Abstract
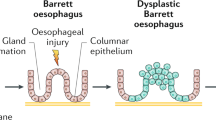
Oesophageal cancer is a global health problem; in 2018 there were more than 572,000 people newly diagnosed with oesophageal cancer worldwide. There are two main histological subtypes of oesophageal cancer, oesophageal adenocarcinoma (EAC) and oesophageal squamous cell carcinoma (ESCC), and there has been a dramatic shift in its epidemiology. While the incidence of EAC and its precursor lesion, Barrett oesophagus, has increased in Western populations over the past four decades, the incidence of ESCC has declined in most parts of the world over the same period. ESCC still accounts for the vast majority of all oesophageal cancer cases diagnosed worldwide each year. Prognosis for patients with oesophageal cancer is strongly related to stage at diagnosis. As most patients are diagnosed with late-stage disease, overall 5-year survival for oesophageal cancer remains <20%. Knowledge of epidemiology and risk factors for oesophageal cancer is essential for public health and clinical decisions about risk stratification, screening and prevention. The goal of this Review is to establish the current epidemiology of oesophageal cancer, with a particular focus on the Western world and the increasing incidence of EAC and Barrett oesophagus.
Key points
-
Oesophageal squamous cell carcinoma (ESCC) remains the most common subtype of oesophageal cancer worldwide; however, in Western populations, the incidence of oesophageal adenocarcinoma (EAC) has increased markedly.
-
The causes of the striking male predominance and racial difference in the incidence of EAC, and its precursor, Barrett oesophagus, remain unknown.
-
The main risk factors for EAC and Barrett oesophagus are gastroesophageal reflux disease, abdominal obesity and cigarette smoking, whereas alcohol consumption and cigarette smoking cause most ESCC cases globally.
-
Patients with EAC who have a prior diagnosis of Barrett oesophagus have better outcomes than patients without a prior diagnosis of Barrett oesophagus; however, this situation represents <10% of patients with EAC.
-
Screening for EAC and Barrett oesophagus needs to go beyond patients with frequent gastroesophageal reflux disease symptoms to include other established risk factors, including obesity and smoking.
-
To date, no screening or surveillance algorithm has sufficient discriminatory accuracy or external validation to support clinical use.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
World Health Organization. Cancer Today: Data visualization tools for exploring the global cancer burden in 2020. https://gco.iarc.fr/today/home (2020).
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 68, 394–424 (2018).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 69, 7–34 (2019).
Thrift, A. P. Barrett’s esophagus and esophageal adenocarcinoma: how common are they really? Dig. Dis. Sci. 63, 1988–1996 (2018).
Anderson, L. A. et al. Survival for oesophageal, stomach and small intestine cancers in Europe 1999-2007: results from EUROCARE-5. Eur. J. Cancer 51, 2144–2157 (2015).
Arnold, M., Soerjomataram, I., Ferlay, J. & Forman, D. Global incidence of oesophageal cancer by histological subtype in 2012. Gut 64, 381–387 (2015).
Pohl, H. & Welch, H. G. The role of overdiagnosis and reclassification in the marked increase of esophageal adenocarcinoma incidence. J. Natl Cancer Inst. 97, 142–146 (2005).
Thrift, A. P. & Whiteman, D. C. The incidence of esophageal adenocarcinoma continues to rise: analysis of period and birth cohort effects on recent trends. Ann. Oncol. 23, 3155–3162 (2012).
Edgren, G., Adami, H. O., Weiderpass, E. & Nyren, O. A global assessment of the oesophageal adenocarcinoma epidemic. Gut 62, 1406–1414 (2013).
Kendall, B. J. & Whiteman, D. C. Temporal changes in the endoscopic frequency of new cases of Barrett’s esophagus in an Australian health region. Am. J. Gastroenterol. 101, 1178–1182 (2006).
Dong, J., Gu, X., El-Serag, H. B. & Thrift, A. P. Underuse of surgery accounts for racial disparities in esophageal cancer survival times: a matched cohort study. Clin. Gastroenterol. Hepatol. 17, 657–665 (2019).
Bray, F. et al. (eds) Cancer Incidence in Five Continents, Vol. XI (Electronic Version) (International Agency for Research on Cancer, 2017).
Arnold, M., Laversanne, M., Brown, L. M., Devesa, S. S. & Bray, F. Predicting the future burden of esophageal cancer by histological subtype: international trends in incidence up to 2030. Am. J. Gastroenterol. 112, 1247–1255 (2017).
Offman, J., Pesola, F. & Sasieni, P. Trends and projections in adenocarcinoma and squamous cell carcinoma of the oesophagus in England from 1971 to 2037. Br. J. Cancer 118, 1391–1398 (2018).
Wang, Q. L., Xie, S. H., Wahlin, K. & Lagergren, J. Global time trends in the incidence of esophageal squamous cell carcinoma. Clin. Epidemiol. 10, 717–728 (2018).
Xie, S. H. & Lagergren, J. A global assessment of the male predominance in esophageal adenocarcinoma. Oncotarget 7, 38876–38883 (2016).
Xie, S. H. & Lagergren, J. The male predominance in esophageal adenocarcinoma. Clin. Gastroenterol. Hepatol. 14, 338–347 (2016).
Thrift, A. P. & El-Serag, H. B. Sex and racial disparity in incidence of esophageal adenocarcinoma: observations and explanations. Clin. Gastroenterol. Hepatol. 14, 330–332 (2016).
Cook, M. B., Chow, W. H. & Devesa, S. S. Oesophageal cancer incidence in the United States by race, sex, and histologic type, 1977–2005. Br. J. Cancer 101, 855–859 (2009).
Cook, M. B., Wild, C. P. & Forman, D. A systematic review and meta-analysis of the sex ratio for Barrett’s esophagus, erosive reflux disease, and nonerosive reflux disease. Am. J. Epidemiol. 162, 1050–1061 (2005).
Xie, S. H. & Lagergren, J. Social group disparities in the incidence and prognosis of oesophageal cancer. U Eur. Gastroenterol. J. 6, 343–348 (2018).
Launoy, G., Bossard, N., Castro, C. & Manfredi, S. Trends in net survival from esophageal cancer in six European Latin countries: results from the SUDCAN population-based study. Eur. J. Cancer Prev. 26, S24–S31 (2017).
Surveillance, Epidemiology, and End Rresults Program. SEER*Stat Database: Incidence - SEER 9 Regs Research Data, Nov 2018 Sub (1975-2016) <Katrina/Rita Population Adjustment> - Linked To County Attributes - Total U.S., 1969-2017 Counties, National Cancer Institute, DCCPS, Surveillance Research Program, released April 2019, based on the2018 submission (National Cancer Institute, 2020).
Thrift, A. P. The epidemic of oesophageal carcinoma: where are we now? Cancer Epidemiol. 41, 88–95 (2016).
Steyerberg, E. W., Earle, C. C., Neville, B. A. & Weeks, J. C. Racial differences in surgical evaluation, treatment, and outcome of locoregional esophageal cancer: a population-based analysis of elderly patients. J. Clin. Oncol. 23, 510–517 (2005).
Spechler, S. J. Clinical practice. Barrett’s esophagus. N. Engl. J. Med. 346, 836–842 (2002).
Ronkainen, J. et al. Prevalence of Barrett’s esophagus in the general population: an endoscopic study. Gastroenterology 129, 1825–1831 (2005).
Cook, M. B. et al. Cancer incidence and mortality risks in a large US Barrett’s oesophagus cohort. Gut 67, 418–529 (2018).
Desai, T. K. et al. The incidence of oesophageal adenocarcinoma in non-dysplastic Barrett’s oesophagus: a meta-analysis. Gut 61, 970–976 (2012).
Rastogi, A. et al. Incidence of esophageal adenocarcinoma in patients with Barrett’s esophagus and high-grade dysplasia: a meta-analysis. Gastrointest. Endosc. 67, 394–398 (2008).
Engel, L. S. et al. Population attributable risks of esophageal and gastric cancers. J. Natl Cancer Inst. 95, 1404–1413 (2003).
Olsen, C. M., Pandeya, N., Green, A. C., Webb, P. M. & Whiteman, D. C. Population attributable fractions of adenocarcinoma of the esophagus and gastroesophageal junction. Am. J. Epidemiol. 174, 582–590 (2011).
Hazelton, W. D. et al. The role of gastroesophageal reflux and other factors during progression to esophageal adenocarcinoma. Cancer Epidemiol. Biomarkers Prev. 24, 1012–1023 (2015).
Pandeya, N., Olsen, C. M. & Whiteman, D. C. Sex differences in the proportion of esophageal squamous cell carcinoma cases attributable to tobacco smoking and alcohol consumption. Cancer Epidemiol. 37, 579–584 (2013).
Sheikh, M. et al. Individual and combined effects of environmental risk factors for esophageal cancer based on results from the Golestan Cohort Study. Gastroenterology 156, 1416–1427 (2019).
Lagergren, J., Bergstrom, R., Lindgren, A. & Nyren, O. Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma. N. Engl. J. Med. 340, 825–831 (1999).
Cook, M. B. et al. Gastroesophageal reflux in relation to adenocarcinomas of the esophagus: a pooled analysis from the Barrett’s and Esophageal Adenocarcinoma Consortium (BEACON). PLoS ONE 9, e103508 (2014).
Thrift, A. P., Kramer, J. R., Qureshi, Z., Richardson, P. A. & El-Serag, H. B. Age at onset of GERD symptoms predicts risk of Barrett’s esophagus. Am. J. Gastroenterol. 108, 915–922 (2013).
Yoshida, N. Inflammation and oxidative stress in gastroesophageal reflux disease. J. Clin. Biochem. Nutr. 40, 13–23 (2007).
Cook, M. B. et al. Cigarette smoking and adenocarcinomas of the esophagus and esophagogastric junction: a pooled analysis from the international BEACON consortium. J. Natl Cancer Inst. 102, 1344–1353 (2010).
Steevens, J., Schouten, L. J., Goldbohm, R. A. & van den Brandt, P. A. Alcohol consumption, cigarette smoking and risk of subtypes of oesophageal and gastric cancer: a prospective cohort study. Gut 59, 39–48 (2010).
Wang, Q. L., Xie, S. H., Li, W. T. & Lagergren, J. Smoking cessation and risk of esophageal cancer by histological type: systematic review and meta-analysis. J. Natl Cancer Inst. 109, djx115 (2017).
Pandeya, N. et al. Associations of duration, intensity, and quantity of smoking with adenocarcinoma and squamous cell carcinoma of the esophagus. Am. J. Epidemiol. 168, 105–114 (2008).
Prabhu, A., Obi, K. O. & Rubenstein, J. H. The synergistic effects of alcohol and tobacco consumption on the risk of esophageal squamous cell carcinoma: a meta-analysis. Am. J. Gastroenterol. 109, 822–827 (2014).
Wang, J. B. et al. Attributable causes of esophageal cancer incidence and mortality in China. PLoS ONE 7, e42281 (2012).
Dar, N. A. et al. Hookah smoking, nass chewing, and oesophageal squamous cell carcinoma in Kashmir, India. Br. J. Cancer 107, 1618–1623 (2012).
Cook, M. B. et al. Cigarette smoking increases risk of Barrett’s esophagus: an analysis of the Barrett’s and Esophageal Adenocarcinoma Consortium. Gastroenterology 142, 744–753 (2012).
Smith, K. J., O’Brien, S. M., Green, A. C., Webb, P. M. & Whiteman, D. C. Current and past smoking significantly increase risk for Barrett’s esophagus. Clin. Gastroenterol. Hepatol. 7, 840–848 (2009).
Edelstein, Z. R., Farrow, D. C., Bronner, M. P., Rosen, S. N. & Vaughan, T. L. Central adiposity and risk of Barrett’s esophagus. Gastroenterology 133, 403–411 (2007).
Anderson, L. A. et al. Risk factors for Barrett’s oesophagus and oesophageal adenocarcinoma: results from the FINBAR study. World J. Gastroenterol. 13, 1585–1594 (2007).
Kubo, A. et al. Cigarette smoking and the risk of Barrett’s esophagus. Cancer Causes Control. 20, 303–311 (2009).
Thrift, A. P., Kramer, J. R., Richardson, P. A. & El-Serag, H. B. No significant effects of smoking or alcohol consumption on risk of Barrett’s esophagus. Dig. Dis. Sci. 59, 108–116 (2014).
Hardikar, S. et al. The role of tobacco, alcohol, and obesity in neoplastic progression to esophageal adenocarcinoma: a prospective study of Barrett’s esophagus. PLoS ONE 8, e52192 (2013).
Coleman, H. G. et al. Tobacco smoking increases the risk of high-grade dysplasia and cancer among patients with Barrett’s esophagus. Gastroenterology 142, 233–240 (2012).
Ramus, J. R., Gatenby, P. A., Caygill, C. P., Watson, A. & Winslet, M. C. The relationship between smoking and severe dysplastic disease in patients with Barrett’s columnar-lined oesophagus. Eur. J. Cancer Prev. 21, 507–510 (2012).
Kong, C. Y. et al. The impact of obesity on the rise in esophageal adenocarcinoma incidence: estimates from a disease simulation model. Cancer Epidemiol. Biomarkers Prev. 20, 2450–2456 (2011).
Kroep, S. et al. Comparing trends in esophageal adenocarcinoma incidence and lifestyle factors between the United States, Spain, and the Netherlands. Am. J. Gastroenterol. 109, 336–343 (2014).
Hoyo, C. et al. Body mass index in relation to oesophageal and oesophagogastric junction adenocarcinomas: a pooled analysis from the international BEACON consortium. Int. J. Epidemiol. 41, 1706–1718 (2012).
Steffen, A. et al. General and abdominal obesity and risk of esophageal and gastric adenocarcinoma in the European Prospective Investigation into Cancer and Nutrition. Int. J. Cancer 137, 646–657 (2015).
Lindkvist, B. et al. Metabolic risk factors for esophageal squamous cell carcinoma and adenocarcinoma: a prospective study of 580,000 subjects within the Me-Can project. BMC Cancer 14, 103 (2014).
El-Serag, H. The association between obesity and GERD: a review of the epidemiological evidence. Dig. Dis. Sci. 53, 2307–2312 (2008).
Thrift, A. P. et al. Obesity and risk of esophageal adenocarcinoma and Barrett’s esophagus: a Mendelian randomization study. J. Natl Cancer Inst. 106, dju252 (2014).
Coleman, H. G., Xie, S. H. & Lagergren, J. The epidemiology of esophageal adenocarcinoma. Gastroenterology 154, 390–405 (2018).
Cook, M. B., Freedman, N. D., Gamborg, M., Sorensen, T. I. & Baker, J. L. Childhood body mass index in relation to future risk of oesophageal adenocarcinoma. Br. J. Cancer 112, 601–607 (2015).
Levi, Z. et al. Body mass index and socioeconomic status measured in adolescence, country of origin, and the incidence of gastroesophageal adenocarcinoma in a cohort of 1 million men. Cancer 119, 4086–4093 (2013).
Singh, S. et al. Central adiposity is associated with increased risk of esophageal inflammation, metaplasia, and adenocarcinoma: a systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 11, 1399–1412 (2013).
Kubo, A. et al. Sex-specific associations between body mass index, waist circumference and the risk of Barrett’s oesophagus: a pooled analysis from the international BEACON consortium. Gut 62, 1684–1691 (2013).
El-Serag, H. B. et al. Visceral abdominal obesity measured by CT scan is associated with an increased risk of Barrett’s oesophagus: a case-control study. Gut 63, 220–229 (2014).
Kendall, B. J., Macdonald, G. A., Prins, J. B., O’Brien, S. & Whiteman, D. C. Total body fat and the risk of Barrett’s oesophagus – a bioelectrical impedance study. Cancer Epidemiol. 38, 266–272 (2014).
Thrift, A. P., Kramer, J. R., Alsarraj, A. & El-Serag, H. B. Fat mass by bioelectrical impedance analysis is not associated with increased risk of Barrett esophagus. J. Clin. Gastroenterol. 48, 218–223 (2014).
Kendall, B. J. et al. Inverse association between gluteofemoral obesity and risk of Barrett’s esophagus in a pooled analysis. Clin. Gastroenterol. Hepatol. 14, 1412–1419 (2016).
Lahmann, P. H., Pandeya, N., Webb, P. M., Green, A. C. & Whiteman, D. C. Body mass index, long-term weight change, and esophageal squamous cell carcinoma: is the inverse association modified by smoking status? Cancer 118, 1901–1909 (2012).
Freedman, N. D. et al. Alcohol intake and risk of oesophageal adenocarcinoma: a pooled analysis from the BEACON consortium. Gut 60, 1029–1037 (2011).
Thrift, A. P. et al. Alcohol and the risk of Barrett’s esophagus: a pooled analysis from the international BEACON consortium. Am. J. Gastroenterol. 109, 1586–1594 (2014).
Lou, Z., Xing, H. & Li, D. Alcohol consumption and the neoplastic progression in Barrett’s esophagus: a systematic review and meta-analysis. PLoS ONE 9, e105612 (2014).
World Cancer Research Fund/American Institute for Cancer Research. Continuous Update Project Report: Diet, Nutrition, Physical Activity and Oesophageal Cancer Project Expert Report (American Institute for Cancer Research, 2018).
Pandeya, N., Williams, G., Green, A. C., Webb, P. M. & Whiteman, D. C. Alcohol consumption and the risks of adenocarcinoma and squamous cell carcinoma of the esophagus. Gastroenterology 136, 1215–1224 (2009).
Crous-Bou, M., Jovani, M., De Vivo, I. & Jacobson, B. C. Gene-environment interactions and the risk of Barrett’s esophagus in three US cohorts. Am. J. Gastroenterol. 114, 893–899 (2019).
Yokoyama, T. et al. Alcohol flushing, alcohol and aldehyde dehydrogenase genotypes, and risk for esophageal squamous cell carcinoma in Japanese men. Cancer Epidemiol. Biomarkers Prev. 12, 1227–1233 (2003).
Wilson, L. F. et al. The impact of reducing alcohol consumption in Australia: an estimate of the proportion of potentially avoidable cancers 2013-2037. Int. J. Cancer 145, 2944–2953 (2019).
Steck, S. E. & Murphy, E. A. Dietary patterns and cancer risk. Nat. Rev. Cancer 20, 125–138 (2020).
Rezende, L. F. M. et al. Physical activity and cancer: an umbrella review of the literature including 22 major anatomical sites and 770 000 cancer cases. Br. J. Sports Med. 52, 826–833 (2018).
Ibiebele, T. I., Hughes, M. C., Whiteman, D. C. & Webb, P. M. Dietary patterns and risk of oesophageal cancers: a population-based case-control study. Br. J. Nutr. 107, 1207–1216 (2012).
Kubo, A. et al. Dietary patterns and the risk of Barrett’s esophagus. Am. J. Epidemiol. 167, 839–846 (2008).
Kubo, A. et al. Dietary antioxidants, fruits, and vegetables and the risk of Barrett’s esophagus. Am. J. Gastroenterol. 103, 1614–1623 (2008).
Li, W. Q. et al. Index-based dietary patterns and risk of esophageal and gastric cancer in a large cohort study. Clin. Gastroenterol. Hepatol. 11, 1130–1136 (2013).
Steevens, J., Schouten, L. J., Goldbohm, R. A. & van den Brandt, P. A. Vegetables and fruits consumption and risk of esophageal and gastric cancer subtypes in the Netherlands Cohort Study. Int. J. Cancer 129, 2681–2693 (2011).
Terry, P., Lagergren, J., Hansen, H., Wolk, A. & Nyren, O. Fruit and vegetable consumption in the prevention of oesophageal and cardia cancers. Eur. J. Cancer Prev. 10, 365–369 (2001).
Thompson, O. M., Beresford, S. A., Kirk, E. A. & Vaughan, T. L. Vegetable and fruit intakes and risk of Barrett’s esophagus in men and women. Am. J. Clin. Nutr. 89, 890–896 (2009).
Li, N. et al. Dietary sugar/starches intake and Barrett’s esophagus: a pooled analysis. Eur. J. Epidemiol. 32, 1007–1017 (2017).
Sun, L., Zhang, Z., Xu, J., Xu, G. & Liu, X. Dietary fiber intake reduces risk for Barrett’s esophagus and esophageal cancer. Crit. Rev. Food Sci. Nutr. 57, 2749–2757 (2017).
Sharp, L., Carsin, A. E., Cantwell, M. M., Anderson, L. A. & Murray, L. J. Intakes of dietary folate and other B vitamins are associated with risks of esophageal adenocarcinoma, Barrett’s esophagus, and reflux esophagitis. J. Nutr. 143, 1966–1973 (2013).
Keszei, A. P., Goldbohm, R. A., Schouten, L. J., Jakszyn, P. & van den Brandt, P. A. Dietary N-nitroso compounds, endogenous nitrosation, and the risk of esophageal and gastric cancer subtypes in the Netherlands Cohort Study. Am. J. Clin. Nutr. 97, 135–146 (2013).
Behrens, G. et al. The association between physical activity and gastroesophageal cancer: systematic review and meta-analysis. Eur. J. Epidemiol. 29, 151–170 (2014).
Singh, S., Devanna, S., Edakkanambeth Varayil, J., Murad, M. H. & Iyer, P. G. Physical activity is associated with reduced risk of esophageal cancer, particularly esophageal adenocarcinoma: a systematic review and meta-analysis. BMC Gastroenterol. 14, 101 (2014).
Petrick, J. L. et al. Association between circulating levels of sex steroid hormones and esophageal adenocarcinoma in the FINBAR study. PLoS ONE 13, e0190325 (2018).
Cook, M. B. et al. Association between circulating levels of sex steroid hormones and Barrett’s esophagus in men: a case-control analysis. Clin. Gastroenterol. Hepatol. 13, 673–682 (2015).
Cook, M. B. et al. Sex steroid hormones in relation to Barrett’s esophagus: an analysis of the FINBAR study. Andrology 5, 240–247 (2017).
Petrick, J. L. et al. Associations between prediagnostic concentrations of circulating sex steroid hormones and esophageal/gastric cardia adenocarcinoma among men. J. Natl Cancer Inst. 111, 34–41 (2019).
Petrick, J. L. & Cook, M. B. Do sex hormones underlie sex differences in cancer incidence? Testing the intuitive in esophageal adenocarcinoma. Am. J. Gastroenterol. 115, 211–213 (2020).
Xie, S. H. et al. Circulating sex hormone levels and risk of esophageal adenocarcinoma in a prospective study in men. Am. J. Gastroenterol. 115, 216–223 (2020).
Xie, S. H. et al. Association between levels of hormones and risk of esophageal adenocarcinoma and Barrett’s esophagus. Clin. Gastroenterol. Hepatol. 18, 2701–2709.e3 (2020).
Lagergren, K., Lagergren, J. & Brusselaers, N. Hormone replacement therapy and oral contraceptives and risk of oesophageal adenocarcinoma: a systematic review and meta-analysis. Int. J. Cancer 135, 2183–2190 (2014).
Cronin-Fenton, D. P. et al. Reproductive and sex hormonal factors and oesophageal and gastric junction adenocarcinoma: a pooled analysis. Eur. J. Cancer 46, 2067–2076 (2010).
Freedman, N. D. et al. The association of menstrual and reproductive factors with upper gastrointestinal tract cancers in the NIH-AARP cohort. Cancer 116, 1572–1581 (2010).
Bodelon, C. et al. Hormonal factors and risks of esophageal squamous cell carcinoma and adenocarcinoma in postmenopausal women. Cancer Prev. Res. 4, 840–850 (2011).
Lu, Y. & Lagergren, J. Reproductive factors and risk of oesophageal cancer, a population-based nested case-control study in Sweden. Br. J. Cancer 107, 564–569 (2012).
Parsonnet, J. et al. Helicobacter pylori infection and the risk of gastric carcinoma. N. Engl. J. Med. 325, 1127–1131 (1991).
Nie, S., Chen, T., Yang, X., Huai, P. & Lu, M. Association of Helicobacter pylori infection with esophageal adenocarcinoma and squamous cell carcinoma: a meta-analysis. Dis. Esophagus 27, 645–653 (2014).
Xie, F. J. et al. Helicobacter pylori infection and esophageal cancer risk: an updated meta-analysis. World J. Gastroenterol. 19, 6098–6107 (2013).
Wang, Z. et al. Helicobacter pylori infection is associated with reduced risk of Barrett’s esophagus: an analysis of the Barrett’s and Esophageal Adenocarcinoma Consortium. Am. J. Gastroenterol. 113, 1148–1155 (2018).
Eross, B. et al. Helicobacter pylori infection reduces the risk of Barrett’s esophagus: a meta-analysis and systematic review. Helicobacter 23, e12504 (2018).
Singh, S., Garg, S. K., Singh, P. P., Iyer, P. G. & El-Serag, H. B. Acid-suppressive medications and risk of oesophageal adenocarcinoma in patients with Barrett’s oesophagus: a systematic review and meta-analysis. Gut 63, 1229–1237 (2014).
Jankowski, J. A. Z. et al. Esomeprazole and aspirin in Barrett’s oesophagus (AspECT): a randomised factorial trial. Lancet 392, 400–408 (2018).
Tan, M. C., El-Serag, H. B., Yu, X. & Thrift, A. P. Acid suppression medications reduce risk of oesophageal adenocarcinoma in Barrett’s oesophagus: a nested case-control study in US male veterans. Aliment. Pharmacol. Ther. 48, 469–477 (2018).
Liao, L. M. et al. Nonsteroidal anti-inflammatory drug use reduces risk of adenocarcinomas of the esophagus and esophagogastric junction in a pooled analysis. Gastroenterology 142, 442–452 (2012).
Thrift, A. P. et al. Nonsteroidal anti-inflammatory drug use is not associated with reduced risk of Barrett’s esophagus. Am. J. Gastroenterol. 111, 1528–1535 (2016).
Vaughan, T. L. et al. Non-steroidal anti-inflammatory drugs and risk of neoplastic progression in Barrett’s oesophagus: a prospective study. Lancet Oncol. 6, 945–952 (2005).
Choi, S. E., Perzan, K. E., Tramontano, A. C., Kong, C. Y. & Hur, C. Statins and aspirin for chemoprevention in Barrett’s esophagus: results of a cost-effectiveness analysis. Cancer Prev. Res. 7, 341–350 (2014).
Sadeghi, S. et al. Aspirin, nonsteroidal anti-inflammatory drugs, and the risks of cancers of the esophagus. Cancer Epidemiol. Biomarkers Prev. 17, 1169–1178 (2008).
Farrow, D. C. et al. Use of aspirin and other nonsteroidal anti-inflammatory drugs and risk of esophageal and gastric cancer. Cancer Epidemiol. Biomarkers Prev. 7, 97–102 (1998).
Alexandre, L. et al. Statin use is associated with reduced risk of histologic subtypes of esophageal cancer: a nested case-control analysis. Gastroenterology 146, 661–668 (2014).
Beales, I. L., Vardi, I., Dearman, L. & Broughton, T. Statin use is associated with a reduction in the incidence of esophageal adenocarcinoma: a case control study. Dis. Esophagus 26, 838–846 (2013).
Nguyen, T., Khalaf, N., Ramsey, D. & El-Serag, H. B. Statin use is associated with a decreased risk of Barrett’s esophagus. Gastroenterology 147, 314–323 (2014).
Nguyen, T., Duan, Z., Naik, A. D., Kramer, J. R. & El-Serag, H. B. Statin use reduces risk of esophageal adenocarcinoma in US veterans with Barrett’s esophagus: a nested case-control study. Gastroenterology 149, 1392–1398 (2015).
Beales, I. L., Dearman, L., Vardi, I. & Loke, Y. Reduced risk of Barrett’s esophagus in statin users: case-control study and meta-analysis. Dig. Dis. Sci. 61, 238–246 (2016).
Singh, S., Singh, A. G., Singh, P. P., Murad, M. H. & Iyer, P. G. Statins are associated with reduced risk of esophageal cancer, particularly in patients with Barrett’s esophagus: a systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 11, 620–629 (2013).
Verbeek, R. E. et al. Familial clustering of Barrett’s esophagus and esophageal adenocarcinoma in a European cohort. Clin. Gastroenterol. Hepatol. 12, 1656–1663.e1 (2014).
Fecteau, R. E. et al. Association between germline mutation in VSIG10L and familial barrett neoplasia. JAMA Oncol. 2, 1333–1339 (2016).
Blaydon, D. C. et al. RHBDF2 mutations are associated with tylosis, a familial esophageal cancer syndrome. Am. J. Hum. Genet. 90, 340–346 (2012).
Becker, J. et al. Supportive evidence for FOXP1, BARX1, and FOXF1 as genetic risk loci for the development of esophageal adenocarcinoma. Cancer Med. 4, 1700–1704 (2015).
Palles, C. et al. Polymorphisms near TBX5 and GDF7 are associated with increased risk for Barrett’s esophagus. Gastroenterology 148, 367–378 (2015).
Levine, D. M. et al. A genome-wide association study identifies new susceptibility loci for esophageal adenocarcinoma and Barrett’s esophagus. Nat. Genet. 45, 1487–1493 (2013).
Gharahkhani, P. et al. Genome-wide association studies in oesophageal adenocarcinoma and Barrett’s oesophagus: a large-scale meta-analysis. Lancet Oncol. 17, 1363–1373 (2016).
Su, Z. et al. Common variants at the MHC locus and at chromosome 16q24.1 predispose to Barrett’s esophagus. Nat. Genet. 44, 1131–1136 (2012).
Lee, E. et al. Pleiotropic analysis of cancer risk loci on esophageal adenocarcinoma risk. Cancer Epidemiol. Biomarkers Prev. 24, 1801–1803 (2015).
Abnet, C. C. et al. A shared susceptibility locus in PLCE1 at 10q23 for gastric adenocarcinoma and esophageal squamous cell carcinoma. Nat. Genet. 42, 764–767 (2010).
Abnet, C. C. et al. Genotypic variants at 2q33 and risk of esophageal squamous cell carcinoma in China: a meta-analysis of genome-wide association studies. Hum. Mol. Genet. 21, 2132–2141 (2012).
Wang, L. D. et al. Genome-wide association study of esophageal squamous cell carcinoma in Chinese subjects identifies susceptibility loci at PLCE1 and C20orf54. Nat. Genet. 42, 759–763 (2010).
Wu, C. et al. Genome-wide association study identifies three new susceptibility loci for esophageal squamous-cell carcinoma in Chinese populations. Nat. Genet. 43, 679–684 (2011).
Wu, C. et al. Joint analysis of three genome-wide association studies of esophageal squamous cell carcinoma in Chinese populations. Nat. Genet. 46, 1001–1006 (2014).
Wu, C. et al. Genome-wide association analyses of esophageal squamous cell carcinoma in Chinese identify multiple susceptibility loci and gene-environment interactions. Nat. Genet. 44, 1090–1097 (2012).
Corley, D. A. et al. Impact of endoscopic surveillance on mortality from Barrett’s esophagus-associated esophageal adenocarcinomas. Gastroenterology 145, 312–319 (2013).
El-Serag, H. B. et al. Surveillance endoscopy is associated with improved outcomes of oesophageal adenocarcinoma detected in patients with Barrett’s oesophagus. Gut 65, 1252–1260 (2016).
Bhat, S. K. et al. Oesophageal adenocarcinoma and prior diagnosis of Barrett’s oesophagus: a population-based study. Gut 64, 20–25 (2015).
Wenker, T. N., Tan, M. C., Liu, Y., El-Serag, H. B. & Thrift, A. P. Prior diagnosis of Barrett’s esophagus is infrequent, but associated with improved esophageal adenocarcinoma survival. Dig. Dis. Sci. 63, 3112–3119 (2018).
Gupta, M. et al. Recurrence of esophageal intestinal metaplasia after endoscopic mucosal resection and radiofrequency ablation of Barrett’s esophagus: results from a US multicenter consortium. Gastroenterology 145, 79–86.e71 (2013).
Orman, E. S., Li, N. & Shaheen, N. J. Efficacy and durability of radiofrequency ablation for Barrett’s esophagus: systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 11, 1245–1255 (2013).
El-Serag, H. B. & Graham, D. Y. Routine polypectomy for colorectal polyps and ablation for Barrett’s esophagus are intellectually the same. Gastroenterology 140, 386–388 (2011).
Hur, C. et al. The cost effectiveness of radiofrequency ablation for Barrett’s esophagus. Gastroenterology 143, 567–575 (2012).
Gordon, L. G. et al. Cost-effectiveness of endoscopic surveillance of non-dysplastic Barrett’s esophagus. Gastrointest. Endosc. 79, 242–256.e6 (2014).
Ross-Innes, C. S. et al. Risk stratification of Barrett’s oesophagus using a non-endoscopic sampling method coupled with a biomarker panel: a cohort study. Lancet Gastroenterol. Hepatol. 2, 23–31 (2017).
Offman, J. et al. Barrett’s Oesophagus Trial 3 (BEST3): study protocol for a randomised controlled trial comparing the Cytosponge-TFF3 test with usual care to facilitate the diagnosis of oesophageal pre-cancer in primary care patients with chronic acid reflux. BMC Cancer 18, 784 (2018).
Thota, P. N. & Chak, A. Is mass screening for Barrett’s esophagus a myth or reality? Clin. Gastroenterol. Hepatol. 17, 610–612 (2019).
Kumar, S. et al. Mass spectrometric analysis of exhaled breath for the identification of volatile organic compound biomarkers in esophageal and gastric adenocarcinoma. Ann. Surg. 262, 981–990 (2015).
Fitzgerald, R. C. et al. Cytosponge-trefoil factor 3 versus usual care to identify Barrett’s oesophagus in a primary care setting: a multicentre, pragmatic, randomised controlled trial. Lancet 396, 333–344 (2020).
Sami, S. S. et al. Acceptability, accuracy, and safety of disposable transnasal capsule endoscopy for Barrett’s esophagus screening. Clin. Gastroenterol. Hepatol. 17, 638–646.e1 (2019).
Taylor, P. R., Abnet, C. C. & Dawsey, S. M. Squamous dysplasia–the precursor lesion for esophageal squamous cell carcinoma. Cancer Epidemiol. Biomarkers Prev. 22, 540–552 (2013).
Domper Arnal, M. J., Ferrández Arenas, Á. & Lanas Arbeloa, Á. Esophageal cancer: risk factors, screening and endoscopic treatment in Western and Eastern countries. World J. Gastroenterol. 21, 7933–7943 (2015).
Chen, Q. et al. Effectiveness evaluation of organized screening for esophageal cancer: a case-control study in Linzhou city, China. Sci. Rep. 6, 35707 (2016).
Rubenstein, J. H. et al. Prediction of Barrett’s esophagus among men. Am. J. Gastroenterol. 108, 353–362 (2013).
Thrift, A. P., Garcia, J. M. & El-Serag, H. B. A multibiomarker risk score helps predict risk for Barrett’s esophagus. Clin. Gastroenterol. Hepatol. 12, 1267–1271 (2014).
Dong, J. et al. Determining risk of Barrett’s esophagus and esophageal adenocarcinoma based on epidemiologic factors and genetic variants. Gastroenterology 154, 1273–1281 (2018).
Rubenstein, J. H. et al. Validation and comparison of tools for selecting individuals to screen for Barrett’s esophagus and early neoplasia. Gastroenterology 158, 2082–2092 (2020).
Thrift, A. P., Kendall, B. J., Pandeya, N., Vaughan, T. L. & Whiteman, D. C. A clinical risk prediction model for Barrett esophagus. Cancer Prev. Res. 5, 1115–1123 (2012).
Rubenstein, J. H. & Thrift, A. P. Risk factors and populations at risk: selection of patients for screening for Barrett’s oesophagus. Best Pract. Res. Clin. Gastroenterol. 29, 41–50 (2015).
Thrift, A. P., Kanwal, F. & El-Serag, H. B. Prediction models for gastrointestinal and liver diseases: too many developed, too few validated. Clin. Gastroenterol. Hepatol. 14, 1678–1680 (2016).
Thrift, A. P., Vaughan, T. L., Anderson, L. A., Whiteman, D. C. & El-Serag, H. B. External validation of the Michigan Barrett’s Esophagus Prediction Tool. Clin. Gastroenterol. Hepatol. 15, 1124–1126 (2017).
Ireland, C. J., Thrift, A. P. & Esterman, A. Risk prediction models for Barrett’s esophagus discriminate well and are generalizable in an external validation study. Dig. Dis. Sci. 65, 2992–2999 (2020).
Thrift, A. P., Kendall, B. J., Pandeya, N. & Whiteman, D. C. A model to determine absolute risk for esophageal adenocarcinoma. Clin. Gastroenterol. Hepatol. 11, 138–144 (2013).
Kunzmann, A. T. et al. Information on genetic variants does not increase identification of individuals at risk of esophageal adenocarcinoma compared to clinical risk factors. Gastroenterology 156, 43–45 (2019).
Kunzmann, A. T. et al. Model for identifying individuals at risk for esophageal adenocarcinoma. Clin. Gastroenterol. Hepatol. 16, 1229–1236 (2018).
Parasa, S. et al. Development and validation of a model to determine risk of progression of Barrett’s esophagus to neoplasia. Gastroenterology 154, 1282–1289 (2017).
Kunzmann, A. T. et al. External validation of a model to determine risk of progression of Barrett’s oesophagus to neoplasia. Aliment. Pharmacol. Ther. 49, 1274–1281 (2019).
Pohl, H., Sirovich, B. & Welch, H. G. Esophageal adenocarcinoma incidence: are we reaching the peak? Cancer Epidemiol. Biomarkers Prev. 19, 1468–1470 (2010).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Additional information
Peer review information
Nature Reviews Gastroenterology & Hepatology thanks G. Falk, S. Xie and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
BEACON: http://beacon.tlvnet.net
Rights and permissions
About this article
Cite this article
Thrift, A.P. Global burden and epidemiology of Barrett oesophagus and oesophageal cancer. Nat Rev Gastroenterol Hepatol 18, 432–443 (2021). https://doi.org/10.1038/s41575-021-00419-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41575-021-00419-3
This article is cited by
-
Correlation between sarcopenia and esophageal cancer: a narrative review
World Journal of Surgical Oncology (2024)
-
Domperidone inhibits cell proliferation via targeting MEK and CDK4 in esophageal squamous cell carcinoma
Cancer Cell International (2024)
-
Somatic mutations of esophageal adenocarcinoma: a comparison between Black and White patients
Scientific Reports (2024)
-
Concise Commentary: It’s All Downhill from Here—How Diagnostic and Therapeutic Advances May Decrease the Incidence Rates of Gastroesophageal Junction and Esophageal Adenocarcinoma
Digestive Diseases and Sciences (2024)
-
High NANOG expression correlates with worse patients’ survival in esophageal adenocarcinoma
BMC Cancer (2023)