Abstract
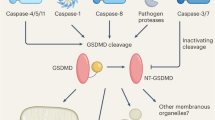
The gasdermins are a family of recently identified pore-forming effector proteins that cause membrane permeabilization and pyroptosis, a lytic pro-inflammatory type of cell death. Gasdermins contain a cytotoxic N-terminal domain and a C-terminal repressor domain connected by a flexible linker. Proteolytic cleavage between these two domains releases the intramolecular inhibition on the cytotoxic domain, allowing it to insert into cell membranes and form large oligomeric pores, which disrupts ion homeostasis and induces cell death. Gasdermin-induced pyroptosis plays a prominent role in many hereditary diseases and (auto)inflammatory disorders as well as in cancer. In this Review, we discuss recent developments in gasdermin research with a focus on mechanisms that control gasdermin activation, pore formation and functional consequences of gasdermin-induced membrane permeabilization.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Sato, H. et al. A new mutation Rim3 resembling Re den is mapped close to retinoic acid receptor α (Rara) gene on mouse chromosome 11. Mamm. Genome 9, 20–25 (1998).
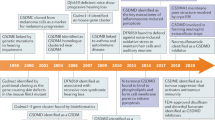
Saeki, N., Kuwahara, Y., Sasaki, H., Satoh, H. & Shiroishi, T. Gasdermin (Gsdm) localizing to mouse Chromosome 11 is predominantly expressed in upper gastrointestinal tract but significantly suppressed in human gastric cancer cells. Mamm. Genome 11, 718–724 (2000). This study names the first gasdermin gene and describes its expression in the mouse.
Van Laer, L. et al. Nonsyndromic hearing impairment is associated with a mutation in DFNA5. Nat. Genet. 20, 194–197 (1998).
Tanaka, S., Mizushina, Y., Kato, Y., Tamura, M. & Shiroishi, T. Functional conservation of Gsdma cluster genes specifically duplicated in the mouse genome. G3 3, 1843–1850 (2013).
Op de Beeck, K. et al. The DFNA5 gene, responsible for hearing loss and involved in cancer, encodes a novel apoptosis-inducing protein. Eur. J. Hum. Genet. 19, 965–973 (2011).
Van Rossom, S. et al. The splicing mutant of the human tumor suppressor protein DFNA5 induces programmed cell death when expressed in the yeast Saccharomyces cerevisiae. Front. Oncol. 2, 77 (2012).
Kayagaki, N. et al. Caspase-11 cleaves gasdermin D for non-canonical inflammasome signaling. Nature 526, 666–671 (2015).
Shi, J. et al. Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature 526, 660–665 (2015). Together with Kayagaki et al. (2015), this paper reveals that GSDMD is cleaved and activated by caspase 1 and caspase 4/5/11 to induce pyroptosis. This paper further shows that other gasdermin family members also bear the pyroptosis-inducing activity in their conserved N-terminal domain.
He, W. T. et al. Gasdermin D is an executor of pyroptosis and required for interleukin-1β secretion. Cell Res. 25, 1285–1298 (2015).
Friedlander, A. M. Macrophages are sensitive to anthrax lethal toxin through an acid-dependent process. J. Biol. Chem. 261, 7123–7126 (1986).
Zychlinsky, A., Prevost, M. C. & Sansonetti, P. J. Shigella flexneri induces apoptosis in infected macrophages. Nature 358, 167–169 (1992).
Hilbi, H., Chen, Y., Thirumalai, K. & Zychlinsky, A. The interleukin 1β-converting enzyme, caspase 1, is activated during Shigella flexneri-induced apoptosis in human monocyte-derived macrophages. Infect. Immun. 65, 5165–5170 (1997).
Cookson, B. T. & Brennan, M. A. Pro-inflammatory programmed cell death. Trends Microbiol. 9, 113–114 (2001).
Broz, P. & Dixit, V. M. Inflammasomes: mechanism of assembly, regulation and signalling. Nat. Rev. Immunol. 16, 407–420 (2016).
Broz, P. Immunology: caspase target drives pyroptosis. Nature 526, 642–643 (2015).
Ding, J. et al. Pore-forming activity and structural autoinhibition of the gasdermin family. Nature 535, 111–116 (2016). This paper reports the first structure of a gasdermin family (that is, GSDMA3) and establishes gasdermin as a new family of pore-forming proteins.
Panganiban, R. A. et al. A functional splice variant associated with decreased asthma risk abolishes the ability of gasdermin B to induce epithelial cell pyroptosis. J. Allergy Clin. Immunol. 142, 1469–1478.e2 (2018).
Rogers, C. et al. Cleavage of DFNA5 by caspase-3 during apoptosis mediates progression to secondary necrotic/pyroptotic cell death. Nat. Commun. 8, 14128 (2017).
Wang, Y. et al. Chemotherapy drugs induce pyroptosis through caspase-3 cleavage of a gasdermin. Nature 547, 99–103 (2017). Together with Rogers et al. (2017), this paper shows that GSDME (DFNA5) is cleaved and activated by caspase 3, which causes pyroptosis directly (Wang et al.) or induces secondary necrosis after apoptosis (Rogers et al.). This paper further shows that GSDME-mediated pyroptosis contributes to the toxicity of chemotherapy drugs given that it is silenced in most cancers but expressed in normal cells.
Lee, B. L. et al. ASC- and caspase-8-dependent apoptotic pathway diverges from the NLRC4 inflammasome in macrophages. Sci. Rep. 8, 3788 (2018).
Chen, K. W. et al. Extrinsic and intrinsic apoptosis activate pannexin-1 to drive NLRP3 inflammasome assembly. EMBO J. 38, e101638 (2019).
Orning, P. et al. Pathogen blockade of TAK1 triggers caspase-8-dependent cleavage of gasdermin D and cell death. Science 362, 1064–1069 (2018).
Sarhan, J. et al. Caspase-8 induces cleavage of gasdermin D to elicit pyroptosis during Yersinia infection. Proc. Natl Acad. Sci. USA 115, E10888–E10897 (2018). Together with Chen et al. (2019) and Orning et al. (2018), this paper reports a new pathway in which caspase 8 controls GSDMD cleavage and activation upon treatment with triggers of extrinsic apoptosis.
Taabazuing, C. Y., Okondo, M. C. & Bachovchin, D. A. Pyroptosis and apoptosis pathways engage in bidirectional crosstalk in monocytes and macrophages. Cell Chem. Biol. 24, 507–514.e4 (2017).
Sollberger, G. et al. Gasdermin D plays a vital role in the generation of neutrophil extracellular traps. Sci. Immunol. 3, pii: eaar6689 (2018).
Kambara, H. et al. Gasdermin D exerts anti-inflammatory effects by promoting neutrophil death. Cell Rep. 22, 2924–2936 (2018).
Chen, K. W. et al. Noncanonical inflammasome signaling elicits gasdermin D-dependent neutrophil extracellular traps. Sci. Immunol. 3, eaar6676 (2018).
Aglietti, R. A. & Dueber, E. C. Recent insights into the molecular mechanisms underlying pyroptosis and gasdermin family functions. Trends Immunol. 38, 261–271 (2017).
Jorgensen, I., Rayamajhi, M. & Miao, E. A. Programmed cell death as a defence against infection. Nat. Rev. Immunol. 17, 151–164 (2017).
Shi, J., Gao, W. & Shao, F. Pyroptosis: gasdermin-mediated programmed necrotic cell death. Trends Biochem. Sci. 42, 245–254 (2017).
Aglietti, R. A. et al. GsdmD p30 elicited by caspase-11 during pyroptosis forms pores in membranes. Proc. Natl Acad. Sci. USA 113, 7858–7863 (2016).
Liu, X. et al. Inflammasome-activated gasdermin D causes pyroptosis by forming membrane pores. Nature 535, 153–158 (2016).
Sborgi, L. et al. GSDMD membrane pore formation constitutes the mechanism of pyroptotic cell death. EMBO J. 35, 1766–1778 (2016). Together with Ding et al. (2016), Aglietti et al. (2016) and Liu et al. (2016), this paper reports that the GSDMD N-terminal domain targets cellular membranes and forms large pores.
Lin, P. H., Lin, H. Y., Kuo, C. C. & Yang, L. T. N-terminal functional domain of Gasdermin A3 regulates mitochondrial homeostasis via mitochondrial targeting. J. Biomed. Sci. 22, 44 (2015).
Shi, P. et al. Loss of conserved Gsdma3 self-regulation causes autophagy and cell death. Biochem. J. 468, 325–336 (2015).
Mulvihill, E. et al. Mechanism of membrane pore formation by human gasdermin-D. EMBO J. 37, e98321 (2018).
Chao, K. L., Kulakova, L. & Herzberg, O. Gene polymorphism linked to increased asthma and IBD risk alters gasdermin-B structure, a sulfatide and phosphoinositide binding protein. Proc. Natl Acad. Sci. USA 114, E1128–E1137 (2017).
Ruan, J., Xia, S., Liu, X., Lieberman, J. & Wu, H. Cryo-EM structure of the gasdermin A3 membrane pore. Nature 557, 62–67 (2018). This study reports the first cryo-electron microscopy structure of the gasdermin A3 (GSDMA3) pore and the conformational changes that the GSDMA3 N-terminal fragment undergoes upon binding to membrane phospholipids.
Evavold, C. L. et al. The pore-forming protein gasdermin D regulates interleukin-1 secretion from living macrophages. Immunity 48, 35–44.e6 (2018).
Heilig, R. et al. The Gasdermin-D pore acts as a conduit for IL-1β secretion in mice. Eur. J. Immunol. 48, 584–592 (2018).
Reboul, C. F., Whisstock, J. C. & Dunstone, M. A. Giant MACPF/CDC pore forming toxins: a class of their own. Biochim. Biophys. Acta 1858, 475–486 (2016).
Tilley, S. J., Orlova, E. V., Gilbert, R. J., Andrew, P. W. & Saibil, H. R. Structural basis of pore formation by the bacterial toxin pneumolysin. Cell 121, 247–256 (2005).
van Pee, K. et al. CryoEM structures of membrane pore and prepore complex reveal cytolytic mechanism of Pneumolysin. eLife 6, e23644 (2017).
Wolf, A. J. et al. Hexokinase is an innate immune receptor for the detection of bacterial peptidoglycan. Cell 166, 624–636 (2016).
Zanoni, I. et al. An endogenous caspase-11 ligand elicits interleukin-1 release from living dendritic cells. Science 352, 1232–1236 (2016).
Mangan, D. F., Welch, G. R. & Wahl, S. M. Lipopolysaccharide, tumor necrosis factor-α, and IL-1β prevent programmed cell death (apoptosis) in human peripheral blood monocytes. J. Immunol. 146, 1541–1546 (1991).
Perera, L. P. & Waldmann, T. A. Activation of human monocytes induces differential resistance to apoptosis with rapid down regulation of caspase-8/FLICE. Proc. Natl Acad. Sci. USA 95, 14308–14313 (1998).
Vigano, E. et al. Human caspase-4 and caspase-5 regulate the one-step non-canonical inflammasome activation in monocytes. Nat. Commun. 6, 8761 (2015).
Gaidt, M. M. et al. Human monocytes engage an alternative inflammasome pathway. Immununity 44, 833–846 (2016).
Chen, K. W. et al. The neutrophil NLRC4 inflammasome selectively promotes IL-1β maturation without pyroptosis during acute Salmonella challenge. Cell Rep. 8, 570–582 (2014).
de Torre-Minguela, C., Barbera-Cremades, M., Gomez, A. I., Martin-Sanchez, F. & Pelegrin, P. Macrophage activation and polarization modify P2X7 receptor secretome influencing the inflammatory process. Sci. Rep. 6, 22586 (2016).
Kayagaki, N. et al. Non-canonical inflammasome activation targets caspase-11. Nature 479, 117–121 (2011).
Ruhl, S. & Broz, P. Caspase-11 activates a canonical NLRP3 inflammasome by promoting K+ efflux. Eur. J. Immunol. 45, 2927–2936 (2015).
Cunha, L. D. et al. AIM2 engages active but unprocessed caspase-1 to induce noncanonical activation of the NLRP3 inflammasome. Cell Rep. 20, 794–805 (2017).
Banerjee, I. et al. Gasdermin D restrains type I interferon response to cytosolic DNA by disrupting ionic homeostasis. Immunity 49, 413–426.e5 (2018). This study shows that GSDMD pores can inhibit cyclic GMP–AMP synthase-dependent interferon production by causing potassium efflux even before inducing cell death.
Kayagaki, N. et al. IRF2 transcriptionally induces GSDMD expression for pyroptosis. Sci. Signal. 12, eaax4917 (2019).
Cooper, S. T. & McNeil, P. L. Membrane repair: mechanisms and pathophysiology. Physiol. Rev. 95, 1205–1240 (2015).
Ruhl, S. et al. ESCRT-dependent membrane repair negatively regulates pyroptosis downstream of GSDMD activation. Science 362, 956–960 (2018). This article shows that calcium influx through GSDMD pores can activate the ESCRT machinery to initiate repair of the damaged membranes.
Schoenauer, R. et al. P2X7 receptors mediate resistance to toxin-induced cell lysis. Biochim. Biophys. Acta 1843, 915–922 (2014).
Rubartelli, A., Cozzolino, F., Talio, M. & Sitia, R. A novel secretory pathway for interleukin-1β, a protein lacking a signal sequence. EMBO J. 9, 1503–1510 (1990).
Andrei, C. et al. The secretory route of the leaderless protein interleukin 1β involves exocytosis of endolysosome-related vesicles. Mol. Biol. Cell 10, 1463–1475 (1999).
MacKenzie, A. et al. Rapid secretion of interleukin-1β by microvesicle shedding. Immunity 15, 825–835 (2001).
Mitra, S. & Sarkar, A. Microparticulate P2X7 and GSDM-D mediated regulation of functional IL-1β release. Purinergic Signal. 15, 119–123 (2019).
Galluzzi, L. et al. Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 25, 486–541 (2018).
Fink, S. L. & Cookson, B. T. Apoptosis, pyroptosis, and necrosis: mechanistic description of dead and dying eukaryotic cells. Infect. Immun. 73, 1907–1916 (2005).
Rogers, C. et al. Gasdermin pores permeabilize mitochondria to augment caspase-3 activation during apoptosis and inflammasome activation. Nat. Commun. 10, 1689 (2019).
Platnich, J. M. et al. Shiga toxin/lipopolysaccharide activates caspase-4 and gasdermin D to trigger mitochondrial reactive oxygen species upstream of the NLRP3 inflammasome. Cell Rep. 25, 1525–1536.e7 (2018).
Baroja-Mazo, A. et al. The NLRP3 inflammasome is released as a particulate danger signal that amplifies the inflammatory response. Nat. Immunol. 15, 738–748 (2014).
Rowczenio, D. M. et al. Late-onset cryopyrin-associated periodic syndromes caused by somatic NLRP3 mosaicism-UK single center experience. Front. Immunol. 8, 1410 (2017).
Mensa-Vilaro, A. et al. Brief report: late-onset cryopyrin-associated periodic syndrome due to myeloid-restricted somatic NLRP3 mosaicism. Arthritis Rheumatol. 68, 3035–3041 (2016).
Xiao, J. et al. Gasdermin D mediates the pathogenesis of neonatal-onset multisystem inflammatory disease in mice. PLOS Biol. 16, e3000047 (2018).
Green, D. R., Oguin, T. H. & Martinez, J. The clearance of dying cells: table for two. Cell Death Differ. 23, 915–926 (2016).
Keller, M., Ruegg, A., Werner, S. & Beer, H. D. Active caspase-1 is a regulator of unconventional protein secretion. Cell 132, 818–831 (2008).
Saeki, N. et al. GASDERMIN, suppressed frequently in gastric cancer, is a target of LMO1 in TGF-β-dependent apoptotic signalling. Oncogene 26, 6488–6498 (2007).
Runkel, F. et al. The dominant alopecia phenotypes Bareskin, Rex-denuded, and Reduced Coat 2 are caused by mutations in gasdermin 3. Genomics 84, 824–835 (2004).
Kumar, S. et al. Gsdma3(I359N) is a novel ENU-induced mutant mouse line for studying the function of Gasdermin A3 in the hair follicle and epidermis. J. Dermatol. Sci. 67, 190–192 (2012).
Zhou, Y. et al. Gsdma3 mutation causes bulge stem cell depletion and alopecia mediated by skin inflammation. Am. J. Pathol. 180, 763–774 (2012).
Tanaka, S. et al. A new Gsdma3 mutation affecting anagen phase of first hair cycle. Biochem. Biophys. Res. Commun. 359, 902–907 (2007).
Ruge, F. et al. Delineating immune-mediated mechanisms underlying hair follicle destruction in the mouse mutant defolliculated. J. Invest. Dermatol. 131, 572–579 (2011).
Carl-McGrath, S., Schneider-Stock, R., Ebert, M. & Rocken, C. Differential expression and localisation of gasdermin-like (GSDML), a novel member of the cancer-associated GSDMDC protein family, in neoplastic and non-neoplastic gastric, hepatic, and colon tissues. Pathology 40, 13–24 (2008).
Sun, Q., Yang, J., Xing, G., Zhang, L. & He, F. Expression of gsdml associates with tumor progression in uterine cervix cancer. Transl Oncol. 1, 73–83 (2008).
Hergueta-Redondo, M. et al. Gasdermin-B promotes invasion and metastasis in breast cancer cells. PLOS ONE 9, e90099 (2014).
Hergueta-Redondo, M. et al. Gasdermin B expression predicts poor clinical outcome in HER2-positive breast cancer. Oncotarget 7, 56295–56308 (2016).
Verlaan, D. J. et al. Allele-specific chromatin remodeling in the ZPBP2/GSDMB/ORMDL3 locus associated with the risk of asthma and autoimmune disease. Am. J. Hum. Genet. 85, 377–393 (2009).
Wu, H. et al. Genetic variation in ORM1-like 3 (ORMDL3) and gasdermin-like (GSDML) and childhood asthma. Allergy 64, 629–635 (2009).
Yu, J. et al. Polymorphisms in GSDMA and GSDMB are associated with asthma susceptibility, atopy and BHR. Pediatr. Pulmonol. 46, 701–708 (2011).
Soderman, J., Berglind, L. & Almer, S. Gene expression-genotype analysis implicates GSDMA, GSDMB, and LRRC3C as contributors to inflammatory bowel disease susceptibility. Biomed. Res. Int. 2015, 834805 (2015).
Zhao, C. N. et al. The association of GSDMB and ORMDL3 gene polymorphisms with asthma: a meta-analysis. Allergy Asthma Immunol. Res. 7, 175–185 (2015).
Das, S., Miller, M. & Broide, D. H. Chromosome 17q21 genes ORMDL3 and GSDMB in asthma and immune diseases. Adv. Immunol. 135, 1–52 (2017).
Watabe, K. et al. Structure, expression and chromosome mapping of MLZE, a novel gene which is preferentially expressed in metastatic melanoma cells. Jpn J. Cancer Res. 92, 140–151 (2001).
Saeki, N. et al. Distinctive expression and function of four GSDM family genes (GSDMA-D) in normal and malignant upper gastrointestinal epithelium. Genes Chromosomes Cancer 48, 261–271 (2009).
Miguchi, M. et al. Gasdermin C is upregulated by inactivation of transforming growth factor β receptor type II in the presence of mutated APC, promoting colorectal cancer proliferation. PLOS ONE 11, e0166422 (2016).
Cerqueira, D. M. et al. Guanylate-binding protein 5 licenses caspase-11 for gasdermin-D mediated host resistance to brucella abortus infection. PLOS Pathog. 14, e1007519 (2018).
Schneider, K. S. et al. The inflammasome drives GSDMD-independent secondary pyroptosis and IL-1 release in the absence of caspase-1 protease activity. Cell Rep. 21, 3846–3859 (2017).
Zhu, Q., Zheng, M., Balakrishnan, A., Karki, R. & Kanneganti, T. D. Gasdermin D promotes AIM2 inflammasome activation and is required for host protection against Francisella novicida. J. Immunol. 201, 3662–3668 (2018).
Monteleone, M. et al. Interleukin-1β maturation triggers its relocation to the plasma membrane for gasdermin-D-dependent and -independent secretion. Cell Rep. 24, 1425–1433 (2018).
Kanneganti, A. et al. GSDMD is critical for autoinflammatory pathology in a mouse model of familial mediterranean fever. J. Exp. Med. 215, 1519–1529 (2018).
Delmaghani, S. et al. Mutations in the gene encoding pejvakin, a newly identified protein of the afferent auditory pathway, cause DFNB59 auditory neuropathy. Nat. Genet. 38, 770–778 (2006).
Collin, R. W. et al. Involvement of DFNB59 mutations in autosomal recessive nonsyndromic hearing impairment. Hum. Mutat. 28, 718–723 (2007).
Mujtaba, G., Bukhari, I., Fatima, A. & Naz, S. A p.C343S missense mutation in PJVK causes progressive hearing loss. Gene 504, 98–101 (2012).
Delmaghani, S. et al. Hypervulnerability to sound exposure through impaired adaptive proliferation of peroxisomes. Cell 163, 894–906 (2015).
Defourny, J. et al. Pejvakin-mediated pexophagy protects auditory hair cells against noise-induced damage. Proc. Natl Acad. Sci. USA 116, 8010–8017 (2019).
Fink, S. L. & Cookson, B. T. Caspase-1-dependent pore formation during pyroptosis leads to osmotic lysis of infected host macrophages. Cell. Microbiol. 8, 1812–1825 (2006).
Martin-Sanchez, F. et al. Inflammasome-dependent IL-1β release depends upon membrane permeabilisation. Cell Death Differ. 23, 1219–1231 (2016).
Tapia, V. S. et al. The three cytokines IL-1β, IL-18, and IL-1α share related but distinct secretory routes. J. Biol. Chem. 294, 8325–8335 (2019).
de Vasconcelos, N. M., Van Opdenbosch, N., Van Gorp, H., Parthoens, E. & Lamkanfi, M. Single-cell analysis of pyroptosis dynamics reveals conserved GSDMD-mediated subcellular events that precede plasma membrane rupture. Cell Death Differ. 26, 146–161 (2019).
Russo, H. M. et al. Active caspase-1 induces plasma membrane pores that precede pyroptotic lysis and are blocked by lanthanides. J. Immunol. 197, 1353–1367 (2016).
Rathkey, J. K. et al. Chemical disruption of the pyroptotic pore-forming protein gasdermin D inhibits inflammatory cell death and sepsis. Sci. Immunol. 3, eaat2738 (2018).
Rieckmann, J. C. et al. Social network architecture of human immune cells unveiled by quantitative proteomics. Nat. Immunol. 18, 583–593 (2017).
Lunny, D. P. et al. Mutations in gasdermin 3 cause aberrant differentiation of the hair follicle and sebaceous gland. J. Invest. Dermatol. 124, 615–621 (2005).
Tamura, M. et al. Members of a novel gene family, GSDM, are expressed exclusively in the epithelium of the skin and gastrointestinal tract in a highly tissue-specific manner. Genomics 89, 618–629 (2007).
Katoh, M. Evolutionary recombination hotspot around GSDML–GSDM locus is closely linked to the oncogenomic recombination hotspot around the PPP1R1B–ERBB2–GRB7 amplicon. Int. J. Oncol. 24, 757–763 (2004).
Fagerberg, L. et al. Analysis of the human tissue-specific expression by genome-wide integration of transcriptomics and antibody-based proteomics. Mol. Cell Proteom. 13, 397–406 (2014).
Katoh, M. Identification and characterization of human DFNA5L, mouse DFNA5l, and rat DFNA5l genes in silico. Int. J. Oncol. 25, 765–770 (2004).
Masumoto, J. et al. Caspy, a zebrafish caspase, activated by ASC oligomerization is required for pharyngeal arch development. J. Biol. Chem. 278, 4268–4276 (2003).
Vincent, W. J., Freisinger, C. M., Lam, P. Y., Huttenlocher, A. & Sauer, J. D. Macrophages mediate flagellin induced inflammasome activation and host defense in zebrafish. Cell. Microbiol. 18, 591–604 (2016).
Ogryzko, N. V. et al. Zebrafish tissue injury causes upregulation of interleukin-1 and caspase-dependent amplification of the inflammatory response. Dis. Model. Mech. 7, 259–264 (2014).
Bird, S., Wang, T., Zou, J., Cunningham, C. & Secombes, C. J. The first cytokine sequence within cartilaginous fish: IL-1β in the small spotted catshark (Scyliorhinus canicula). J. Immunol. 168, 3329–3340 (2002).
Tyrkalska, S. D. et al. Neutrophils mediate Salmonella Typhimurium clearance through the GBP4 inflammasome-dependent production of prostaglandins. Nat. Commun. 7, 12077 (2016).
Yang, D. et al. Sensing of cytosolic LPS through caspy2 pyrin domain mediates noncanonical inflammasome activation in zebrafish. Nat. Commun. 9, 3052 (2018).
Busch-Nentwich, E., Sollner, C., Roehl, H. & Nicolson, T. The deafness gene dfna5 is crucial for ugdh expression and HA production in the developing ear in zebrafish. Development 131, 943–951 (2004).
Liu, W., Kinnefors, A., Bostrom, M., Edin, F. & Rask-Andersen, H. Distribution of pejvakin in human spiral ganglion: an immunohistochemical study. Cochlear Implant. Int. 14, 225–231 (2013).
Martinon, F., Burns, K. & Tschopp, J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-β. Mol. Cell. 10, 417–426 (2002).
Kersey, P. J. et al. Ensembl Genomes 2018: an integrated omics infrastructure for non-vertebrate species. Nucleic Acids Res. 46, D802–D808 (2018).
Zerbino, D. R. et al. Ensembl 2018. Nucleic Acids Res. 46, D754–D761 (2018).
Carithers, L. J. & Moore, H. M. The Genotype-Tissue Expression (GTEx) project. Biopreserv. Biobank 13, 307–308 (2015).
Schmiedel, B. J. et al. Impact of genetic polymorphisms on human immune cell gene expression. Cell 175, 1701–1715.e16 (2018).
Acknowledgements
P.B. was supported by grants from the Swiss National Science Foundation (175576) and the European Research Council (ERC-2017-CoG 770988, InflamCellDeath). P.P. was supported by grants from Sociedad Española de Reumatología Pediátrica, Ministerio de Economia, Industria y Competitividad–Fondo Europeo de Desarrollo Regional (SAF2017–88276-R), Fundación Séneca (20859/PI/18) and the European Research Council (ERC-2013-CoG 614578). F.S. was supported by grants from China NSFC (81788101), MOST of China (2017YFA0505900 and 2016YFA0501500) and the Chinese Academy of Sciences (XDB08020202).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- Inflammasome
-
A multi-protein signalling complex assembled in the cytosol upon detection of host-derived or pathogen-derived danger signals that promotes cytokine release, pyroptotic cell death and inflammation.
- Caspases
-
A family of cysteine-dependent aspartate proteases that play a central role in cell death and inflammation by processing their substrates at specific aspartate residues.
- Necrosis
-
A type of cell death in which the integrity of the plasma membrane is lost and the constituents of the cell are released.
- NLRP3
-
(Nucleotide-binding oligomerization domain, leucine-rich repeat and pyrin domain-containing 3). A cytoplasmic sensor that assembles an inflammasome. NLRP3 is a broad-spectrum sensor of pathogen-associated and damage-associated molecular patterns.
- Pannexin-1 channels
-
Plasma membrane channels permeable to ions and small metabolites such as ATP.
- NETosis
-
A unique form of neutrophil cell death characterized by the release of DNA, histone and granular contents into the extracellular space.
- Liposomes
-
Artificial spherical vesicles with at least one lipid bilayer composed of phospholipids.
- Mitophagy
-
The selective removal of mitochondria by autophagy under conditions of nutrient starvation or mitochondrial stress.
- ASC foci
-
(Apoptosis-associated speck-like protein containing a caspase recruitment domain foci). These multimeric protein aggregates result from helical fibril formation that is initiated by the homo-oligomerization of inflammasome proteins.
- Familial Mediterranean fever
-
A genetically autosomal recessive inherited autoinflammatory disease arising from mutations in the MEFV gene, which encodes the protein pyrin, and is characterized by self-limited attacks of fever and serositis.
- Pyrin
-
An inflammasome-forming cytoplasmic sensor of bacteria-induced RHO modifications and disruption of actin cytoskeleton dynamics. Pyrin is encoded by the MEFV gene.
- Peroxisomes
-
Organelles of eukaryotic cells involved in the catabolism of different fatty acids and the reduction of reactive oxygen species.
- NLRP1
-
(Nucleotide-binding oligomerization domain, leucine-rich repeat and pyrin domain-containing 1). A cytoplasmic sensor that assembles an inflammasome. NLRP1 is activated by the N-end rule proteosomal degradation pathway.
- NLRC4
-
(Nucleotide-binding oligomerization domain, leucine-rich repeat and caspase recruitment domain-containing 4). An inflammasome scaffold protein that oligomerizes upon binding to the inflammasome sensor — the NLR family apoptosis inhibitory protein (NAIP) — and forms a platform to activate caspase 1.
- Toll-like receptor
-
A class of pattern recognition receptor located at endosomal and plasma membranes that senses pathogen-associated and damage-associated molecular patterns and initiates signalling pathways to induce inflammation.
Rights and permissions
About this article
Cite this article
Broz, P., Pelegrín, P. & Shao, F. The gasdermins, a protein family executing cell death and inflammation. Nat Rev Immunol 20, 143–157 (2020). https://doi.org/10.1038/s41577-019-0228-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41577-019-0228-2
This article is cited by
-
Elucidating the role of Pyroptosis in papillary thyroid cancer: prognostic, immunological, and therapeutic perspectives
Cancer Cell International (2024)
-
Smart responsive Fe/Mn nanovaccine triggers liver cancer immunotherapy via pyroptosis and pyroptosis-boosted cGAS-STING activation
Journal of Nanobiotechnology (2024)
-
Transcriptome-wide association studies associated with Crohn’s disease: challenges and perspectives
Cell & Bioscience (2024)
-
(Pro)renin receptor mediates tubular epithelial cell pyroptosis in diabetic kidney disease via DPP4-JNK pathway
Journal of Translational Medicine (2024)
-
Multiomics characterization of pyroptosis in the tumor microenvironment and therapeutic relevance in metastatic melanoma
BMC Medicine (2024)