Abstract
The realization that autoantibodies can contribute to dysfunction of the brain has brought about a paradigm shift in neurological diseases over the past decade, offering up important novel diagnostic and therapeutic opportunities. Detection of specific autoantibodies to neuronal or glial targets has resulted in a better understanding of central nervous system autoimmunity and in the reclassification of some diseases previously thought to result from infectious, ‘idiopathic’ or psychogenic causes. The most prominent examples, such as aquaporin 4 autoantibodies in neuromyelitis optica or NMDAR autoantibodies in encephalitis, have stimulated an entire field of clinical and experimental studies on disease mechanisms and immunological abnormalities. Also, these findings inspired the search for additional autoantibodies, which has been very successful to date and has not yet reached its peak. This Review summarizes this rapid development at a point in time where preclinical studies have started delivering fundamental new data for mechanistic understanding, where new technologies are being introduced into this field, and — most importantly — where the first specifically tailored immunotherapeutic approaches are emerging.
Similar content being viewed by others
Introduction
Our increased understanding of the roles of autoantibodies in neurological disease has markedly changed clinical practice in neurology and psychiatry over the past decade. Autoantibodies have enabled the reclassification of diseases; for example, the discovery of antibodies targeting the water channel aquaporin 4 (AQP4) in patients with neuromyelitis optica (NMO) allowed NMO to be distinguished from multiple sclerosis1. The most obvious change to clinical practice from the pre-antibody era has been seen in patients with antibodies to brain neuropil, such as those seen in anti-N-methyl-d-aspartate receptor (NMDAR) encephalitis2. Previously, the often young patients presenting with new-onset psychosis, hyperkinesia, amnesia or vegetative dysfunction were often suspected of having drug-induced psychosis, malignant catatonia, encephalitis of unknown aetiology or encephalitis lethargica. However, we now routinely determine antibodies to the NMDAR to verify NMDAR encephalitis, which has become the most common autoimmune encephalitis. Most importantly, the ongoing discovery of antibodies targeting the surface of neuronal or glial cells has brought about important diagnostic and therapeutic opportunities, and in addition to antipsychotic medication, tranquilizers and psychotherapy, patients today receive immediate immunotherapy including B cell depletion and antibody removal3,4.
More than 25 new types of autoantibody have been described recently in patients with neurological disease. Several of these are turning out to be directly pathogenic and, thus, represent new clinical entities. Antibody discoveries are still ongoing, with fundamental implications for our understanding of autoimmune diseases and for clinical decision-making. This Review therefore provides an overview of the relevant autoantibodies and their associated clinical syndromes, discusses the known and suspected factors leading to such humoral autoimmunity, describes the mechanisms of how antibodies cause disease and reviews evolving concepts in antibody-selective immunotherapy. The Review specifically mentions the expansion of antibody-mediated pathology into unexpected areas, such as neurodegeneration and brain development, where humoral autoimmunity may contribute in a subtle way to symptoms of memory, cognition and psychiatric dysfunction.
Growing interest in antibody-mediated neurological disease not only stems from the diagnostic and therapeutic implications. The availability of monospecific disease-defining target epitopes offers major opportunities to immunologists and neuroscientists, using these specific entities as models for understanding more general mechanisms of autoimmunity. The past decade of important clinical observations should now be followed by a decade of thorough basic science, addressing the emerging questions regarding immune checkpoint dysregulation, antibody pathogenicity, the homeostatic brain antibody repertoire and target-specific immunotherapy.
Autoantibodies in neurological diseases
The expanding autoantibody spectrum
Antibody-associated neurological diseases are characterized by a wide range of clinical symptoms, including memory impairment, behavioural abnormalities, seizures, psychosis, movement disorders or vegetative dysfunction. Traditionally, over the past decade, antibodies in neurological diseases were referred to as malignancy-associated onconeuronal antibodies, that is, those binding to nuclear or cytoplasmic proteins such as Hu, Ma and Yo5. These neurological conditions are primarily mediated by cytotoxic T cells. By contrast, the present review focuses on the antibodies that are directly pathogenic after binding to conformational surface-expressed central nervous system (CNS) antigens, such as NMDARs6, γ-aminobutyric acid (GABA) receptors7,8, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors9, metabotropic glutamate receptor type 5 (mGluR5)10, immunoglobulin-like cell adhesion molecule 5 (IgLON5)11, myelin oligodendrocyte glycoprotein (MOG)12, leucine-rich glioma-inactivated protein 1 (LGI1)13,14,15 or CASPR2 (also known as CNTNAP2)16 (Table 1).
The presence of antibodies targeting cell surface-expressed molecules in cerebrospinal fluid (CSF) or serum generally indicates certain clinical syndromes (Box 1). A major step was the discovery that NMO, a relapsing disease with inflammatory demyelinating lesions in the spinal cord and optic nerve, is characterized by autoantibodies to AQP4 (ref.1). Shortly thereafter, the discovery of NMDAR-targeting antibodies that cause multistage encephalitis often manifesting with psychosis2,6 demonstrated together with further antibodies that autoimmune encephalitides are at least as common as infectious encephalitides in northern hemisphere countries17,18. Many additional autoimmune encephalitides were subsequently described in the past decade, which were shown to differ with respect to the type of antibody, clinical syndrome and mode of antibody-mediated pathogenicity, as well as in other respects (Table 1). Some antibodies strongly indicate particular clinical presentations, such as new-onset psychosis, fever and movement disorder in young patients with NMDAR encephalitis19 or faciobrachial dystonic seizures and hyponatraemia in older men with LGI1-specific antibodies20. Although this Review largely ignores acetylcholine receptor (AChR) autoantibodies in patients with myasthenia gravis because of their peripheral action at the neuromuscular junction, findings from functional antibody assessments21 and animal models22 as well as from treatment trials23 in myasthenia gravis have suggested important research questions for the more recently identified antibody-mediated neurological diseases.
Interestingly, this new field has also reached veterinary medicine with obvious overlap, likely related to highly conserved receptor epitopes and similar mechanisms of immune function (and dysfunction) across species24. For example, cats with LGI1-specific autoantibodies show complex orofacial movements25 reminiscent of human patients with faciobrachial dystonic seizures20. Glial fibrillary acidic protein (GFAP)-binding antibodies can be detected in dogs with necrotizing meningoencephalitis26, a lethal canine equivalent to the human GFAP antibody-associated autoimmune meningoencephalitis27. Knut, the famous polar bear from the Berlin Zoo, drowned in 2011 following seizures due to the first known animal case of NMDAR encephalitis28; this raised public awareness of this new disease group in both humans and other mammals. Indeed, a high prevalence of NMDAR autoantibodies has since been seen in other mammals29. Time (and ongoing research) will tell whether these ‘natural’ autoimmune animal models can be helpful to better understand and treat human disease. For example, the finding of complement deposition in cats with LGI1 autoantibodies25 may instruct the consideration of complement-depleting therapy also in human patients with LGI1 encephalitis.
Discoveries with clinical implications
It is likely that more autoantibodies that are yet to be discovered are involved in neurological disease. The past 2–3 years alone have resulted in the identification of numerous novel targets, such as ATP1A3, CPT1C, flotillin 1 and flotillin 2, NBCE1 (also known as SLC4A4), RGS8, syntaxin 1B (STX1B), ROCK2, GlURD2 (also known as GRID2), PDE10A, AP3B2, neurochondrin, drebrin (DBN1) or septin 5 (refs30,31,32,33,34,35,36,37). Most of these newly identified antigens are intracellular epitopes and often point to paraneoplastic neurological syndromes. Thus, the clinical relevance of these antibodies relates to their role as disease biomarkers rather than causing or aggravating disease. Although the recently discovered meningoencephalomyelitis-associated GFAP antibody also targets an intracellular protein, it stands out because of its relative commonness and frequent response to immunotherapy27,38. Discovery of these novel antigens was mostly done with immunoprecipitation and mass spectrometry, but advanced strategies are increasingly involved, such as phage display for the characterization of Kelch-like protein 11 (KLHL11) antibodies39.
Many new antibodies were immediately implemented in clinical algorithms owing to their high diagnostic yield and therapeutic implications. For most individual antibodies, the fixed cell-based assay has become the gold standard and is commercially available for clinical routine. For this assay, HEK293 cells are transfected to express the antigen of interest in the native conformation including mammalian post-translational modifications, and antibody binding is visualized with fluorescently labelled secondary antibodies (Fig. 1a,b). In research laboratories, anti-neuronal autoantibodies can be detected via binding to cultured primary rodent neurons (Fig. 1c,d). Alternatively, indirect immunofluorescence on rodent or monkey brain sections (‘tissue-based assay’) can indicate novel autoantibodies. In particular, the use of unfixed murine sections can detect autoreactivity to surface-expressed antigens, and the staining patterns and subcellular localization sometimes suggest the yet to be defined targets, some of which might soon become part of the clinical diagnostic panel (Fig. 1e–l).
a,b | Cell-based assays show high sensitivity for defined autoantigens. Antigens of interest (here leucine-rich glioma-inactivated 1 (LGI1)) are expressed in HEK293 cells, which are fixed, incubated with a patient sample containing autoantibodies (orange) and visualized with fluorescently labelled secondary antibodies (green). c,d | Using the same labelling technique, live neurons can be incubated with a patient sample for detection of autoantibodies. Enlarged insert: synaptic clusters of bound N-methyl-d-aspartate receptor (NMDAR) autoantibodies (green). e–l | Tissue-based assays using rodent brain sections detect autoantibody binding to a large variety of target epitopes on neurons, glia cells and endothelium. Examples include autoantibodies to NMDAR (parts e,f), GAD (part g), Hu protein (part h), amphiphysin (part i), flotillin (part j), glial fibrillary acidic protein (GFAP) (part k) and an as yet undefined antigen on catecholaminergic fibres around brain arteries in a patient with immunotherapy-responsive dementia (part l).
Reasons for autoantibody development
Defects in the immune checkpoints
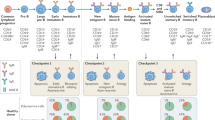
The risk of developing anti-neuronal humoral autoimmunity relates to dysregulated central and peripheral negative selection of B cells, with potentially dysregulated mechanisms of tolerance including clonal deletion, B cell receptor (BCR) editing and anergy40,41. As an early step, immature B cells undergo these processes when their surface-expressed IgM recognizes a self-antigen in the bone marrow42 (Fig. 2a,b). In patients with neuromyelitis optica spectrum disorder (NMOSD), elevated frequencies of autoreactive B cells that have recently emigrated from the bone marrow indicate dysfunctional central B cell tolerance43. Peripheral tolerance checkpoints, including those that occur during B cell maturation in the spleen and following antigen-mediated activation to enter the germinal centre, are only partly understood44. B cells clonally expand in the germinal centre, class switch, undergo affinity maturation and become memory B cells or plasma cells (Fig. 2h).
a–c | Emerging data suggest defective B cell tolerance checkpoints in several antibody-mediated neurological diseases, increasing autoreactive immature B cells (part a) that are not removed (part b) but can be activated and enter germinal centres (part c). d,e | Several established or suspected triggers related to tissue injury or infection (part d) lead to transport of autoantigen to lymph nodes (part e). f | Germinal centres form after stimulation of autoreactive B cells with their antigen together with T cell help. T cells might be activated by the same autoantigen, a viral antigen or yet to be determined unrelated antigens. g | Enhanced T cell activation by immune checkpoint inhibitors is a recent trigger for antibody-mediated neurological disease. h | Affinity-matured and class-switched B cells become plasmablasts and secrete large amounts of autoantibodies. i,j | In a possible alternative route, autoreactive extrafollicular B cells recognize neuronal autoantigens via their B cell receptor (BCR) and are simultaneously stimulated via virus-induced B cell-intrinsic Toll-like receptor (TLR) activation, leading to maturation even without T cell help. k | In many autoantibody-mediated neurological diseases, plasma cells migrate into the brain and release large amounts of autoantibodies. CNS, central nervous system; TCR, T cell receptor.
In patients with GABAA receptor encephalitis45,46, NMOSD43 or LGI1 encephalitis14, memory B cells and plasma cells show extensive somatic hypermutation, in line with the general evidence that germinal centres are a central site of autoreactive B cell activation47. By contrast, antibodies in patients with NMDAR encephalitis are frequently unmutated, pointing to disease-specific alterations48,49,50. Although ‘back-mutation’ of NMDAR autoantibodies to germline sequences preserved binding to the antigen49, reverting LGI1 and AQP4 antibodies to the germ line demonstrated loss of binding14,43. These findings suggest that somatic hypermutations are required and bring in de novo autoreactivity against LGI1 and AQP4 from non-autoreactive precursor B cells. Thus, post-germinal centre checkpoints, including apoptosis or BCR editing, may be dysfunctional in these patients. Although back-mutated AQP4 antibodies lost binding to AQP4, they were commonly polyreactive/self-reactive (non-AQP4), suggesting that they derived from the accumulating pool of autoreactive mature naive B cells resulting from defective tolerance. Ongoing work will have to clarify whether reverted LGI1 (or further neurological) autoantibodies show similar polyreactivity that would point to impaired B cell tolerance checkpoints.
It is unclear whether already established genetic B cell checkpoint defects leading to primary immunodeficiency disorders can also underlie humoral anti-neuronal autoimmunity, given the — somewhat counterintuitive — overlap with autoimmunity51. Selected clinical cases point to a possible link between immune-mediated limbic encephalitis and immune deficiency52, but systematic research is needed to support a robust connection.
Finally, antibody-mediated neurological disease can occur as a rare adverse event during tumour therapy with anti-CTLA4 and anti-PD1/anti-PDL1 checkpoint inhibitors. Through the unleashing of T cells and stimulation of autoantibody production, checkpoint inhibitors can induce neurological autoimmunity (Fig. 2g), often with antibodies to Ma2, Hu, Yo, GABAB receptor or NMDAR53. Early administration of high-dose steroids or immunosuppression can lead to marked improvement. Symptoms can resemble typical paraneoplastic neurological disorders, making it difficult for clinicians to distinguish whether they result from the tumour or the checkpoint inhibitor treatment5.
Tumour-associated and post-viral antibodies
Ectopic expression of neuronal antigens in malignant tumours can trigger an antitumour immune response cross-reacting with the cognate CNS protein, often leading to devastating paraneoplastic neurological syndromes5,54. The most common forms include subacute sensory neuronopathy, paraneoplastic cerebellar degeneration and limbic encephalitis, and typical underlying malignancies are small cell lung cancer, breast cancer and ovarian cancer. The disease is mainly caused by cytotoxic T cells with consequent permanent tissue destruction. In a very similar (‘facultatively paraneoplastic’) way, antibody-mediated neurological disease can also arise from underlying tumours (Fig. 2d–f). A common association is NMDAR-expressing ovarian teratomas in 20–40% of women with NMDAR encephalitis6,55. Teratomas contained dense B cell and T cell infiltrates and dysplastic neurons expressing NMDARs, indicating peripheral initiation of the immune process56,57.
Viral infections are another reproducible trigger of antibody-mediated encephalitis. First observations of frequent NMDAR autoantibodies in patients with herpes simplex encephalitis (HSE) led to the hypothesis of autoimmune-mediated clinical symptoms after HSE58. Prospective studies confirmed this hypothesis and determined a frequency of almost one-third of patients with HSE developing NMDAR encephalitis thereafter59,60. Experimental mice inoculated intranasally with HSV-1 developed serum NMDAR antibodies61. Autoimmunity can also involve autoantibodies to GABAA, AMPA, dopamine D2 receptors and as yet undefined antigens62, and underlying viruses seem to encompass many groups, such as Epstein–Barr virus, varicella-zoster virus, human herpesvirus 6, HIV, hepatitis viruses and Japanese encephalitis B virus. This suggests that virus-induced autoantibody generation could be a broad mechanism of pathology in autoimmune neurological disease63. It also includes the recent COVID-19 pandemic, where severely ill patients with neurological symptoms can have CSF autoantibodies to neuronal and astrocytic epitopes in the absence of SARS-CoV-2 in the brain64, but more investigation is needed to prove a causal relationship.
Several mechanisms may contribute to the loss of B cell tolerance and generation of autoantibodies to synaptic antigens in the context of viral infections, such as co-engagement of the BCR together with co-stimulatory signals from pathogen-associated molecular patterns. A possible scenario is the release of brain-restricted ‘neo-antigens’ (for example, NMDAR protein) after virus-induced tissue destruction and apoptosis (Fig. 2d,e). Virus molecules or debris antigens from necrotic tissue (such as nucleic acids) are co-presented to NMDAR antibody-specific B cells in local lymph nodes (Fig. 2i) and induce B cell-intrinsic Toll-like receptor (TLR) signalling, which together with BCR ligation may allow escape from tolerance44,65,66 (Fig. 2j). Alternatively, B cells may simultaneously capture NMDAR protein and virus. Presenting viral proteins to virus-specific T cells could stimulate T cell help with consecutive B cell proliferation, class switch and antibody production (Fig. 2f), as shown for MOG-specific B cells and influenza antigen, linking infection and autoimmunity67.
A role for molecular mimicry between viral and self-antigens has not been confirmed in antibody-mediated encephalitis, unlike in other autoimmune diseases68. It is also as yet unclear whether genetic susceptibility contributes to the risk of virus-induced autoimmune encephalitis, for example, via B cell-intrinsic TLR signalling, similar to inborn errors of innate immunity as genetic risk factors for HSE69 and against the background of predominant HLA haplotypes in autoimmune encephalitis (Box 2).
Defective tolerance or naturally occurring autoantibodies?
In most autoimmune diseases, non-autoreactive B cell precursors transform into B cells producing autoreactive antibodies through massive somatic hypermutation70. Therefore, it was all the more surprising that the first studies of CSF-derived NMDAR autoantibodies showed few or no hypermutations in the memory B cell and plasma cell pool48,50, but were of high affinity and already pathogenic in the germline configuration49. On the one hand, this antibody property in NMDAR encephalitis is reminiscent of autoimmune regulator (AIRE) deficiency, where high-affinity unmutated autoreactive antibodies are present already in the naive B cell compartment71. On the other hand — although speculative and requiring further research — germline antibodies may be comparable with ‘naturally occurring autoantibodies’, which are innate-like unmutated antibodies that serve as a first line of antibody-mediated defence independent of T cells and an adaptive immune response72.
There are several potential advantages for retaining strongly self-reactive antibodies in the repertoire. One hypothetical possibility is that there could be an important physiological role of naturally occurring antibodies to NMDAR; for instance, they may facilitate the clearance of NMDAR protein released from apoptotic neurons. This may be limited to a certain window, for example, during brain development or neurodegeneration. Such a functional role was recently suggested by the finding that NMDAR autoantibodies may act as endogenous NMDAR antagonists and reduce stress-induced depression and anxiety29; however, these findings await independent confirmation.
Alternatively, anti-neuronal natural antibodies might exert beneficial effects via cross-reactivity with abundant pathogens or tumour cells. The common association with certain viruses and ovarian teratomas in NMDAR encephalitis may support this hypothesis, which has been conceptually shown for other antibodies, such as self-reactive germline antibodies produced by IGHV4-34 B cells that bind both commensal bacteria and autoantigens73. Molecular mimicry between (as yet undefined) pathogens and NMDAR protein may lead to the maintenance of NMDAR-reactive B cells in the repertoire in an anergic state. In this way, such pathogens cannot escape from antibody responses as anergic B cells could be reactivated by undergoing clonal redemption, that is, mutating to increase their affinity for the foreign antigen by reducing self-recognition74.
Role of T cells
Information on how T cells regulate autoantibody-producing B cells in antibody-mediated neurological diseases is scarce. The strong HLA association in some diseases (such as LGI1 or IgLON5) supports T cell-dependent pathways (Box 2). Indeed, emerging data from patients with NMOSD suggest that the same antigen, AQP4, is recognized by T helper cells and by B cells75. T cell responses to AQP4 seem tightly regulated by central and peripheral tolerance, suggesting dysfunctional thymic deletion of autoreactive T cells may occur in NMO76. Injection of encephalitogenic AQP4-specific T cells into rats led to their CNS infiltration, indicating a role of AQP4-specific T cells for NMO77. The observation that the antibody response in NMDAR encephalitis requires continuous repopulation from germinal centre reactions56 indicates the need for T cell help. T cell–B cell interaction may not take place in germinal centres in the lymph nodes or spleen but at extrafollicular sites, potentially also in ectopic lymph node-like follicles in the brain or ovarian teratomas78. On the other hand, the low number of NMDAR autoantibody hypermutations48,50 may indicate extrafollicular B cell activation with T cell-independent B cell activation pathways, for example, via TLR engagement (Box 3).
In contrast to NMOSD, the search for NMDAR-specific T helper cells in NMDAR encephalitis led to the unexpected observation that their number is significantly reduced and they were functionally altered, questioning the concept that NMDAR-specific T cells contribute to disease79. It is unclear, however, whether non-NMDAR-specific T cells are involved. One possibility is the above-described simultaneous capture of NMDAR and viral protein by B cells, which then receive help from virus-specific T cells. In another possible scenario, B cells internalize multimolecular complexes via the membrane-bound NMDAR antibody (BCR), and process and present non-NMDAR peptides to T helper cells. In this way, functionally complexed but structurally unrelated antigens (Fig. 2f) might contribute to NMDAR autoantibody generation, similar to findings in coeliac disease where exogenous T cell antigens (gluten) result in specific antibodies to the autoantigen transglutaminase 2 (TG2) by involvement of gluten–TG2 complexes after T cell help80. There, self-tolerance to TG2 is regulated by the absence of T cell help and not by B cell negative selection81.
T cells might further play a role in antibody-mediated neurological diseases by connecting the microbiome to humoral immune responses. T cells from NMOSD cross-reacted with a Clostridium perfringens ABC transporter75, and C. perfringens was enriched in the gut microbiota of patients with NMOSD82. No overabundance of certain bacterial taxa was observed in NMDAR encephalitis, indicating disease-specific effects83.
Pathogenicity of autoantibodies
Principal disease mechanisms
It has become clear that the clinical symptoms seen in patients with antibody-mediated neurological diseases not only relate to the distribution of the antibody’s target protein. The variable clinical pictures associated with the same antibody suggest multiple contributing factors, which are being actively investigated. These include biophysical antibody properties, such as affinity or glycosylation patterns. Also, the local anatomical enrichment of B cells in the brain (Box 1) may explain clinical differences, given that plasma cells can secrete thousands of antibody molecules per second, potentially resulting in high local gradients84. Several established pathogenic antibody mechanisms help explain the multifaceted clinical phenotype and will instruct novel treatment strategies, as discussed below.
An important advancement in the research of neurological autoantibody-mediated effects has been the single-cell assessment of humoral autoimmunity with the development of recombinant monoclonal human disease-specific autoantibodies, which are currently becoming an essential experimental component85. Monoclonal antibodies allow for the exact dosing of experimental conditions, the analysis of immunological mechanisms at single-cell level, exclusion of effects from other antibodies present in human samples, epitope mapping via crystallography, antibody engineering for improved microscopy and the development of novel therapies13,14,48,49,50,86,87,88,89.
Cross-linking and target receptor internalization
Cross-linking and target receptor internalization is common for antibodies to ionotropic ion channels. NMDAR autoantibodies led to rapid reduction of neuronal surface NMDARs owing to internalization90,91 (Fig. 3a). In addition, single-molecule microscopy techniques demonstrated that disruption of NMDAR–EphB2 interactions led to loss of synaptic NMDAR localization92. Synaptic AMPA receptor clusters decreased after incubation with AMPA receptor antibodies, leading to reduction of AMPA receptor-mediated miniature excitatory postsynaptic currents93,94. Antibody-mediated internalization of glycine receptors disrupted glycinergic neurotransmission95,96, and GABAA receptor antibodies reduced the synaptic and extra-synaptic density of GABAA receptors97. Similarly, neurexin 3α antibodies caused reduction of neurexin 3α levels and the total number of synapses in primary neurons98, and patient-derived antibodies decreased surface-expressed IgLON5 leading to cytoskeletal changes in hippocampal neurons99,100.
a | N-methyl-d-aspartate receptor (NMDAR) autoantibodies lead to receptor cross-linking, internalization and degradation, thus reducing the number of NMDARs on the neuronal surface. b | Aquaporin 4 (AQP4) autoantibodies bind to clustered AQP4 and induce complement-dependent cytotoxicity. c | Binding of GABAB receptor autoantibodies directly block receptor signalling. d | Leucine-rich glioma-inactivated 1 (LGI1) autoantibodies induce neuronal dysfunction by interrupting the trans-synaptic binding of LGI1 to its postsynaptic receptor ADAM22 (and likewise ADAM23 at the presynaptic site; not shown). e | Myelin oligodendrocyte glycoprotein (MOG) autoantibodies target the myelin sheath of axons and induce FcR-mediated antibody-dependent cellular cytotoxicity. f | Selected autoantibodies, such as to synapsin, are internalized via neuronal FcRs and lead to target inactivation intracellularly. AMPA, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid; GABA, γ-aminobutyric acid.
Complement activation
Complement activation seems to be restricted to a few autoimmune encephalitides101. AQP4 antibodies activated the complement cascade by binding to AQP4 assembled in orthogonal arrays of particles and multivalent interaction of organized antibody clusters with the initial complement component C1q (ref.102) (Fig. 3b). Clinical effects of complement inhibition strongly support the role for pathogenesis103.
Stimulatory or inhibitory effects on receptors
Receptor stimulation or inhibition can mediate rapid antibody effects. GABAB receptor antibodies did not change the surface expression of synaptic receptors but blocked the function of the receptor8 (Fig. 3c). Direct antagonistic actions on glycine receptors contribute also to the pathogenic mechanism of GlyR autoantibodies95. Patient samples containing DPPX antibodies caused an increase in action potential firing in preparations of gut nerve plexus104.
Disrupted protein–protein interaction
Antibody-mediated disruption of protein–protein interaction can interfere with normal neuronal function. LGI1 antibodies efficiently blocked the interaction of LGI1 with its receptors ADAM22 and ADAM23, disrupting the trans-synaptic ADAM22–ADAM23 protein complex with presynaptic and postsynaptic implications14,15 (Fig. 3d). LGI1 (and CASRPR2) antibodies are frequently of the IgG4 class, that is, they do not activate complement but block enzymatic activity or protein–protein interactions105.
Antibody-dependent cellular cytotoxicity
Antibody-dependent cellular cytotoxicity results from IgG-Fc binding to FcγRs on phagocytes and natural killer cells, which leads to degranulation and destruction of the antibody-bound target structure (Fig. 3e). MOG antibody-containing sera induced natural killer cell activation to surface-expressed MOG106. Also, pathological effects of AQP4 antibodies were eliminated by removing antibody-dependent cellular cytotoxicity effector function107.
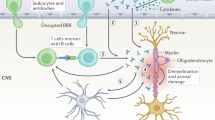
Blood–brain barrier integrity
The integrity of the blood–brain barrier can be reduced in antibody-mediated neurological disease. Patients with NMO have antibodies in their serum that target GRP78 (glucose-regulated protein 78), which is expressed on brain microvascular endothelial cells, and they show decreased claudin 5 expression, which facilitates the transit of AQP4-specific IgG into the brain parenchyma and brain dysfunction108. Similar effects were observed with GRP78 autoantibodies in Lambert–Eaton myasthenic syndrome, suggesting that antibody-mediated disruption of the blood–brain barrier may represent a broader principle of pathology109. It is tempting to speculate that GRP78 antibodies — which are common in patients with cancer110 — may help drive the cognitive deficits seen in patients with cancer who have serum NMDAR autoantibodies of different isotypes111, and these autoantibodies may also be involved in other antibody-mediated diseases.
Antibody uptake into neurons
Only recently was the uptake of antibodies by neurons recognized as a mechanism to induce neuronal dysfunction with as yet limited experimental confirmation. Intrathecally injected amphiphysin-specific antibodies induced changes of vesicle-associated proteins in presynaptic nerve terminals112, which may relate to the transient extracellular exposure of amphiphysin during synaptic vesicle recycling113. Synapsin-specific antibodies inactivated their target after internalization by clathrin-dependent endocytosis into primary neurons114 (Fig. 3f). FcRs are highly expressed in neurons and could similarly internalize tau-specific antibodies in a mouse model of Alzheimer disease115.
Informative animal models
Animal models of antibody-mediated neurological diseases are being developed in parallel with clinical discoveries. Passive immunization is ideal for analysing the pathogenic effects of a given autoantibody, including molecular synaptic changes and behavioural abnormalities. By contrast, active immunization models are needed to explain early steps in the immunological cascade and the role of other immune cells, and for preclinical testing of B cell-targeting immunotherapies.
Starting with first attempts using a single injection of CSF or purified serum IgG from patients, various passive transfer models have become available. Administration of mGluR1 antibodies into the subarachnoid space of mice resulted in transient ataxia116. In NMO, AQP4-IgG was initially administered into experimental autoimmune encephalomyelitis animals, leading to spinal cord inflammation and complement deposition, but little demyelination87. Passive immunization of AQP4 autoantibodies in rodents was also combined with complement injection or was targeted into the spinal cord, optic nerve or retina, resulting in better recapitulation of disease117. Passive intrathecal transfer of CSF or monoclonal antibodies from patients with NMDAR encephalitis resulted in decreased NMDAR density in the hippocampus, hypofunction in NMDAR-mediated synaptic transmission, behavioural changes compatible with human disease and a low threshold for seizures118,119,120. Abnormalities of memory and behaviour were reversible and paralleled normalization of synaptic NMDARs, and no complement deposition was detected. Transfer of human AMPA receptor autoantibodies into mice impaired long-term synaptic plasticity and affected learning and memory9. Intrathecal transfer of amphiphysin antibodies led to decreased presynaptic inhibition due to disturbed GABAergic inhibition113.
Further refined models used recombinant monoclonal human autoantibodies, allowing intrathecal administration of defined autoantibody concentrations85. A human NMDAR-specific monoclonal antibody decreased synaptic density and caused reversible memory impairment50. Intraperitoneally injected hybridoma-derived monoclonal human NMDAR antibodies generated from the blood of a patient with NMDAR encephalitis resulted in increased wheel running activity121 and intraperitoneal injection of a monoclonal AQP4 antibody caused CNS pathology122. Similarly, intrathecal administration of monoclonal human GABAA receptor antibodies resulted in severe catatonia and epileptic seizures in mice, paralleled by characteristic electrophysiological changes45.
Active immunization models are much scarcer. Immunization with conformationally stabilized NMDAR tetramers induced murine encephalitis mimicking core behavioural and pathologic aspects of the disease, such as seizures, behavioural changes and immune cell infiltration into the hippocampus123. Both antibody-producing cells releasing NMDAR autoantibodies and T cells were essential123. AQP4 antibody production was induced with active immunization in Lewis rats using AQP4 mimotopes, which mimic parts of the extracellular loops of AQP4 (ref.124). Some work was also done with T cell transfer models in which highly encephalitogenic AQP4268–285-specific T cells infiltrated the CNS and reproduced some clinical features of NMO77. Despite obvious progress in the development of informative animal models, further refinements are needed as most models cover only a portion of the clinical phenotype or disease mechanisms.
Fetal brain development
Pathogenic anti-neuronal autoantibodies may be highly relevant also for the developing brain during pregnancy, when the blood–brain barrier is not fully developed. Antibodies crossing into the brain may cause a range of neurodevelopmental abnormalities and neuropsychiatric diseases, such as autism and attention deficit hyperactivity disorder125. An example of materno-fetal autoantibody transfer is arthrogryposis multiplex congenita, a severe neuromuscular developmental disorder due to antibodies to the fetal AChR. Milder forms present with AChR inactivation syndrome126.
Based on data generated in mice, other groups found impaired cortical development due to antibodies to NR2B (also known as GlUN2B or GRIN2B) in murine models of maternal lupus127 or developmental delay and behavioural abnormalities in the murine offspring after gestational treatment with human IgG from mothers who had a child with autism spectrum disorder (ASD)128. Another well-characterized anti-neuronal antibody, CASPR2, was identified in the mother of a child with autism and caused cortical plate thinning and an ASD-like phenotype in murine offspring129, similar to what is seen in genetic models of CASPR2 deficiency130. Similar synaptic and behavioural abnormalities in the offspring were seen after materno-fetal transfer of purified IgG from patients with CASPR2 antibodies131.
Given the high prevalence of NR1-NMDAR autoantibodies in blood samples of controls132 and the developmental deficits induced by transient NMDAR blockage133, we recently analysed the effect of human NR1 autoantibodies for brain development in a pregnant mouse model134. Indeed, monoclonal human NMDAR autoantibodies administered to pregnant dams resulted in massive enrichment in the fetal brain, impaired neurodevelopmental reflexes and electrophysiological changes in the offspring. Behavioural abnormalities included hyperactivity, lower anxiety and impaired sensorimotor gating, and reduced brain volumes persisted into adulthood134. In support, assessment of women with NMDAR encephalitis during pregnancy and short-term follow-up of the babies demonstrated >50% preterm deliveries and occurrence of child death135. In a related pregnancy model, injection of NMDAR antibody-containing human IgG similarly resulted in profound synaptic, cortical and behavioural abnormalities, which, however, reversed during adulthood136.
The possibility that transient antibody exposure during brain development might lead to lifelong psychiatric morbidity has broad medical and ethical implications. Of importance, low or even sub-threshold titres of maternal autoantibodies (that is, those that appear negative in routine autoantibody assays) may accumulate in the fetal brain to levels sufficient for permanent synaptic dysfunction134. Although not yet supported by case series, materno-fetal antibody transfer and consequent neuropsychiatric disease might be preventable by immunotherapy. In addition, it would be an entirely new concept in psychiatry and neurology to treat healthy persons (asymptomatic pregnant mothers) in a preventive attempt for their children, which might be facilitated by the ongoing development of antibody-specific treatments.
Autoantibodies in neurodegenerative diseases
Link between autoantibodies and neurodegeneration
Several well-established autoantibodies that define acute encephalopathies can occur in patients with slowly progressing cognitive decline or movement disorders mimicking classical neurodegenerative diseases. For example, encephalopathy caused by LGI1 antibodies can resemble the characteristic clinical picture of Alzheimer disease137, and antibodies specific for voltage-gated potassium channels can lead to suspected frontotemporal dementia138 or unspecified ‘reversible dementia’139, GFAP-specific antibodies have suspected involvement in Parkinson disease140, GABAB receptor-specific antibodies may cause symptoms mimicking amyotrophic lateral sclerosis (ALS)141 and several cell surface-binding autoantibodies have been shown to phenocopy Creutzfeldt–Jakob disease142. In this first category, the symptoms are caused by autoantibodies, but the unusual clinical presentation mimicking a neurodegenerative disease regularly leads to delayed diagnosis and incorrect treatments.
The second category comprises autoantibodies in established, pathology-proven neurodegenerative diseases, which may develop secondary to neurodegeneration. The antibody’s role awaits scientific clarification as to whether they are mere bystanders of a degenerative process or whether their pathogenic function can shape the disease. For example, in ALS there is a relatively strong inflammatory response including antibody deposition143, considered to contribute to disease progression and to additionally drive neurodegeneration144. Mice deficient in the most common ALS-associated gene, C9orf72, show a strong autoimmune phenotype with increased B cell activation145,146. Also, patients with ALS can have well-characterized autoantibodies, such as to LRP4, which bind to neuronal surfaces147.
The third category is even more intriguing as the traditional border between autoimmunity and neurodegenerative diseases becomes blurry. Patients with IgLON5-specific autoantibodies suffer from a sleep disorder with abnormal movements and cognitive decline11. It was considered an autoimmune disease, given cytoskeletal changes and reduced expression of IgLON5 after experimental antibody incubation and clinical improvement with immunotherapy99,148,149. However, neuropathological studies revealed deposition of hyperphosphorylated tau protein characteristic for neurodegeneration, indicative of a new ‘tauopathy’11. The example suggests a continuum between autoimmune encephalitis and autoimmune dementia, stimulating the fascinating — yet still controversial — idea that some autoantibodies may be primary drivers of neurodegeneration and that, vice versa, selective immunotherapy can delay, halt or even prevent a neurodegenerative disease. Along these lines, the potential causative role of autoantibodies after cerebral ischaemia for the development of post-stroke dementia150 and the role of synaptic autoantibodies for cognitive impairment in patients with cancer awaits confirmation111.
Antibodies associated with neurodegenerative diseases
Recently, more and more autoantibodies to proteins involved in neurodegeneration have been isolated from the pool of naturally occurring antibodies in healthy subjects, with these studies aiming for the identification of antibodies that modify turnover of target proteins or inhibit fibre aggregation. The human β-amyloid antibody with potent clearing capabilities, aducanumab, showed promise in early clinical trials151. Although phase III trials were stopped prematurely152, the company intends to seek regulatory approval based on data reanalysis. The reduction of high-affinity antibodies to α-synuclein in Parkinson disease in one study suggested a beneficial role for clearance of toxic proteins153. The inherent potential of this antibody source for the development of new treatments explains ongoing activity, such as the isolation of autoantibodies to hyperphosphorylated tau154, ALS-related misfolded SOD1 (ref.155) and α-synuclein and/or β-amyloid156.
The common finding of low-level naturally occurring antibodies targeting neurodegenerative proteins suggest an interesting new concept, which may also be true for synaptic autoantigens. ‘Smouldering’ low-level antibodies might continuously modify synaptic proteins, prune ion channel expression or prevent protein aggregation. Although clearly pathogenic at the molecular level, such antibodies might not cause clinically detectable effects in isolation. For example, antibodies to synapsin or prion protein can modify their targets, but are detectable in a broader spectrum of seemingly unlinked clinical conditions as well as in some healthy controls114,157. Thus, in neurodegenerative diseases, humoral autoimmunity may not be exclusively present or absent but, rather, subtly alter the progress of protein aggregation, misfolding and degeneration.
A risk factor modulating brain disease?
Given the above-mentioned effects of anti-neuronal autoantibodies on synaptic function and the possibility of smouldering humoral autoimmunity, then the prevalence of antibodies, their titres and duration in the CSF, certain antibody combinations and pathogenicity would together add a measurable risk to the development of neurological disease, reminiscent of a polygenic risk score or a metabolic profile. This hypothesis is supported by emerging concepts that disease is often not conferred by a single autoantibody but, rather, by a broader repertoire, such as demonstrated for GPCR-specific autoantibodies158. Individual serum autoantibody signatures can be stable over time regarding the number of reactivities and antigen specificity159.
It is highly appealing to work towards and confirm similar profiles for the autoantibody repertoire in the CSF. The antibody composition will have long-term influence on brain function and, vice versa, being influenced itself by the history of immunological events. Our current approach of cloning and determining specificity of single B cells and plasma cells in the CSF of patients with antibody-mediated neurological disease, patients with ‘classical’ neurodegenerative disorders and healthy subjects might generate distinct clusters of disease or brain state (‘brain antibody-omics’). Together with anticipated technical innovations for the identification of ‘difficult’ 3D conformational epitopes, antibody profiling in neurodegenerative diseases might yield novel exciting developments regarding biomarkers, mechanistic pathological understanding and treatment targets.
Emerging immunotherapy concepts
A powerful armamentarium for antibody reduction
Immunotherapies in antibody-mediated neurological diseases are widely used (Fig. 4a) and must be rapidly administered to prevent persisting deficits, in particular of memory and behaviour160. Common first-line regimes consist of pulsed intravenous methylprednisolone plus therapeutic apheresis or intravenous immunoglobulins55. In particular, depleting anti-CD20 monoclonal antibodies, such as rituximab161,162, have become the mainstay in many neuroimmunological departments, and further anti-CD20 monoclonals are available, such as ofatumumab, ublituximab, obinutuzumab and rituximab’s humanized version ocrelizumab163. Despite its efficacy, discontinuation may lead to relapses, indicating that B cell depletion cannot restore early immune checkpoints and that autoreactive B cells reoccur164.
a | A large variety of treatment approaches are currently available (green, red) or in clinical development (blue), focusing on different targets along the B cell lineage. b | An exciting new route is the development of highly antibody-selective immunotherapies. Chimeric autoantibody receptor (CAAR) T cells are engineered to detect and deplete B cells monospecific for a given autoantibody, such as N-methyl-d-aspartate receptor (NMDAR) autoantibodies, via extracellular presentation of the target autoantigen. c | The non-pathogenic aquaporin 4 (AQP4)-selective antibody ‘aquaporumab’ lacks complement-dependent cytotoxicity (CDC)/antibody-dependent cellular cytotoxicity (ADCC) effector functions and can outcompete pathogenic AQP4 patient antibodies. d | In an experimental approach using anti-CD138 antibodies conjugated to an autoantigen, binding of pathogenic autoantibodies may induce CDC/ADCC-mediated depletion of long-lived plasma cells. CAR, chimeric antigen receptor; FcRn, neonatal Fc receptor; i.v., intravenous.
The most recent developments include several promising immunotherapies for NMOSD. Three powerful studies have demonstrated clinical benefit with complement inactivation using eculizumab103, IL-6R inhibition using satralizumab165 and B cell depletion with the anti-CD19 antibody inebilizumab166. The proteasome inhibitor bortezomib that induces plasma cell apoptosis showed beneficial effects in NMOSD and NMDAR encephalitis167,168 and provided the rationale for a currently recruiting multicentre placebo-controlled double-blinded clinical trial of bortezomib in patients with autoimmune encephalitis169. In a patient with refractory CASPR2 encephalitis and a patient with refractory NMDAR encephalitis, targeting CD38 with daratumumab resulted in a profound decline in autoantibody titres, suggesting another new option to deplete plasma cells170,171.
Numerous further immunotherapy approaches are in more experimental phases of the pipeline, such as inactivation of autoantibodies with bacterial enzymes that either cleave or enzymatically deglycosylate immunoglobulins172,173. Also, inhibition of the neonatal Fc receptor (FcRn) with efgartigimod or rozanolixizumab reduced IgG concentrations in phase II trials in patients with myasthenia gravis174,175. Experiences from rheumatological diseases might also translate into antibody-mediated neurological diseases, such as early work with a CD40L-targeting non-antibody protein that inhibited human B cell activation and plasma cell differentiation176 or with anti-CD19/anti-CD20 chimeric antigen receptor (CAR) T cells177. Finally, the promising safety and efficacy data from autologous stem cell transplantation in multiple sclerosis178 may inspire similar studies in carefully selected patients with neurological disease.
Highly selective novel immunotherapies
Although efficient immunotherapies have become available for antibody-mediated neurological disease, they are usually not selective for the isolated depletion of disease-driving antibodies, thus having relevant side effects from global immunosuppression. An exciting new development aims for genetically engineered antibody-specific immunotherapy sparing beneficial antibodies. Related to the concept of CAR T cells for haematological malignancies, chimeric autoantibody receptor (CAAR) T cells were recently introduced to target autoimmune B cells in the blistering skin disease pemphigus vulgaris179. CAAR T cells expressed the skin autoantigen desmoglein 3 as the extracellular domain instead of a CAR antibody fragment and depleted antigen-specific autoreactive B cells in vitro and in vivo179. Similarly, CAAR T cells expressing domains of the coagulation factor VIII termed BAR (B cell antibody receptor) specifically depleted neutralizing antibody-producing B cells in haemophilic mice180. It is obvious that this strategy should be developed also in neurological conditions, such as we do for NMDAR autoantibodies (Fig. 4b). CAAR T cells may persist and prevent recurrence of pathogenic B cells, ensuring long-term remission, but safety issues have to be answered before initiation of clinical trials, such as ‘safety switches’ inducing apoptosis.
Other experimental antibody-selective immunotherapies include ‘aquaporumab’, an engineered high-affinity monoclonal AQP4 antibody without CDC/antibody-dependent cellular cytotoxicity effector functions, sterically blocking binding of lower-affinity polyclonal serum IgG181 (Fig. 4c). An entirely different approach depleted antigen-specific long-lived plasma cells in a murine model secreting ovalbumin (OVA) antibodies using an anti-CD138 antibody conjugated with OVA182 (Fig. 4d). If successful also with a human autoantigen, the principle could be amenable to diverse autoantigens. In the remote future, restoring immune tolerance via antigen-specific ‘tolerization’ might be an innovative approach to suppress antibody-mediated neurological diseases183.
Conclusion
Antibody-mediated neurological diseases are a rapidly growing group of variable clinical entities with multifaceted manifestation and often profound response to treatment. The underlying autoantibodies directly confer pathogenicity by targeting single ion channels or receptors responsible for brain function. The diverse mechanisms of disease include antibody-mediated receptor internalization, complement activation, disrupted protein–protein interaction and signalling. The far-reaching clinical and scientific implications relate to emerging evidence that humoral autoimmunity participates in a much larger spectrum of neurological diseases than previously thought, ranging from encephalitis and psychosis to movement disorders, neurodegenerative diseases and neurodevelopmental abnormalities during pregnancy. Future research should now clarify the underlying molecular mechanisms of both neurological pathology and immunological dysfunction, including the role of incomplete B cell checkpoints, altered BCR repertoires, B cell homing, T cell help, affinity maturation and the homeostatic antibody repertoire in the brain. New technologies will have to be included, such as adaptive immune receptor repertoire sequencing or high-throughput identification of conformational antigen epitopes. Given the defined monospecific targets, antibody-mediated neurological diseases might be the perfect models for innovative future antigen-specific treatments that could avoid the serious adverse effects seen with chronic immunosuppressive agents.
References
Lennon, V. A., Kryzer, T. J., Pittock, S. J., Verkman, A. S. & Hinson, S. R. IgG marker of optic-spinal multiple sclerosis binds to the aquaporin-4 water channel. J. Exp. Med. 202, 473–477 (2005).
Dalmau, J. et al. Paraneoplastic anti-N-methyl-d-aspartate receptor encephalitis associated with ovarian teratoma. Ann. Neurol. 61, 25–36 (2007). This article presents the first identification of the NMDAR NR1 subunit as the target in a new type of autoimmune encephalitis, which has now become the most common antibody-mediated form.
Pollak, T. A. et al. Autoimmune psychosis: an international consensus on an approach to the diagnosis and management of psychosis of suspected autoimmune origin. Lancet Psychiatry 7, 93–108 (2019).
Graus, F. et al. A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol. 15, 391–404 (2016). This first international consensus paper on the clinical diagnostic criteria for autoimmune encephalitis symbolizes the transformation of this new development into clinical pathways.
Graus, F. & Dalmau, J. Paraneoplastic neurological syndromes in the era of immune-checkpoint inhibitors. Nat. Rev. Clin. Oncol. 16, 535–548 (2019).
Dalmau, J. et al. An update on anti-NMDA receptor encephalitis for neurologists and psychiatrists: mechanisms and models. Lancet Neurol. 18, 1045–1057 (2019).
Petit-Pedrol, M. et al. Encephalitis with refractory seizures, status epilepticus, and antibodies to the GABAA receptor: a case series, characterisation of the antigen, and analysis of the effects of antibodies. Lancet Neurol. 13, 276–286 (2014).
Lancaster, E. et al. Antibodies to the GABAB receptor in limbic encephalitis with seizures: case series and characterisation of the antigen. Lancet Neurol. 9, 67–76 (2010).
Haselmann, H. et al. Human autoantibodies against the AMPA receptor subunit GluA2 induce receptor reorganization and memory dysfunction. Neuron 100, 91–105.e9 (2018).
Spatola, M. et al. Encephalitis with mGluR5 antibodies: symptoms and antibody effects. Neurology 90, e1964–e1972 (2018).
Sabater, L. et al. A novel non-rapid-eye movement and rapid-eye-movement parasomnia with sleep breathing disorder associated with antibodies to IgLON5: a case series, characterisation of the antigen, and post-mortem study. Lancet Neurol. 13, 575–586 (2014). This study identifies IgLON5 antibodies in a novel tauopathy, pointing to potentially broader overlapping mechanisms between antibody-mediated autoimmunity and neurodegenerative diseases.
Reindl, M. & Waters, P. Myelin oligodendrocyte glycoprotein antibodies in neurological disease. Nat. Rev. Neurol. 15, 89–102 (2019).
Fukata, Y. et al. LGI1–ADAM22–MAGUK configures transsynaptic nanoalignment for synaptic transmission and epilepsy prevention. Proc. Natl Acad. Sci. USA 118, e2022580118 (2021).
Kornau, H. C. et al. Human cerebrospinal fluid monoclonal LGI1 autoantibodies increase neuronal excitability. Ann. Neurol. 87, 405–418 (2020).
Petit-Pedrol, M. et al. LGI1 antibodies alter Kv1.1 and AMPA receptors changing synaptic excitability, plasticity and memory. Brain 141, 3144–3159 (2018).
Dawes, J. M. et al. Immune or genetic-mediated disruption of CASPR2 causes pain hypersensitivity due to enhanced primary afferent excitability. Neuron 97, 806–822.e10 (2018).
Dubey, D. et al. Autoimmune encephalitis epidemiology and a comparison to infectious encephalitis. Ann. Neurol. 83, 166–177 (2018).
Gable, M. S., Sheriff, H., Dalmau, J., Tilley, D. H. & Glaser, C. A. The frequency of autoimmune N-methyl-d-aspartate receptor encephalitis surpasses that of individual viral etiologies in young individuals enrolled in the california encephalitis project. Clin. Infect Dis. 54, 899–904 (2012).
Al-Diwani, A. et al. The psychopathology of NMDAR-antibody encephalitis in adults: a systematic review and phenotypic analysis of individual patient data. Lancet Psychiatry 6, 235–246 (2019).
Irani, S. R. et al. Faciobrachial dystonic seizures precede Lgi1 antibody limbic encephalitis. Ann. Neurol. 69, 892–900 (2011).
Fichtner, M. L., Jiang, R., Bourke, A., Nowak, R. J. & O’Connor, K. C. Autoimmune pathology in myasthenia gravis disease subtypes is governed by divergent mechanisms of immunopathology. Front. Immunol. 11, 776 (2020).
Toyka, K. V., Brachman, D. B., Pestronk, A. & Kao, I. Myasthenia gravis: passive transfer from man to mouse. Science 190, 397–399 (1975).
Newsom-Davis, J., Wilson, S. G., Vincent, A. & Ward, C. D. Long-term effects of repeated plasma exchange in myasthenia gravis. Lancet 1, 464–468 (1979).
Devinsky, O. et al. A cross-species approach to disorders affecting brain and behaviour. Nat. Rev. Neurol. 14, 677–686 (2018).
Pakozdy, A. et al. Suspected limbic encephalitis and seizure in cats associated with voltage-gated potassium channel (VGKC) complex antibody. J. Vet. Intern. Med. 27, 212–214 (2013).
Matsuki, N. et al. Prevalence of autoantibody in cerebrospinal fluids from dogs with various CNS diseases. J. Vet. Med. Sci. 66, 295–297 (2004).
Fang, B. et al. Autoimmune glial fibrillary acidic protein astrocytopathy: a novel meningoencephalomyelitis. JAMA Neurol. 73, 1297–1307 (2016).
Prüss, H. et al. Anti-NMDA receptor encephalitis in the polar bear (Ursus maritimus) Knut. Sci. Rep. 5, 12805 (2015).
Pan, H. et al. Uncoupling the widespread occurrence of anti-NMDAR1 autoantibodies from neuropsychiatric disease in a novel autoimmune model. Mol. Psychiatry 24, 1489–1501 (2019).
Honorat, J. A. et al. Autoimmune septin-5 cerebellar ataxia. Neurol. Neuroimmunol. Neuroinflamm 5, e474 (2018).
Scharf, M. et al. A spectrum of neural autoantigens, newly identified by histo-immunoprecipitation, mass spectrometry, and recombinant cell-based indirect immunofluorescence. Front. Immunol. 9, 1447 (2018).
Popkirov, S. et al. Rho-associated protein kinase 2 (ROCK2): a new target of autoimmunity in paraneoplastic encephalitis. Acta Neuropathol. Commun. 5, 40 (2017).
Zekeridou, A. et al. Phosphodiesterase 10A IgG: a novel biomarker of paraneoplastic neurologic autoimmunity. Neurology 93, e815–e822 (2019).
Honorat, J. A. et al. Autoimmune gait disturbance accompanying adaptor protein-3B2-IgG. Neurology 93, e954–e963 (2019).
Miske, R. et al. Neurochondrin is a neuronal target antigen in autoimmune cerebellar degeneration. Neurol. Neuroimmunol. Neuroinflamm. 4, e307 (2016).
Pitsch, J. et al. Drebrin autoantibodies in patients with seizures and suspected encephalitis. Ann. Neurol. 87, 869–884 (2020).
Berridge, G. et al. Glutamate receptor δ2 serum antibodies in pediatric opsoclonus myoclonus ataxia syndrome. Neurology 91, e714–e723 (2018).
Flanagan, E. P. et al. Glial fibrillary acidic protein immunoglobulin G as biomarker of autoimmune astrocytopathy: analysis of 102 patients. Ann. Neurol. 81, 298–309 (2017).
Mandel-Brehm, C. et al. Kelch-like protein 11 antibodies in seminoma-associated paraneoplastic encephalitis. N. Engl. J. Med. 381, 47–54 (2019). This article identifies KLHL11 autoantibodies indicating both the continuously ongoing discovery of further autoantibodies and the increasing technical refinement to identify undetermined epitopes.
Wardemann, H. et al. Predominant autoantibody production by early human B cell precursors. Science 301, 1374–1377 (2003).
Cyster, J. G. & Allen, C. D. C. B cell responses: cell interaction dynamics and decisions. Cell 177, 524–540 (2019).
Nemazee, D. Mechanisms of central tolerance for B cells. Nat. Rev. Immunol. 17, 281–294 (2017).
Cotzomi, E. et al. Early B cell tolerance defects in neuromyelitis optica favour anti-AQP4 autoantibody production. Brain 142, 1598–1615 (2019).
Rawlings, D. J., Metzler, G., Wray-Dutra, M. & Jackson, S. W. Altered B cell signalling in autoimmunity. Nat. Rev. Immunol. 17, 421–436 (2017).
Kreye, J. et al. Encephalitis patient derived monoclonal GABAA receptor antibodies cause catatonia and epileptic seizures. Preprint at bioRxiv https://doi.org/10.1101/2021.01.28.428602 (2021).
Brändle, S. M. et al. Cross-reactivity of a pathogenic autoantibody to a tumor antigen in GABAA receptor encephalitis. Proc. Natl Acad. Sci. USA 118, e1916337118 (2021).
Vinuesa, C. G., Sanz, I. & Cook, M. C. Dysregulation of germinal centres in autoimmune disease. Nat. Rev. Immunol. 9, 845–857 (2009).
Kreye, J. et al. Human cerebrospinal fluid monoclonal N-methyl-d-aspartate receptor autoantibodies are sufficient for encephalitis pathogenesis. Brain 139, 2641–2652 (2016). This article presents the first cloning and recombinant production of monoclonal human NMDAR NR1 autoantibodies, enabling understanding of antibody pathogenicity and immune mechanisms at a molecular level.
Wenke, N. K. et al. N-Methyl-d-aspartate receptor dysfunction by unmutated human antibodies against the NR1 subunit. Ann. Neurol. 85, 771–776 (2019).
Malviya, M. et al. NMDAR encephalitis: passive transfer from man to mouse by a recombinant antibody. Ann. Clin. Transl Neurol. 4, 768–783 (2017).
Fischer, A., Provot, J., Jais, J. P., Alcais, A. & Mahlaoui, N. Autoimmune and inflammatory manifestations occur frequently in patients with primary immunodeficiencies. J. Allergy Clin. Immunol. 140, 1388–1393.e8 (2017).
Akman, C. I., Patterson, M. C., Rubinstein, A. & Herzog, R. Limbic encephalitis associated with anti-GAD antibody and common variable immune deficiency. Dev. Med. Child. Neurol. 51, 563–567 (2009).
Vogrig, A. et al. Central nervous system complications associated with immune checkpoint inhibitors. J. Neurol. Neurosurg. Psychiatry 91, 772–778 (2020).
Darnell, R. B. & Posner, J. B. Paraneoplastic syndromes involving the nervous system. N. Engl. J. Med. 349, 1543–1554 (2003).
Titulaer, M. J. et al. Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: an observational cohort study. Lancet Neurol. 12, 157–165 (2013).
Makuch, M. et al. N-Methyl-d-aspartate receptor antibody production from germinal center reactions: therapeutic implications. Ann. Neurol. 83, 553–561 (2018).
Day, G. S., Laiq, S., Tang-Wai, D. F. & Munoz, D. G. Abnormal neurons in teratomas in NMDAR encephalitis. JAMA Neurol. 71, 717–724 (2014).
Prüss, H. et al. N-Methyl-d-aspartate receptor antibodies in herpes simplex encephalitis. Ann. Neurol. 72, 902–911 (2012). This study shows a high rate of NMDAR autoantibodies in patients with herpes encephalitis and suggests a causal relationship related to virus-induced humoral autoimmunity.
Armangue, T. et al. Herpes simplex virus encephalitis is a trigger of brain autoimmunity. Ann. Neurol. 75, 317–323 (2014).
Armangue, T. et al. Frequency, symptoms, risk factors, and outcomes of autoimmune encephalitis after herpes simplex encephalitis: a prospective observational study and retrospective analysis. Lancet Neurol. 17, 760–772 (2018). This prospective study demonstrates that 27% of patients with herpes encephalitis develop immunotherapy-responsive NMDAR encephalitis, usually within 2 months.
Linnoila, J. et al. Mouse model of anti-NMDA receptor post-herpes simplex encephalitis. Neurol. Neuroimmunol. Neuroinflamm. 6, e529 (2018).
Linnoila, J. J., Binnicker, M. J., Majed, M., Klein, C. J. & McKeon, A. CSF herpes virus and autoantibody profiles in the evaluation of encephalitis. Neurol. Neuroimmunol. Neuroinflamm. 3, e245 (2016).
Prüss, H. Postviral autoimmune encephalitis: manifestations in children and adults. Curr. Opin. Neurol. 30, 327–333 (2017).
Franke, C. et al. High frequency of cerebrospinal fluid autoantibodies in COVID-19 patients with neurological symptoms. Brain Behav. Immun. 93, 415–419 (2020).
Kuraoka, M. et al. BCR and endosomal TLR signals synergize to increase AID expression and establish central B cell tolerance. Cell Rep. 18, 1627–1635 (2017).
Kolhatkar, N. S. et al. Altered BCR and TLR signals promote enhanced positive selection of autoreactive transitional B cells in Wiskott–Aldrich syndrome. J. Exp. Med. 212, 1663–1677 (2015).
Sanderson, N. S. et al. Cocapture of cognate and bystander antigens can activate autoreactive B cells. Proc. Natl Acad. Sci. USA 114, 734–739 (2017).
Kain, R. et al. Molecular mimicry in pauci-immune focal necrotizing glomerulonephritis. Nat. Med. 14, 1088–1096 (2008).
Lafaille, F. G. et al. Human SNORA31 variations impair cortical neuron-intrinsic immunity to HSV-1 and underlie herpes simplex encephalitis. Nat. Med. 25, 1873–1884 (2019).
Amara, K. et al. Monoclonal IgG antibodies generated from joint-derived B cells of RA patients have a strong bias toward citrullinated autoantigen recognition. J. Exp. Med. 210, 445–455 (2013).
Meyer, S. et al. AIRE-deficient patients harbor unique high-affinity disease-ameliorating autoantibodies. Cell 166, 582–595 (2016).
Kearney, J. F., Patel, P., Stefanov, E. K. & King, R. G. Natural antibody repertoires: development and functional role in inhibiting allergic airway disease. Annu. Rev. Immunol. 33, 475–504 (2015).
Schickel, J. N. et al. Self-reactive VH4-34-expressing IgG B cells recognize commensal bacteria. J. Exp. Med. 214, 1991–2003 (2017).
Burnett, D. L., Reed, J. H., Christ, D. & Goodnow, C. C. Clonal redemption and clonal anergy as mechanisms to balance B cell tolerance and immunity. Immunol. Rev. 292, 61–75 (2019).
Varrin-Doyer, M. et al. Aquaporin 4-specific T cells in neuromyelitis optica exhibit a TH17 bias and recognize Clostridium ABC transporter. Ann. Neurol. 72, 53–64 (2012).
Sagan, S. A. et al. Tolerance checkpoint bypass permits emergence of pathogenic T cells to neuromyelitis optica autoantigen aquaporin-4. Proc. Natl Acad. Sci. USA 113, 14781–14786 (2016).
Zeka, B. et al. Aquaporin 4-specific T cells and NMO-IgG cause primary retinal damage in experimental NMO/SD. Acta Neuropathol. Commun. 4, 82 (2016).
Dabner, M. et al. Ovarian teratoma associated with anti-N-methyl d-aspartate receptor encephalitis: a report of 5 cases documenting prominent intratumoral lymphoid infiltrates. Int J. Gynecol. Pathol. 31, 429–437 (2012).
Dao, L. M. et al. Decreased inflammatory cytokine production of antigen-specific CD4+ T cells in NMDA receptor encephalitis. J. Neurol. https://doi.org/10.1007/s00415-020-10371-y (2021).
Iversen, R. & Sollid, L. M. Autoimmunity provoked by foreign antigens. Science 368, 132–133 (2020).
du Pre, M. F. et al. B cell tolerance and antibody production to the celiac disease autoantigen transglutaminase 2. J. Exp. Med. 217, e20190860 (2020).
Cree, B. A., Spencer, C. M., Varrin-Doyer, M., Baranzini, S. E. & Zamvil, S. S. Gut microbiome analysis in neuromyelitis optica reveals overabundance of Clostridium perfringens. Ann. Neurol. 80, 443–447 (2016).
Herken, J. et al. Normal gut microbiome in NMDA receptor encephalitis. Neurol. Neuroimmunol. Neuroinflamm. 6, e632 (2019).
Nutt, S. L., Hodgkin, P. D., Tarlinton, D. M. & Corcoran, L. M. The generation of antibody-secreting plasma cells. Nat. Rev. Immunol. 15, 160–171 (2015).
Zou, A., Ramanathan, S., Dale, R. C. & Brilot, F. Single-cell approaches to investigate B cells and antibodies in autoimmune neurological disorders. Cell Mol. Immunol. 18, 294–306 (2021).
Takata, K. et al. Characterization of pathogenic monoclonal autoantibodies derived from muscle-specific kinase myasthenia gravis patients. JCI Insight 4, e127167 (2019).
Bennett, J. L. et al. Intrathecal pathogenic anti-aquaporin-4 antibodies in early neuromyelitis optica. Ann. Neurol. 66, 617–629 (2009). This study demonstrates cloning of intrathecal AQP4 autoantibodies, thereby paving the way for monoclonal human autoantibody technology to become state of the art in antibody-mediated neurological diseases.
Kreye, J. et al. A therapeutic non-self-reactive SARS-CoV-2 antibody protects from lung pathology in a COVID-19 hamster model. Cell 183, 1058–1069 (2020).
Ramberger, M. et al. Distinctive binding properties of human monoclonal LGI1 autoantibodies determine pathogenic mechanisms. Brain 143, 1731–1745 (2020).
Jezequel, J. et al. Dynamic disorganization of synaptic NMDA receptors triggered by autoantibodies from psychotic patients. Nat. Commun. 8, 1791 (2017). This study demonstrates different molecular mechanisms on synaptic transmission by NMDAR autoantibodies derived from patients with schizophrenia versus healthy controls.
Moscato, E. H. et al. Acute mechanisms underlying antibody effects in anti-N-methyl-d-aspartate receptor encephalitis. Ann. Neurol. 76, 108–119 (2014).
Ladepeche, L. et al. NMDA receptor autoantibodies in autoimmune encephalitis cause a subunit-specific nanoscale redistribution of NMDA receptors. Cell Rep. 23, 3759–3768 (2018).
Lai, M. et al. AMPA receptor antibodies in limbic encephalitis alter synaptic receptor location. Ann. Neurol. 65, 424–434 (2009).
Peng, X. et al. Cellular plasticity induced by anti-α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor encephalitis antibodies. Ann. Neurol. 77, 381–398 (2015).
Crisp, S. J. et al. Glycine receptor autoantibodies disrupt inhibitory neurotransmission. Brain 142, 3398–3410 (2019).
Carvajal-Gonzalez, A. et al. Glycine receptor antibodies in PERM and related syndromes: characteristics, clinical features and outcomes. Brain 137, 2178–2192 (2014).
Pettingill, P. et al. Antibodies to GABAA receptor α1 and γ2 subunits: clinical and serologic characterization. Neurology 84, 1233–1241 (2015).
Gresa-Arribas, N. et al. Human neurexin-3α antibodies associate with encephalitis and alter synapse development. Neurology 86, 2235–2242 (2016).
Landa, J. et al. Effects of IgLON5 antibodies on neuronal cytoskeleton: a link between autoimmunity and neurodegeneration. Ann. Neurol. 88, 1023–1027 (2020).
Sabater, L., Planaguma, J., Dalmau, J. & Graus, F. Cellular investigations with human antibodies associated with the anti-IgLON5 syndrome. J. Neuroinflammation 13, 226 (2016).
Bien, C. G. et al. Immunopathology of autoantibody-associated encephalitides: clues for pathogenesis. Brain 135, 1622–1638 (2012).
Soltys, J. et al. Membrane assembly of aquaporin-4 autoantibodies regulates classical complement activation in neuromyelitis optica. J. Clin. Invest. 129, 2000–2013 (2019). This study analyses the molecular mechanisms that are required for AQP4 autoantibodies to activate complement, helping to understand organ-specific damage.
Pittock, S. J. et al. Eculizumab in aquaporin-4-positive neuromyelitis optica spectrum disorder. N. Engl. J. Med. 381, 614–625 (2019).
Piepgras, J. et al. Anti-DPPX encephalitis: pathogenic effects of antibodies on gut and brain neurons. Neurology 85, 890–897 (2015).
Koneczny, I. A new classification system for IgG4 autoantibodies. Front. Immunol. 9, 97 (2018).
Brilot, F. et al. Antibodies to native myelin oligodendrocyte glycoprotein in children with inflammatory demyelinating central nervous system disease. Ann Neurol 66, 833–842 (2009).
Asavapanumas, N. & Verkman, A. S. Neuromyelitis optica pathology in rats following intraperitoneal injection of NMO-IgG and intracerebral needle injury. Acta Neuropathol. Commun. 2, 48 (2014).
Shimizu, F. et al. Glucose-regulated protein 78 autoantibody associates with blood–brain barrier disruption in neuromyelitis optica. Sci. Transl Med. 9, eaai9111 (2017).
Shimizu, F. et al. GRP78 antibodies damage the blood–brain barrier and relate to cerebellar degeneration in Lambert–Eaton myasthenic syndrome. Brain 142, 2253–2264 (2019).
Mintz, P. J. et al. Fingerprinting the circulating repertoire of antibodies from cancer patients. Nat. Biotechnol. 21, 57–63 (2003).
Bartels, F. et al. Neuronal autoantibodies associated with cognitive impairment in melanoma patients. Ann. Oncol. 30, 823–829 (2019).
Werner, C. et al. Human autoantibodies to amphiphysin induce defective presynaptic vesicle dynamics and composition. Brain 139, 365–379 (2016).
Geis, C. et al. Stiff person syndrome-associated autoantibodies to amphiphysin mediate reduced GABAergic inhibition. Brain 133, 3166–3180 (2010).
Rocchi, A. et al. Autoantibodies to synapsin I sequestrate synapsin I and alter synaptic function. Cell Death Dis. 10, 864 (2019).
Congdon, E. E., Gu, J., Sait, H. B. & Sigurdsson, E. M. Antibody uptake into neurons occurs primarily via clathrin-dependent Fcγ receptor endocytosis and is a prerequisite for acute tau protein clearance. J. Biol. Chem. 288, 35452–35465 (2013).
Sillevis Smitt, P. et al. Paraneoplastic cerebellar ataxia due to autoantibodies against a glutamate receptor. N. Engl. J. Med. 342, 21–27 (2000).
Duan, T. & Verkman, A. S. Experimental animal models of aquaporin-4-IgG-seropositive neuromyelitis optica spectrum disorders: progress and shortcomings. Brain Pathol. 30, 13–25 (2020).
Planaguma, J. et al. Human N-methyl d-aspartate receptor antibodies alter memory and behaviour in mice. Brain 138, 94–109 (2015). This article presents the first experiments demonstrating reversible behavioural and memory changes in mice after intrathecal administration of NMDAR antibody-containing CSF.
Wright, S. et al. Epileptogenic effects of NMDAR antibodies in a passive transfer mouse model. Brain 138, 3159–3167 (2015).
Wright, S. K. et al. In vitro characterisation and neurosteroid treatment of an N-methyl-d-aspartate receptor antibody-mediated seizure model. Preprint at bioRxiv https://doi.org/10.1101/2020.12.22.423962 (2021).
Sharma, R. et al. Monoclonal antibodies from a patient with anti-NMDA receptor encephalitis. Ann. Clin. Transl Neurol. 5, 935–951 (2018).
Hillebrand, S. et al. Circulating AQP4-specific auto-antibodies alone can induce neuromyelitis optica spectrum disorder in the rat. Acta Neuropathol. 137, 467–485 (2019).
Jones, B. E. et al. Autoimmune receptor encephalitis in mice induced by active immunization with conformationally stabilized holoreceptors. Sci. Transl Med. 11, eaaw0044 (2019). This article presents the first active immunization model of NMDAR encephalitis using conformationally stabilized NMDAR that resembles the human phenotype and may facilitate the development of new treatments.
Tsymala, I. et al. Induction of aquaporin 4-reactive antibodies in Lewis rats immunized with aquaporin 4 mimotopes. Acta Neuropathol. Commun. 8, 49 (2020).
Gata-Garcia, A. & Diamond, B. Maternal antibody and ASD: clinical data and animal models. Front. Immunol. 10, 1129 (2019).
Hacohen, Y. et al. Fetal acetylcholine receptor inactivation syndrome: a myopathy due to maternal antibodies. Neurol. Neuroimmunol. Neuroinflamm. 2, e57 (2014).
Lee, J. Y. et al. Neurotoxic autoantibodies mediate congenital cortical impairment of offspring in maternal lupus. Nat. Med. 15, 91–96 (2009).
Braunschweig, D. et al. Maternal autism-associated IgG antibodies delay development and produce anxiety in a mouse gestational transfer model. J. Neuroimmunol. 252, 56–65 (2012).
Brimberg, L. et al. Caspr2-reactive antibody cloned from a mother of an ASD child mediates an ASD-like phenotype in mice. Mol. Psychiatry 21, 1663–1671 (2016). This article shows that monoclonal CASPR2 autoantibodies generated from a mother of a child with ASD mimicked an ASD phenotype in mice and thus demonstrates the vast potential role of materno-fetal antibody transfer for lifelong neuropsychiatric morbidity.
Penagarikano, O. et al. Absence of CNTNAP2 leads to epilepsy, neuronal migration abnormalities, and core autism-related deficits. Cell 147, 235–246 (2011).
Coutinho, E. et al. Persistent microglial activation and synaptic loss with behavioral abnormalities in mouse offspring exposed to CASPR2-antibodies in utero. Acta Neuropathol. 134, 567–583 (2017).
Hammer, C. et al. Neuropsychiatric disease relevance of circulating anti-NMDA receptor autoantibodies depends on blood–brain barrier integrity. Mol. Psychiatry 19, 1143–1149 (2013).
Stefani, M. R. & Moghaddam, B. Transient N-methyl-d-aspartate receptor blockade in early development causes lasting cognitive deficits relevant to schizophrenia. Biol. Psychiatry 57, 433–436 (2005).
Jurek, B. et al. Human gestational N-methyl-d-aspartate receptor autoantibodies impair neonatal murine brain function. Ann. Neurol. 86, 656–670 (2019).
Joubert, B. et al. Pregnancy outcomes in anti-NMDA receptor encephalitis: case series. Neurol. Neuroimmunol. Neuroinflamm. 7, e668 (2020).
Garcia-Serra, A. et al. Placental transfer of NMDAR antibodies causes reversible alterations in mice. Neurol. Neuroimmunol. Neuroinflamm. 8, e915 (2020).
Marquetand, J. et al. Slowly progressive LGI1 encephalitis with isolated late-onset cognitive dysfunction: a treatable mimic of Alzheimer’s disease. Eur. J. Neurol. 23, e28–e29 (2016).
McKeon, A. et al. Potassium channel antibody associated encephalopathy presenting with a frontotemporal dementia like syndrome. Arch. Neurol. 64, 1528–1530 (2007).
Reintjes, W., Romijn, M. D., Hollander, D., Ter Bruggen, J. P. & van Marum, R. J. Reversible dementia: two nursing home patients with voltage-gated potassium channel antibody-associated limbic encephalitis. J. Am. Med. Dir. Assoc. 16, 790–794 (2015).
Tomczak, A. et al. A case of GFAP-astroglial autoimmunity presenting with reversible parkinsonism. Mult. Scler. Relat. Disord. 39, 101900 (2019).
Schumacher, H., Meyer, T. & Prüss, H. GABAB receptor encephalitis in a patient diagnosed with amyotrophic lateral sclerosis. BMC Neurol. 19, 41 (2019).
Maat, P. et al. Pathologically confirmed autoimmune encephalitis in suspected Creutzfeldt–Jakob disease. Neurol. Neuroimmunol. Neuroinflamm. 2, e178 (2015).
May, C. et al. Highly immunoreactive IgG antibodies directed against a set of twenty human proteins in the sera of patients with amyotrophic lateral sclerosis identified by protein array. PLoS ONE 9, e89596 (2014).
Liu, J. & Wang, F. Role of neuroinflammation in amyotrophic lateral sclerosis: cellular mechanisms and therapeutic implications. Front. Immunol. 8, 1005 (2017).
O’Rourke, J. G. et al. C9orf72 is required for proper macrophage and microglial function in mice. Science 351, 1324–1329 (2016).
Atanasio, A. et al. C9orf72 ablation causes immune dysregulation characterized by leukocyte expansion, autoantibody production, and glomerulonephropathy in mice. Sci. Rep. 6, 23204 (2016).
Tüzün, E. et al. LRP4 antibody positive amyotrophic lateral sclerosis patients display neuropil-reactive IgG and enhanced serum complement levels. Immunol. Lett. 203, 54–56 (2018).
Gaig, C. et al. Clinical manifestations of the anti-IgLON5 disease. Neurology 88, 1736–1743 (2017).
Honorat, J. A. et al. IgLON5 antibody: neurological accompaniments and outcomes in 20 patients. Neurol. Neuroimmunol. Neuroinflamm. 4, e385 (2017).
Doyle, K. P. & Buckwalter, M. S. Does B lymphocyte-mediated autoimmunity contribute to post-stroke dementia? Brain Behav. Immun. 64, 1–8 (2017).
Sevigny, J. et al. The antibody aducanumab reduces Aβ plaques in Alzheimer’s disease. Nature 537, 50–56 (2016).
Long, J. M. & Holtzman, D. M. Alzheimer disease: an update on pathobiology and treatment strategies. Cell 179, 312–339 (2019).
Brudek, T. et al. Autoimmune antibody decline in Parkinson’s disease and multiple system atrophy; a step towards immunotherapeutic strategies. Mol. Neurodegener. 12, 44 (2017).
Pascual, G. et al. Immunological memory to hyperphosphorylated tau in asymptomatic individuals. Acta Neuropathol. 133, 767–783 (2017).
Maier, M. et al. A human-derived antibody targets misfolded SOD1 and ameliorates motor symptoms in mouse models of amyotrophic lateral sclerosis. Sci. Transl Med. 10, eaah3924 (2018).
Albus, A., Jordens, M., Moller, M. & Dodel, R. Encoding the sequence of specific autoantibodies against β-amyloid and α-synuclein in neurodegenerative diseases. Front. Immunol. 10, 2033 (2019).
Frontzek, K. et al. Autoantibodies against the prion protein in individuals with PRNP mutations. Neurology 95, e2028–e2037 (2020).
Cabral-Marques, O. et al. GPCR-specific autoantibody signatures are associated with physiological and pathological immune homeostasis. Nat. Commun. 9, 5224 (2018). This study analyses GPCR-specific autoantibodies, and strengthens the concept that a ‘network’ of functionally related autoantibodies can provide homeostatic functions, which may also apply to anti-neuronal autoantibodies.
Neiman, M. et al. Individual and stable autoantibody repertoires in healthy individuals. Autoimmunity 52, 1–11 (2019).
Finke, C. et al. Evaluation of cognitive deficits and structural hippocampal damage in encephalitis with leucine-rich, glioma-inactivated 1 antibodies. JAMA Neurol. 74, 50–59 (2017).
Damato, V., Evoli, A. & Iorio, R. Efficacy and safety of rituximab therapy in neuromyelitis optica spectrum disorders: a systematic review and meta-analysis. JAMA Neurol. 73, 1342–1348 (2016).
Lee, W. J. et al. Rituximab treatment for autoimmune limbic encephalitis in an institutional cohort. Neurology 86, 1683–1691 (2016).
Graf, J. et al. Targeting B cells to modify MS, NMOSD, and MOGAD: Part 1. Neurol. Neuroimmunol. Neuroinflamm. 8, e918 (2021).
Chamberlain, N. et al. Rituximab does not reset defective early B cell tolerance checkpoints. J. Clin. Invest. 126, 282–287 (2016).
Yamamura, T. et al. Trial of satralizumab in neuromyelitis optica spectrum disorder. N. Engl. J. Med. 381, 2114–2124 (2019).
Cree, B. A. C. et al. Inebilizumab for the treatment of neuromyelitis optica spectrum disorder (N-MOmentum): a double-blind, randomised placebo-controlled phase 2/3 trial. Lancet 394, 1352–1363 (2019).
Zhang, C. et al. Safety and efficacy of bortezomib in patients with highly relapsing neuromyelitis optica spectrum disorder. JAMA Neurol. 74, 1010–1012 (2017). This article presents the successful depletion of long-lived plasma cells by proteasome inhibition, demonstrating the potential as a new treatment in antibody-mediated neurological diseases.
Scheibe, F. et al. Bortezomib for treatment of therapy-refractory anti-NMDA receptor encephalitis. Neurology 88, 366–370 (2017).
Wickel, J. et al. Generate-Boost: study protocol for a prospective, multicenter, randomized controlled, double-blinded phase II trial to evaluate efficacy and safety of bortezomib in patients with severe autoimmune encephalitis. Trials 21, 625 (2020).
Scheibe, F. et al. Daratumumab treatment for therapy-refractory anti-CASPR2 encephalitis. J. Neurol. 267, 317–323 (2020).
Ratuszny, D. et al. Case report: daratumumab in a patient with severe refractory anti-NMDA receptor encephalitis. Front. Neurol. 11, 602102 (2020).
Tradtrantip, L., Asavapanumas, N. & Verkman, A. S. Therapeutic cleavage of anti-aquaporin-4 autoantibody in neuromyelitis optica by an IgG-selective proteinase. Mol. Pharmacol. 83, 1268–1275 (2013).
Tradtrantip, L., Ratelade, J., Zhang, H. & Verkman, A. S. Enzymatic deglycosylation converts pathogenic neuromyelitis optica anti-aquaporin-4 immunoglobulin G into therapeutic antibody. Ann. Neurol. 73, 77–85 (2013).
Bril, V. et al. Efficacy and safety of rozanolixizumab in moderate to severe generalized myasthenia gravis: a phase 2 randomized control trial. Neurology 96, e853–e865 (2021).
Howard, J. F. Jr. et al. Randomized phase 2 study of FcRn antagonist efgartigimod in generalized myasthenia gravis. Neurology 92, e2661–e2673 (2019).
Karnell, J. L. et al. A CD40L-targeting protein reduces autoantibodies and improves disease activity in patients with autoimmunity. Sci. Transl Med. 11, eaar6584 (2019).
Kansal, R. et al. Sustained B cell depletion by CD19-targeted CAR T cells is a highly effective treatment for murine lupus. Sci. Transl Med. 11, eaav1648 (2019).
Burt, R. K. et al. Effect of nonmyeloablative hematopoietic stem cell transplantation vs continued disease-modifying therapy on disease progression in patients with relapsing-remitting multiple sclerosis: a randomized clinical trial. JAMA 321, 165–174 (2019).
Ellebrecht, C. T. et al. Reengineering chimeric antigen receptor T cells for targeted therapy of autoimmune disease. Science 353, 179–184 (2016). This article presents the development of CAAR T cells to treat a DSG3 antibody-mediated skin disease, indicating that the CAAR concept should also be explored to specifically deplete autoantibodies that cause neurological diseases.
Parvathaneni, K. & Scott, D. W. Engineered FVIII-expressing cytotoxic T cells target and kill FVIII-specific B cells in vitro and in vivo. Blood Adv. 2, 2332–2340 (2018).
Duan, T., Tradtrantip, L., Phuan, P. W., Bennett, J. L. & Verkman, A. S. Affinity-matured ‘aquaporumab’ anti-aquaporin-4 antibody for therapy of seropositive neuromyelitis optica spectrum disorders. Neuropharmacology 162, 107827 (2020).
Cheng, Q. et al. Selective depletion of plasma cells in vivo based on the specificity of their secreted antibodies. Eur. J. Immunol. 50, 284–291 (2020).
Steinman, L. et al. Restoring immune tolerance in neuromyelitis optica: Part I. Neurol. Neuroimmunol. Neuroinflamm. 3, e276 (2016).
Irani, S. R. et al. Antibodies to Kv1 potassium channel-complex proteins leucine-rich, glioma inactivated 1 protein and contactin-associated protein-2 in limbic encephalitis, Morvan’s syndrome and acquired neuromyotonia. Brain 133, 2734–2748 (2010).
Lai, M. et al. Investigation of LGI1 as the antigen in limbic encephalitis previously attributed to potassium channels: a case series. Lancet Neurol. 9, 776–785 (2010).
Hutchinson, M. et al. Progressive encephalomyelitis, rigidity, and myoclonus: a novel glycine receptor antibody. Neurology 71, 1291–1292 (2008).
Lancaster, E. et al. Antibodies to metabotropic glutamate receptor 5 in the Ophelia syndrome. Neurology 77, 1698–1701 (2011).
Boronat, A. et al. Encephalitis and antibodies to dipeptidyl-peptidase-like protein-6, a subunit of Kv4.2 potassium channels. Ann. Neurol. 73, 120–128 (2013).
Dale, R. C. et al. Antibodies to surface dopamine-2 receptor in autoimmune movement and psychiatric disorders. Brain 135, 3453–3468 (2012).
Folli, F. et al. Autoantibodies to a 128-kd synaptic protein in three women with the stiff-man syndrome and breast cancer. N. Engl. J. Med. 328, 546–551 (1993).
Piepgras, J. et al. Intrathecal immunoglobulin A and G antibodies to synapsin in a patient with limbic encephalitis. Neurol. Neuroimmunol. Neuroinflamm. 2, e169 (2015).
Sun, B., Ramberger, M., O’Connor, K. C., Bashford-Rogers, R. J. M. & Irani, S. R. The B cell immunobiology that underlies CNS autoantibody-mediated diseases. Nat. Rev. Neurol. 16, 481–492 (2020).
Reiber, H., Ungefehr, S. & Jacobi, C. The intrathecal, polyspecific and oligoclonal immune response in multiple sclerosis. Mult. Scler. 4, 111–117 (1998).
Prüss, H. et al. Retrospective analysis of NMDA receptor antibodies in encephalitis of unknown origin. Neurology 75, 1735–1739 (2010). This retrospective analysis of archived CSF demonstrates that NMDAR encephalitis can account for 1% of young patients on a neuro-ICU. The study suggests that patients with other yet to be defined autoantibodies may still be overlooked.
Gresa-Arribas, N. et al. Antibody titres at diagnosis and during follow-up of anti-NMDA receptor encephalitis: a retrospective study. Lancet Neurol. 13, 167–177 (2014).
van Sonderen, A. et al. Anti-LGI1 encephalitis: clinical syndrome and long-term follow-up. Neurology 87, 1449–1456 (2016).
Dendrou, C. A., Petersen, J., Rossjohn, J. & Fugger, L. HLA variation and disease. Nat. Rev. Immunol. 18, 325–339 (2018).
Mueller, S. H. et al. Genetic predisposition in anti-LGI1 and anti-NMDA receptor encephalitis. Ann. Neurol. 83, 863–869 (2018).
Binks, S. et al. Distinct HLA associations of LGI1 and CASPR2-antibody diseases. Brain 141, 2263–2271 (2018).
Estrada, K. et al. A whole-genome sequence study identifies genetic risk factors for neuromyelitis optica. Nat. Commun. 9, 1929 (2018).
Yoshimura, S. et al. Distinct genetic and infectious profiles in Japanese neuromyelitis optica patients according to anti-aquaporin 4 antibody status. J. Neurol. Neurosurg. Psychiatry 84, 29–34 (2013).
Shu, Y. et al. HLA class II allele DRB1*16:02 is associated with anti-NMDAR encephalitis. J. Neurol. Neurosurg. Psychiatry 90, 652–658 (2019).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Additional information
Peer review information
Nature Reviews Immunology thanks A. McKeon, A. Vincent and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Neuromyelitis optica
-
(NMO). An autoimmune inflammatory disorder predominantly affecting the optic nerves and spinal cord, caused by autoantibodies to the water channel aquaporin 4 (AQP4).
- Neuropil
-
A dense unmyelinated fibre network of neuronal processes in the brain, containing synapses, axons and dendrites, and making up large parts of the brain’s grey matter.
- NMDAR encephalitis
-
The most common autoimmune encephalitis. Inflammation in the brain is caused by autoantibodies to the N-methyl-d-aspartate receptor (NMDAR) and can lead to psychosis, amnesia, epileptic seizures, abnormal movements and reduced levels of consciousness.
- Faciobrachial dystonic seizures
-
Characteristic abnormal involuntary movements associated with leucine-rich glioma-inactivated protein 1 (LGI1) autoantibodies, typically as frequent, brief stereotyped seizures predominantly affecting one arm and the ipsilateral face.
- Paraneoplastic neurological syndromes
-
Diverse neurological abnormalities in the context of cancer, caused by cytotoxic T cells (and to a lesser degree by antibodies) cross-reacting with antigens in the nervous system that are ectopically expressed in the tumour.
- Blood–brain barrier
-
The border between blood vessels and brain parenchyma, composed of a dense network of endothelial cells, pericytes and astrocytes. The intact blood–brain barrier prevents soluble molecules, such as (auto)antibodies, entering the brain.
- Post-stroke dementia
-
Largely unexplained progressive cognitive decline affecting 30% of stroke survivors and being an important health issue. A potential relation to anti-neuronal autoantibodies is under investigation.
- ‘Brain antibody-omics’
-
A term coined by the author that underlies the (yet to be proven) hypothesis that autoantibodies are present also in the healthy brain, and that their composition and fluctuations can contribute to several brain diseases including dementia.
- Conformational epitopes
-
Structures composed of different parts of a protein, in contrast to linear epitopes composed of continuous amino acids in a line. Many pathogenic anti-neuronal autoantibodies bind only conformational epitopes.
- Therapeutic apheresis
-
Extracorporeal elimination of antibodies in autoimmune diseases, either with plasma exchange (blood plasma is removed and replaced) or immunoadsorption (antibodies removed with an adsorber column and blood reinfused).
Rights and permissions
About this article
Cite this article
Prüss, H. Autoantibodies in neurological disease. Nat Rev Immunol 21, 798–813 (2021). https://doi.org/10.1038/s41577-021-00543-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41577-021-00543-w
This article is cited by
-
Serum autoantibodies against α7-nicotinic receptors in subgroups of patients with bipolar disorder or schizophrenia: clinical features and link with peripheral inflammation
Translational Psychiatry (2024)
-
Cognitive deficits associated with novel intrathecal anti-nuclear antibodies
Molecular Psychiatry (2024)
-
Unique association of anti-GABAA receptor encephalitis and myasthenia gravis in a patient with type A thymoma
Neurological Sciences (2024)
-
Diagnosis and management of autoimmune diseases in the ICU
Intensive Care Medicine (2024)
-
Distinct features of B cell receptors in neuromyelitis optica spectrum disorder among CNS inflammatory demyelinating diseases
Journal of Neuroinflammation (2023)