Abstract
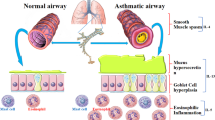
Cytokines play a key role in orchestrating and perpetuating the chronic airway inflammation in asthma and chronic obstructive pulmonary disease (COPD), making them attractive targets for treating these disorders. Asthma and some cases of COPD are mainly driven by type 2 immune responses, which comprise increased airway eosinophils, T helper 2 (TH2) cells and group 2 innate lymphoid cells (ILC2s) and the secretion of IL-4, IL-5 and IL-13. Clinical trials of antibodies that block these interleukins have shown reduced acute exacerbations and oral corticosteroid use and improvements in lung function and symptoms in selected patients. More recent approaches that block upstream cytokines, such as thymic stromal lymphopoietin (TSLP), show promise in improving patient outcome. Importantly, the clinical trials in cytokine blockade have highlighted the crucial importance of patient selection for the successful use of these expensive therapies and the need for biomarkers to better predict drug responses.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Barnes, P. J. et al. Chronic obstructive pulmonary disease. Nat. Rev. Primers 1, 15076 (2015).
Postma, D. S. & Rabe, K. F. The asthma-COPD overlap syndrome. N. Engl. J. Med. 373, 1241–1249 (2015).
Barnes, P. J. Asthma-COPD overlap. Chest 149, 7–8 (2016).
Price, D., Fletcher, M. & van der Molen, T. Asthma control and management in 8,000 European patients: the REcognise Asthma and LInk to Symptoms and Experience (REALISE) survey. NPJ Prim. Care Respir. Med. 24, 14009 (2014).
Gross, N. J. & Barnes, P. J. New therapies for asthma and chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 195, 159–166 (2017).
Chung, K. F. Targeting the interleukin pathway in the treatment of asthma. Lancet 386, 1086–1096 (2015).
Lefaudeux, D. et al. U-BIOPRED clinical adult asthma clusters linked to a subset of sputum omics. J. Allergy Clin. Immunol. 139, 1797–1807 (2017).
Barnes, P. J. Cellular and molecular mechanisms of asthma and COPD. Clin. Sci. 131, 1541–1558 (2017).
Ebbo, M., Crinier, A., Vely, F. & Vivier, E. Innate lymphoid cells: major players in inflammatory diseases. Nat. Rev. Immunol. 17, 665–678 (2017).
Barnes, P. J. Pathophysiology of allergic inflammation. Immunol. Rev. 242, 31–50 (2011).
Hogg, J. C. et al. The nature of small-airway obstruction in chronic obstructive pulmonary disease. N. Engl. J. Med. 350, 2645–2653 (2004).
Brusselle, G. G., Joos, G. F. & Bracke, K. R. New insights into the immunology of chronic obstructive pulmonary disease. Lancet 378, 1015–1026 (2011).
McDonough, J. E. et al. Small-airway obstruction and emphysema in chronic obstructive pulmonary disease. N. Engl. J. Med. 365, 1567–1575 (2011).
Galban, C. J. et al. Computed tomography-based biomarker provides unique signature for diagnosis of COPD phenotypes and disease progression. Nat. Med. 18, 1711–1715 (2012).
Lange, P. et al. Lung-function trajectories leading to chronic obstructive pulmonary disease. N. Engl. J. Med. 373, 111–122 (2015).
Barnes, P. J. Mechanisms of development of multimorbidity in the elderly. Eur. Respir. J. 45, 790–806 (2015).
Barnes, P. J. Glucocorticosteroids. Handb. Exp. Pharmacol. 237, 93–115 (2017).
Barnes, P. J. Corticosteroid resistance in patients with asthma and chronic obstructive pulmonary disease. J. Allergy Clin. Immunol. 131, 636–645 (2013).
Ito, K. et al. Decreased histone deacetylase activity in chronic obstructive pulmonary disease. N. Engl. J. Med. 352, 1967–1976 (2005).
Hew, M. et al. Relative corticosteroid insensitivity of peripheral blood mononuclear cells in severe asthma. Am. J. Respir. Crit. Care Med. 174, 134–141 (2006).
Molfino, N. A., Gossage, D., Kolbeck, R., Parker, J. M. & Geba, G. P. Molecular and clinical rationale for therapeutic targeting of interleukin-5 and its receptor. Clin. Exp. Allergy 42, 712–737 (2012).
Leckie, M. J. et al. Effects of an interleukin-5 blocking monoclonal antibody on eosinophils, airway hyperresponsiveness and the late asthmatic response. Lancet 356, 2144–2148 (2000). This is the first study of an anti-IL-5 antibody in patients with asthma and shows a marked reduction in blood and sputum eosinophils.
Flood-Page, P. et al. A study to evaluate safety and efficacy of mepolizumab in patients with moderate persistent asthma. Am. J. Respir. Crit. Care Med. 176, 1062–1071 (2007).
Haldar, P. et al. Mepolizumab and exacerbations of refractory eosinophilic asthma. N. Engl. J. Med. 360, 973–984 (2009).
Nair, P. et al. Mepolizumab for prednisone-dependent asthma with sputum eosinophilia. N. Engl. J. Med. 360, 985–993 (2009).
Pavord, I. D. et al. Mepolizumab for severe eosinophilic asthma (DREAM): a multicentre, double-blind, placebo-controlled trial. Lancet 380, 651–659 (2012). This is a large study of an anti-IL-5 antibody in patients with severe eosinophilic asthma that shows an ~50% reduction in acute exacerbations.
Bel, E. H. et al. Oral glucocorticoid-sparing effect of mepolizumab in eosinophilic asthma. N. Engl. J. Med. 371, 1189–1197 (2014).
Ortega, H. G. et al. Mepolizumab treatment in patients with severe eosinophilic asthma. N. Engl. J. Med. 371, 1198–1207 (2014).
Powell, C., Milan, S. J., Dwan, K., Bax, L. & Walters, N. Mepolizumab versus placebo for asthma. Cochrane Database Syst. Rev. 7, CD010834 (2015).
Chupp, G. L. et al. Efficacy of mepolizumab add-on therapy on health-related quality of life and markers of asthma control in severe eosinophilic asthma (MUSCA): a randomised, double-blind, placebo-controlled, parallel-group, multicentre, phase 3b trial. Lancet Respir. Med. 5, 390–400 (2017).
Wechsler, M. E. et al. Mepolizumab or placebo for eosinophilic granulomatosis with polyangiitis. N. Engl. J. Med. 376, 1921–1932 (2017).
Kim, S., Marigowda, G., Oren, E., Israel, E. & Wechsler, M. E. Mepolizumab as a steroid-sparing treatment option in patients with Churg-Strauss syndrome. J. Allergy Clin. Immunol. 125, 1336–1343 (2010).
Castro, M. et al. Reslizumab for poorly controlled, eosinophilic asthma: a randomized, placebo-controlled study. Am. J. Respir. Crit. Care Med. 184, 1125–1132 (2011).
Castro, M. et al. Reslizumab for inadequately controlled asthma with elevated blood eosinophil counts: results from two multicentre, parallel, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet Respir. Med. 3, 355–366 (2015).
Bjermer, L. et al. Reslizumab for inadequately controlled asthma with elevated blood eosinophil levels: a randomized phase 3 study. Chest 150, 789–798 (2016).
Brusselle, G., Germinaro, M., Weiss, S. & Zangrilli, J. Reslizumab in patients with inadequately controlled late-onset asthma and elevated blood eosinophils. Pulm. Pharmacol. Ther. 43, 39–45 (2017).
Corren, J., Weinstein, S., Janka, L., Zangrilli, J. & Garin, M. Phase 3 study of reslizumab in patients with poorly controlled asthma: effects across a broad range of eosinophil counts. Chest 150, 799–810 (2016).
Ghazi, A., Trikha, A. & Calhoun, W. J. Benralizumab—a humanized mAb to IL-5Ralpha with enhanced antibody-dependent cell-mediated cytotoxicity — a novel approach for the treatment of asthma. Expert Opin. Biol. Ther. 12, 113–118 (2012).
Kolbeck, R. et al. MEDI-563, a humanized anti-IL-5 receptor alpha mAb with enhanced antibody-dependent cell-mediated cytotoxicity function. J. Allergy Clin. Immunol. 125, 1344–1353 (2010). This paper explains the mechanism of action of benralizumab, an IL-5Rα-specific antibody that induces cytotoxicity.
Castro, M. et al. Benralizumab, an anti-interleukin 5 receptor alpha monoclonal antibody, versus placebo for uncontrolled eosinophilic asthma: a phase 2b randomised dose-ranging study. Lancet Respir. Med. 2, 879–890 (2014).
Khorasanizadeh, M., Eskian, M., Assa’ad, A. H., Camargo, C. A. Jr & Rezaei, N. Efficacy and safety of benralizumab, a monoclonal antibody against IL-5Rα, in uncontrolled eosinophilic asthma. Int. Rev. Immunol. 35, 294–311 (2016).
Bleecker, E. R. et al. Efficacy and safety of benralizumab for patients with severe asthma uncontrolled with high-dosage inhaled corticosteroids and long-acting beta2-agonists (SIROCCO): a randomised, multicentre, placebo-controlled phase 3 trial. Lancet 388, 2115–2127 (2016).
FitzGerald, J. M. et al. Benralizumab, an anti-interleukin-5 receptor alpha monoclonal antibody, as add-on treatment for patients with severe, uncontrolled, eosinophilic asthma (CALIMA): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet 388, 2128–2141 (2016).
Nair, P. et al. Oral glucocorticoid-sparing effect of benralizumab in severe asthma. N. Engl. J. Med. 376, 2448–2458 (2017).
Ferguson, G. T. et al. Benralizumab for patients with mild to moderate, persistent asthma (BISE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Respir. Med. 5, 568–576 (2017).
Nowak, R. M. et al. A randomized trial of benralizumab, an antiinterleukin 5 receptor alpha monoclonal antibody, after acute asthma. Am. J. Emerg. Med. 33, 14–20 (2015).
Brightling, C. E. et al. Benralizumab for chronic obstructive pulmonary disease and sputum eosinophilia: a randomised, double-blind, placebo-controlled, phase 2a study. Lancet Respir. Med. 2, 891–901 (2014).
Dasgupta, A. et al. A pilot randomised clinical trial of mepolizumab in COPD with eosinophilic bronchitis. Eur. Respir. J. 49, 1602486 (2017).
Pavord, I. D. et al. Mepolizumab for eosinophilic chronic obstructive pulmonary disease. N. Engl. J. Med. 377, 1613–1629 (2017). Reference 48 and 49 are two large studies of an anti-IL-5 therapy in patients with COPD that show a small reduction in exacerbations in patients with elevated blood eosinophils.
Leung, E., Al Efraij, K. & FitzGerald, J. M. The safety of mepolizumab for the treatment of asthma. Expert Opin. Drug Safety 16, 397–404 (2017).
Lugogo, N. et al. Long-term efficacy and safety of mepolizumab in patients with severe eosinophilic asthma: a multi-center, open-label, phase IIIb study. Clin. Ther. 38, 2058–2070.e1 (2016).
Cabon, Y. et al. Comparison of anti-interleukin-5 therapies in patients with severe asthma: global and indirect meta-analyses of randomized placebo-controlled trials. Clin. Exp. Allergy 47, 129–138 (2017). This is a meta-analysis of published studies of anti-IL-5 antibody treatment of severe asthma.
Larose, M. C. et al. Correlation between CCL26 production by human bronchial epithelial cells and airway eosinophils: Involvement in patients with severe eosinophilic asthma. J. Allergy Clin. Immunol. 136, 904–913 (2015).
Kasaian, M. T. & Miller, D. K. IL-13 as a therapeutic target for respiratory disease. Biochem. Pharmacol. 76, 147–155 (2008).
Antoniu, S. A. Pitrakinra, a dual IL-4/IL-13 antagonist for the potential treatment of asthma and eczema. Curr. Opin. Invest. Drugs 11, 1286–1294 (2010).
Chiba, Y., Todoroki, M., Nishida, Y., Tanabe, M. & Misawa, M. A novel STAT6 inhibitor AS1517499 ameliorates antigen-induced bronchial hypercontractility in mice. Am. J. Respir. Cell Mol. Biol. 41, 516–524 (2009).
Izuhara, K. et al. Roles of periostin in respiratory disorders. Am. J. Respir. Crit. Care Med. 19, 949–956 (2016).
Corren, J. et al. Lebrikizumab treatment in adults with asthma. N. Engl. J. Med. 365, 1088–1098 (2011).
Hanania, N. A. et al. Lebrikizumab in moderate-to-severe asthma: pooled data from two randomised placebo-controlled studies. Thorax 70, 748–756 (2015). References 58 and 59 are two large studies of an anti-IL-13 antibody that demonstrate little clinical benefit in patients with severe type 2 asthma.
Piper, E. et al. A phase II placebo-controlled study of tralokinumab in moderate-to-severe asthma. Eur. Respir. J. 41, 330–338 (2013).
Brightling, C. E. et al. Efficacy and safety of tralokinumab in patients with severe uncontrolled asthma: a randomised, double-blind, placebo-controlled, phase 2b trial. Lancet. Resp. Med. 3, 692–701 (2015).
Wenzel, S. et al. Dupilumab in persistent asthma with elevated eosinophil levels. N. Engl. J. Med. 368, 2455–2466 (2013).
Wenzel, S. et al. Dupilumab efficacy and safety in adults with uncontrolled persistent asthma despite use of medium-to-high-dose inhaled corticosteroids plus a long-acting beta2 agonist: a randomised double-blind placebo-controlled pivotal phase 2b dose-ranging trial. Lancet 388, 31–44 (2016). This is a large clinical trial of an anti-IL-4Rα antibody in patients with moderate to severe asthma, which shows marked improvement of symptoms and lung function and reduced exacerbations, even in patients without elevated blood eosinophils.
Blauvelt, A. et al. Long-term management of moderate-to-severe atopic dermatitis with dupilumab and concomitant topical corticosteroids (LIBERTY AD CHRONOS): a 1-year, randomised, double-blinded, placebo-controlled, phase 3 trial. Lancet 389, 2287–2303 (2017).
Kraft, M. & Worm, M. Dupilumab in the treatment of moderate-to-severe atopic dermatitis. Expert Rev. Clin. Immunol. 13, 301–310 (2017).
Bachert, C. et al. Effect of subcutaneous dupilumab on nasal polyp burden in patients with chronic sinusitis and nasal polyposis: a randomized clinical trial. JAMA 315, 469–479 (2016).
Mitchell, P. D. & O’Byrne, P. M. Epithelial-derived cytokines in asthma. Chest 151, 1338–1344 (2017).
Ying, S. et al. Expression and cellular provenance of thymic stromal lymphopoietin and chemokines in patients with severe asthma and chronic obstructive pulmonary disease. J. Immunol. 181, 2790–2798 (2008).
Gauvreau, G. M. et al. Effects of an anti-TSLP antibody on allergen-induced asthmatic responses. N. Engl. J. Med. 370, 2102–2110 (2014).
Corren, J. et al. Tezepelumab in adults with uncontrolled asthma. N. Engl. J. Med. 377, 936–946 (2017). This large study shows good efficacy of an anti-TSLP antibody in patients with severe asthma, with improvement in symptoms and lung function and reduced exacerbations, blood eosinophils and FeNO.
Nagarkar, D. R. et al. Thymic stromal lymphopoietin activity is increased in nasal polyps of patients with chronic rhinosinusitis. J. Allergy Clin. Immunol. 132, 593–600 (2013).
Verstraete, K. et al. Structure and antagonism of the receptor complex mediated by human TSLP in allergy and asthma. Nat. Commun. 8, 14937 (2017).
Xu, M. & Dong, C. IL-25 in allergic inflammation. Immunol. Rev. 278, 185–191 (2017).
Mitchell, P. D. & O’Byrne, P. M. Biologics and the lung: TSLP and other epithelial cell-derived cytokines in asthma. Pharmacol. Ther. 169, 104–112 (2017).
Cheng, D. et al. Epithelial interleukin-25 is a key mediator in Th2-high, corticosteroid-responsive asthma. Am. J. Respir. Crit. Care Med. 190, 639–648 (2014).
Christianson, C. A. et al. Persistence of asthma requires multiple feedback circuits involving type 2 innate lymphoid cells and IL-33. J. Allergy Clin. Immunol. 136, 59–68 (2015).
Kubo, M. Innate and adaptive type 2 immunity in lung allergic inflammation. Immunol. Rev. 278, 162–172 (2017).
Prefontaine, D. et al. Increased IL-33 expression by epithelial cells in bronchial asthma. J. Allergy Clin. Immunol. 125, 752–754 (2010).
Qiu, C. et al. Anti-interleukin-33 inhibits cigarette smoke-induced lung inflammation in mice. Immunology 138, 76–82 (2013).
Xia, J. et al. Increased IL-33 expression in chronic obstructive pulmonary disease. Am. J. Physiol. Lung Cell Mol. Physiol. 308, L619–L627 (2015).
Vlahos, R., Bozinovski, S., Hamilton, J. A. & Anderson, G. P. Therapeutic potential of treating chronic obstructive pulmonary disease (COPD) by neutralising granulocyte macrophage-colony stimulating factor (GM-CSF). Pharmacol. Ther. 112, 106–115 (2006).
Molfino, N. A. et al. Phase 2, randomised placebo-controlled trial to evaluate the efficacy and safety of an anti-GM-CSF antibody (KB003) in patients with inadequately controlled asthma. BMJ Open 6, e007709 (2016).
Barnes, P. J. Role of GATA-3 in allergic diseases. Curr. Mol. Med. 8, 330–334 (2008).
Finotto, S. et al. Treatment of allergic airway inflammation and hyperresponsiveness by antisense-induced local blockade of GATA-3 expression. J. Exp. Med. 193, 1247–1260 (2001).
Maneechotesuwan, K. et al. Suppression of GATA-3 nuclear import and phosphorylation: a novel mechanism of corticosteroid action in allergic disease. PLoS Med. 6, e1000076 (2009).
Krug, N. et al. Allergen-induced asthmatic responses modified by a GATA3-specific DNAzyme. N. Engl. J. Med. 372, 1987–1995 (2015). This study shows that an inhaled GATA3-blocking DNAzyme modestly reduces the response to inhaled allergen in patients with mild asthma.
Berry, M. A. et al. Evidence of a role of tumor necrosis factor alpha in refractory asthma. N. Engl. J. Med. 354, 697–708 (2006).
Howarth, P. H. et al. Tumour necrosis factor (TNFalpha) as a novel therapeutic target in symptomatic corticosteroid dependent asthma. Thorax 60, 1012–1018 (2005).
Erin, E. M. et al. The effects of a monoclonal antibody directed against tumour necrosis factor-alpha in asthma. Am. J. Respir. Crit. Care Med. 174, 753–762 (2006).
Wenzel, S. E. et al. A randomized, double-blind, placebo-controlled study of TNF-α blockade in severe persistent asthma. Am. J. Respir. Crit. Care Med. 179, 549–558 (2009). This large study demonstrates a lack of effect and side effects of anti-TNF therapy in patients with severe asthma.
Rennard, S. I. et al. The safety and efficacy of infliximab in moderate to severe chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 175, 926–934 (2007). This study shows a lack of clinical effect and serious side effects with an anti-TNF therapy in patients with severe COPD.
Suissa, S., Ernst, P. & Hudson, M. TNF-alpha antagonists and the prevention of hospitalisation for chronic obstructive pulmonary disease. Pulm. Pharmacol. Ther. 21, 234–238 (2008).
Dentener, M. A. et al. Effect of infliximab on local and systemic inflammation in chronic obstructive pulmonary disease: a pilot study. Respiration 76, 275–282 (2008).
Al-Ramli, W. et al. T(H)17-associated cytokines (IL-17A and IL-17F) in severe asthma. J. Allergy Clin. Immunol. 123, 1185–1187 (2009).
Ricciardolo, F. L. M. et al. Identification of IL-17F/frequent exacerbator endotype in asthma. J. Allergy Clin. Immunol. 140, 395–406 (2017).
Alcorn, J. F., Crowe, C. R. & Kolls, J. K. TH17 cells in asthma and COPD. Annu. Rev. Physiol. 72, 495–516 (2010).
Di Stefano, A. et al. Th17-related cytokine expression is increased in the bronchial mucosa of stable COPD patients. Clin. Exp. Immunol 157, 316–324 (2009).
Maneechotesuwan, K., Kasetsinsombat, K., Wongkajornsilp, A. & Barnes, P. J. Decreased indoleamine 2,3-dioxygenase activity and IL-10/IL-17 A ratio in patients with COPD. Thorax 68, 330–337 (2013).
Shen, N., Wang, J., Zhao, M., Pei, F. & He, B. Anti-interleukin-17 antibodies attenuate airway inflammation in tobacco-smoke-exposed mice. Inhal. Toxicol. 23, 212–218 (2011).
McKinley, L. et al. TH17 cells mediate steroid-resistant airway inflammation and airway hyperresponsiveness in mice. J. Immunol. 181, 4089–4097 (2008).
Busse, W. W. et al. Randomized, double-blind, placebo-controlled study of brodalumab, a human anti-IL-17 receptor monoclonal antibody, in moderate to severe asthma. Am. J. Respir. Crit. Care Med. 188, 1294–1302 (2013). This study shows a lack of clinical effect of an anti-IL-17R antibody in patients with severe asthma.
Eich, A. et al. A randomized, placebo-controlled phase 2 trial of CNTO 6785 in chronic obstructive pulmonary disease. COPD 14, 476–483 (2017).
Duvallet, E., Semerano, L., Assier, E., Falgarone, G. & Boissier, M. C. Interleukin-23: a key cytokine in inflammatory diseases. Ann. Med. 43, 503–511 (2011).
Fujii, U. et al. IL-23 is essential for the development of elastase-induced pulmonary inflammation and emphysema. Am. J. Respir. Cell. Mol. Biol. 55, 697–707 (2016).
Benson, J. M. et al. Therapeutic targeting of the IL-12/23 pathways: generation and characterization of ustekinumab. Nat. Biotechnol. 29, 615–624 (2011).
Papp, K. A. et al. Risankizumab versus ustekinumab for moderate-to-severe plaque psoriasis. N. Engl. J. Med. 376, 1551–1560 (2017).
US National Library of Medicine. ClincalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02443298 (2018).
Sousa, A. R., Lane, S. J., Nakhosteen, J. A., Lee, T. H. & Poston, R. N. Expression of interleukin-1 beta (IL-1β) and interleukin-1 receptor antagonist (IL-1ra) on asthmatic bronchial epithelium. Am. J. Respir. Crit. Care Med. 154, 1061–1066 (1996).
Kim, R. Y. et al. Inflammasomes in COPD and neutrophilic asthma. Thorax 70, 1199–1201 (2015).
Di Stefano, A. et al. Innate immunity but not NLRP3 inflammasome activation correlates with severity of stable COPD. Thorax 69, 516–524 (2014).
Rogliani, P., Calzetta, L., Ora, J. & Matera, M. G. Canakinumab for the treatment of chronic obstructive pulmonary disease. Pulm Pharmacol. Ther. 31, 15–27 (2015).
Ridker, P. M. et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 377, 1119–1131 (2017).
Novick, D., Kim, S., Kaplanski, G. & Dinarello, C. A. Interleukin-18, more than a Th1 cytokine. Semin. Immunol. 25, 439–448 (2013).
Rovina, N. et al. Interleukin-18 in induced sputum: association with lung function in chronic obstructive pulmonary disease. Respir. Med. 103, 1056–1062 (2009).
Dima, E. et al. Implication of interleukin (IL)-18 in the pathogenesis of chronic obstructive pulmonary disease (COPD). Cytokine 74, 313–317 (2015).
Briend, E. et al. IL-18 associated with lung lymphoid aggregates drives IFNgamma production in severe COPD. Respir. Res. 18, 159 (2017).
Mistry, P. et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of single-dose antiinterleukin- 18 mAb GSK1070806 in healthy and obese subjects. Int. J. Clin. Pharmacol. Ther. 52, 867–879 (2014).
Grubek-Jaworska, H. et al. IL-6 and IL-13 in induced sputum of COPD and asthma patients: correlation with respiratory tests. Respiration 84, 101–107 (2012).
Nishimoto, N. & Kishimoto, T. Inhibition of IL-6 for the treatment of inflammatory diseases. Curr. Opin. Pharmacol. 4, 386–391 (2004).
Paul-Pletzer, K. Tocilizumab: blockade of interleukin-6 signaling pathway as a therapeutic strategy for inflammatory disorders. Drugs Today 42, 559–576 (2006).
Donnelly, L. E. & Barnes, P. J. Chemokine receptors as therapeutic targets in chronic obstructive pulmonary disease. Trends Pharmacol. Sci. 27, 546–553 (2006).
Erin, E. M., Williams, T. J., Barnes, P. J. & Hansel, T. T. Eotaxin receptor (CCR3) antagonism in asthma and allergic disease. Curr. Drug Targets. Inflamm. Allergy 1, 201–214 (2002).
Das, A. M. et al. Selective inhibition of eosinophil influx into the lung by small molecule CC chemokine receptor 3 antagonists in mouse models of allergic inflammation. J. Pharmacol. Exp. Ther. 318, 411–417 (2006).
Ying, S. et al. Eosinophil chemotactic chemokines (eotaxin, eotaxin-2, RANTES, monocyte chemoattractant protein-3 (MCP-3), and MCP-4), and C-C chemokine receptor 3 expression in bronchial biopsies from atopic and nonatopic (Intrinsic) asthmatics. J. Immunol. 163, 6321–6329 (1999).
Pease, J. E. & Horuk, R. Recent progress in the development of antagonists to the chemokine receptors CCR3 and CCR4. Expert Opin. Drug Discov. 9, 467–483 (2014).
Sabroe, I. et al. A small molecule antagonist of chemokine receptors CCR1 and CCR3. Potent inhibition of eosinophil function and CCR3-mediated HIV-1 entry. J. Biol. Chem. 275, 25985–25992 (2000).
Gauvreau, G. M. et al. Antisense therapy against CCR3 and the common beta chain attenuates allergen-induced eosinophilic responses. Am. J. Respir. Crit. Care Med. 177, 952–958 (2008).
Donnelly, L. E. & Barnes, P. J. Chemokine receptor CXCR2 antagonism to prevent airway inflammation. Drugs Future 36, 465–472 (2011).
Traves, S. L., Culpitt, S., Russell, R. E. K., Barnes, P. J. & Donnelly, L. E. Elevated levels of the chemokines GRO-α and MCP-1 in sputum samples from COPD patients. Thorax 57, 590–595 (2002).
Jatakanon, A. et al. Neutrophilic inflammation in severe persistent asthma. Am. J. Respir. Crit. Care Med. 160, 1532–1539 (1999).
Holz, O. et al. SCH527123, a novel CXCR2 antagonist, inhibits ozone-induced neutrophilia in healthy subjects. Eur. Respir. J. 35, 564–570 (2010).
Leaker, B. R., Barnes, P. J. & O’Connor, B. Inhibition of LPS-induced airway neutrophilic inflammation in healthy volunteers with an oral CXCR2 antagonist. Respir. Res. 14, 137 (2013).
Lazaar, A. L. et al. SB-656933, a novel CXCR2 selective antagonist, inhibits ex vivo neutrophil activation and ozone-induced airway inflammation in humans. Br. J. Clin. Pharmacol 72, 282–293 (2011).
Rennard, S. I. et al. CXCR2 antagonist MK-7123- a phase 2 proof-of-concept trial for chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 191, 1001–1011 (2015). This large study shows an overall lack of clinical benefit of an oral CXCR2 antagonist in patients with COPD.
Nair, P. et al. Safety and efficacy of a CXCR2 antagonist in patients with severe asthma and sputum neutrophils: a randomized, placebo-controlled clinical trial. Clin. Exp. Allergy 42, 1097–1103 (2012). This study shows a lack of clinical benefit of a CXCR2 antagonist in severe neutrophilic asthma.
O’Byrne, P. M. et al. Efficacy and safety of a CXCR2 antagonist, AZD5069, in patients with uncontrolled persistent asthma: a randomised, double-blind, placebo-controlled trial. Lancet Respir. Med. 4, 797–806 (2016).
Busch-Petersen, J. et al. Danirixin: a Reversible and Selective Antagonist of the CXC Chemokine Receptor 2. J. Pharmacol. Exp. Ther. 362, 338–346 (2017).
Kirsten, A. M. et al. The safety and tolerability of oral AZD5069, a selective CXCR2 antagonist, in patients with moderate-to-severe COPD. Pulm Pharmacol. Ther. 31, 36–41 (2015).
Costa, C. et al. CXCR3 and CCR5 chemokines in the induced sputum from patients with COPD. Chest 133, 26–33 (2008).
Pilette, C., Francis, J. N., Till, S. J. & Durham, S. R. CCR4 ligands are up-regulated in the airways of atopic asthmatics after segmental allergen challenge. Eur. Respir. J. 23, 876–884 (2004).
Costa, C. et al. Enhanced monocyte migration to CXCR3 and CCR5 chemokines in COPD. Eur. Respir. J. 47, 1093–1102 (2016).
Kerstjens, H. A., Bjermer, L., Eriksson, L., Dahlstrom, K. & Vestbo, J. Tolerability and efficacy of inhaled AZD4818, a CCR1 antagonist, in moderate to severe COPD patients. Respir. Med. 104, 1297–1303 (2010).
Woollard, S. M. & Kanmogne, G. D. Maraviroc: a review of its use in HIV infection and beyond. Drug Design Dev. Ther. 9, 5447–5468 (2015).
Vijayanand, P. et al. Chemokine receptor 4 plays a key role in T cell recruitment into the airways of asthmatic patients. J. Immunol. 184, 4568–4574 (2010).
Cahn, A. et al. Safety, tolerability, pharmacokinetics and pharmacodynamics of GSK2239633, a CC-chemokine receptor 4 antagonist, in healthy male subjects: results from an open-label and from a randomised study. BMC Pharmacol. Toxicol. 14, 14 (2013).
ten Brinke, A., Zwinderman, A. H., Sterk, P. J., Rabe, K. F. & Bel, E. H. “Refractory” eosinophilic airway inflammation in severe asthma: effect of parenteral corticosteroids. Am. J. Respir. Crit. Care Med. 170, 601–605 (2004).
Pavord, I. D. et al. Blood eosinophils and inhaled corticosteroid/long-acting beta-2 agonist efficacy in COPD. Thorax 71, 118–125 (2016).
Acknowledgements
P.J.B. holds a National Institute for Health Research Senior Investigator Award.
Competing interests
P.J.B. has served on Scientific Advisory Boards of AstraZeneca, Boehringer-Ingelheim, Chiesi, GlaxoSmithKline, Glenmark, Johnson & Johnson, Napp, Novartis, Pfizer, ProSonix, RespiVert, Teva and Zambon and has received research funding from AstraZeneca, Boehringer-Ingelheim, Chiesi, Novartis and Takeda.
Reviewer information
Nature Reviews Immunology thanks Sally Wenzel and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s noteSpringer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- U-BIOPRED
-
Unbiased Biomarkers for the Prediction of Respiratory Disease Outcomes is a European project investigating mechanisms of severe asthma.
- Type 2 immunity
-
A type of immunity that provides protection against parasitic infections but is also activated in allergic diseases such as asthma. It is orchestrated by T helper 2 (TH2) cells and group 2 innate lymphoid cells (ILC2s) through the secretion of IL-4, IL-5, IL-9 and IL-13, which are regulated by the transcription factor GATA3. Type 2 inflammation is characterized by eosinophils, mast cells and alternatively activated macrophages. Upstream cytokines from epithelial cells that regulate TH2 cells and ILC2s include the alarmins IL-25, IL-33 and thymic stromal lymphopoietin (TSLP).
- T helper 2 (TH2) cells
-
A subset of CD4+ T helper cells that has an important role in humoral immunity and in allergic responses. TH2 cells express the transcription factors GATA3 and signal transducer and activator of transcription 6 (STAT6) and produce cytokines such as IL-4, IL-5, IL-9 and IL-13, which regulate IgE synthesis, eosinophil proliferation, mast cell proliferation and airway hyperresponsiveness, respectively.
- Innate lymphoid cells
-
Immune cells that do not express antigen specific B or T cell receptors. Three main subtypes are recognized depending on the transcription factors expressed and the pattern of cytokines they release.
- Airway hyperresponsiveness
-
Increased airway narrowing in response to various external stimuli that is a physiological characteristic of patients with asthma.
- Type 1 immunity
-
A type of immunity that provides protection against microbial infections and is orchestrated by T helper 1 cells, cytotoxic T cells and group 1 innate lymphoid cells and the transcription factor T-bet, which regulates the secretion of IFNγ. Type 1 immunity is associated with increased pro-inflammatory macrophage activation.
- Type 3 immunity
-
A type of immunity that is directed against microorganisms, in particular fungi, and orchestrated by T helper 17 cells and group 3 innate lymphoid cells, which express RORγt and secrete IL-17 and IL-22, leading to neutrophilic inflammation.
- TH17 cells
-
A subset of CD4+ T helper cells that express the transcription factor RORγt and secrete IL-17A, IL-17F and IL-22. They are involved in neutrophilic inflammatory responses and are important in responses to bacteria and viruses, as well as autoimmune diseases.
- GATA3
-
A transcription factor expressed predominantly by T helper 2 cells and group 2 innate lymphoid cells that is important for their differentiation and secretion of characteristic type 2 cytokines (IL-4, IL-5, IL-9 and IL-13).
- Forced expired volume in 1 second
-
(FEV1). A measure of airflow. Decreases in FEV1 reflect airway obstruction in asthma and chronic obstructive pulmonary disease and are a measure of disease severity.
- Fractional exhaled nitric oxide
-
(FeNO). The fractional content in exhaled breath of nitric oxide gas, which is a non-invasive biomarker of type 2 inflammation.
- Alarmin
-
A type of molecule that is released from damaged cells and activates the immune system. In airway disease, alarmins released from airway epithelial cells include IL-25, IL-33 and thymic stromal lymphopoietin (TSLP), which activate type 2 immunity.
- NLRP3 inflammasome
-
Intracellular multiprotein complex containing NOD-, LRR- and pyrin domain-containing protein 3 (NLRP3), which leads to activation of caspase 1 and the generation of IL-1β and IL-18 from precursors. It is a component of the innate immune system and activated by several inflammatory signals.
- Muckle–Wells syndrome
-
Rare autosomal dominant disease that causes periodic fever and is associated with NLRP3 inflammasome activation and increased production of IL-1β.
Rights and permissions
About this article
Cite this article
Barnes, P.J. Targeting cytokines to treat asthma and chronic obstructive pulmonary disease. Nat Rev Immunol 18, 454–466 (2018). https://doi.org/10.1038/s41577-018-0006-6
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41577-018-0006-6
This article is cited by
-
Vitamin D and the microbiota connection: understanding its potential to improve COPD outcomes
The Egyptian Journal of Bronchology (2024)
-
Effect of YuPingFeng granules on clinical symptoms of stable COPD: study protocol for a multicenter, double-blind, and randomized controlled trial
BMC Complementary Medicine and Therapies (2024)
-
Ferroptotic alveolar epithelial type II cells drive TH2 and TH17 mixed asthma triggered by birch pollen allergen Bet v 1
Cell Death Discovery (2024)
-
Immunological factors linked to geographical variation in vaccine responses
Nature Reviews Immunology (2024)
-
Extracts of Phlomoides umbrosa Turczaninow alleviate allergic airway inflammation in lipopolysaccharide-stimulated RAW 264.7 cells and ovalbumin-induced hyper-responsiveness mouse model
Food Science and Biotechnology (2024)