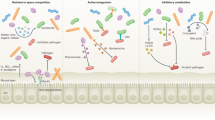
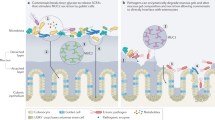
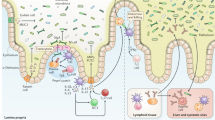
Abstract
The intestinal mucosal barrier is composed of epithelial cells that are protected by an overlying host-secreted mucous layer and functions as the first line of defence against pathogenic and non-pathogenic microorganisms. Some microorganisms have evolved strategies to either survive in the mucosal barrier or circumvent it to establish infection. In this Review, we discuss the current state of knowledge of the complex interactions of commensal microorganisms with the intestinal mucosal barrier, and we discuss strategies used by pathogenic microorganisms to establish infection by either exploiting different epithelial cell lineages or disrupting the mucous layer, as well as the role of defects in mucus production in chronic disease.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Savage, D. C. Microbial ecology of the gastrointestinal tract. Annu. Rev. Microbiol. 31, 107–133 (1977).
Qin, J. et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65 (2010).
Koenig, J. E. et al. Succession of microbial consortia in the developing infant gut microbiome. Proc. Natl Acad. Sci. USA 108, 4578–4585 (2011).
Palmer, C., Bik, E. M., DiGiulio, D. B., Relman, D. A. & Brown, P. O. Development of the human infant intestinal microbiota. PLoS Biol. 5, e177 (2007).
Yassour, M. et al. Natural history of the infant gut microbiome and impact of antibiotic treatment on bacterial strain diversity and stability. Sci. Transl. Med. 8, 343ra81–343ra81 (2016).
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, 559–563 (2014). This study demonstrates that the gut microbiota reacts quickly to shifts in diet that alter the amount of plant material versus animal protein and that similar taxa are responsive to diet across individual humans.
Sonnenburg, E. D. et al. Diet-induced extinctions in the gut microbiota compound over generations. Nature 529, 212–215 (2016). This proof-of-principle study demonstrates that prolonged dietary fibre starvation in mice results in reduced diversity within a generation, and this loss of diversity becomes irreparable and more severe over multiple generations.
El Kaoutari, A., Armougom, F., Gordon, J. I., Raoult, D. & Henrissat, B. The abundance and variety of carbohydrate-active enzymes in the human gut microbiota. Nat. Rev. Microbiol. 11, 497–504 (2013).
Furusawa, Y. et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 504, 446–450 (2013). This study demonstrates that the bacterial metabolite butyrate induces the differentiation of T reg cells and, furthermore, is able to ameliorate colitis in mice, providing novel insight into the interactions between the microbiota and host immune homeostasis.
Kim, M. H., Kang, S. G., Park, J. H., Yanagisawa, M. & Kim, C. H. Short-chain fatty acids activate GPR41 and GPR43 on intestinal epithelial cells to promote inflammatory responses in mice. Gastroenterology 145, 396–406 (2013).
Kaiko, G. E. et al. The colonic crypt protects stem cells from microbiota-derived metabolites. Cell 165, 1708–1720 (2016).
den Besten, G. et al. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 54, 2325–2340 (2013).
Louis, P., Hold, G. L. & Flint, H. J. The gut microbiota, bacterial metabolites and colorectal cancer. Nat. Rev. Microbiol. 12, 661–672 (2014).
Turner, J. R. Intestinal mucosal barrier function in health and disease. Nat. Rev. Immunol. 9, 799–809 (2009).
Peterson, L. W. & Artis, D. Intestinal epithelial cells: regulators of barrier function and immune homeostasis. Nat. Rev. Immunol. 14, 141–153 (2014).
Johansson, M. E. V. & Hansson, G. C. Immunological aspects of intestinal mucus and mucins. Nat. Rev. Immunol. 16, 639–649 (2016).
Gerbe, F., Legraverend, C. & Jay, P. The intestinal epithelium tuft cells: specification and function. Cell. Mol. Life Sci. 69, 2907–2917 (2012).
Barr, J. J. et al. Bacteriophage adhering to mucus provide a non-host-derived immunity. Proc. Natl Acad. Sci. USA 110, 10771–10776 (2013).
Pabst, O. New concepts in the generation and functions of IgA. Nat. Rev. Immunol. 12, 821–832 (2012).
Kommineni, S. et al. Bacteriocin production augments niche competition by enterococci in the mammalian gastrointestinal tract. Nature 526, 719–722 (2015).
Tailford, L. E., Crost, E. H., Kavanaugh, D. & Juge, N. Mucin glycan foraging in the human gut microbiome. Front. Genet. 6, 81 (2015).
McGuckin, M. A., Lindén, S. K., Sutton, P. & Florin, T. H. Mucin dynamics and enteric pathogens. Nat. Rev. Microbiol. 9, 265–278 (2011).
Cameron, E. & Sperandio, V. Frenemies: signaling and nutritional integration in pathogen-microbiota-host interactions. Cell Host Microbe 18, 275–284 (2015).
Tanoue, T., Atarashi, K. & Honda, K. Development and maintenance of intestinal regulatory T cells. Nat. Rev. Immunol. 16, 295–309 (2016).
Iliev, I. D. & Leonardi, I. Fungal dysbiosis: immunity and interactions at mucosal barriers. Nat. Rev. Immunol. 17, 635–646 (2017).
Darwich, A. S., Aslam, U., Ashcroft, D. M. & Rostami-Hodjegan, A. Meta-analysis of the turnover of intestinal epithelia in preclinical animal species and humans. Drug Metab. Dispos. 42, 2016–2022 (2014).
Johansson, M. E. V., Larsson, J. M. H. & Hansson, G. C. The two mucus layers of colon are organized by the MUC2 mucin, whereas the outer layer is a legislator of host-microbial interactions. Proc. Natl Acad. Sci. USA 108, 4659–4665 (2011).
Li, H. et al. The outer mucus layer hosts a distinct intestinal microbial niche. Nat. Commun. 6, 8292 (2015). This study separates the microbial populations and physiological responses of mucus-associated and luminal bacteria, demonstrating distinct microbiota and behaviours in these niches.
Wrzosek, L. et al. Bacteroides thetaiotaomicron and Faecalibacterium prausnitzii influence the production of mucus glycans and the development of goblet cells in the colonic epithelium of a gnotobiotic model rodent. BMC Biol. 11, 61 (2013).
Birchenough, G. M. H., Nystrom, E. E. L., Johansson, M. E. V. & Hansson, G. C. A sentinel goblet cell guards the colonic crypt by triggering Nlrp6-dependent Muc2 secretion. Science 352, 1535–1542 (2016). This is a novel study that reveals differentiation of crypt goblet cells into distinct roles, including a TLR-expressing sentinel goblet cell that senses microbial pathogen-associated molecular patterns.
Johansson, M. E. V. et al. Normalization of host intestinal mucus layers requires long-term microbial colonization. Cell Host Microbe 18, 582–592 (2015).
Velcich, A. et al. Colorectal cancer in mice genetically deficient in the mucin Muc2. Science 295, 1726–1729 (2002). This is a foundational study in which MUC2 was eliminated in mice to demonstrate that deficiency of MUC2 leads to development of colorectal cancer.
Van der Sluis, M. et al. Muc2-deficient mice spontaneously develop colitis, indicating that MUC2 is critical for colonic protection. Gastroenterology 131, 117–129 (2006).
Johansson, M. E. V., Sjövall, H. & Hansson, G. C. The gastrointestinal mucus system in health and disease. Nat. Rev. Gastroenterol. Hepatol. 10, 352–361 (2013).
Earle, K. A. et al. Quantitative imaging of gut microbiota spatial organization. Cell Host Microbe 18, 478–488 (2015). This is a seminal study that both innovates microscopic techniques for visualizing gut bacteria and shows low fibre-induced thinning of the colonic mucous layer.
Desai, M. S. et al. A dietary fiber-deprived gut microbiota degrades the colonic mucus barrier and enhances pathogen susceptibility. Cell 167, 1339–1353 (2016). This study demonstrates that dietary fibre deprivation leads to mucus thinning and increased pathogen susceptibility in mice.
Porter, N. T. & Martens, E. C. The critical roles of polysaccharides in gut microbial ecology and physiology. Annu. Rev. Microbiol. 71, 349–369 (2017).
Koropatkin, N. M., Cameron, E.a & Martens, E. C. How glycan metabolism shapes the human gut microbiota. Nat. Rev. Microbiol. 10, 323–335 (2012).
Hutkins, R. W. et al. Prebiotics: why definitions matter. Curr. Opin. Biotechnol. 37, 1–7 (2016).
Martens, E. C. et al. Recognition and degradation of plant cell wall polysaccharides by two human gut symbionts. PLoS Biol. 9, e1001221 (2011).
Cuskin, F. et al. Human gut bacteroidetes can utilize yeast mannan through a selfish mechanism. Nature 517, 165–169 (2015).
Larsbrink, J. et al. A discrete genetic locus confers xyloglucan metabolism in select human gut bacteroidetes. Nature 506, 498–502 (2014).
Ndeh, D. et al. Complex pectin metabolism by gut bacteria reveals novel catalytic functions. Nature 544, 65–70 (2017).
Sonnenburg, E. D. et al. Specificity of polysaccharide use in intestinal bacteroides species determines diet-induced microbiota alterations. Cell 141, 1241–1252 (2010).
Nihira, T. et al. Discovery of β-1,4-d-mannosyl- N-acetyl-d-glucosamine phosphorylase involved in the metabolism of N-glycans. J. Biol. Chem. 288, 27366–27374 (2013).
Martens, E. C., Kelly, A. G., Tauzin, A. S. & Brumer, H. The devil lies in the details: how variations in polysaccharide fine-structure impact the physiology and evolution of gut microbes. J. Mol. Biol. 426, 3851–3865 (2014).
Martens, E. C., Chiang, H. C. & Gordon, J. I. Mucosal glycan foraging enhances fitness and transmission of a saccharolytic human gut bacterial symbiont. Cell Host Microbe 4, 447–457 (2008). This is a study that initially probes the Bacteroides mechanisms of host O -linked and N -linked glycan degradation, linking these functions to colonization and transmission between generations.
Nakjang, S., Ndeh, D. A., Wipat, A., Bolam, D. N. & Hirt, R. P. A novel extracellular metallopeptidase domain shared by animal host-associated mutualistic and pathogenic microbes. PLoS ONE 7, e30287 (2012).
Noach, I. et al. Recognition of protein-linked glycans as a determinant of peptidase activity. Proc. Natl Acad. Sci. USA 114, 1–10 (2017).
Owen, C. D. et al. Unravelling the specificity and mechanism of sialic acid recognition by the gut symbiont Ruminococcus gnavus. Nat. Commun. 8, 2196 (2017).
Crost, E. H. et al. The mucin-degradation strategy of Ruminococcus gnavus: the importance of intramolecular trans-sialidases. Gut Microbes 7, 302–312 (2016).
Davis, C. P. & Savage, D. C. Habitat, succession, attachment, and morphology of segmented, filamentous microbes indigenous to the murine gastrointestinal tract. Infect. Immun. 10, 948–956 (1974).
Lee, S. M. et al. Bacterial colonization factors control specificity and stability of the gut microbiota. Nature 501, 426–429 (2013). This is a novel study showing that the order of gut colonization affects the ability to compete, even among isogenic strains of the same species, and that functions that govern crypt colonization contribute to colonization advantage.
Whitaker, W. R., Shepherd, E. S. & Sonnenburg, J. L. Tunable expression tools enable single-cell strain distinction in the gut microbiome. Cell 169, 538–546.e12 (2017).
Pedron, T. et al. A crypt-specific core microbiota resides in the mouse colon. mBio 3, e00116–e00112 (2012).
Sonnenberg, G. F. et al. Innate lymphoid cells promote anatomical containment of lymphoid-resident commensal bacteria. Science 336, 1321–1325 (2012).
Vaishnava, S. et al. The antibacterial lectin RegIIIγ promotes the spatial segregation of microbiota and host in the intestine. Science 334, 255–258 (2011). This paper demonstrates the importance of the antimicrobial protein REG3γ to avoid close bacterial contact with small intestinal epithelium.
Hase, K. et al. Uptake through glycoprotein 2 of FimH+ bacteria by M cells initiates mucosal immune response. Nature 462, 226–230 (2009).
Clark, M. A., Jepson, M. A., Simmons, N. L. & Hirts, B. H. Preferential interaction of Salmonella typhimurium with mouse Peyer's patch M cells. Res. Microbiol. 145, 543–552 (1994).
Rescigno, M. et al. Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nat. Immunol. 2, 361–367 (2001).
Marra, A. & Isberg, R. R. Invasin-dependent and invasin-independent pathways for translocation of Yersinia pseudotuberculosis across the Peyer’s patch intestinal epithelium. Infect. Immun. 65, 3412–3421 (1997).
Rankin, S., Isberg, R. R. & Leong, J. M. The integrin-binding domain of invasin is sufficient to allow bacterial entry into mammalian cells. Infect. Immun. 60, 3909–3912 (1992).
Clark, M. A., Hirst, B. H. & Jepson, M. A. M-Cell surface β1 integrin expression and invasin-mediated targeting of Yersinia pseudotuberculosis to mouse Peyer’s patch M cells. Infect. Immun. 66, 1237–1243 (1998).
Secott, T. E., Lin, T. L. & Wu, C. C. Mycobacterium avium subsp. paratuberculosis fibronectin attachment protein facilitates M-cell targeting and invasion through a fibronectin bridge with host integrins. Infect. Immun. 72, 3724–3732 (2004).
Shen, Y., Naujokas, M., Park, M. & Ireton, K. InIB-dependent internalization of Listeria is mediated by the Met receptor tyrosine kinase. Cell 103, 501–510 (2000).
Mengaud, J., Ohayon, H., Gounon, P., Mege, R. M. & Cossart, P. E-Cadherin is the receptor for internalin, a surface protein required for entry of L. monocytogenes into epithelial cells. Cell 84, 923–932 (1996).
Sousa, S. et al. Src, cortactin and Arp2/3 complex are required for E-cadherin-mediated internalization of Listeria into cells. Cell. Microbiol. 9, 2629–2643 (2007).
Kocks, C. et al. L. monocytogenes-induced actin assembly requires the actA gene product, a surface protein. Cell 68, 521–531 (1992).
Silva, A. J., Pham, K. & Benitez, J. A. Haemagglutinin/protease expression and mucin gel penetration in El Tor biotype Vibrio cholerae. Microbiology 149, 1883–1891 (2003).
Wu, Z., Nybom, P. & Magnusson, K.-E. Distinct effects of Vibrio cholerae haemagglutinin/protease on the structure and localization of the tight junction-associated proteins occludin and ZO-1. Cell. Microbiol. 2, 11–17 (2000).
Fasano, A. et al. Vibrio cholerae produces a second enterotoxin, which affects intestinal tight junctions. Proc. Natl Acad. Sci. USA 88, 5242–5246 (1991).
Saitoh, Y. et al. Tight junctions. Structural insight into tight junction disassembly by Clostridium perfringens enterotoxin. Science 347, 775–778 (2015).
Amat, C. B., Motta, J.-P., Chadee, K. & Buret, A. G. Giardia duodenalis directly depletes mucins in intestinal goblet cells. FASEB J 30, 162.1–162.1 (2016).
Maia-Brigagão, C., Morgado-Díaz, J. A. & De Souza, W. Giardia disrupts the arrangement of tight, adherens and desmosomal junction proteins of intestinal cells. Parasitol. Int. 61, 280–287 (2012).
Teoh, Da, Kamieniecki, D., Pang, G. & Buret, A. G. Giardia lamblia rearranges F-actin and alpha-actinin in human colonic and duodenal monolayers and reduces transepithelial electrical resistance. J. Parasitol. 86, 800–806 (2000).
Nikitas, G. et al. Transcytosis of Listeria monocytogenes across the intestinal barrier upon specific targeting of goblet cell accessible E-cadherin. J. Exp. Med. 208, 2263–2277 (2011).
Zhou, Z. et al. Lipid A modifications in polymyxin-resistant Salmonella typhimurium: PmrA-dependent 4-amino-4-deoxy- L-arabinose, and phosphoethanolamine incorporation. J. Biol. Chem. 276, 43111–43121 (2001).
Gunn, J. S. et al. PmrA-PmrB-regulated genes necessary for 4-aminoarabinose lipid A modification and polymyxin resistance. Mol. Microbiol. 27, 1171–1182 (1998).
Vaara, M. et al. Characterization of the lipopolysaccharide from the polymyxin-resistant pmrA mutants of Salmonella typhimurium. FEBS Lett. 129, 145–149 (1981).
Peschel, A. How do bacteria resist human antimicrobial peptides? Trends Microbiol. 10, 179–186 (2002).
Llobet, E., Tomas, J. M. & Bengoechea, J. A. Capsule polysaccharide is a bacterial decoy for antimicrobial peptides. Microbiology 154, 3877–3886 (2008).
Tan, Y., Zanoni, I., Cullen, T. W., Goodman, A. L. & Kagan, J. C. Mechanisms of toll-like receptor 4 endocytosis reveal a common immune-evasion strategy used by pathogenic and commensal bacteria. Immunity 43, 909–922 (2015).
Cullen, T. W. et al. Antimicrobial peptide resistance mediates resilience of prominent gut commensals during inflammation. Scince 347, 170–175 (2015).
Kim, Y. G. et al. Neonatal acquisition of Clostridia species protects against colonization by bacterial pathogens. Science 356, 315–319 (2017).
Byndloss, M. X. et al. Microbiota-activated PPAR-γ signaling inhibits dysbiotic Enterobacteriaceae expansion. Science 357, 570–575 (2017).
Bergstrom, K. S. B. et al. Muc2 protects against lethal infectious colitis by disassociating pathogenic and commensal bacteria from the colonic mucosa. PLoS Pathog. 6, e1000902 (2010).
Hasnain, S. Z. et al. Mucin gene deficiency in mice impairs host resistance to an enteric parasitic infection. Gastroenterology 138, 1763–1771 (2010).
Turner, J. E., Stockinger, B. & Helmby, H. IL-22 mediates goblet cell hyperplasia and worm expulsion in intestinal helminth infection. PLoS Pathog. 9, 1–7 (2013).
Zheng, Y. et al. Interleukin-22 mediates early host defense against attaching and effacing bacterial pathogens. Nat. Med. 14, 282–289 (2008).
Behnsen, J. et al. The cytokine IL-22 promotes pathogen colonization by suppressing related commensal bacteria. Immunity 40, 262–273 (2014).
Erdem, A. L., Avelino, F., Xicohtencatl-Cortes, J. & Girón, J. A. Host protein binding and adhesive properties of H6 and H7 flagella of attaching and effacing Escherichia coli. J. Bacteriol. 189, 7426–7435 (2007).
Lawley, T. D. et al. Antibiotic treatment of Clostridium difficile carrier mice triggers a supershedder state, spore-mediated transmission, and severe disease in immunocompromised hosts. Infect. Immun. 77, 3661–3669 (2009).
Tasteyre, A., Barc, M. C., Collignon, A., Boureau, H. & Karjalainen, T. Role of FliC and FliD flagellar proteins of Clostridium difficile in adherence and gut colonization. Infect. Immun. 69, 7937–7940 (2001).
Purcell, E. B., McKee, R. W., McBride, S. M., Waters, C. M. & Tamayo, R. Cyclic diguanylate inversely regulates motility and aggregation in Clostridium difficile. J. Bacteriol. 194, 3307–3316 (2012).
Li, S., Konstantinov, S. R., Smits, R. & Peppelenbosch, M. P. Bacterial biofilms in colorectal cancer initiation and progression. Trends Mol. Med. 23, 18–30 (2017).
Dejea, C. M. et al. Patients with familial adenomatous polyposis harbor colonic biofilms containing tumorigenic bacteria. Science 359, 592–597 (2018).
Grys, T. E., Siegel, M. B., Lathem, W. W. & Welch, R. A. The StcE protease contributes to intimate adherence of enterohemorrhagic Escherichia coli O157:H7 to host cells. Infect. Immun. 73, 1295–1303 (2005).
Hews, C. L. et al. The StcE metalloprotease of enterohaemorrhagic Escherichia coli reduces the inner mucus layer and promotes adherence to human colonic epithelium ex vivo. Cell. Microbiol. 19, e12717 (2017).
Valeri, M. et al. Pathogenic E. coli exploits SslE mucinase activity to translocate through the mucosal barrier and get access to host cells. PLoS One 10, 1–14 (2015).
Moncada, D., Keller, K. & Chadee, K. Entamoeba histolytica cysteine proteinases disrupt the polymeric structure of colonic mucin and alter its protective function. Infect. Immun. 71, 838–844 (2003). This is a study that reveals that pathogens can target critical sites in the MUC2 network to dissolve its important barrier function.
Lidell, M. E., Moncada, D. M., Chadee, K. & Hansson, G. C. Entamoeba histolytica cysteine proteases cleave the MUC2 mucin in its C-terminal domain and dissolve the protective colonic mucus gel. Proc. Natl Acad. Sci. USA 103, 9298–9303 (2006).
Chadee, K., Petri, W. A., Innes, D. J. & Ravdin, J. I. Rat and human colonic mucins bind to and inhibit adherence lectin of Entamoeba histolytica. J. Clin. Invest. 80, 1245–1254 (1987).
Hasnain, S. Z., McGuckin, M. A., Grencis, R. K. & Thornton, D. J. Serine protease(s) secreted by the nematode Trichuris muris degrade the mucus barrier. PLoS Negl. Trop. Dis. 6, e1856 (2012).
Nusrat, A. Turner, J. R., Verkade, P., Madara, L. & Parkos, C. A. Clostridium difficile toxins disrupt epithelial barrier function by altering membrane microdomain localization of tight junction proteins. Infect. Immun. 69, 1329–1336 (2001).
Guttman, J. A. et al. Attaching and effacing pathogen-induced tight junction disruption in vivo. Cell Microbiol 8, 634–645 (2006).
Simonovic, I., Rosenberg, J., Koutsouris, A. & Hecht, G. Enteropathogenic Escherichia coli dephosphorylates and dissociates occludin from intestinal epithelial tight junctions. Cell. Microbiol. 2, 305–315 (2000).
Elmi, A. et al. Campylobacter jejuni outer membrane vesicle-associated proteolytic activity promotes bacterial invasion by mediating cleavage of intestinal epithelial cell E-cadherin and occludin. Cell. Microbiol. 18, 561–572 (2016).
Ng, K. M. et al. Microbiota-liberated host sugars facilitate post-antibiotic expansion of enteric pathogens. Nature 502, 96–99 (2013). This study connects the mucin-foraging activity of commensal B. thetaiotaomicron with the release of sialic acid, which benefits the pathogens C. difficile and S . Typhimurium.
Pacheco, A. R. et al. Fucose sensing regulates bacterial intestinal colonization. Nature 492, 113–117 (2012).
Bry, L., Falk, P. G., Midtvedt, T. & Gordon, J. I. A model of host-microbial interactions in an open mammalian ecosystem. Science 273, 1380–1383 (1996).
Engevik, M. A. et al. Loss of NHE3 alters gut microbiota composition and influences bacteroides thetaiotaomicron growth. Am. J. Physiol. Gastrointest. Liver Physiol. 305, G697–G711 (2013).
Schumann, M., Siegmund, B., Schulzke, J. D. & Fromm, M. Celiac disease: role of the epithelial barrier. Cell. Mol. Gastroenterol. Hepatol. 3, 150–162 (2017).
Wang, Y. et al. Cosmc is an essential chaperone for correct protein O-glycosylation. Proc. Natl Acad. Sci. USA 107, 9228–9233 (2010).
Bergstrom, K. et al. Core 1– and 3–derived O-glycans collectively maintain the colonic mucus barrier and protect against spontaneous colitis in mice. Mucosal Immunol. 10, 91–103 (2017).
Fu, J. et al. Loss of intestinal core 1–derived O-glycans causes spontaneous colitis in mice. J. Clin. Invest. 121, 1657–1666 (2011). This is a critical study that demonstrates that defects in MUC2 glycosylation in mice, which are similar to those observed in some humans, promote spontaneous inflammation.
Johansson, M. E. V. et al. Bacteria penetrate the normally impenetrable inner colon mucus layer in both murine colitis models and patients with ulcerative colitis. Gut 63, 281–291 (2014).
Pullan, R. D. et al. Thickness of adherent mucus gel on colonic mucosa in humans and its relevance to colitis. Gut 35, 353–359 (1994).
Schroeder, B. O. et al. Bifidobacteria or fiber protects against diet-induced microbiota-mediated colonic mucus deterioration. Cell Host Microbe 23, 27–40.e7 (2017). This study demonstrates that a low-fibre, Western-style diet promotes increased permeability of the inner mucous layer that can be reversed by prebiotics and probiotics.
Palm, N. W. et al. Immunoglobulin A coating identifies colitogenic bacteria in inflammatory bowel disease. Cell 158, 1000–1010 (2014). This is an important study establishing that a subset of microbiota species is coated with IgA and that these species elicit increased host inflammatory responses when separately introduced into germ-free mice.
Bergstrom, K. S. B. & Xia, L. Mucin-type O-glycans and their roles in intestinal homeostasis. Glycobiology 23, 1026–1037 (2013).
Ju, T. et al. Human tumor antigens Tn and sialyl Tn arise from mutations in Cosmc. Cancer Res. 68, 1636–1646 (2008).
An, G. et al. Increased susceptibility to colitis and colorectal tumors in mice lacking core 3–derived O-glycans. J. Exp. Med. 204, 1417–1429 (2007).
Sommer, F. et al. Altered mucus glycosylation in core 1 O-glycan-deficient mice affects microbiota composition and intestinal architecture. PLoS One 9, e85254 (2014).
Zhu, W. et al. Precision editing of the gut microbiota ameliorates colitis. Nature 553, 208–211 (2018).
Zou, J. et al. Fiber-mediated nourishment of gut microbiota protects against diet-induced obesity by restoring IL-22-mediated colonic health. Cell Host Microbe 23, 41–53 (2017).
Macpherson, aJ., McCoy, K. D., Johansen, F.-E. & Brandtzaeg, P. The immune geography of IgA induction and function. Mucosal Immunol. 1, 11–22 (2008).
Moor, K. et al. High-avidity IgA protects the intestine by enchaining growing bacteria. Nature 544, 498–502 (2017).
Okumura, R. et al. Lypd8 promotes the segregation of flagellated microbiota and colonic epithelia. Nature 532, 117–121 (2016).
Propheter, D. C., Chara, A. L., Harris, T. A., Ruhn, K. A. & Hooper, L. V. Resistin-like molecule β is a bactericidal protein that promotes spatial segregation of the microbiota and the colonic epithelium. Proc. Natl Acad. Sci. 114, 11027–11033 (2017).
Akira, S. & Takeda, K. Toll-like receptor signalling. Nat. Rev. Immunol. 4, 499–511 (2004).
Fukata, M. Toll-like receptor-4 is required for intestinal response to epithelial injury and limiting bacterial translocation in a murine model of acute colitis. Am. J. Physiol. Gastrointest. Liver Physiol. 288, G1055–G1065 (2005).
Danne, C. et al. A large polysaccharide produced by helicobacter hepaticus induces an anti-inflammatory gene signature in macrophages. Cell Host Microbe 22, 733–745.e5 (2017).
Chung, L. et al. Bacteroides fragilis toxin coordinates a pro-carcinogenic inflammatory cascade via targeting of colonic epithelial cells. Cell Host Microbe 23, 203–214.e5 (2018).
Frantz, A. L. et al. Targeted deletion of MyD88 in intestinal epithelial cells results in compromised antibacterial immunity associated with downregulation of polymeric immunoglobulin receptor, mucin-2, and antibacterial peptides. Mucosal Immunol. 5, 501–512 (2012).
Xie, L. et al. Targeting of MyD88 homodimerization by novel synthetic inhibitor TJ-M2010-5 in preventing colitis-associated colorectal cancer. J. Natl Cancer Inst. 108, 1–12 (2016).
Carvalho, F. A. et al. Transient inability to manage proteobacteria promotes chronic gut inflammation in TLR5-deficient mice. Cell Host Microbe 12, 139–152 (2012).
Zhan, Y. et al. Gut microbiota protects against gastrointestinal tumorigenesis caused by epithelial injury. Cancer Res. 73, 7199–7210 (2013).
Smith, P. M. et al. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 341, 569–573 (2013).
Singh, N. et al. Activation of the receptor (Gpr109a) for niacin and the commensal metabolite butyrate suppresses colonic inflammation and carcinogenesis. Immunity 40, 128–139 (2014).
Zhang, M. et al. Butyrate inhibits interleukin-17 and generates Tregs to ameliorate colorectal colitis in rats. BMC Gastroenterol. 16, 84 (2016).
Ivanov, I. I. et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 139, 485–498 (2009).
Fukuda, S. et al. Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature 469, 543–547 (2011).
Howitt, M. R. et al. Tuft cells, taste-chemosensory cells, orchestrate parasite type 2 immunity in the gut. Science 351, 1329–1333 (2016).
Gerbe, F. et al. Intestinal epithelial tuft cells initiate type 2 mucosal immunity to helminth parasites. Nature 529, 226–230 (2016).
von Moltke, J., Ji, M., Liang, H.-E. & Locksley, R. M. Tuft-cell-derived IL-25 regulates an intestinal ILC2-epithelial response circuit. Nature 529, 221–225 (2016).
Holmén Larsson, J. M., Thomsson, K. A., Rodríguez-Piñeiro, A. M., Karlsson, H. & Hansson, G. C. Studies of mucus in mouse stomach, small intestine, and colon. III. Gastrointestinal Muc5ac and Muc2 mucin O-glycan patterns reveal a regiospecific distribution. Am. J. Physiol. Gastrointest. Liver Physiol. 305, G357–G363 (2013).
Bennett, E. P. et al. Control of mucin-type O-glycosylation: a classification of the polypeptide GalNAc-transferase gene family. Glycobiology 22, 736–756 (2012).
Brockhausen, I., Schachter, H., S. P. Essentials of Glycobiology (Cold Spring Harbor Laboratory Press, 2009).
Lawhon, S. D., Maurer, R., Suyemoto, M. & Altier, C. Intestinal short-chain fatty acids alter Salmonella typhimurium invasion gene expression and virulence through BarA/SirA. Mol. Microbiol. 46, 1451–1464 (2002).
Gantois, I. et al. Butyrate specifically down-regulates Salmonella pathogenicity island 1 gene expression. Appl. Environ. Microbiol. 72, 946–949 (2006).
Hung, C. C. et al. The intestinal fatty acid propionate inhibits Salmonella invasion through the post-translational control of HilD. Mol. Microbiol. 87, 1045–1060 (2013).
Ferreyra, J. A. et al. Gut microbiota-produced succinate promotes C. difficile infection after antibiotic treatment or motility disturbance. Cell Host Microbe 16, 770–777 (2014).
Curtis, M. M. et al. The gut commensal Bacteroides thetaiotaomicron exacerbates enteric infection through modification of the metabolic landscape. Cell Host Microbe 16, 759–769 (2014).
Tobe, T., Nakanishi, N. & Sugimoto, N. Activation of motility by sensing short-chain fatty acids via two steps in a flagellar gene regulatory cascade in enterohemorrhagic Escherichia coli. Infect. Immun. 79, 1016–1024 (2011).
Acknowledgements
Work in the authors’ laboratories was supported by National Institutes of Health R01 and R21 (GM099513 and AI128120) awards (to E.C.M.) and an Innovator Award from the Kenneth Rainin Foundation. Work in their laboratories was also supported by the following grants (to M.S.D.): Luxembourg National Research Fund (FNR) CORE (C15/BM/10318186); FNR AFR Bilateral (11228353); Luxembourg Ministry of Higher Education and Research support (DM-Muc); and Personalized Medicine Consortium of Luxembourg Pump Prime (Die-IBD).
Author information
Authors and Affiliations
Contributions
E.C.M., M.N. and M.S.D. researched data for the article, discussed the content, wrote the article, and reviewed and edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Commensal
-
Refers to symbiotic microorganisms that do not seem to harm (pathogens) or benefit (mutualists) their host. Commensal is arguably an imprecise term to apply to a microorganism, especially considering that any microorganism may display behaviours that are both beneficial and detrimental.
- Microbiota
-
A community of microorganisms, such as the taxonomically diverse and densely populated assemblage of species that inhabits the human gut.
- Inflammatory bowel disease
-
(IBD). A group of chronic diseases characterized by episodes of relapsing and remitting inflammation. Crohn’s disease and ulcerative colitis are the most common types of IBDs and are thought to develop from a combination of host genetic predisposition, including defects in the gut mucosal and immunological barriers, and environmental factors, such as diet and the microbiota.
- Tight junctions
-
Multi-protein complexes that form near the apical ends of intestinal epithelial cells, providing tight, water-impermeable and ion-impermeable seals between cells. Proteins that are included in tight junctions include claudins and occludins, which span the membrane and are anchored together outside the cell and are connected to the intracellular cytoskeleton.
- Mucous layer
-
Mucus, composed mostly of secreted mucin glycoproteins and other substances, is secreted by goblet cells and overlies the intestinal epithelium, forming an adherent and insoluble inner layer and a looser outer layer in the colon.
- Tuft cells
-
Taste-chemosensory cells that are present in the intestinal epithelium and have an important role in initiating a type 2 immune response to clear parasitic infection. They have a unique appearance that includes a tubulovesicular system and an apical bundle of microfilaments that are attached to a tuft of long, lumen-facing microvilli.
- Glycoproteins
-
Class of proteins comprising oligosaccharide chains connected to a backbone polypeptide, sometimes thousands of amino acids long, which is modified with glycans in the endoplasmic reticulum. Mucins and other glycoproteins contain O-linked and N-linked glycans attached to serine or threonine and asparagine, respectively.
- Secretory IgA
-
Immunoglobulin A (IgA) is the most abundant antibody isotype in the human body and occurs in two subtypes: IgA1 and IgA2, which are distinguished by their heavy chains. IgA1 is predominant in the serum, and IgA2 is most abundant in the intestinal tract to bind pathogens by reducing mobility and decreasing proliferation.
- Carbohydrate-active enzymes
-
(CAZymes). This term encompasses several different groups of enzymes: glycoside hydrolases, polysaccharide lyases, carbohydrate esterases and glycosyltransferases. These groups, and hundreds of more specific enzyme families that they collectively contain, are involved in building and degrading polysaccharides and glycans.
- Prebiotics
-
Dietary supplements (most often oligosaccharide or polysaccharide fibres) that selectively increase certain taxonomic groups in the gut microbiota and also exert a positive impact on host health.
- Hidden Markov models
-
A statistical approach that is often used in bioinformatics to identify hidden, but meaningful, amino acid sequence signals in proteins and can be used to group them into families with similar function.
- Interleukin-22
-
(IL-22). A cytokine that is part of the IL-10 cytokine family that is produced by activated natural killer and T cells and, in the gut, is responsible for activating epithelial cell regeneration, mucin production and antimicrobial peptide secretion.
- Innate lymphoid cells
-
(ILCs). A group of innate immune cells that are characterized by the absence of antigen-specific B or T cell receptors.
- Colonization resistance
-
A phenomenon by which the presence of a healthy microbiota inhibits invasion by external pathogens. In the gut, this state has been ascribed to the inhibition of many enteric pathogens, which are more adept at gaining an infection foothold after a major perturbation, such as antibiotic treatment.
- Lectin
-
A broad family of plant, animal and bacterial proteins defined by their ability to bind to certain carbohydrates with moderate to high affinity.
- Type III secretion systems
-
(T3SSs). Needle-like complexes related to the bacterial flagellum that are an important virulence factor of Gram-negative bacteria. They enable pathogenic bacteria to inject effector proteins into the cytoplasm of the host target cell and modify a variety of cellular responses.
- Molecular chaperone
-
Type of protein that assists in the folding and unfolding of other proteins. They prevent aggregation by binding to non-native structures and therefore support the folding process.
Rights and permissions
About this article
Cite this article
Martens, E.C., Neumann, M. & Desai, M.S. Interactions of commensal and pathogenic microorganisms with the intestinal mucosal barrier. Nat Rev Microbiol 16, 457–470 (2018). https://doi.org/10.1038/s41579-018-0036-x
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41579-018-0036-x
This article is cited by
-
Gut microbial features may influence antiviral IgG levels after vaccination against viral respiratory infectious diseases: the evidence from two-sample bidirectional mendelian randomization
BMC Infectious Diseases (2024)
-
Characterization of intestinal fungal community diversity in people living with HIV/AIDS (PLWHA)
AIDS Research and Therapy (2024)
-
Lung microbiome: new insights into the pathogenesis of respiratory diseases
Signal Transduction and Targeted Therapy (2024)
-
Neonatal intestinal mucus barrier changes in response to maturity, inflammation, and sodium decanoate supplementation
Scientific Reports (2024)
-
Electroacupuncture Improving Intestinal Barrier Function in Rats with Irritable Bowel Syndrome Through Regulating Aquaporins
Digestive Diseases and Sciences (2024)