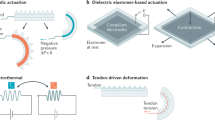
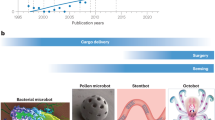
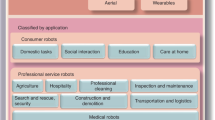
Abstract
Soft robotics enables the design of soft machines and devices at different scales. The compliance and mechanical properties of soft robots make them especially interesting for medical applications. Depending on the level of interaction with humans, different levels of biocompatibility and biomimicry are required for soft materials used in robots. In this Review, we investigate soft robots for biomedical applications, including soft tools for surgery, diagnosis and drug delivery, wearable and assistive devices, prostheses, artificial organs and tissue-mimicking active simulators for training and biomechanical studies. We highlight challenges regarding durability and reliability, and examine traditional and novel soft and active materials as well as different actuation strategies. Finally, we discuss future approaches and applications in the field.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Wang, L. & Iida, F. Deformation in soft-matter robotics: a categorization and quantitative characterization. IEEE Robot. Autom. Mag. 22, 125–139 (2015).
Laschi, C., Mazzolai, B. & Cianchetti, M. Soft robotics: Technologies and systems pushing the boundaries of robot abilities. Sci. Robot. 1, eaah3690 (2016).
Cianchetti, M. & Laschi, C. Pleasant to the Touch: By emulating nature, scientists hope to find innovative new uses for soft robotics in health-care technology. IEEE Pulse 7, 34–37 (2016).
McEvoy, M. A. & Correll, N. Materials that couple sensing, actuation, computation, and communication. Science 347, 1261689 (2015).
Manti, M., Cacucciolo, V. & Cianchetti, M. Stiffening in soft robotics: a review of the state of the art. IEEE Robot. Autom. Mag. 23, 93–106 (2016).
Pfeifer, R., Lungarella, M. & Iida, F. The challenges ahead for bio-inspired ‘soft’ robotics. Commun. ACM 55, 76 (2012).
Rané, A., Tan, G. Y. & Tewari, A. K. Laparo-endoscopic single-site surgery in urology: is robotics the missing link? BJU Int. 104, 1041–1043 (2009).
Vitiello, V., Lee, S.-L., Cundy, T. P. & Yang, G.-Z. Emerging robotic platforms for minimally invasive surgery. IEEE Rev. Biomed. Eng. 6, 111–126 (2013).
Vyas, L., Aquino, D., Kuo, C.-H., Dai, J. S. & Dasgupta, P. Flexible robotics. BJU Int. 107, 187–189 (2011).
Loeve, A., Breedveld, P. & Dankelman, J. Scopes too flexible…and too stiff. IEEE Pulse 1, 26–41 (2010).
Le, H. M., Do, T. N. & Phee, S. J. A survey on actuators-driven surgical robots. Sens. Actuators A Phys. 247, 323–354 (2016).
Ikuta, K., Tsukamoto, M. & Hirose, S. in Proceedings. 1988 IEEE International Conference on Robotics and Automation 427–430 (Philadelphia, PA, USA, 1988).
Menciassi, A. et al. Shape memory alloy clamping devices of a capsule for monitoring tasks in the gastrointestinal tract. J. Micromech. Microeng. 15, 2045–2055 (2005).
Robinson, G. & Davies, J. B. C. in Proceedings 1999 IEEE International Conference on Robotics and Automation 2849–2854 (Detroit, MI, USA, 1999).
Burgner-Kahrs, J., Caleb Rucker, D. & Choset, H. Continuum robots for medical applications: a survey. IEEE Trans. Rob. 31, 1261–1280 (2015).
Polygerinos, P. et al. Soft Robotics: Review of fluid-driven intrinsically soft devices; manufacturing, sensing, control, and applications in human-robot interaction. Adv. Eng. Mater. 19, 1700016 (2017).
Phee, L. et al. Analysis and development of locomotion devices for the gastrointestinal tract. IEEE Trans. Biomed. Eng. 49, 613–616 (2002).
Bertetto, A. M. et al. A novel fluidic bellows manipulator. J. Robot. Mechatron. 16, 604–612 (2004).
Volder, M. D. et al. Production and characterization of a hydraulic microactuator. J. Micromech. Microeng. 15, S15–S21 (2005).
Howell, L. L., Magleby, S. P. & Olsen, B. M. Handbook of Compliant Mechanisms. (John Wiley & Sons, 2013).
De Greef, A., Lambert, P. & Delchambre, A. Towards flexible medical instruments: review of flexible fluidic actuators. Precis. Eng. 33, 311–321 (2009).
Comber, D. B., Slightam, J. E., Gervasi, V. R., Neimat, J. S. & Barth, E. J. Design, additive manufacture, and control of a pneumatic MR-compatible needle driver. IEEE Trans. Rob. 32, 138–149 (2016).
Suzumori, K. Elastic materials producing compliant robots. Rob. Auton. Syst. 18, 135–140 (1996).
Suzumori, K., Koga, A., Kondo, F. & Haneda, R. Integrated flexible microactuator systems. Robotica 14, 493 (1996).
Ranzani, T., Gerboni, G., Cianchetti, M. & Menciassi, A. A bioinspired soft manipulator for minimally invasive surgery. Bioinspir. Biomim. 10, 035008 (2015).
Arezzo, A. et al. Total mesorectal excision using a soft and flexible robotic arm: a feasibility study in cadaver models. Surg. Endosc. 31, 264–273 (2016).
Konishi, S., Kawai, F. & Cusin, P. Thin flexible end-effector using pneumatic balloon actuator. Sens. Actuators A Phys. 89, 28–35 (2001).
Noritsugu, T. et al. Development of pneumatic rotary soft actuator made of silicone rubber. J. Robot. Mechatron. 13, 17–22 (2001).
Yang, Q. et al. in IEEE Conference on Robotics, Automation and Mechatronics, 2004 https://doi.org/10.1109/RAMECH.2004.1438950 (Singapore, 2004).
Suzumori, K., Iikura, S. & Tanaka, H. in [1991] Proceedings. IEEE Micro Electro Mechanical Systems 204–209 (Nara, Japan, 1991).
Low, J.-H., Delgado-Martinez, I. & Yeow, C.-H. Customizable soft pneumatic chamber–gripper devices for delicate surgical manipulation. J. Med. Device. 8, 044504 (2014).
Gerboni, G. et al. A novel linear elastic actuator for minimally invasive surgery: development of a surgical gripper. Smart Mater. Struct. 25, 105025 (2016).
Ranzani, T., Cianchetti, M., Gerboni, G., De Falco, I. & Menciassi, A. A. Soft modular manipulator for minimally invasive surgery: design and characterization of a single module. IEEE Trans. Rob. 32, 187–200 (2016).
Lazeroms, M. et al. A hydraulic forceps with force-feedback for use in minimally invasive surgery. Mechatronics 6, 437–446 (1996).
Gerboni, G. et al. Modular soft mechatronic manipulator for minimally invasive surgery (MIS): overall architecture and development of a fully integrated soft module. Meccanica 50, 2865–2878 (2015).
Moers, A. J. M., De Volder, M. F. L. & Reynaerts, D. Integrated high pressure microhydraulic actuation and control for surgical instruments. Biomed. Microdevices 14, 699–708 (2012).
Ikuta, K., Ichikawa, H., Suzuki, K. & Yamamoto, T. in 2003 IEEE International Conference on Robotics and Automation https://doi.org/10.1109/robot.2003.1241991 (Taipei, Taiwan, 2004)
Ruzzu, A., Bade, K., Fahrenberg, J. & Maas, D. Positioning system for catheter tips based on an active microvalve system. J. Micromech. Microeng. 8, 161–164 (1998).
Sun, Y., Song, S., Liang, X. & Ren, H. A. Miniature soft robotic manipulator based on novel fabrication methods. IEEE Robot. Autom. Lett. 1, 617–623 (2016).
Gorissen, B., Vincentie, W., Al-Bender, F., Reynaerts, D. & De Volder, M. Modeling and bonding-free fabrication of flexible fluidic microactuators with a bending motion. J. Micromech. Microeng. 23, 045012 (2013).
Diodato, A. et al. Soft robotic manipulator for improving dexterity in minimally invasive surgery. Surg. Innov. 25, 69–76 (2018).
Karki, S. et al. Thin films as an emerging platform for drug delivery. Asian J. Pharm. Sci. 11, 559–574 (2016).
Borodkin, S. & Tucker, F. E. Drug release from hydroxypropyl cellulose-polyvinyl acetate films. J. Pharm. Sci. 63, 1359–1364 (1974).
Fusco, S. et al. An integrated microrobotic platform for on-demand, targeted therapeutic interventions. Adv. Mater. 26, 952–957 (2014).
Breger, J. C. et al. Self-folding thermo-magnetically responsive soft microgrippers. ACS Appl. Mater. Interfaces 7, 3398–3405 (2015).
Ricotti, L. et al. Biohybrid actuators for robotics: A review of devices actuated by living cells. Sci. Robot. 2, eaaq0495 (2017).
Tefertiller, C., Pharo, B., Evans, N. & Winchester, P. Efficacy of rehabilitation robotics for walking training in neurological disorders: a review. J. Rehabil. Res. Dev. 48, 387–416 (2011).
Herr, H. M. & Kornbluh, R. D. in Proc. SPIE 5385, Smart Structures and Materials 2004: Electroactive Polymer Actuators and Devices (EAPAD) https://doi.org/10.1117/12.544510 (San Diego, CA, USA, 2004).
Gordon, K. E., Sawicki, G. S. & Ferris, D. P. Mechanical performance of artificial pneumatic muscles to power an ankle–foot orthosis. J. Biomech. 39, 1832–1841 (2006).
do Nascimento, B. G., Vimieiro, C. B. S., Nagem, D. A. P. & Pinotti, M. Hip orthosis powered by pneumatic artificial muscle: voluntary activation in absence of myoelectrical signal. Artif. Organs 32, 317–322 (2008).
Kawamura, T., Takanaka, K., Nakamura, T. & Osumi, H. in 2013 IEEE 13th International Conference on Rehabilitation Robotics (ICORR) https://doi.org/10.1109/ICORR.2013.6650350 (Seattle, WA, USA, 2013).
Pittaccio, S. et al. SHADE: a shape-memory-activated device promoting ankle dorsiflexion. J. Mater. Eng. Perform. 18, 824–830 (2009).
Pittaccio, S. & Viscuso, S. An EMG-controlled SMA device for the rehabilitation of the ankle joint in post-acute stroke. J. Mater. Eng. Perform. 20, 666–670 (2011).
Stirling, L. et al. Applicability of shape memory alloy wire for an active, soft orthotic. J. Mater. Eng. Perform. 20, 658–662 (2011).
Park, Y.-L. et al. Design and control of a bio-inspired soft wearable robotic device for ankle–foot rehabilitation. Bioinspir. Biomim. 9, 016007 (2014).
Wehner, M. et al. in 2013 IEEE International Conference on Robotics and Automation 3362–3369 (Karlsruhe, Germany, 2013).
Asbeck, A. T., De Rossi, S. M. M., Holt, K. G. & Walsh, C. J. A biologically inspired soft exosuit for walking assistance. Int. J. Rob. Res. 34, 744–762 (2015).
Awad, L. N. et al. A soft robotic exosuit improves walking in patients after stroke. Sci. Transl Med. 9, eaai9084 (2017).
Mengüç, Y. et al. Wearable soft sensing suit for human gait measurement. Int. J. Rob. Res. 33, 1748–1764 (2014).
Tsagarakis, N. G. & Caldwell, D. G. Development and control of a ‘soft-actuated’ exoskeleton for use in physiotherapy and training. Autonom. Robots 15, 21–33 (2003).
Villoslada, A., Flores, A., Copaci, D., Blanco, D. & Moreno, L. High-displacement flexible shape memory alloy actuator for soft wearable robots. Rob. Auton. Syst. 73, 91–101 (2015).
Copaci, D., Cano, E., Moreno, L. & Blanco, D. New design of a soft robotics wearable elbow exoskeleton based on shape memory alloy wire actuators. Appl. Bion. Biomech. 2017, 1605101 (2017).
Galiana, I., Hammond, F. L., Howe, R. D. & Popovic, M. B. in 2012 IEEE/RSJ International Conference on Intelligent Robots and Systems 317–322 (Vilamoura, Portugal, 2012).
Natividad, R. F. & Yeow, C. H. in 2016 6th IEEE International Conference on Biomedical Robotics and Biomechatronics (BioRob) 989–993 (Singapore, 2016).
Kim, D. H., Heo, S.-H. & Park, H.-S. in 2017 International Conference on Rehabilitation Robotics (ICORR) 1326–1330 (London, UK, 2017).
Maeder-York, P. et al. Biologically inspired soft robot for thumb rehabilitation. J. Med. Device. 8, 020934 (2014).
Carpi, F., Mannini, A. & De Rossi, D. Proc. SPIE 6927, Electroactive Polymer Actuators and Devices (EAPAD) 2008 https://doi.org/10.1117/12.774644 (San Diego, CA, USA, 2008).
Delph, M. A. et al. in 2013 IEEE 13th International Conference on Rehabilitation Robotics (ICORR) https://doi.org/10.1109/ICORR.2013.6650426 (Seattle, WA, USA, 2013).
In, H., Kang, B. B., Sin, M. & Cho, K.-J. Exo-Glove: a wearable robot for the hand with a soft tendon routing system. IEEE Robot. Autom. Mag. 22, 97–105 (2015).
Kang, B. B. et al. in 2016 IEEE International Conference on Robotics and Automation (ICRA) 3750–3755 (Stockholm, Sweden, 2016).
Connelly, L. et al. A pneumatic glove and immersive virtual reality environment for hand rehabilitative training after stroke. IEEE Trans. Neural Syst. Rehabil. Eng. 18, 551–559 (2010).
Guo, S. et al. in 2015 IEEE International Conference on Mechatronics and Automation (ICMA) 2197–2202 (Beijing, China, 2015).
Polygerinos, P., Wang, Z., Galloway, K. C., Wood, R. J. & Walsh, C. J. Soft robotic glove for combined assistance and at-home rehabilitation. Robot. Auton. Syst. 73, 135–143 (2015).
Ilievski, F., Mazzeo, A. D., Shepherd, R. F., Chen, X. & Whitesides, G. M. Soft robotics for chemists. Angew. Chem. Int. Ed. 50, 1890–1895 (2011).
Yeo, J. C. et al. Flexible and stretchable strain sensing actuator for wearable soft robotic applications. Adv. Mater. Technol. 1, 1600018 (2016).
Yap, H. K., Lim, J. H., Nasrallah, F. & Yeow, C.-H. Design and preliminary feasibility study of a soft robotic glove for hand function assistance in stroke survivors. Front. Neurosci. 11, 547 (2017).
Manto, M. et al. Dynamically responsive intervention for tremor suppression. IEEE Eng. Med. Biol. Mag. 22, 120–132 (2003).
Ansari, Y. et al. Towards the development of a soft manipulator as an assistive robot for personal care of elderly people. Int. J. Adv. Rob. Syst. 14, 172988141668713 (2017).
Pfeifer, R. & Bongard, J. How the Body Shapes the Way We Think: A New View of Intelligence. (MIT Press, 2006).
Deimel, R. & Brock, O. A novel type of compliant and underactuated robotic hand for dexterous grasping. Int. J. Rob. Res. 35, 161–185 (2015).
Galloway, K. C. et al. Soft robotic grippers for biological sampling on deep reefs. Soft Robot. 3, 23–33 (2016).
Mutlu, R., Alici, G., in het Panhuis, P. & Spinks, G. M. 3D printed flexure hinges for soft monolithic prosthetic fingers. Soft Robot. 3, 120–133 (2016).
Thompson-Bean, E., Das, R. & McDaid, A. Methodology for designing and manufacturing complex biologically inspired soft robotic fluidic actuators: prosthetic hand case study. Bioinspir. Biomim. 11, 066005 (2016).
Cheng, N. et al. Prosthetic jamming terminal device: a case study of untethered soft robotics. Soft Robot. 3, 205–212 (2016).
Ogawa, A., Obinata, G., Hase, K., Dutta, A. & Nakagawa, M. in 2008 30th Annual International Conference of the IEEE Engineering in Medicine and Biology Society 330–333 (Vancouver, BC, Canada, 2008).
Paterno, L., Ibrahimi, M., Gruppioni, E., Menciassi, A. & Ricotti, L. Sockets for limb prostheses: a review of existing technologies and open challenges. IEEE Trans. Biomed. Engineer. https://doi.org/10.1109/TBME.2017.2775100 (2018).
Roche, E. T. et al. A bioinspired soft actuated material. Adv. Mater. 26, 1200–1206 (2014).
Roche, E. T. et al. Soft robotic sleeve supports heart function. Sci. Transl Med. 9, eaaf3925 (2017).
Payne, C. J. et al. An implantable extracardiac soft robotic device for the failing heart: mechanical coupling and synchronization. Soft Robot. 4, 241–250 (2017).
Horvath, M. A. et al. An intracardiac soft robotic device for augmentation of blood ejection from the failing right ventricle. Ann. Biomed. Eng. 45, 2222–2233 (2017).
Cohrs, N. H. et al. A soft total artificial heart-first concept evaluation on a hybrid mock circulation. Artif. Organs 41, 948–958 (2017).
Schumacher, C. M., Loepfe, M., Fuhrer, R., Grass, R. N. & Stark, W. J. 3D printed lost-wax casted soft silicone monoblocks enable heart-inspired pumping by internal combustion. RSC Adv. 4, 16039–16042 (2014).
Mac Murray, B. C. et al. Poroelastic foams for simple fabrication of complex soft robots. Adv. Mater. 27, 6334–6340 (2015).
Irwin, D. E., Kopp, Z. S., Agatep, B., Milsom, I. & Abrams, P. Worldwide prevalence estimates of lower urinary tract symptoms, overactive bladder, urinary incontinence and bladder outlet obstruction. BJU Int. 108, 1132–1138 (2011).
Chonan, S. et al. Development of an artificial urethral valve using SMA actuators. Smart Mater. Struct. 6, 410–414 (1997).
Weiss, F. M., Deyhle, H., Kovacs, G. & Müller, B. in Proc. SPIE 8340, Electroactive Polymer Actuators and Devices (EAPAD) 2012 https://doi.org/10.1117/12.914649 (San Diego, CA, USA, 2012).
Hached, S. et al. Novel, wirelessly controlled, and adaptive artificial urinary sphincter. IEEE/ASME Trans. Mechatron. 20, 3040–3052 (2015).
Lamraoui, H. et al. Development of a novel artificial urinary sphincter: a versatile automated device. IEEE/ASME Trans. Mechatron. 15, 916–924 (2010).
Baldoli, I. et al. A novel simulator for mechanical ventilation in newborns: MEchatronic REspiratory System SImulator for Neonatal Applications. Proc. Inst. Mech. Eng. H 229, 581–591 (2015).
Kim, S., Kim, P., Park, C.-Y. & Choi, S.-B. A new tactile device using magneto-rheological sponge cells for medical applications: experimental investigation. Sens. Actuators A Phys. 239, 61–69 (2016).
Someya, Y. et al. in 2016 International Symposium on Micro-NanoMechatronics and Human Science (MHS) https://doi.org/10.1109/MHS.2016.7824208 (Nagoya, Japan, 2016).
Manti, M., Cianchetti, M., Nacci, A., Ursino, F. & Laschi, C. in 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) 3623–3626 (2015).
Zhu, M., Xu, W. & Cheng, L. K. Esophageal peristaltic control of a soft-bodied swallowing robot by the central pattern generator. IEEE/ASME Trans. Mechatron. 22, 91–98 (2017).
Lu, X., Xu, W. & Li, X. A. Soft robotic tongue — mechatronic design and surface reconstruction. IEEE/ASME Trans. Mechatron. 22, 2102–2110 (2017).
Wehner, M. et al. Pneumatic energy sources for autonomous and wearable soft robotics. Soft Robot. 1, 263–274 (2014).
Roseman, J. M. et al. Hybrid integrated biological–solid-state system powered with adenosine triphosphate. Nat. Commun. 6, 10070 (2015).
Menciassi, A. Swell findings in hydrogels. N. Engl. J. Med. 378, 864–865 (2018).
Acknowledgements
The authors acknowledge support from the European Commission through the Hybrid Heart (#767195) and I-SUPPORT (#643666) projects.
Author information
Authors and Affiliations
Contributions
C.L. and M.C. conceived the topic of the Review. M.C. analysed aspects related to prostheses, wearable devices and artificial organs. C.L. and M.C. analysed aspects related to assistive robots and body-part simulators. A.M. and P.D. analysed aspects related to surgery and drug delivery. All authors equally contributed to the writing and revising of the paper. All authors contributed to the discussion to draw conclusions and perspectives.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Intiutive Surgical: http://www.intuitivesurgical.com/ Ovesco: http://www.ovesco.com The Lee Company: http://www.theleeco.com/ Soft Robotics: https://www.softroboticsinc.com/ HybridHeart: http://www.hybridheart.eu/ BR Biomedical Pvt Ltd: http://www.brbiomedicals.com The Bionic Humanoid: http://www.jst.go.jp/impact/bionichumanoids/en/index.html
Rights and permissions
About this article
Cite this article
Cianchetti, M., Laschi, C., Menciassi, A. et al. Biomedical applications of soft robotics. Nat Rev Mater 3, 143–153 (2018). https://doi.org/10.1038/s41578-018-0022-y
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41578-018-0022-y
This article is cited by
-
Silicone-based highly stretchable multifunctional fiber pumps
Scientific Reports (2024)
-
Network of cyano-p-aramid nanofibres creates ultrastiff and water-rich hydrospongels
Nature Materials (2024)
-
Biodegradable suture development-based albumin composites for tissue engineering applications
Scientific Reports (2024)
-
Self-healable and conductive mussel inspired PVA/borax@PDA–LiTFSI hydrogel-based self-adhesive for human motion sensor
Polymer Bulletin (2024)
-
Bioinspired nanomaterials for wearable sensing and human-machine interfacing
Nano Research (2024)