite, often measured as a reduction in host fitness.
- Defensive symbiosis
-
An interaction in which the symbiont protects the host (via direct or indirect mechanisms) against natural enemies, such as microbial parasites and eukaryotic parasitoids.
- Pathobiont
-
Any organism that can cause harm to its host, but normally lives as a harmless symbiont.
Rights and permissions
About this article
Cite this article
Drew, G.C., Stevens, E.J. & King, K.C. Microbial evolution and transitions along the parasite–mutualist continuum. Nat Rev Microbiol 19, 623–638 (2021). https://doi.org/10.1038/s41579-021-00550-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41579-021-00550-7
This article is cited by
-
Commensal lifestyle regulated by a negative feedback loop between Arabidopsis ROS and the bacterial T2SS
Nature Communications (2024)
-
The defensome of complex bacterial communities
Nature Communications (2024)
-
Wastewater irrigation and Trichoderma colonization in tomato plants: effects on plant traits, antioxidant activity, and performance of the insect pest Macrosiphum euphorbiae
Environmental Science and Pollution Research (2024)
-
The Feather Moss Hylocomium splendens Affects the Transcriptional Profile of a Symbiotic Cyanobacterium in Relation to Acquisition and Turnover of Key Nutrients
Microbial Ecology (2024)
-
The genus Sodalis as a resource for understanding the multifaceted evolution of bacterial symbiosis in insects
Symbiosis (2024)