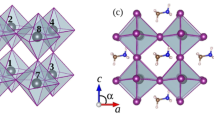
Abstract
Materials with the perovskite ABX3 structure play a major role across materials chemistry and physics as a consequence of their ubiquity and wide range of useful properties. ReO3-type structures can be described as ABX3 perovskites in which the A-cation site is unoccupied, giving rise to the general composition BX3, where B is typically a cation and X is a bridging anion. The chemical diversity of such structures is extensive, ranging from simple oxides and fluorides, such as WO3 and AlF3, to complex structures in which the bridging anion is polyatomic, such as in the Prussian blue-related cyanides Fe(CN)3 and CoPt(CN)6. The same ReO3-type structure is found in metal–organic frameworks, for example, In(im)3 (im = imidazolate) and the well-known MOF-5 structure, where the B-site cation is polyatomic. The extended 3D connectivity and openness of this structure type leads to compounds with interesting and often unusual properties. Notable among these properties are negative thermal expansion (for example, ScF3), photocatalysis (for example, CoSn(OH)6), thermoelectricity (for example, CoAs3) and superconductivity in a phase that is controversially described as SH3 with a doubly interpenetrating ReO3 structure. We present an account of this exciting family of materials and discuss future opportunities in the area.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Kojima, A., Teshima, K., Shirai, Y. & Miyasaka, T. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J. Am. Chem. Soc. 131, 6050–6051 (2009).
von Hippel, A. Ferroelectricity, domain structure, and phase transitions of barium titanate. Rev. Mod. Phys. 22, 221–237 (1950).
Callaghan, A., Moeller, C. W. & Ward, R. Magnetic interactions in ternary ruthenium oxides. Inorg. Chem. 5, 1572–1576 (1966).
Wu, M. K. et al. Superconductivity at 93 K in a new mixed-phase Y–Ba–Cu–O compound system at ambient pressure. Phys. Rev. Lett. 58, 908–910 (1987).
von Helmolt, R., Wecker, J., Holzapfel, B., Schultz, L. & Samwer, K. Giant negative magnetoresistance in perovskite-like La2/3Ba1/3MnOx ferromagnetic films. Phys. Rev. Lett. 71, 2331–2333 (1993).
Moreira dos Santos, A. et al. Evidence for the likely occurrence of magnetoferroelectricity in the simple perovskite, BiMnO3. Solid State Commun. 122, 49–52 (2002).
Cheetham, A. K. et al. Multiferroic behavior associated with an order–disorder hydrogen bonding transition in metal-organic frameworks (MOFs) with the perovskite ABX3 architecture. J. Am. Chem. Soc. 131, 13625–13627 (2009).
Ye, H.-Y. et al. Metal-free three-dimensional perovskite ferroelectrics. Science 361, 151–155 (2018).
Li, W. et al. Chemically diverse and multifunctional hybrid organic–inorganic perovskites. Nat. Rev. Mater. 2, 16099 (2017).
Woodward, P. M. Octahedral tilting in perovskites. II. Structure stabilizing forces. Acta Crystallogr. B 53, 44–66 (1997).
Glazer, A. M. The classification of tilted octahedra in perovskites. Acta Crystallogr. B 28, 3384–3392 (1972).
Woodward, P. M. Octahedral tilting in perovskites. I. Geometrical considerations. Acta Crystallogr. B 53, 32–43 (1997).
Howard, C. J. & Stokes, H. T. Group-theoretical analysis of octahedral tilting in perovskites. Acta Crystallogr. B 54, 782–789 (1998).
Duyker, S. G., Hill, J. A., Howard, C. J. & Goodwin, A. L. Guest-activated forbidden tilts in a molecular perovskite analogue. J. Am. Chem. Soc. 138, 11121–11123 (2016).
Boström, H. L. B., Hill, J. A. & Goodwin, A. L. Columnar shifts as symmetry-breaking degrees of freedom in molecular perovskites. Phys. Chem. Chem. Phys. 18, 31881–31894 (2016).
Li, H., Eddaoudi, M., O’Keeffe, M. & Yaghi, O. M. Design and synthesis of an exceptionally stable and highly porous metal–organic framework. Nature 402, 276–279 (1999).
Zheng, H. et al. Nanostructured tungsten oxide—properties, synthesis, and applications. Adv. Funct. Mater. 21, 2175–2196 (2011).
Ferretti, A., Rogers, D. B. & Goodenough, J. B. The relation of the electrical conductivity in single crystals of rhenium trioxide to the conductivities of Sr2MgReO6 and NaxWO3. J. Phys. Chem. Solids 26, 2007–2011 (1965).
Mattheiss, L. F. Band structure and Fermi surface of ReO3. Phys. Rev. 181, 987–1000 (1969).
Tanisaki, S. Crystal structure of monoclinic tungsten trioxide at room temperature. J. Phys. Soc. Jpn 15, 573–581 (1960).
Honig, J. M., Dimmock, J. O. & Kleiner, W. H. ReO3 band structure in the tight-binding approximation. J. Chem. Phys. 50, 5232–5242 (1969).
Sleight, A. & Gillson, J. Preparation and properties of alkali rhenium bronzes and a WO3–ReO3 solid solution. Solid State Commun. 4, 601–602 (1966).
Chatterji, T., Hansen, T. C., Brunelli, M. & Henry, P. F. Negative thermal expansion of ReO3 in the extended temperature range. Appl. Phys. Lett. 94, 241902 (2009).
Purans, J. et al. X-ray absorption spectroscopy study of local dynamics and thermal expansion in ReO3. Phys. Rev. B 92, 014302 (2015).
Takenaka, K. Negative thermal expansion materials: technological key for control of thermal expansion. Sci. Technol. Adv. Mater. 13, 013001 (2012).
Rodriguez, E. E. et al. The role of static disorder in negative thermal expansion in ReO3. J. Appl. Phys. 105, 114901 (2009).
Lind, C. Two decades of negative thermal expansion research: where do we stand? Materials 5, 1125–1154 (2012).
Corà, F., Stachiotti, M. G., Catlow, C. R. A. & Rodriguez, C. O. Transition metal oxide chemistry: electronic structure study of WO3, ReO3, and NaWO3. J. Chem. Phys. B 101, 3945–3952 (1997).
Bozin, E. S., Chatterji, T. & Billinge, S. J. L. Local structure of ReO3 at ambient pressure from neutron total-scattering study. Phys. Rev. B 86, 3–6 (2012).
Jorgensen, J.-E., Jorgensen, J. D., Batlogg, B., Remeika, J. P. & Axe, J. D. Order parameter and critical exponent for the pressure-induced phase transitions in ReO3. Phys. Rev. B 33, 4793–4798 (1986).
Schirber, J. E. & Morosin, B. “Compressibility collapse” transition in ReO3. Phys. Rev. Lett. 42, 1485–1487 (1979).
Biswas, K. et al. Pressure-induced phase transitions in nanocrystalline ReO3. J. Phys. Condens. Matter. 19, 436214 (2007).
Muthu, D. V. S. et al. Pressure-induced structural phase transitions and phonon anomalies in ReO3: Raman and first-principles study. Phys. Rev. B 91, 224308 (2015).
Cava, R., Santoro, A., Murphy, D., Zahurak, S. & Roth, R. The structures of lithium-inserted metal oxides: LiReO3 and Li2ReO3. J. Solid State Chem. 42, 251–262 (1982).
Cava, R., Santoro, A., Murphy, D., Zahurak, S. & Roth, R. The structures of the lithium inserted metal oxides Li0.2ReO3 and Li0.36WO3. J. Solid State Chem. 50, 121–128 (1983).
Bashian, N. H. et al. Correlated polyhedral rotations in the absence of polarons during electrochemical insertion of lithium in ReO3. ACS Energy Lett. 3, 2513–2519 (2018).
Santato, C., Odziemkowski, M., Ulmann, M. & Augustynski, J. Crystallographically oriented mesoporous WO3 films: synthesis, characterization, and applications. J. Am. Chem. Soc. 123, 10639–10649 (2001).
Cronin, J., Tarico, D., Tonazzi, J., Agrawal, A. & Kennedy, S. Microstructure and properties of sol–gel deposited WO3 coatings for large area electrochromic windows. Sol. Energy Mater. Sol. Cell 29, 371–386 (1993).
Sun, M., Xu, N., Cao, Y. W., Yao, J. N. & Wang, E. G. Nanocrystalline tungsten oxide thin film: preparation, microstructure, and photochromic behavior. J. Mater. Sci. 15, 927–933 (2000).
Lee, S.-H. et al. Crystalline WO3 nanoparticles for highly improved electrochromic applications. Adv. Mater. 18, 763–766 (2006).
Baeck, S.-H., Choi, K.-S., Jaramillo, T., Stucky, G. & McFarland, E. Enhancement of photocatalytic and electrochromic properties of electrochemically fabricated mesoporous WO3 thin films. Adv. Mater. 15, 1269–1273 (2003).
Aird, A. & Salje, E. K. H. Sheet superconductivity in twin walls: experimental evidence of. J. Phys. Condens. Matter. 10, L377–L380 (1998).
Wang, L., Teleki, A., Pratsinis, S. E. & Gouma, P. I. Ferroelectric WO3 nanoparticles for acetone selective detection. Chem. Mater. 20, 4794–4796 (2008).
Antonik, M. et al. Microstructural effects in WO3 gas-sensing films. Thin Solid Films 256, 247–252 (1995).
Galatsis, K., Li, Y., Wlodarski, W. & Kalantar-zadeh, K. Sol–gel prepared MoO3–WO3 thin-films for O2 gas sensing. Sens. Actuat. B Chem. 77, 478–483 (2001).
Li, X.-L., Lou, T.-J., Sun, X.-M. & Li, Y.-D. Highly sensitive WO3 hollow-sphere gas sensors. Inorg. Chem. 43, 5442–5449 (2004).
Ponzoni, A., Comini, E., Ferroni, M. & Sberveglieri, G. Nanostructured WO3 deposited by modified thermal evaporation for gas-sensing applications. Thin Solid Films 490, 81–85 (2005).
Ma, M. et al. Dual oxygen and tungsten vacancies on a WO3 photoanode for enhanced water oxidation. Angew. Chem. Int. Ed. 55, 11819–11823 (2016).
Zhang, J., Liu, Z. & Liu, Z. Novel WO3/Sb2S3 heterojunction photocatalyst based on WO3 of different morphologies for enhanced efficiency in photoelectrochemical water splitting. ACS Appl. Mater. Interfaces 8, 9684–9691 (2016).
Li, W. et al. WO3 nanoflakes for enhanced photoelectrochemical conversion. ACS Nano 8, 11770–11777 (2014).
Hou, Y., Zuo, F., Dagg, A. P., Liu, J. & Feng, P. Branched WO3 nanosheet array with layered C3N4 heterojunctions and CoOx nanoparticles as a flexible photoanode for efficient photoelectrochemical water oxidation. Adv. Mater. 26, 5043–5049 (2014).
Su, J., Feng, X., Sloppy, J. D., Guo, L. & Grimes, C. A. Vertically aligned WO3 nanowire arrays grown directly on transparent conducting oxide coated glass: synthesis and photoelectrochemical properties. Nano Lett. 11, 203–208 (2011).
Li, N. et al. Aqueous synthesis and visible-light photochromism of metastable h-WO3 hierarchical nanostructures. Eur. J. Inorg. Chem. 2015, 2804–2812 (2015).
Chen, Z. et al. Hierarchical nanostructured WO3 with biomimetic proton channels and mixed ionic–electronic conductivity for electrochemical energy storage. Nano Lett. 15, 6802–6808 (2015).
Yoon, S. et al. Development of a high-performance anode for lithium ion batteries using novel ordered mesoporous tungsten oxide materials with high electrical conductivity. Phys. Chem. Chem. Phys. 13, 11060 (2011).
Girish Kumar, S. & Koteswara Rao, K. Tungsten-based nanomaterials (WO3 & Bi2WO6): modifications related to charge carrier transfer mechanisms and photocatalytic applications. Appl. Surf. Sci. 355, 939–958 (2015).
Kida, T. et al. WO3 nanolamella gas sensor: porosity control using SnO2 nanoparticles for enhanced NO2 sensing. Langmuir 30, 2571–2579 (2014).
Amano, F., Ishinaga, E. & Yamakata, A. Effect of particle size on the photocatalytic activity of WO3 particles for water oxidation. J. Phys. Chem. C 117, 22584–22590 (2013).
Chen, D. & Ye, J. Hierarchical WO3 hollow shells: dendrite, sphere, dumbbell, and their photocatalytic properties. Adv. Funct. Mater. 18, 1922–1928 (2008).
Wang, H., Dong, X., Peng, S., Dong, L. & Wang, Y. Improvement of thermoelectric properties of WO3 ceramics by ZnO addition. J. Alloy. Compd 527, 204–209 (2012).
Kieslich, G. et al. Using crystallographic shear to reduce lattice thermal conductivity: high temperature thermoelectric characterization of the spark plasma sintered Magnéli phases WO2.90 and WO2.722. Phys. Chem. Chem. Phys. 15, 15399 (2013).
Kim, S.-J. et al. Mesoporous WO3 nanofibers with protein-templated nanoscale catalysts for detection of trace biomarkers in exhaled breath. ACS Nano 10, 5891–5899 (2016).
Shendage, S. et al. Sensitive and selective NO2 gas sensor based on WO3 nanoplates. Sens. Actuators B 240, 426–433 (2017).
Rao, P. M. et al. Simultaneously efficient light absorption and charge separation in WO3/BiVO4 core/shell nanowire photoanode for photoelectrochemical water oxidation. Nano Lett. 14, 1099–1105 (2014).
Tordjman, M., Weinfeld, K. & Kalish, R. Boosting surface charge-transfer doping efficiency and robustness of diamond with WO3 and ReO3. Appl. Phys. Lett. 111, 111601 (2017).
Juza, R. & Hahn, H. Über die Kristallstrukturen von Cu3N, GaN und InN Metallamide und Metallnitride. Z. Anorg. Allg. Chem. 239, 282–287 (1938).
Paniconi, G. et al. Structural chemistry of Cu3N powders obtained by ammonolysis reactions. Solid State Sci. 9, 907–913 (2007).
Zhao, J., You, S., Yang, L. & Jin, C. Structural phase transition of Cu3N under high pressure. Solid State Commun. 150, 1521–1524 (2010).
Birkett, M. et al. Atypically small temperature-dependence of the direct band gap in the metastable semiconductor copper nitride Cu3N. Phys. Rev. B 95, 115201 (2017).
Terada, S., Tanaka, H. & Kubota, K. Heteroepitaxial growth of Cu3N thin films. J. Cryst. Growth 94, 567–568 (1989).
Asano, M., Umeda, K. & Tasaki, A. Cu3N thin film for a new light recording media. Jpn J. Appl. Phys. 29, 1985–1986 (1990).
Juza, R. & Hahn, H. Kupfernitrid metallamide und metallnitride. VII. Z. Anorg. Allg. Chem. 241, 172–178 (1939).
Zakutayev, A. et al. Defect tolerant semiconductors for solar energy conversion. J. Phys. Chem. Lett. 5, 1117–1125 (2014).
Kim, Y., Wieder, B. J., Kane, C. L. & Rappe, A. M. Dirac line nodes in inversion-symmetric crystals. Phys. Rev. Lett. 115, 036806 (2015).
Pereira, N., Dupont, L., Tarascon, J. M., Klein, L. C. & Amatucci, G. G. Electrochemistry of Cu3N with lithium. J. Electrochem. Soc. 150, A1273 (2003).
Wu, H. & Chen, W. Copper nitride nanocubes: size-controlled synthesis and application as cathode catalyst in alkaline fuel cells. J. Am. Chem. Soc. 133, 15236–15239 (2011).
Fischer, D. & Jansen, M. Synthesis and structure of Na3N. Angew. Chem. Int. Ed. 41, 1755–1756 (2002).
Vajenine, G. V. Plasma-assisted synthesis and properties of Na3N. Inorg. Chem. 46, 5146–5148 (2007).
Vajenine, G. V., Hoch, C., Dinnebier, R. E., Senyshyn, A. & Niewa, R. A temperature-dependent structural study of anti-ReO3-type Na3N: to distort or not to distort? Z. Anorg. Allg. Chem. 636, 94–99 (2010).
Kim, D. et al. Type-II Dirac line node in strained Na3N. Phys. Rev. Mater. 2, 104203 (2018).
Zintl, E. & Brauer, G. Konstitution des Lithiumnitrids. Z. Elektrochem. 41, 102–107 (1935).
Fischer, D., Cancarevic, Z., Schön, J. C. & Jansen, M. Zur synthese und struktur von K3N. Z Anorg. Allg. Chem. 630, 156–160 (2004).
Greve, B. K. et al. Pronounced negative thermal expansion from a simple structure: cubic ScF3. J. Am. Chem. Soc. 132, 15496–15498 (2010).
Groult, H. et al. Nano-CoF3 prepared by direct fluorination with F2 gas: application as electrode material in Li-ion battery. J. Fluor. Chem. 196, 117–127 (2017).
Chaudhuri, S., Chupas, P. J., Wilson, M., Madden, P. & Grey, C. P. Study of the nature and mechanism of the rhombohedral-to-cubic phase transition in α-AlF3 with molecular dynamics simulations. J. Phys. Chem. B 108, 3437–3445 (2004).
Hepworth, M. A., Jack, K. H., Peacock, R. D. & Westland, G. J. The crystal structures of the trifluorides of iron, cobalt, ruthenium, rhodium, palladium and iridium. Acta Crystallogr. 10, 63–69 (2002).
Daniel, P., Bulou, A., Leblanc, M., Rousseau, M. & Nouet, J. Structural and vibrational study of VF3. Mater. Res. Bull. 25, 413–420 (1990).
Siegel, S. The structure of TiF3. Acta Crystallogr. 9, 684–684 (1956).
Shannon, R. D. T. & Prewitt, C. T. Effective ionic radii in oxides and fluorides. Acta Crystallogr. B 25, 925–946 (1969).
Shannon, R. D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr. A 32, 751–767 (1976).
Hu, L. et al. New insights into the negative thermal expansion: direct experimental evidence for the “guitar-string” effect in cubic ScF3. J. Am. Chem. Soc. 138, 8320–8323 (2016).
Piskunov, S. et al. Interpretation of unexpected behavior of infrared absorption spectra of ReO3 beyond the quasiharmonic approximation. Phys. Rev. B 93, 214101 (2016).
Bhandia, R., Siegrist, T., Besara, T. & Schmiedeshoff, G. M. Grüneisen divergence near the structural quantum phase transition in ScF3. Philos. Mag. 99, 631–643 (2019).
Yang, C. et al. Size effects on negative thermal expansion in cubic ScF3. Appl. Phys. Lett. 109, 023110 (2016).
Hu, L. et al. Localized symmetry breaking for tuning thermal expansion in ScF3 nanoscale frameworks. J. Am. Chem. Soc. 140, 4477–4480 (2018).
Wang, T. et al. Tunable thermal expansion and magnetism in Zr-doped ScF3. Appl. Phys. Lett. 109, 181901 (2016).
Chen, J. et al. Tunable thermal expansion in framework materials through redox intercalation. Nat. Commun. 8, 14441 (2017).
Goodwin, A. L., Chapman, K. W. & Kepert, C. J. Guest-dependent negative thermal expansion in nanoporous Prussian blue analogues MIIPtIV(CN)6·xH2O (0 ≤ x ≤ 2; M = Zn, Cd). J. Am. Chem. Soc. 127, 17980–17981 (2005).
Duan, N., Kameswari, U. & Sleight, A. W. Further contraction of ZrW2O8. J. Am. Chem. Soc. 121, 10432–10433 (2002).
Phillips, A. E., Goodwin, A. L., Halder, G. J., Southon, P. D. & Kepert, C. J. Nanoporosity and exceptional negative thermal expansion in single-network cadmium cyanide. Angew. Chem. Inter. Ed. 47, 1396–1399 (2008).
Phillips, A. E., Halder, G. J., Chapman, K. W., Goodwin, A. L. & Kepert, C. J. Zero thermal expansion in a flexible, stable framework: tetramethylammonium copper(I) zinc(II) cyanide. J. Am. Chem. Soc. 132, 10–11 (2010).
Carey, T., Tang, C. C., Hriljac, J. A. & Anderson, P. A. Chemical control of thermal expansion in cation-exchanged zeolite A. Chem. Mater. 26, 1561–1566 (2014).
Arai, H., Okada, S., Sakurai, Y. & Yamaki, J. I. Cathode performance and voltage estimation of metal trihalides. J. Power Sources 68, 716–719 (1997).
Nishijima, M. et al. Cathode properties of metal trifluorides in Li and Na secondary batteries. J. Power Sources 190, 558–562 (2009).
Zhou, M., Zhao, L., Doi, T., Okada, S. & Yamaki, J. I. Thermal stability of FeF3 cathode for Li-ion batteries. J. Power Sources 195, 4952–4956 (2010).
Takami, T. et al. Role of the particle size of Fe nanoparticles in the capacity of FeF3 batteries. AIP Adv. 9, 045301 (2019).
Yang, Z. et al. Atomistic insights into FeF3 nanosheet: an ultrahigh-rate and long-life cathode material for Li-ion batteries. ACS Appl. Mater. Interfaces 10, 3142–3151 (2018).
Jiang, J., Li, L., Xu, M., Zhu, J. & Li, C. M. FeF3 thin nickel ammine nitrate matrix: smart configurations and applications as superior cathodes for Li-ion batteries. ACS Appl. Mater. Interfaces 8, 16240–16247 (2016).
Kim, T. et al. A cathode material for lithium-ion batteries based on graphitized carbon-wrapped FeF3 nanoparticles prepared by facile polymerization. J. Mater. Chem. 4, 14857–14864 (2016).
Li, C., Gu, L., Tsukimoto, S., van Aken, P. A. & Maier, J. Low-temperature ionic-liquid-based synthesis of nanostructured iron-based fluoride cathodes for lithium batteries. Adv. Mater. 22, 3650–3654 (2010).
Yang, Z., Pei, Y., Wang, X., Liu, L. & Su, X. First principles study on the structural, magnetic and electronic properties of Co-doped FeF3. Comput. Theor. Chem. 980, 44–48 (2012).
Liu, L. et al. Excellent cycle performance of Co-doped FeF3/C nanocomposite cathode material for lithium-ion batteries. J. Mater. Chem. 22, 17539 (2012).
Liu, L. et al. A comparison among FeF3·3H2O, FeF3·0.33H2O and FeF3 cathode materials for lithium ion batteries: structural, electrochemical, and mechanism studies. J. Power Sources 238, 501–515 (2013).
Kitajou, A. et al. Novel synthesis and electrochemical properties of perovskite-type NaFeF3 for sodium-ion battery. J. Power Sources 198, 389–392 (2012).
Ruchaud, N., Mirambet, C., Fournes, L., Grannec, J. & Soubeyroux, J. L. Determination of the cationic arrangement in Sn2F6 from neutron powder diffraction. Z. Anorg. Allg. Chem. 590, 173–180 (1990).
Köhl, P., Reinen, D., Decher, G. & Wanklyn, B. Strukturelle Modifikationen von FeZrF6. Z. Kristallogr. Cryst. Mater. 153, 211–220 (1980).
Mayer, H., Reinen, D. & Heger, G. Struktur und Bindung in Übergangsmetall-fluoriden MIIMeIVF6. J. Solid. State Chem. 50, 213–224 (1983).
Bachmann, B. & Müller, B. G. Zur synthese und kristallstruktur von LiPdAlF6 und PdZrF6. Z. Anorg. Allg. Chem. 619, 189–192 (1993).
Rodriguez, V. & Couzi, M. Structural phase transition in the ordered fluorides M IIZrF6 (M II = Co, Zn). III. Landau theory. J. Phys. Condens. Matter 2, 7395–7406 (1990).
Gerasimenko, A. V., Gaivoronskaya, K. A., Slobodyuk, A. B. & Didenko, N. A. Magnesium hexafluoridozirconates MgZrF6·5H2O, MgZrF6·2H2O, and MgZrF6: structures, phase transitions, and internal mobility of water molecules. Z. Anorg. Allg. Chem. 643, 1785–1792 (2017).
Friebel, C., Pebler, J., Steffens, F., Weber, M. & Reinen, D. Phase transitions in CuZrF6 and CrZrF6: a Mössbauer and EPR study of local and cooperative Jahn–Teller distortions. J. Solid State Chem. 46, 253–264 (1983).
Schmidt, R., Kraus, M. & Müller, B. G. Neue fluorozirconate und -hafnate mit V2+ und Ti2+. Z. Anorg. Allg. Chem. 627, 2344–2350 (2001).
Le Mercier, T., Chassaing, J., Bizot, D. & Quarton, M. Structural, spectroscopic and magnetic studies of VIIMIVF6 compounds with MIV = Zr, Nb. Mater. Res. Bull. 27, 259–267 (1992).
Hester, B. R., dos Santos, A. M., Molaison, J. J., Hancock, J. C. & Wilkinson, A. P. Synthesis of defect perovskites (He2−xϒx)(CaZr)F6 by inserting helium into the negative thermal expansion material CaZrF6. J. Am. Chem. Soc. 139, 13284–13287 (2017).
De, I., Desai, V. P. & Chakravarty, A. S. Magnetic properties of some complexes of Mo5+. Phys. Rev. B 8, 3769–3772 (1973).
Llorente, S. et al. Synthesis and crystal structure of CuIIMoIVF6 and CrIINbIVF6 (LT form). Z. Anorg. Allg. Chem. 624, 1538–1542 (1998).
Goubard, F. et al. Fluorocomplexes of niobium IV: the magnetic structure of VNbF6. J. Magn. Magn. Mater. 146, 129–132 (1995).
Yang, C. et al. Large positive thermal expansion and small band gap in double-ReO3-type compound NaSbF6. Inorg. Chem. 56, 4990–4995 (2017).
Gupta, M. K., Singh, B., Mittal, R. & Chaplot, S. L. Negative thermal expansion behavior in ReO3. Phys. Rev. B 98, 014301 (2018).
Hancock, J. C. et al. Large negative thermal expansion and anomalous behavior on compression in cubic ReO3-type AIIBIVF6: CaZrF6 and CaHfF6. Chem. Mater. 27, 3912–3918 (2015).
Ticknor, J. O. et al. Zero thermal expansion and abrupt amorphization on compression in anion excess ReO3-type cubic YbZrF7. Chem. Mater. 30, 3071–3077 (2018).
Baxter, S. J., Hester, B. R., Wright, B. R. & Wilkinson, A. P. Controlling the negative thermal expansion and response to pressure in ReO3-type fluorides by the deliberate introduction of excess fluoride: Mg1−xZr1+xF6+2x, x = 0.15, 0.30, 0.40, and 0.50. Chem. Mater. 31, 3440–3448 (2019).
Reddy, M., Madhavi, S., Subba Rao, G. & Chowdari, B. Metal oxyfluorides TiOF2 and NbO2F as anodes for Li-ion batteries. J. Power Sources 162, 1312–1321 (2006).
Pérez-Flores, J. C. et al. VO2F: a new transition metal oxyfluoride with high specific capacity for Li ion batteries. J. Mater. Chem. A 3, 20508–20515 (2015).
Frevel, L. K. & Rinn, H. W. The crystal structure of NbO2F and TaO2F. Acta Crystallogr. 9, 626–627 (1956).
Poulain, M., Lucas, J. & Tilley, R. J. A structural study of a nonstoichiometric niobium–zirconium oxyfluoride with the ReO3 type structure. J. Solid State Chem. 17, 331–337 (1976).
Pierce, J. W. & Vlasse, M. The crystal structures of two oxyfluorides of molybdenum. Acta Crystallogr. B 27, 158–163 (1971).
Nakhal, S. & Lerch, M. New transition metal oxide fluorides with ReO3-type structure. Z. Naturforsch. B Chem. Sci. 71, 457–461 (2016).
Nakhal, S., Bredow, T. & Lerch, M. Syntheses and crystal structures of New ReO3 type-derived transition metal oxide fluorides. Z. Anorg. Allg. Chem. 641, 1036–1042 (2015).
Dabachi, J., Body, M., Galven, C., Boucher, F. & Legein, C. Preparation-dependent composition and O/F ordering in NbO2F and TaO2F. Inorg. Chem. 56, 5219–5232 (2017).
Liu, K., Li, K., Peng, Q. & Zhang, C. A brief review on key technologies in the battery management system of electric vehicles. Front. Mech. Eng. 14, 47–64 (2019).
Cambaz, M. A. et al. Vanadium oxyfluoride/few-layer graphene composite as a high-performance cathode material for lithium batteries. Inorg. Chem. 55, 3789–3796 (2016).
Chen, R. et al. Lithiation-driven structural transition of VO2F into disordered rock-salt LixVO2F. RSC Adv. 6, 65112–65118 (2016).
Wang, X. et al. Structural changes in a high-energy density VO2F cathode upon heating and Li cycling. ACS Appl. Mater. Interfaces 1, 4514–4521 (2018).
Mitchell, R. H., Welch, M. D. & Chakhmouradian, A. R. Nomenclature of the perovskite supergroup: a hierarchical system of classification based on crystal structure and composition. Mineral. Mag. 81, 411–461 (2017).
Xue, X. & Kanzaki, M. High-pressure δ-Al(OH)3 and δ-AlOOH phases and isostructural hydroxides/oxyhydroxides: new structural insights from high-resolution 1H and 27Al NMR. J. Phys. Chem. B 111, 13156–13166 (2007).
Welch, M. D. & Kleppe, A. K. Polymorphism of the hydroxide perovskite Ga(OH)3 and possible proton-driven transformational behaviour. Phys. Chem. Miner. 43, 515–526 (2016).
Au-Yeung, S. C. F., Denes, G., Greedan, J. E., Eaton, D. R. & Birchall, T. A novel synthetic route to “iron trihydroxide, Fe(OH)3”: characterization and magnetic properties. Inorg. Chem. 23, 1513–1517 (1984).
Au-Yeung, S. C. F. et al. The preparation and characterization of iron trihydroxide, Fe(OH)3. Can. J. Chem. 63, 3378–3385 (1985).
Schubert, K. & Seitz, A. Kristallstruktur von Sc(OH)3 und In(OH)3. Z. Anorg. Allg. Chem. 256, 226–238 (1948).
Mullica, D., Beall, G., Milligan, W., Korp, J. & Bernal, I. The crystal structure of cubic In(OH)3 by X-ray and neutron diffraction methods. J. Inorg. Nucl. Chem. Lett. 41, 277–282 (1979).
Mullica, D. & Milligan, W. Structural refinement of cubic Lu(OH)3. J. Inorg. Nucl. Chem. 42, 223–227 (1980).
Mullica, D., Sappenfield, E., Gable, D. & Tims, T. Crystal structural analyses of 1:3 (Lu, In)(OH)3 and 1:3 (Yb, In)(OH)3. J. Less-Common Met. 152, 157–163 (1989).
Birch, W. D., Pring, A., Reller, A. & Schmalle, H. Bernalite: a new ferric hydroxide with perovskite structure. Naturwissenschaften 79, 509–511 (1992).
Li, B. et al. In2O3 hollow microspheres: synthesis from designed In(OH)3 precursors and applications in gas sensors and photocatalysis. Langmuir 22, 9380–9385 (2006).
Morgenstern-Badarau, I. Effet Jahn–Teller et structure cristalline de l’hydroxyde CuSn(OH)6. J. Solid State Chem. 17, 399–406 (1976).
Neilson, J. R., Kurzman, J. A., Seshadri, R. & Morse, D. E. Ordering double perovskite hydroxides by kinetically controlled aqueous hydrolysis. Inorg. Chem. 50, 3003–3009 (2011).
Mizoguchi, H., Bhuvanesh, N. S. P., Kim, Y.-I., Ohara, S. & Woodward, P. M. Hydrothermal crystal growth and structure determination of double hydroxides LiSb(OH)6, BaSn(OH)6, and SrSn(OH)6. Inorg. Chem. 53, 10570–10577 (2014).
Nakayama, N., Kosuge, K., Kachi, S., Shinjo, T. & Takada, T. Magnetic properties of FeSn(OH)6 and its oxidation product, FeSnO(OH)5. Mater. Res. Bull. 13, 17–22 (1978).
Xu, R., Deng, B., Min, L., Xu, H. & Zhong, S. CuSn(OH)6 submicrospheres: room-temperature synthesis and weak antiferromagnetic behavior. Mater. Lett. 65, 733–735 (2010).
Wu, J. M. & Chen, Y. N. The surface plasmon resonance effect on the enhancement of photodegradation activity by Au/ZnSn(OH)6 nanocubes. Dalton Trans. 44, 16294–16303 (2015).
Wang, L. et al. Single-crystalline ZnSn(OH)6 hollow cubes via self-templated synthesis at room temperature and their photocatalytic properties. J. Mater. Chem. 21, 4352 (2011).
Gao, Y. et al. Perovskite hydroxide CoSn(OH)6 nanocubes for efficient photoreduction of CO2 to CO. ACS Sustain. Chem. Eng. 6, 781–786 (2018).
Chen, D. et al. Preferential cation vacancies in perovskite hydroxide for the oxygen evolution reaction. Angew. Chem. Int. Ed. 57, 8691–8696 (2018).
Shinagawa, T., Garcia-Esparza, A. T. & Takanabe, K. Insight on Tafel slopes from microkinetic analysis of aqueous electrocatalysis for energy conversion. Sci. Rep. 5, 13801 (2015).
Li, B.-Q. et al. Regulating p-block metals in perovskite nanodots for efficient electrocatalytic water oxidation. Nat. Commun. 8, 934 (2017).
Welch, M. D., Crichton, W. A. & Ross, N. L. Compression of the perovskite-related mineral bernalite Fe(OH)3 to 9 GPa and a reappraisal of its structure. Mineral. Mag. 69, 309–315 (2005).
Oftedal, I. Die Kristallstruktur von Skutterudit und Speiskobalt-chloanthit. Z. Kristallogr. Cryst. Mater. 66, 517–546 (1928).
Mandel, N. & Donohue, J. The refinement of the crystal structure of skutterudite, CoAs3. Acta Crystallogr. B 27, 2288–2289 (1971).
von Schnering, H. G. Homoatomic bonding of main group elements. Angew. Chem. Int. Ed. 20, 33–51 (1981).
Jeitschko, W. & Braun, D. LaFe4P12 with filled CoAs3-type structure and isotypic lanthanoid–transition metal polyphosphides. Acta Crystallogr. B 33, 3401–3406 (1977).
Meisner, G. Superconductivity and magnetic order in ternary rare earth transition metal phosphides. Phys. C. 108, 763–764 (1981).
Sales, B. C., Mandrus, D. & Williams, R. K. Filled skutterudite antimonides: a new class of thermoelectric materials. Science 272, 1325–1328 (1996).
Caillat, T., Borshchevsky, A. & Fleurial, J. Properties of single crystalline semiconducting CoSb3. J. Appl. Phys. 80, 4442–4449 (1996).
Snyder, G. J. & Toberer, E. S. Complex thermoelectric materials. Nat. Mater. 7, 105–114 (2008).
Gaultois, M. W. et al. Data-driven review of thermoelectric materials: performance and resource considerations. Chem. Mater. 25, 2911–2920 (2013).
Shi, X. et al. Multiple-filled skutterudites: high thermoelectric figure of merit through separately optimizing electrical and thermal transports. J. Am. Chem. Soc. 133, 7837–7846 (2011).
Khan, A. U. et al. Nano-micro-porous skutterudites with 100% enhancement in ZT for high performance thermoelectricity. Nano Energy 31, 152–159 (2017).
Buser, H. J., Schwarzenbach, D., Petter, W. & Ludi, A. The crystal structure of Prussian blue: Fe4[Fe(CN)6]3.xH2O. Inorg. Chem. 16, 2704–2710 (1977).
Chapman, K. W., Chupas, P. J. & Kepert, C. J. Compositional dependence of negative thermal expansion in the Prussian blue analogues MIIPtIV(CN)6 (M = Mn, Fe, Co, Ni, Cu, Zn, Cd). J. Am. Chem. Soc. 128, 7009–7014 (2006).
Gao, Q. et al. Low-frequency phonon driven negative thermal expansion in cubic GaFe(CN)6 Prussian blue analogues. Inorg. Chem. 57, 10918–10924 (2018).
Behera, J. N., D’Alessandro, D. M., Soheilnia, N. & Long, J. R. Synthesis and characterization of ruthenium and iron–ruthenium Prussian blue analogues. Chem. Mater. 21, 1922–1926 (2009).
Williams, D., Pleune, B., Leinenweber, K. & Kouvetakis, J. Synthesis and structural properties of the binary framework C–N compounds of Be, Mg, Al, and Tl. J. Solid State Chem. 159, 244–250 (2001).
Brousseau, L. C., Kouvetakis, W. D. & O’Keeffe, M. Synthetic routes to Ga(CN)3 and MGa(CN)4 (M = Li, Cu) framework structures. J. Am. Chem. Soc. 119, 6292–6296 (1997).
Williams, D. J., Partin, D. E., Lincoln, F. J., Kouvetakis, J. & O’Keeffe, M. The disordered crystal structures of Zn(CN)2 and Ga(CN)3. J. Solid State Chem. 134, 164–169 (1997).
Williams, D., Kouvetakis, J. & O’Keeffe, M. Synthesis of nanoporous cubic In(CN)3 and In1–xGax(CN)3 and corresponding inclusion compounds. Inorg. Chem. 37, 4617–4620 (1998).
Shi, N. et al. Negative thermal expansion in cubic FeFe(CN)6 Prussian blue analogues. Dalton Trans. 48, 3658–3663 (2019).
Yang, J., Wang, H., Lu, L., Shi, W. & Zhang, H. Large-scale synthesis of Berlin green Fe[Fe(CN)6] microcubic crystals. Cryst. Growth Des. 6, 2438–2440 (2006).
Gao, Q. et al. Switching between giant positive and negative thermal expansions of a YFe(CN)6-based Prussian blue analogue induced by guest species. Angew. Chem. Int. Ed. 56, 9023–9028 (2017).
Kumar, A., Yusuf, S. M. & Keller, L. Structural and magnetic properties of Fe[Fe(CN)6]·4H2O. Phys. Rev. B 71, 054414 (2005).
Ohba, M. & Oˉkawa, H. Synthesis and magnetism of multi-dimensional cyanide-bridged bimetallic assemblies. Coordin. Chem. Rev. 198, 313–328 (2000).
Ferlay, S., Mallah, T., Ouahe`s, R., Veillet, P. & Verdaguer, M. A room-temperature organometallic magnet based on Prussian blue. Nature 378, 701–703 (1995).
Ohkoshi, S. I., Arai, K. I., Sato, Y. & Hashimoto, K. Humidity-induced magnetization and magnetic pole inversion in a cyano-bridged metal assembly. Nat. Mater. 3, 857–861 (2004).
Lu, Y., Wang, L., Cheng, J. & Goodenough, J. B. Prussian blue: a new framework of electrode materials for sodium batteries. Chem. Commun. 48, 6544 (2012).
Zhang, J. et al. FeFe(CN)6 nanocubes as a bipolar electrode material in aqueous symmetric sodium-ion batteries. Chem. Plus. Chem. 82, 1170–1173 (2017).
Wu, X. et al. Single-crystal FeFe(CN)6 nanoparticles: a high capacity and high rate cathode for Na-ion batteries. J. Mater. Chem. A 1, 10130 (2013).
Wu, X. et al. Low defect FeFe(CN)6 framework as stable host material for high performance Li-ion batteries. ACS Appl. Mater. Interfaces 8, 23706–23712 (2016).
Shadike, Z. et al. Long life and high-rate Berlin green FeFe(CN)6 cathode material for a non-aqueous potassium-ion battery. J. Mater. Chem. A 5, 6393–6398 (2017).
Cliffe, M. J. et al. Strongly coloured thiocyanate frameworks with perovskite-analogue structures. Chem. Sci. 10, 793–801 (2019).
Ravnsbæk, D. B. et al. Thermal polymorphism and decomposition of Y(BH4)3. Inorg. Chem. 49, 3801–3809 (2010).
Ley, M. B. et al. Complex hydrides for hydrogen storage-new perspectives. Mater. Today 17, 122–128 (2014).
Mohtadi, R. & Orimo, S.-i. The renaissance of hydrides as energy materials. Nat. Rev. Mater. 2, 16091 (2017).
Yan, Y. et al. Dehydriding and rehydriding properties of yttrium borohydride Y(BH4)3 prepared by liquid-phase synthesis. Int. J. Hydrog. Energy 34, 5732–5736 (2009).
Olsen, J. E. et al. Structure and thermal properties of composites with RE-borohydrides (RE = La, Ce, Pr, Nd, Sm, Eu, Gd, Tb, Er, Yb or Lu) and LiBH4. RSC Adv. 4, 1570–1582 (2014).
Ley, M. B., Jørgensen, M., Cˇerný, R., Filinchuk, Y. & Jensen, T. R. From M(BH4)3 (M = La, Ce) borohydride frameworks to controllable synthesis of porous hydrides and ion conductors. Inorg. Chem. 55, 9748–9756 (2016).
GharibDoust, S. P. et al. Synthesis, structure, and polymorphic transitions of praseodymium(III) and neodymium(III) borohydride, Pr(BH4)3 and Nd(BH4)3. Dalton Trans. 47, 8307–8319 (2018).
Grinderslev, J. B., Møller, K. T., Bremholm, M. & Jensen, T. R. Trends in synthesis, crystal structure, and thermal and magnetic properties of rare-earth metal borohydrides. Inorg. Chem. 58, 5503–5517 (2019).
Ley, M. B. et al. LiCe(BH4)3Cl, a new lithium-ion conductor and hydrogen storage material with isolated tetranuclear anionic clusters. Chem. Mater. 24, 1654–1663 (2012).
Latroche, M. et al. Full-cell hydride-based solid-state Li batteries for energy storage. Int. J. Hydrog. Energy 44, 7875–7887 (2019).
Maouel, H. A., Alonzo, V., Roisnel, T., Rebbah, H. & Le Fur, E. The first three-dimensional vanadium hypophosphite. Acta Crystallogr. A 65, i36–i38 (2009).
Evans, H. A. et al. Polymorphism in M(H2PO2)3 (M = V, Al, Ga) compounds with the perovskite-related ReO3 structure. Chem. Commun. 55, 2964–2967 (2019).
Yang, W. et al. Carbon nanotube reinforced polylactide/basalt fiber composites containing aluminium hypophosphite: thermal degradation, flame retardancy and mechanical properties. RSC Adv. 5, 105869–105879 (2015).
Bermúdez-García, J. M. et al. Giant barocaloric effect in the ferroic organic–inorganic hybrid [TPrA][Mn(dca)3] perovskite under easily accessible pressures. Nat. Commun. 8, 15715 (2017).
Jain, P., Dalal, N. S., Toby, B. H., Kroto, H. W. & Cheetham, A. K. Order–disorder antiferroelectric phase transition in a hybrid inorganic–organic framework with the perovskite architecture. J. Am. Chem. Soc. 130, 10450–10451 (2008).
Jain, P. et al. Multiferroic behavior associated with an order–disorder hydrogen bonding transition in metal–organic frameworks (MOFs) with the perovskite ABX3 architecture. J. Am. Chem. Soc. 131, 13625–13627 (2009).
Wang, Z., Hu, K., Gao, S. & Kobayashi, H. Formate-based magnetic metal–organic frameworks templated by protonated amines. Adv. Mater. 22, 1526–1533 (2010).
Goldschmidt, V. M. Die Gesetze der Krystallochemie. Naturwissenschaften 14, 477–485 (1926).
Kieslich, G., Sun, S. & Cheetham, A. K. Solid-state principles applied to organic–inorganic perovskites: new tricks for an old dog. Chem. Sci. 5, 4712–4715 (2014).
Kieslich, G., Sun, S. & Cheetham, A. K. An extended tolerance factor approach for organic–inorganic perovskites. Chem. Sci. 6, 3430–3433 (2015).
Seifert, H. On the existence of a vanadium (IV) formate. J. Inorg. Nucl. Chem. 27, 1269–1270 (1965).
Tian, Y.-Q., Zhao, Y.-M., Xu, H.-J. & Chi, C.-Y. CO2 template synthesis of metal formates with a ReO3 net. Inorg. Chem. 46, 1612–1616 (2007).
Paredes-García, V. et al. Structural and magnetic characterization of the tridimensional network [Fe(HCO2)3]·nHCO2H. New J. Chem. 37, 2120 (2013).
Görne, A. L. et al. Ammonothermal synthesis, crystal structure, and properties of the ytterbium(II) and ytterbium(III) amides and the first two rare-earth-metal guanidinates, YbC(NH)3 and Yb(CN3H4)3. Inorg. Chem. 55, 6161–6168 (2016).
Hu, K.-L., Kurmoo, M., Wang, Z. & Gao, S. Metal–organic perovskites: synthesis, structures, and magnetic properties of [C(NH2)3][MII(HCOO)3] (M = Mn, Fe, Co, Ni, Cu, and Zn; C(NH2)3 = guanidinium). Chem. Eur. J. 15, 12050–12064 (2009).
Evans, H. A. et al. Hydrogen bonding controls the structural evolution in perovskite-related hybrid platinum(IV) iodides. Inorg. Chem. 57, 10375–10382 (2018).
Müller-Buschbaum, K. & Mokaddem, Y. Three-dimensional networks of lanthanide 1,2,4-triazolates: [Yb(Tz)3] and [Eu2(Tz)5(TzH)2], the first 4f networks with complete nitrogen coordination. Chem. Commun. 2006, 2060–2062 (2006).
Rybak, J.-C., Rekawka, A. & Müller-Buschbaum, K. Utilizing a metal melt of gallium for the synthesis of the homoleptic 1,2,4-triazolate dense framework [Ga(Tz)3]. Z. Anorg. Allg. Chem. 639, 2382–2385 (2013).
Schweinefuß, M. E. et al. Indium imidazolate frameworks with differently distorted ReO3-type structures: syntheses, structures, phase transitions, and crystallization studies. Cryst. Growth Des. 14, 4664–4673 (2014).
Matsumoto, K. et al. A peanut-shaped polyaromatic capsule: solvent-dependent transformation and electronic properties of a non-contacted fullerene dimer. Angew. Chem. Inter. Ed. 58, 8463–8467 (2019).
Abrahams, B. F., Hoskins, B. F., Robson, R. & Slizys, D. A. α-Polonium coordination networks constructed from bis(imidazole) ligands. CrystEngComm 4, 478–482 (2002).
Eddaoudi, M. Systematic design of pore size and functionality in isoreticular MOFs and their application in methane storage. Science 295, 469–472 (2002).
Yaghi, O. M. et al. Reticular synthesis and the design of new materials. Nature 423, 705–714 (2003).
Férey, G. Hybrid porous solids: past, present, future. Chem. Soc. Rev. 37, 191–214 (2008).
Lock, N. et al. Elucidating negative thermal expansion in MOF-5. J. Phys. Chem. C 114, 16181–16186 (2010).
Deng, H. et al. Multiple functional groups of varying ratios in metal–organic frameworks. Science 327, 846–850 (2010).
Kong, X. et al. Mapping of functional groups in metal–organic frameworks. Science 341, 882–885 (2013).
Perry, J. J. IV, Perman, J. A. & Zaworotko, M. J. Design and synthesis of metal–organic frameworks using metal–organic polyhedra as supermolecular building blocks. Chem. Soc. Rev. 38, 1400 (2009).
Li, J.-R., Timmons, D. J. & Zhou, H.-C. Interconversion between molecular polyhedra and metal–organic frameworks. J. Am. Chem. Soc. 131, 6368–6369 (2009).
Biswas, S. et al. A cubic coordination framework constructed from benzobistrazolate ligands and zinc ions having selective gas sorption properties. Dalton Trans. 33, 6487–6495 (2009).
Go´mez-Gualdro´n, D. A., Wilmer, C. E., Farha, O. K., Hupp, J. T. & Snurr, R. Q. Exploring the limits of methane storage and delivery in nanoporous materials. J. Phys. Chem. C 118, 6941–6951 (2014).
Wilmer, C. E. et al. Large-scale screening of hypothetical metal–organic frameworks. Nat. Chem. 4, 83–89 (2012).
He, J., Yu, J., Zhang, Y., Pan, Q. & Xu, R. Synthesis, structure, and luminescent property of a heterometallic metal–organic framework constructed from rod-shaped secondary building blocks. Inorg. Chem. 44, 9279–9282 (2005).
Henke, S., Schneemann, A., Wütscher, A. & Fischer, R. A. Directing the breathing behavior of pillared-layered metal–organic frameworks via a systematic library of functionalized linkers bearing flexible substituents. J. Am. Chem. Soc. 134, 9464–9474 (2012).
Zhu, A.-X. et al. Coordination network that reversibly switches between two nonporous polymorphs and a high surface area porous phase. J. Am. Chem. Soc. 140, 15572–15576 (2018).
Tan, J. C. & Cheetham, A. K. Mechanical properties of hybrid inorganic–organic framework materials: establishing fundamental structure–property relationships. Chem. Soc. Rev. 40, 1059–1080 (2011).
Nowacki, W. Die Kristallstruktur von ScF3. Z. Kristallogr. Cryst. Mater. 101, 273–283 (1939).
Drozdov, A. P., Eremets, M. I., Troyan, I. A., Ksenofontov, V. & Shylin, S. I. Conventional superconductivity at 203 kelvin at high pressures in the sulfur hydride system. Nature 525, 73–76 (2015).
Duan, D. et al. Pressure-induced metallization of dense (H2S)2H2 with high-Tc superconductivity. Sci. Rep. 4, 6968 (2014).
Einaga, M. et al. Crystal structure of the superconducting phase of sulfur hydride. Nat. Phys. 12, 835–838 (2016).
Gordon, E. E. et al. Structure and composition of the 200 K-superconducting phase of H2S at ultrahigh pressure: the perovskite (SH−)(H3S+). Angew. Chem. Int. Ed. 55, 3682–3684 (2016).
Majumdar, A., Tse, J. S. & Yao, Y. Mechanism for the structural transformation to the modulated superconducting phase of compressed hydrogen sulfide. Sci. Rep. 9, 5023 (2019).
Momma, K. & Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 44, 1272–1276 (2011).
Rosi, N. L., Eddaoudi, M., Kim, J., O’Keeffe, M. & Yaghi, O. M. Advances in the chemistry of metal–organic frameworks. CrystEngComm 4, 401–404 (2002).
Acknowledgements
H.A.E. thanks the National Research Council (USA) for financial support through the Research Associate Program. A.K.C. thanks the Ras al Khaimah Centre for Advanced Materials for financial support. H.A.E and R.S. at UC Santa Barbara were supported by the US Department of Energy, Office of Science, Basic Energy Sciences under award number DE-SC-0012541.
Author information
Authors and Affiliations
Contributions
H.A.E. wrote most of the Inorganic systems section. A.K.C. mapped out the original concept of the article and wrote the Introduction and most of the Metal–organic frameworks section. Y.W. contributed significantly to the Metal–organic frameworks section. R.S. worked extensively on the figures and captions, and provided input throughout the review.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Evans, H.A., Wu, Y., Seshadri, R. et al. Perovskite-related ReO3-type structures. Nat Rev Mater 5, 196–213 (2020). https://doi.org/10.1038/s41578-019-0160-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41578-019-0160-x
This article is cited by
-
Electrochemical Proton Storage: From Fundamental Understanding to Materials to Devices
Nano-Micro Letters (2022)