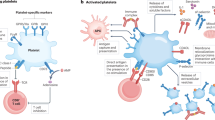
Abstract
Platelets are small anucleate cellular fragments that are released by megakaryocytes and safeguard vascular integrity through a process termed ‘haemostasis’. However, platelets have important roles beyond haemostasis as they contribute to the initiation and coordination of intravascular immune responses. They continuously monitor blood vessel integrity and tightly coordinate vascular trafficking and functions of multiple cell types. In this way platelets act as ‘patrolling officers of the vascular highway’ that help to establish effective immune responses to infections and cancer. Here we discuss the distinct biological features of platelets that allow them to shape immune responses to pathogens and tumour cells, highlighting the parallels between these responses.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Zhang, X., Zhuchenko, O., Kuspa, A. & Soldati, T. Social amoebae trap and kill bacteria by casting DNA nets. Nat. Commun. 7, 10938 (2016).
Chen, G., Zhuchenko, O. & Kuspa, A. Immune-like phagocyte activity in the social amoeba. Science 317, 678–681 (2007).
Monahan-earley, R., Dvorak, A. M. & Aird, W. C. Evolutionary origins of the blood vascular system and endothelium. J. Thromb. Haemost. 11, 46–66 (2013).
Cines, B. D. B. et al. Endothelial cells in physiology and in the pathophysiology of vascular disorders by. Blood 91, 3527–3561 (1998).
Levin, J. The evolution of mammalian platelets. in Platelets 3rd edn (ed. Michelson, A. D.) 3–25 (Academic Press, 2013).
Semple, J. W., Italiano, J. E. & Freedman, J. Platelets and the immune continuum. Nat. Rev. Immunol. 11, 264–274 (2011). In this review, the authors comprehensively outline some of the key properties of platelets that enable them to contribute to immunity.
Kapur, R., Zufferey, A., Boilard, E. & Semple, J. W. Nouvelle cuisine: platelets served with inflammation. J. Immunol. 194, 5579–5587 (2015).
Nurden, A. T. The biology of the platelet with special reference to inflammation, wound healing and immunity. Front. Biosci. Landmark 23, 726–751 (2018).
Haemmerle, M., Stone, R. L., Menter, D. G., Afshar-Kharghan, V. & Sood, A. K. The platelet lifeline to cancer: challenges and opportunities. Cancer Cell 33, 965–983 (2018).
Hotchkiss, R. S. & Moldawer, L. L. Parallels between cancer and infectious disease. N. Engl. J. Med. 371, 380–383 (2014).
Jacqueline, C. et al. Infections and cancer: the ‘fifty shades of immunity’ hypothesis. BMC Cancer 17, 1–11 (2017).
Schreiber, R. D., Old, L. J. & Smyth, M. J. Cancer immunoediting: integrating suppression and promotion. Science 331, 1565–1570 (2011).
Karin, M., Lawrence, T. & Nizet, V. Innate immunity gone awry: linking microbial infections to chronic inflammation and cancer. Cell 124, 823–835 (2006).
Balkwill, F. & Mantovani, A. Inflammation and cancer: back to Virchow? Lancet 357, 539–545 (2001).
Dvorak, H. F. Tumours: wounds that do not heal–redux. Cancer Immunol. Res. 3, 1–11 (2015).
Haas, S. et al. Inflammation-induced emergency megakaryopoiesis driven by hematopoietic stem cell-like megakaryocyte progenitors. Cell Stem Cell 17, 422–434 (2015).
Lefrançais, E. et al. The lung is a site of platelet biogenesis and a reservoir for haematopoietic progenitors. Nature 544, 105–109 (2017). In this study, the authors provide evidence that the lungs are primary sites of terminal platelet production.
Sanjuan-Pla, A. et al. Platelet-biased stem cells reside at the apex of the haematopoietic stem-cell hierarchy. Nature 502, 232–236 (2013). In this study, the authors identify a molecularly and functionally distinct mouse HSC subset primed for platelet-specific gene expression with enhanced propensity for short-term and long-term reconstitution of platelets.
Shin, J. Y., Hu, W., Naramura, M. & Park, C. Y. High c-Kit expression identifies hematopoietic stem cells with impaired self-renewal and megakaryocytic bias. J. Exp. Med. 211, 217–231 (2014).
Grinenko, T. et al. Clonal expansion capacity defines two consecutive developmental stages of long-term hematopoietic stem cells. J. Exp. Med. 211, 209–215 (2014).
Nakamura-Ishizu, A. et al. Thrombopoietin metabolically primes hematopoietic stem cells to megakaryocyte-lineage differentiation. Cell Rep. 25, 1772–1785.e6 (2018).
Hill, R. J., Warren, M. K. & Levin, J. Stimulation of thrombopoiesis in mice by human recombinant interleukin 6. J. Clin. Invest. 85, 1242–1247 (1990).
Bailey, S. E. R., Ukoumunne, O. C., Shephard, E. & Hamilton, W. How useful is thrombocytosis in predicting an underlying cancer in primary care? A systematic review. Fam. Pract. 34, 4–10 (2017).
Varki, A. Review in translational hematology Trousseau’ s syndrome: multiple definitions and multiple mechanisms. Blood 110, 1723–1729 (2010).
McAllister, S. S. & Weinberg, R. A. The tumour-induced systemic environment as a critical regulator of cancer progression and metastasis. Nat. Cell Biol. 16, 717–727 (2014).
Stone, R. L. et al. Paraneoplastic thrombocytosis in ovarian cancer. N. Engl. J. Med. 366, 610–618 (2012).
Nishimura, S. et al. IL-1α induces thrombopoiesis through megakaryocyte rupture in response to acute platelet needs. J. Cell Biol. 209, 453–466 (2015).
Machlus, K. R. et al. CCL5 derived from platelets increases megakaryocyte proplatelet formation. Blood 127, 921–926 (2016).
Massberg, S. et al. Immunosurveillance by hematopoietic progenitor cells trafficking through blood, lymph, and peripheral tissues. Cell 131, 994–1008 (2007).
Zhang, L. et al. A novel role of sphingosine 1-phosphate receptor S1pr1 in mouse thrombopoiesis. J. Exp. Med. 209, 2165–2181 (2012).
Chang, H. Y. et al. Quantifying platelet margination in diabetic blood flow. Biophys. J. 115, 1371–1382 (2018).
Wong, C. H. Y., Jenne, C. N., Petri, B., Chrobok, N. L. & Kubes, P. Nucleation of platelets with blood-borne pathogens on Kupffer cells precedes other innate immunity and contributes to bacterial clearance. Nat. Immunol. 14, 785–792 (2013). This study identifies a novel surveillance mechanism by which platelets survey macrophages that rapidly converts to a critical host response to blood-borne bacteria.
Chen, J. & López, J. A. Interactions of platelets with subendothelium and endothelium. Microcirculation 12, 235–246 (2005).
Sreeramkumar, V. et al. Neutrophils scan for activated platelets to initiate inflammation. Science 346, 1234–1238 (2014). The authors demonstrate that neutrophils scan the inflamed vasculature for activated platelets and show that the neutrophils’ bipolarity allows the integration of signals from the endothelium and the circulation.
Weinbaum, S., Tarbell, J. M. & Damiano, E. R. The structure and function of the endothelial glycocalyx layer. Annu. Rev. Biomed. Eng. 9, 121–167 (2007).
Becker, B. F., Jacob, M., Leipert, S., Salmon, A. H. J. & Chappell, D. Degradation of the endothelial glycocalyx in clinical settings: searching for the sheddases. Br. J. Clin. Pharmacol. 80, 389–402 (2015).
Andonegui, G. et al. Mice that exclusively express TLR4 on endothelial cells can efficiently clear a lethal systemic gram-negative bacterial infection. J. Clin. Invest. 119, 1921–1930 (2009).
Zullo, J. A. et al. Exocytosis of endothelial lysosome-related organelles hair-triggers a patchy loss of glycocalyx at the onset of sepsis. Am. J. Pathol. 186, 248–258 (2016).
Kalagara, T. et al. The endothelial glycocalyx anchors von Willebrand factor fibers to the vascular endothelium. Blood Adv. 2, 2347–2357 (2018).
Pober, J. S. & Sessa, W. C. Evolving functions of endothelial cells in inflammation. Nat. Rev. Immunol. 7, 803–815 (2007).
Wagner, D. D. & Frenette, P. S. The vessel wall and its interactions. Blood 111, 5271–5281 (2008).
Atkinson, B. T. et al. Laser-induced endothelial cell activation supports fibrin formation. Blood 116, 4675–4683 (2010).
Henn, V. et al. CD40 ligand on activated platelets triggers an inflammatory reaction of endothelial cells. Nature 391, 591–594 (1998).
Denis, M. M. et al. Escaping the nuclear confines: signal-dependent pre-mRNA splicing in anucleate platelets. Cell 122, 379–391 (2005). In this study, the authors identify signal-dependent pre-mRNA splicing in anucleate platelets.
Lindemann, S. et al. Activated platelets mediate inflammatory signaling by regulated interleukin 1β synthesis. J. Cell Biol. 154, 485–490 (2001).
Hottz, E. D. et al. Platelets mediate increased endothelium permeability in dengue through NLRP3-inflammasome activation. Blood 122, 3405–3414 (2013).
Weis, S. M. & Cheresh, D. A. Tumour angiogenesis: molecular pathways and therapeutic targets. Nat. Med. 17, 1359–1370 (2011).
Kim, Y. J., Borsig, L., Varki, N. M. & Varki, A. P-selectin deficiency attenuates tumour growth and metastasis. Proc. Natl Acad. Sci. USA 95, 9325–9330 (1998).
Qi, C. et al. P-selectin-mediated platelet adhesion promotes tumour growth. Oncotarget 6, 6584–6596 (2015).
Brooks, P., Clark, R. & Cheresh, D. Requirement of vascular integrin alpha v beta 3 for angiogenesis. Science 264, 569–571 (1994).
Sipkins, D. A. et al. Detection of tumour angiogenesis in vivo by alphaVbeta3-targeted magnetic resonance imaging. Nat. Med. 4, 623–626 (1998).
Contrino, J., Hair, G., Kreutzer, D. L. & Rickles, F. R. In situ detection of tissue factor in vascular endothelial cells: correlation with the malignant phenotype of human breast disease. Nat. Med. 2, 209 (1996).
Weis, S. M. & Cheresh, D. A. Pathophysiological consequences of VEGF-induced vascular permeability. Nature 437, 497–504 (2005).
Kisucka, J. et al. Platelets and platelet adhesion support angiogenesis while preventing excessive hemorrhage. Proc. Natl Acad. Sci. USA 103, 855–860 (2006).
Ho-Tin-Noé, B., Goerge, T. & Wagner, D. D. Platelets: guardians of tumour vasculature. Cancer Res. 69, 5623–5626 (2009). The authors provide a comprehensive review of activated platelets as crucial regulators of tumour vascular homeostasis preventing tumour haemorrhage.
Italiano, J. E. et al. Angiogenesis is regulated by a novel mechanism: pro- and antiangiogenic proteins are organized into separate platelet α granules and differentially released. Blood 111, 1227–1233 (2008). The authors demonstrate that separate packaging of angiogenesis regulators into pharmacologically and morphologically distinct populations of α-granules in megakaryocytes and platelets provides a mechanism by which platelets can locally stimulate or inhibit angiogenesis.
Kuznetsov, H. S. et al. Identification of luminal breast cancers that establish a tumour-supportive macroenvironment defined by proangiogenic platelets and bone marrow-derived cells. Cancer Discov. 2, 1150–1165 (2012).
Klement, G. et al. Platelets actively sequester angiogenesis regulators. Blood 113, 2835–2842 (2009). This study identifies the ability of platelets to selectively take up angiogenesis regulators in cancer-bearing hosts.
Medzhitov, R. & Janeway Jr., C. A. Decoding the pattern of self and nonself by the innate immune system. Science 296, 298–300 (2002).
Takeuchi, O. & Akira, S. Pattern recognition receptors and inflammation. Cell 140, 805–820 (2010).
Aslam, R. et al. Platelet Toll-like receptor expression modulates lipopolysaccharide-induced thrombocytopenia and tumour necrosis factor-α production in vivo. Blood 107, 637–641 (2005).
Shiraki, R. et al. Expression of Toll-like receptors on human platelets. Thromb. Res. 113, 379–385 (2004).
Koupenova, M., Clancy, L., Corkrey, H. A. & Freedman, J. E. Circulating platelets as mediators of immunity, inflammation, and thrombosis. Circ. Res. 122, 337–351 (2018).
Yeaman, M. R. Platelets: at the nexus of antimicrobial defence. Nat. Rev. Microbiol. 12, 426–437 (2014). This review provides a comprehensive overview of the antimicrobial roles of platelets, which are mediated both directly and indirectly to integrate innate and adaptive immune responses to pathogens.
Mebius, M. M. et al. Interference with the host haemostatic system by schistosomes. PLOS Pathog. 9, 1–8 (2013).
McMorran, B. J. et al. New insights into the protective power of platelets in malaria infection. Commun. Integr. Biol. 6, e23653 1–4 (2013).
Assinger, A. Platelets and infection - an emerging role of platelets in viral infection. Front. Immunol. 5, 10–12 (2014).
Boilard, E. et al. Influenza virus H1N1 activates platelets through FcγRIIA signaling and thrombin generation. Blood 123, 2854–2863 (2014).
Zapata, J. C., Cox, D. & Salvato, M. S. The role of platelets in the pathogenesis of viral hemorrhagic fevers. PLOS Negl. Trop. Dis. 8, e2858 1–12 (2014).
Zahn, A., Jennings, N., Ouwehand, W. H. & Allain, J. P. Hepatitis C virus interacts with human platelet glycoprotein VI. J. Gen. Virol. 87, 2243–2251 (2006).
Gavrilovskaya, I. N., Gorbunova, E. E. & Mackow, E. R. Pathogenic hantaviruses direct the adherence of quiescent platelets to infected endothelial cells. J. Virol. 84, 4832–4839 (2010).
Koupenova, M. et al. Platelet-TLR7 mediates host survival and platelet count during viral infection in the absence of platelet-dependent thrombosis. Blood 124, 791–802 (2014).
Auerbach, D. J. et al. Identification of the platelet-derived chemokine CXCL4/PF-4 as a broad-spectrum HIV-1 inhibitor. Proc. Natl Acad. Sci. USA 109, 9569–9574 (2012).
Von Hundelshausen, P. & Weber, C. Platelets as immune cells: bridging inflammation and cardiovascular disease. Circ. Res. 100, 27–40 (2007).
Cocchi, F. et al. Identification the major HIV-suppressive factors produced by. Science 270, 1811–1815 (1995).
Kraemer, B. F. et al. Novel anti-bacterial activities of β-defensin 1 in human platelets: suppression of pathogen growth and signaling of neutrophil extracellular trap formation. PLOS Pathog. 7, e1002355 1–9 (2011).
Wilson, S. S., Wiens, M. E. & Smith, J. G. Antiviral mechanisms of human defensins. J. Mol. Biol. 425, 4965–4980 (2013).
Shannon, O. Platelet interaction with bacterial toxins and secreted products. Platelets 26, 302–308 (2015).
Waller, A. K. et al. Staphylococcus aureus lipoteichoic acid inhibits platelet activation and thrombus formation via the Paf receptor. J. Infect. Dis. 208, 2046–2057 (2013).
White, J. G. Platelets are covercytes, not phagocytes: uptake of bacteria involves channels of the open canalicular system. Platelets 16, 121–131 (2005).
Gaertner, F. et al. Migrating platelets are mechano-scavengers that collect and bundle bacteria. Cell 171, 1368–1382 (2017). This study shows that platelets use their ability to migrate to act as cellular scavengers, scanning the vascular surface for potential invaders and collecting deposited bacteria.
Ali, R. A., Wuescher, L. M., Dona, K. R. & Worth, R. G. Platelets mediate host defence against Staphylococcus aureus through direct bactericidal activity and by enhancing macrophage activities. J. Immunol. 198, 344–351 (2017).
Fitzgerald, J. R., Foster, T. J. & Cox, D. The interaction of bacterial pathogens with platelets. Nat. Rev. Microbiol. 4, 445–457 (2006).
Engelmann, B. & Massberg, S. Thrombosis as an intravascular effector of innate immunity. Nat. Rev. Immunol. 13, 34–45 (2013).
Shannon, O., Uekötter, A. & Flock, J.-I. Extracellular fibrinogen binding protein, Efb, from Staphylococcus aureus as an antiplatelet agent in vivo. Thromb. Haemost. 93, 927–931 (2005).
Veloso, T. R. et al. Prophylaxis of experimental endocarditis with antiplatelet and antithrombin agents: a role for long-term prevention of infective endocarditis in humans? J. Infect. Dis. 211, 72–79 (2015).
Sexton, T. R. et al. Ticagrelor reduces thromboinflammatory markers in patients with pneumonia. JACC Basic to Transl. Sci. 3, 435–449 (2018). In this study, the authors show that patients with pneumonia receiving ticagrelor required less supplemental oxygen and lung function test results trended towards improvement.
McDonald, B., Urrutia, R., Yipp, B. G., Jenne, C. N. & Kubes, P. Intravascular neutrophil extracellular traps capture bacteria from the bloodstream during sepsis. Cell Host Microbe 12, 324–333 (2012).
McDonald, B. et al. Platelets and neutrophil extracellular traps collaborate to promote intravascular coagulation during sepsis in mice. Blood 129, 1357–1367 (2017).
Clemetson, K. Functional expression of CCR1, CCR3, CCR4, and CXCR4 chemokine receptors on human platelets. Blood 96, 4046–4054 (2000).
Sauter, R. J. et al. Functional relevance of the anaphylatoxin receptor C3aR for platelet function and arterial thrombus formation marks an intersection point between innate immunity and thrombosis. Circulation 138, 1720–1735 (2018).
Subramaniam, S. et al. Distinct contributions of complement factors to platelet activation and fibrin formation in venous thrombus development. Blood 129, 2291–2302 (2017).
Sarris, M. & Sixt, M. Navigating in tissue mazes: chemoattractant interpretation in complex environments. Curr. Opin. Cell Biol. 36, 93–102 (2015).
Lowenhaupt, R. W., Miller, M. A. & Glueck, H. I. Platelet migration and chemotaxis demonstrated in vitro. Thromb. Res. 3, 477–487 (1973).
Kraemer, B. F. et al. PI3 kinase-dependent stimulation of platelet migration by stromal cell-derived factor 1 (SDF-1). J. Mol. Med. 88, 1277–1288 (2010).
Pitchford, S. C. et al. Allergen induces the migration of platelets to lung tissue in allergic asthma. Am. J. Respir. Crit. Care Med. 177, 604–612 (2008).
Feng, D., Nagy, J. A., Pyne, K., Dvorak, H. F. & Dvorak, A. M. Platelets exit venules by a transcellular pathway at sites of F–Met peptide–induced acute inflammation in guinea pigs. Int. Arch. Allergy Immunol. 116, 188–195 (1998).
Czapiga, M., Gao, J.-L., Kirk, A. & Lekstrom-Himes, J. Human platelets exhibit chemotaxis using functional N-formyl peptide receptors. Exp. Hematol. 33, 73–84 (2005).
Cho, M. S. et al. Role of ADP receptors on platelets in the growth of ovarian cancer. Blood 130, 1235–1242 (2017).
Cho, M. S. et al. Platelets increase the proliferation of ovarian cancer cells. Blood 120, 4869–4872 (2012).
Wong, S. Y. & Hynes, R. O. Lymphatic or hematogenous dissemination: how does a metastatic tumour cell decide? Cell Cycle 5, 812–817 (2006).
Brown, M. et al. Lymph node blood vessels provide exit routes for metastatic tumour cell dissemination in mice. Science 359, 1408–1411 (2018).
Dongre, A. & Weinberg, R. A. New insights into the mechanisms of epithelial–mesenchymal transition and implications for cancer. Nat. Rev. Mol. Cell Biol. 20, 69–84 (2019).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Karnoub, A. E. et al. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 449, 557–563 (2007).
Condeelis, J. & Pollard, J. W. Macrophages: obligate partners for tumour cell migration, invasion, and metastasis. Cell 124, 263–266 (2006).
Li, R. et al. Presence of intratumoural platelets is associated with tumour vessel structure and metastasis. BMC Cancer 14, 1–10 (2014).
Haemmerle, M. et al. FAK regulates platelet extravasation and tumour growth after antiangiogenic therapy withdrawal. J Clin Invest 126, 1885–1896 (2016).
Varon, D. & Shai, E. Role of platelet-derived microparticles in angiogenesis and tumour progression. Discov. Med. 8, 237–241 (2009).
Labelle, M., Begum, S. & Hynes, R. O. Direct signaling between platelets and cancer cells induces an epithelial-mesenchymal-like transition and promotes metastasis. Cancer Cell 20, 576–590 (2011).
Boucharaba, A. et al. Platelet-derived lysophosphatidic acid supports the progression of osteolytic bone metastases in breast cancer. J. Clin. Invest. 114, 1714–1725 (2004).
Patrignani, P. & Patrono, C. Aspirin and cancer. J. Am. Coll. Cardiol. 68, 967–976 (2016).
Michael, J. V. et al. Platelet microparticles infiltrating solid tumours transfer miRNAs that suppress tumour growth. Blood 130, 567–580 (2017). The authors show platelet-derived microparticles may shuttle platelet-derived microRNA to tumour cells in solid tumours, regulating tumour cell gene expression and modulating tumour progression.
Reymond, N., D’Água, B. B. & Ridley, A. J. Crossing the endothelial barrier during metastasis. Nat. Rev. Cancer 13, 858–870 (2013).
Thomas, G. M. et al. Tissue factor expressed by circulating cancer cell-derived microparticles drastically increases the incidence of deep vein thrombosis in mice. J. Thromb. Haemost. 13, 1310–1319 (2015). The authors demonstrate in mice that tissue factor expressed on tumour microparticles contributes to the increased incidence of cancer-associated venous thrombosis.
Hisada, Y. & Mackman, N. Cancer-associated pathways and biomarkers of venous thrombosis. Blood 130, 1499–1506 (2017).
Stark, K. et al. Distinct pathogenesis of pancreatic cancer microvesicle-associated venous thrombosis identifies new antithrombotic targets in vivo. Arterioscler. Thromb. Vasc. Biol. 38, 772–786 (2018).
Egan, K., Cooke, N. & Kenny, D. Living in shear: platelets protect cancer cells from shear induced damage. Clin. Exp. Metastasis 31, 697–704 (2014).
Nieswandt, B., Hafner, M., Echtenacher, B. & Männel, D. N. Lysis of tumour cells by natural killer cells in mice is impeded by platelets. Cancer Res. 59, 1295–1300 (1999).
Placke, T. et al. Platelet-derived MHC class I confers a pseudonormal phenotype to cancer cells that subverts the antitumour reactivity of natural killer immune cells. Cancer Res. 72, 440–448 (2012).
Kopp, H. G., Placke, T. & Salih, H. R. Platelet-derived transforming growth factor-beta down-regulates NKG2D thereby inhibiting natural killer cell antitumour reactivity. Cancer Res. 69, 7775–7783 (2009).
Xu, X. R., Yousef, G. M. & Ni, H. Cancer and platelet crosstalk: opportunities and challenges of aspirin and other antiplatelet agents. Blood 131, 1777–1789 (2018).
Hyslop, S. R. & Josefsson, E. C. Undercover agents: targeting tumours with modified platelets. Trends Cancer 3, 235–246 (2017).
Broadley, S. P. et al. Dual-track clearance of circulating bacteria balances rapid restoration of blood sterility with induction of adaptive immunity. Cell Host Microbe 20, 36–48 (2016).
Zeng, Z. et al. CRIg functions as a macrophage pattern recognition receptor to directly bind and capture blood-borne gram-positive bacteria. Cell Host Microbe 20, 99–106 (2016).
Massberg, S. et al. Reciprocal coupling of coagulation and innate immunity via neutrophil serine proteases. Nat. Med. 16, 887–896 (2010).
Clark, S. R. et al. Platelet TLR4 activates neutrophil extracellular traps to ensnare bacteria in septic blood. Nat. Med. 13, 463–469 (2007). In this study, the authors find that platelets support NET formation during sepsis.
Gaertner, F. & Massberg, S. Blood coagulation in immunothrombosis—At the frontline of intravascular immunity. Semin. Immunol. 28, 561–569 (2016).
Pamer, E. G. Immune responses to listeria monocytogenes. Nat. Rev. Immunol. 4, 812–823 (2004).
Verschoor, A. et al. A platelet-mediated system for shuttling blood-borne bacteria to CD8α+ dendritic cells depends on glycoprotein GPIb and complement C3. Nat. Immunol. 12, 1194–1201 (2011).
Zufferey, A. et al. Mature murine megakaryocytes present antigen-MHC class I molecules to T cells and transfer them to platelets. Blood Adv. 1, 1773–1785 (2017).
Chapman, L. M. et al. Platelets present antigen in the context of MHC class I. J. Immunol. 189, 916–923 (2012).
Kaplan, R. N. et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438, 820–827 (2005).
Hiratsuka, S., Watanabe, A., Aburatani, H. & Maru, Y. Tumour-mediated upregulation of chemoattractants and recruitment of myeloid cells predetermines lung metastasis. Nat. Cell Biol. 8, 1369–1375 (2006).
Qian, B.-Z. et al. CCL2 recruits inflammatory monocytes to facilitate breast-tumour metastasis. Nature 475, 222–225 (2011).
Chen, Q., Zhang, X. H.-F. & Massagué, J. Macrophage binding to receptor VCAM-1 transmits survival signals in breast cancer cells that invade the lungs. Cancer Cell 20, 538–549 (2011).
Best, M. G., Wesseling, P. & Wurdinger, T. Tumour-educated platelets as a noninvasive biomarker source for cancer detection and progression monitoring. Cancer Res. 78, 3407–3412 (2018).
Wang, R. et al. Electron cryotomography reveals ultrastructure alterations in platelets from patients with ovarian cancer. Proc. Natl. Acad. Sci. 112, 14266–14271 (2015).
Gay, L. J. & Felding-Habermann, B. Contribution of platelets to tumour metastasis. Nat. Rev. Cancer 11, 123–134 (2011). In this comprehensive review, the authors summarize the evidence indicating that the activation of platelets and the coagulation system have a crucial role in the metastatic progression of cancer.
Labelle, M. & Hynes, R. O. The initial hours of metastasis: the importance of cooperative host-tumour cell interactions during hematogenous dissemination. Cancer Discov. 2, 1091–1099 (2012).
Coupland, L. A., Chong, B. H. & Parish, C. R. Platelets and P-selectin control tumour cell metastasis in an organ-specific manner and independently of NK cells. Cancer Res. 72, 4662–4671 (2012).
de Bruijne-Admiraal, L. G., Modderman, P. W., Von dem Borne, A. E. & Sonnenberg, A. P-selectin mediates Ca2+-dependent adhesion of activated platelets to many different types of leukocytes: detection by flow cytometry. Blood 80, 134–142 (1992).
Neumann, F. J. et al. Induction of cytokine expression in leukocytes by binding of thrombin-stimulated platelets. Circulation 95, 2387–2394 (1997).
Martins, P. A. et al. Platelet binding to monocytes increases the adhesive properties of monocytes by up-regulating the expression and functionality of β1 and β2 integrins. J. Leukoc. Biol. 79, 499–507 (2006).
Pircher, J. et al. Cathelicidins prime platelets to mediate arterial thrombosis and tissue inflammation. Nat. Commun. 9, 1523 (2018).
Zuchtriegel, G. et al. Platelets guide leukocytes to their sites of extravasation. PLOS Biol. 14, e1002459 1–28 (2016).
Koenen, R. R. et al. Disrupting functional interactions between platelet chemokines inhibits atherosclerosis in hyperlipidemic mice. Nat. Med. 15, 97–103 (2009).
Grommes, J. et al. Disruption of platelet-derived chemokine heteromers prevents neutrophil extravasation in acute lung injury. Am. J. Respir. Crit. Care Med. 185, 628–636 (2012).
Vajen, T. et al. Blocking CCL5-CXCL4 heteromerization preserves heart function after myocardial infarction by attenuating leukocyte recruitment and NETosis. Sci. Rep. 8, 1–11 (2018).
Xiang, B. et al. Platelets protect from septic shock by inhibiting macrophage-dependent inflammation via the cyclooxygenase 1 signalling pathway. Nat. Commun. 4, 2657 (2013).
Lax, S. et al. Platelet CLEC-2 protects against lung injury via effects of its ligand podoplanin on inflammatory alveolar macrophages in the mouse. Am. J. Physiol. Lung Cell. Mol. Physiol. 313, L1016–L1029 ajplung.00023.2017 (2017).
Rayes, J. et al. The podoplanin-CLEC-2 axis inhibits inflammation in sepsis. Nat. Commun. 8, 2239 (2017).
Hitchcock, J. R. et al. Inflammation drives thrombosis after salmonella infection via CLEC-2 on platelets. J. Clin. Invest. 125, 4429–4446 (2015). The authors report that infection-driven thrombosis follows local inflammation, upregulation of podoplanin and platelet activation.
Coffelt, S. B., Wellenstein, M. D. & De Visser, K. E. Neutrophils in cancer: neutral no more. Nat. Rev. Cancer 16, 431–446 (2016).
Tran, D. Q. et al. GARP (LRRC32) is essential for the surface expression of latent TGF- on platelets and activated FOXP3+ regulatory T cells. Proc. Natl Acad. Sci. USA 106, 13445–13450 (2009).
Rachidi, S. et al. Platelets subvert T cell immunity against cancer via GARP-TGFβ axis. Sci. Immunol. 2, eaai7911 (2017).
Wang, C. et al. In situ activation of platelets with checkpoint inhibitors for post-surgical cancer immunotherapy. Nat. Biomed. Eng. 1, 0011 (2017).
Gros, A. et al. Single platelets seal neutrophil-induced vascular breaches via GPVI during immune-complex-mediated inflammation in mice. Blood 126, 1017–1026 (2015).
Ho-Tin-Noé, B., Boulaftali, Y. & Camerer, E. Platelets and vascular integrity: how platelets prevent bleeding in inflammation. Blood 131, 277–288 (2018).
Boulaftali, Y. et al. Platelet ITAM signaling is critical for vascular integrity in infammation. J. Clin. Invest. 123, 908–916 (2013).
Herzog, B. H. et al. Podoplanin maintains high endothelial venule integrity by interacting with platelet CLEC-2. Nature 502, 105 (2013).
Iannacone, M. et al. Platelets prevent IFN-alpha/beta-induced lethal hemorrhage promoting CTL-dependent clearance of lymphocytic choriomeningitis virus. Proc. Natl. Acad. Sci. 105, 629–634 (2008).
Kleinschnitz, C. et al. Targeting platelets in acute experimental stroke: impact of glycoprotein Ib, VI, and IIb/IIIa blockade on infarct size, functional outcome, and intracranial bleeding. Circulation 115, 2323–2330 (2007).
Im, J. H. et al. Coagulation facilitates tumour cell spreading in the pulmonary vasculature during early metastatic colony formation. Cancer Res. 64, 8613–8619 (2004).
Palumbo, J. S. et al. Platelets and fibrin(ogen) increase metastatic potential by impeding natural killer cell–mediated elimination of tumour cells. Blood 105, 178–185 (2005).
Machlus, K. R. & Italiano, J. E. The incredible journey: from megakaryocyte development to platelet formation. J. Cell Biol. 201, 785–796 (2013).
Brown, E., Carlin, L. M., Nerlov, C., Lo Celso, C. & Poole, A. W. Multiple membrane extrusion sites drive megakaryocyte migration into bone marrow blood vessels. Life Sci. Alliance 1, e201800061 (2018).
Bender, M. et al. Microtubule sliding drives proplatelet elongation and is dependent on cytoplasmic dynein. Blood 125, 860–868 (2015).
Junt, T. et al. Dynamic visualization of thrombopoiesis within bone marrow. Science 317, 1767–1770 (2007).
Stegner, D. et al. Thrombopoiesis is spatially regulated by the bone marrow vasculature. Nat. Commun. 8, 127 (2017). The authors provide evidence that megakaryocytes are largely sessile cells in close contact with the vasculature and homogeneously distributed in the bone marrow.
Thon, J. N. et al. Microtubule and cortical forces determine platelet size during vascular platelet production. Nat. Commun. 3, 852 (2012).
Kile, B. T. The role of apoptosis in megakaryocytes and platelets. Br. J. Haematol. 165, 217–226 (2014).
Olson, T. S. et al. Megakaryocytes promote murine osteoblastic HSC niche expansion and stem cell engraftment after radioablative conditioning. Blood 121, 5238–5249 (2013).
Bruns, I. et al. Megakaryocytes regulate hematopoietic stem cell quiescence through CXCL4 secretion. Nat. Med. 20, 1315–1320 (2014).
Zhao, M. et al. Megakaryocytes maintain homeostatic quiescence and promote post-injury regeneration of hematopoietic stem cells. Nat. Med. 20, 1321–1326 (2014).
Pinho, S. et al. Lineage-biased hematopoietic stem cells are regulated by distinct niches. Dev. Cell 44, 634–641 (2018).
Hérault, A. et al. Myeloid progenitor cluster formation drives emergency and leukaemic myelopoiesis. Nature 544, 53–58 (2017).
Palumbo, J. S. et al. Factor XIII transglutaminase supports hematogenous tumour cell metastasis through a mechanism dependent on natural killer cell function. J. Thromb. Haemost. 6, 812–819 (2008).
Labelle, M., Begum, S. & Hynes, R. O. Platelets guide the formation of early metastatic niches. Proc. Natl Acad. Sci. USA 111, E3053–E3061 (2014).
Läubli, H., Spanaus, K.-S. & Borsig, L. Selectin-mediated activation of endothelial cells induces expression of CCL5 and promotes metastasis through recruitment of monocytes. Blood 114, 4583–4591 (2009).
Haemmerle, M. et al. Platelets reduce anoikis and promote metastasis by activating YAP1 signaling. Nat. Commun. 8, 310 (2017). This study shows that platelets increase cancer metastasis by activating YAP1 through a RhoA signalling pathway.
Ward, Y. et al. Platelets promote metastasis via binding tumour CD97 leading to bidirectional signaling that coordinates transendothelial migration. Cell Rep. 23, 808–822 (2018).
Schumacher, D., Strilic, B., Sivaraj, K., Wettschureck, N. & Offermanns, S. Platelet-derived nucleotides promote tumour-cell transendothelial migration and metastasis via P2Y2 receptor. Cancer Cell 24, 130–137 (2013).
Martín-Granado, V. et al. C3G promotes a selective release of angiogenic factors from activated mouse platelets to regulate angiogenesis and tumour metastasis. Oncotarget 8, 110994–111011 (2017).
Acknowledgements
This study was supported by SFB914 (S.M.), SFB1123 (S.M.), SFB1312 (S.M.) and the Deutsches Zentrum für Herz-Kreislaufforschung (S.M. and F.G.). F.G. received funding from the European Union’s Horizon 2020 research and innovation programme under Marie Skłodowska-Curie grant agreement no. 747687.
Reviewer information
Nature Reviews Immunology thanks E. Josefsson and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Coelomic circulation
-
The circulatory system of the main body cavity.
- Haemolymph
-
Fluid that circulates in the interior of the arthropod body that is analogous to the blood in vertebrates.
- Thrombopoietin
-
(TPO). A glycoprotein hormone produced by the liver and kidney. TPO binds to and activates TPO receptor (CD110) on haematopoietic stem and progenitor cells, which is necessary for megakaryocyte proliferation and maturation.
- Ticagrelor
-
A reversible inhibitor of the ADP receptor subtype P2Y12 used to treat patients with acute coronary syndromes.
- P2Y12
-
A subtype of the platelet ADP receptor family that triggers strong platelet activation.
- Neutrophil extracellular traps
-
Web-like structures consisting of extracellular DNA strands decorated with antimicrobial proteins such as histones and neutrophil proteases.
- Factor XIII
-
A transglutaminase that crosslinks and stabilizes the fibrin meshwork
- Clopidogrel
-
An irreversible inhibitor of the platelet ADP receptor subtype P2Y12 used clinically to prevent thrombotic events in patients receiving percutaneous coronary intervention for coronary atherosclerosis.
Rights and permissions
About this article
Cite this article
Gaertner, F., Massberg, S. Patrolling the vascular borders: platelets in immunity to infection and cancer. Nat Rev Immunol 19, 747–760 (2019). https://doi.org/10.1038/s41577-019-0202-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41577-019-0202-z
This article is cited by
-
Preoperative prediction of microsatellite instability status in colorectal cancer based on a multiphasic enhanced CT radiomics nomogram model
BMC Medical Imaging (2024)
-
The dynamic role of platelets in cancer progression and their therapeutic implications
Nature Reviews Cancer (2024)
-
Expanding applications of allogeneic platelets, platelet lysates, and platelet extracellular vesicles in cell therapy, regenerative medicine, and targeted drug delivery
Journal of Biomedical Science (2023)
-
Nanomedicine in cancer therapy
Signal Transduction and Targeted Therapy (2023)
-
Lipid remodeling in megakaryocyte differentiation and platelet biogenesis
Nature Cardiovascular Research (2023)