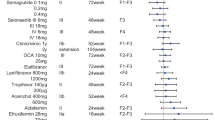
Abstract
Our understanding of nonalcoholic fatty liver disease pathophysiology continues to advance rapidly. Accordingly, the field has moved from describing the clinical phenotype through the presence of nonalcoholic steatohepatitis (NASH) and degree of fibrosis to deep phenotyping with a description of associated comorbidities, genetic polymorphisms and environmental influences that could be associated with disease progression. These insights have fuelled a robust therapeutic pipeline across a variety of new targets to resolve steatohepatitis or reverse fibrosis, or both. Additionally, some of these therapies have beneficial effects that extend beyond the liver, such as effects on glycaemic control, lipid profile and weight loss. In addition, emerging therapies for NASH cirrhosis would have to demonstrate either reversal of fibrosis with associated reduction in portal hypertension or at least delay the progression with eventual decrease in liver-related outcomes. For non-cirrhotic NASH, it is the expectation that reversal of fibrosis by one stage or resolution of NASH with no worsening in fibrosis will need to be accompanied by overall survival benefits. In this Review, we summarize NASH therapies that have progressed to phase II and beyond. We also discuss some of the potential clinical challenges with the use of these new therapies when approved.
Key points
-
There are several novel treatments currently under development for treatment of non-cirrhotic and cirrhotic nonalcoholic steatohepatitis.
-
Resolution of steatohepatitis and reversal of fibrosis are two important histological end points in the ongoing clinical trials.
-
Emerging therapies are beginning to differentiate through extra-hepatic benefits such as improvement in glycaemic control, lipid profile and weight loss.
-
Safety and tolerability of the emerging therapies will determine compliance that would result in sustained benefits and, ultimately, improvement in long-term survival.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Chalasani, N. et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology 67, 328–357 (2018).
Kleiner, D. E. et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 41, 1313–1321 (2005).
Shirazi, F., Wang, J. & Wong, R. J. Nonalcoholic steatohepatitis becomes the leading indication for liver transplant registrants among US adults born between 1945 and 1965. J. Clin. Exp. Hepatol. 10, 30–36 (2020).
Younossi, Z. M. Non-alcoholic fatty liver disease – a global public health perspective. J. Hepatol. 70, 531–544 (2019).
Shingina, A. et al. Future trends in demand for liver transplant: birth cohort effects among patients with NASH and HCC. Transplantation 103, 140–148 (2019).
Parrish, N. F. et al. The changing face of liver transplantation in the United States: the effect of HCV antiviral eras on transplantation trends and outcomes. Transpl. Direct 5, e427 (2019).
Haldar, D. et al. Outcomes of liver transplantation for non-alcoholic steatohepatitis: a European Liver Transplant Registry study. J. Hepatol. 71, 313–322 (2019).
Calzadilla-Bertot, L. et al. Increasing incidence of nonalcoholic steatohepatitis as an indication for liver transplantation in Australia and New Zealand. Liver Transpl. 25, 25–34 (2019).
Noureddin, M. et al. NASH leading cause of liver transplant in women: updated analysis of indications for liver transplant and ethnic and gender variances. Am. J. Gastroenterol. 113, 1649–1659 (2018).
Holmer, M. et al. Nonalcoholic fatty liver disease is an increasing indication for liver transplantation in the Nordic countries. Liver Int. 38, 2082–2090 (2018).
Parthasarathy, G., Revelo, X. & Malhi, H. Pathogenesis of nonalcoholic steatohepatitis: an overview. Hepatol. Commun. 4, 478–492 (2020).
Noureddin, M. & Sanyal, A. J. Pathogenesis of NASH: the impact of multiple pathways. Curr. Hepatol. Rep. 17, 350–360 (2018).
Friedman, S. L., Neuschwander-Tetri, B. A., Rinella, M. & Sanyal, A. J. Mechanisms of NAFLD development and therapeutic strategies. Nat. Med. 24, 908–922 (2018).
Chalasani, N. et al. Genome-wide association study identifies variants associated with histologic features of nonalcoholic fatty liver disease. Gastroenterology 139, 1567–1576.E6 (2010).
Eslam, M., Valenti, L. & Romeo, S. Genetics and epigenetics of NAFLD and NASH: Clinical impact. J. Hepatol. 68, 268–279 (2018).
Unalp-Arida, A. & Ruhl, C. E. PNPLA3 I148M and liver fat and fibrosis scores predict liver disease mortality in the United States population. Hepatology 71, 820–834 (2020).
Trepo, E. & Valenti, L. Update on NAFLD genetics: from new variants to the clinic. J. Hepatol. 72, 1196–1209 (2020).
Singh, S. et al. Fibrosis progression in nonalcoholic fatty liver vs nonalcoholic steatohepatitis: a systematic review and meta-analysis of paired-biopsy studies. Clin. Gastroenterol. Hepatol. 13, 643–654.E9 (2015).
Kleiner, D. E. et al. Association of histologic disease activity with progression of nonalcoholic fatty liver disease. JAMA Netw. Open 2, e1912565 (2019).
Sanyal, A. J. et al. The natural history of advanced fibrosis due to nonalcoholic steatohepatitis: data from the simtuzumab trials. Hepatology 70, 1913–1927 (2019).
Dulai, P. S. et al. Increased risk of mortality by fibrosis stage in nonalcoholic fatty liver disease: systematic review and meta-analysis. Hepatology 65, 1557–1565 (2017).
Vilar-Gomez, E. et al. Fibrosis severity as a determinant of cause-specific mortality in patients with advanced nonalcoholic fatty liver disease: a multi-national cohort study. Gastroenterology 155, 443–457.e17 (2018).
Ekstedt, M. et al. Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology 61, 1547–1554 (2015).
Angulo, P. et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology 149, 389–397.e10 (2015).
Harrison, S. A. et al. Selonsertib for patients with bridging fibrosis or compensated cirrhosis due to NASH: results from randomized phase III STELLAR trials. J. Hepatol. 73, 26–39 (2020).
Harrison, S. A. et al. Simtuzumab is ineffective for patients with bridging fibrosis or compensated cirrhosis caused by nonalcoholic steatohepatitis. Gastroenterology 155, 1140–1153 (2018).
Neuschwander-Tetri, B. A. et al. Clinical, laboratory and histological associations in adults with nonalcoholic fatty liver disease. Hepatology 52, 913–924 (2010).
Vuppalanchi, R. & Chalasani, N. Nonalcoholic fatty liver disease and nonalcoholic steatohepatitis: selected practical issues in their evaluation and management. Hepatology 49, 306–317 (2009).
Shamseddeen, H. et al. Spontaneous fluctuations in liver biochemistries in patients with compensated NASH cirrhosis: implications for drug hepatotoxicity monitoring. Drug Saf. 43, 281–290 (2020).
Ajmera, V. H. et al. Clinical utility of an increase in magnetic resonance elastography in predicting fibrosis progression in NAFLD. Hepatology 71, 849–860 (2020).
Vuppalanchi, R. et al. Performance characteristics of vibration-controlled transient elastography for evaluation of nonalcoholic fatty liver disease. Hepatology 67, 134–144 (2018).
Younossi, Z. M. et al. Diagnostic modalities for nonalcoholic fatty liver disease, nonalcoholic steatohepatitis, and associated fibrosis. Hepatology 68, 349–360 (2018).
Siddiqui, M. S. et al. Case definitions for inclusion and analysis of endpoints in clinical trials for nonalcoholic steatohepatitis through the lens of regulatory science. Hepatology 67, 2001–2012 (2018).
Rinella, M. E., Tacke, F., Sanyal, A. J., Anstee, Q. M. & participants of the AASLD/EASL Workshop. Report on the AASLD/EASL joint workshop on clinical trial endpoints in NAFLD. Hepatology 70, 1424–1436 (2019).
US Food and Drug Administration. Noncirrhotic nonalcholic steatohepatitis with liver fibrosis: developing drugs for treatment – guidance for industry (FDA, 2018).
US Food and Drug Administration. Noncirrhotic nonalcholic steatohepatitis with compensated cirrhosis: developing drugs for treatment – guidance for industry (FDA, 2019).
Siemens Healthineers. FDA grants breakthrough device designation to Siemens Healthineers Enhanced Liver Fibrosis (ELF™) Test. Siemens https://www.siemens-healthineers.com/en-us/press-room/press-releases/elftest.html (2018).
Garcia-Tsao, G. et al. Randomized placebo-controlled trial of emricasan in non-alcoholic steatohepatitis (NASH) cirrhosis with severe portal hypertension. J. Hepatol. 72, 885–895 (2020).
Garcia-Tsao, G. et al. Emricasan (IDN-6556) lowers portal pressure in patients with compensated cirrhosis and severe portal hypertension. Hepatology 69, 717–728 (2019).
Harrison, S. A. et al. A blood-based biomarker panel (NIS4) for non-invasive diagnosis of non-alcoholic steatohepatitis and liver fibrosis: a prospective derivation and global validation study. Lancet Gastroenterol. Hepatol. 5, 970–985 (2020).
Eslam, M., Sanyal, A. J., George, J. & International Consensus Panel. MAFLD: A consensus-driven proposed nomenclature for metabolic associated fatty liver disease. Gastroenterology 158, 1999–2014.e1 (2020).
World Health Organization. Tenfold increase in childhood and adolescent obesity in four decades: new study by Imperial College London and WHO (WHO, 2017).
Younossi, Z. M. et al. Epidemiology of chronic liver diseases in the USA in the past three decades. Gut 69, 564–568 (2020).
Cusi, K. et al. Long-term pioglitazone treatment for patients with nonalcoholic steatohepatitis and prediabetes or type 2 diabetes mellitus: a randomized trial. Ann. Intern. Med. 165, 305–315 (2016).
Sanyal, A. J. et al. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N. Engl. J. Med. 362, 1675–1685 (2010).
Keli, S. O., Hertog, M. G., Feskens, E. J. & Kromhout, D. Dietary flavonoids, antioxidant vitamins, and incidence of stroke: the Zutphen study. Arch. Intern. Med. 156, 637–642 (1996).
Kubota, Y. et al. Dietary intakes of antioxidant vitamins and mortality from cardiovascular disease: the Japan Collaborative Cohort Study (JACC) study. Stroke 42, 1665–1672 (2011).
Cheng, P. et al. Vitamin E intake and risk of stroke: a meta-analysis. Br. J. Nutr. 120, 1181–1188 (2018).
Klein, E. A. et al. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 306, 1549–1556 (2011).
Kim, Y. et al. Relation of vitamin E and selenium exposure to prostate cancer risk by smoking status: a review and meta-analysis. Anticancer. Res. 35, 4983–4996 (2015).
Lippman, S. M. et al. Effect of selenium and vitamin E on risk of prostate cancer and other cancers: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 301, 39–51 (2009).
Hampp, C. & Pippins, J. Pioglitazone and bladder cancer: FDA’s assessment. Pharmacoepidemiol. Drug Saf. 26, 117–118 (2017).
Mehtala, J. et al. Pioglitazone use and risk of bladder cancer: a systematic literature review and meta-analysis of observational studies. Diabetol. Int. 10, 24–36 (2019).
Tang, H. et al. Pioglitazone and bladder cancer risk: a systematic review and meta-analysis. Cancer Med. 7, 1070–1080 (2018).
Ali, A. H., Carey, E. J. & Lindor, K. D. Recent advances in the development of farnesoid X receptor agonists. Ann. Transl. Med. 3, 5 (2015).
Carr, R. M. & Reid, A. E. FXR agonists as therapeutic agents for non-alcoholic fatty liver disease. Curr. Atheroscler. Rep. 17, 16 (2015).
Neuschwander-Tetri, B. A. et al. Farnesoid X nuclear receptor ligand obeticholic acid for non-cirrhotic, non-alcoholic steatohepatitis (FLINT): a multicentre, randomised, placebo-controlled trial. Lancet 385, 956–965 (2015).
Younossi, Z. et al. Positive results from REGENERATE: a phase 3 international, randomized, placebo-controlled study evaluating obeticholic acid treatment for NASH [abstract GS-06]. J. Hepatol. 70, E5 (2019).
Younossi, Z. M. et al. Obeticholic acid for the treatment of non-alcoholic steatohepatitis: interim analysis from a multicentre, randomised, placebo-controlled phase 3 trial. Lancet 394, 2184–2196 (2019).
Siddiqui, M. S. et al. Impact of obeticholic acid on the lipoprotein profile in patients with non-alcoholic steatohepatitis. J. Hepatol. 72, 25–33 (2020).
Al-Dury, S. et al. Obeticholic acid may increase the risk of gallstone formation in susceptible patients. J. Hepatol. 71, 986–991 (2019).
Ratziu, V. et al. REGENERATE: design of a pivotal, randomised, phase 3 study evaluating the safety and efficacy of obeticholic acid in patients with fibrosis due to nonalcoholic steatohepatitis. Contemp. Clin. Trials 84, 105803 (2019).
Intercept Pharmaceuticals. Intercept receives complete response letter from FDA for obeticholic acid for the treatment of fibrosis due to NASH. Intercept https://ir.interceptpharma.com/news-releases/news-release-details/intercept-receives-complete-response-letter-fda-obeticholic-acid (2020).
Alemi, F. et al. The TGR5 receptor mediates bile acid-induced itch and analgesia. J. Clin. Invest. 123, 1513–1530 (2013).
Macdonald, G. A. & Prins, J. B. Peroxisomal fatty acid metabolism, peroxisomal proliferator-activated receptors and non-alcoholic fatty liver disease. J. Gastroenterol. Hepatol. 19, 1335–1337 (2004).
Staels, B. et al. Hepatoprotective effects of the dual peroxisome proliferator-activated receptor alpha/delta agonist, GFT505, in rodent models of nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. Hepatology 58, 1941–1952 (2013).
Grygiel-Górniak, B. Peroxisome proliferator-activated receptors and their ligands: nutritional and clinical implications – a review. Nutr. J. 13, 17 (2014).
Rotman, Y. & Sanyal, A. J. Current and upcoming pharmacotherapy for non-alcoholic fatty liver disease. Gut 66, 180–190 (2017).
Chen, J., Montagner, A., Tan, N. S. & Wahli, W. Insights into the role of PPARβ/δ in NAFLD. Int. J. Mol. Sci. 19, 1893 (2018).
Regnier, M. et al. Insights into the role of hepatocyte PPARα activity in response to fasting. Mol. Cell Endocrinol. 471, 75–88 (2018).
Odegaard, J. I. et al. Alternative M2 activation of Kupffer cells by PPARδ ameliorates obesity-induced insulin resistance. Cell Metab. 7, 496–507 (2008).
Cariou, B. et al. Dual peroxisome proliferator-activated receptor α/δ agonist GFT505 improves hepatic and peripheral insulin sensitivity in abdominally obese subjects. Diabetes Care 36, 2923–2930 (2013).
Ratziu, V. et al. Elafibranor, an agonist of the peroxisome proliferator-activated receptor-α and -δ, induces resolution of nonalcoholic steatohepatitis without fibrosis worsening. Gastroenterology 150, 1147–1159.e5 (2016).
Erstad, D. J. et al. Molecular magnetic resonance imaging accurately measures the antifibrotic effect of EDP-305, a novel farnesoid X receptor agonist. Hepatol. Commun. 2, 821–835 (2018).
GENFIT. GENFIT: announces results from interim analysis of RESOLVE-IT phase 3 trial of elafibranor in adults with NASH and fibrosis. GENFIT https://ir.genfit.com/news-releases/news-release-details/genfit-announces-results-interim-analysis-resolve-it-phase-3 (2020).
Harrison S. A. et al. RESOLVE-IT® phase 3 trial of elafibranor in NASH: final results of the week 72 interim surrogate efficacy analysis (Poster). Hepatology 72 (2020).
Taylor, N. P. Genfit cans phase 3 NASH trial after failing interim analysis. Fierce Biotech https://www.fiercebiotech.com/biotech/genfit-cans-phase-3-nash-trial-after-failing-interim-analysis (2020).
Sinha, R. A., Bruinstroop, E., Singh, B. K. & Yen, P. M. Nonalcoholic fatty liver disease and hypercholesterolemia: roles of thyroid hormones, metabolites, and agonists. Thyroid 29, 1173–1191 (2019).
Sinha, R. A., Singh, B. K. & Yen, P. M. Direct effects of thyroid hormones on hepatic lipid metabolism. Nat. Rev. Endocrinol. 14, 259–269 (2018).
Sinha, R. A. et al. Thyroid hormone stimulates hepatic lipid catabolism via activation of autophagy. J. Clin. Invest. 122, 2428–2438 (2012).
Pagadala, M. R. et al. Prevalence of hypothyroidism in nonalcoholic fatty liver disease. Dig. Dis. Sci. 57, 528–534 (2012).
Liangpunsakul, S. & Chalasani, N. Is hypothyroidism a risk factor for non-alcoholic steatohepatitis? J. Clin. Gastroenterol. 37, 340–343 (2003).
Kim, D. et al. Subclinical hypothyroidism and low-normal thyroid function are associated with nonalcoholic steatohepatitis and fibrosis. Clin. Gastroenterol. Hepatol. 16, 123–131.e1 (2018).
Kelly, M. J. et al. Discovery of 2-[3,5-dichloro-4-(5-isopropyl-6-oxo-1,6-dihydropyridazin-3-yloxy)phenyl]-3,5-dioxo-2,3,4,5-tetrahydro[1,2,4]triazine-6-carbonitrile (MGL-3196), a highly selective thyroid hormone receptor beta agonist in clinical trials for the treatment of dyslipidemia. J. Med. Chem. 57, 3912–3923 (2014).
Loomba, R. et al. VK2809, a novel liver-directed thyroid receptor beta agonist, significantly reduces liver fat in patients with non-alcoholic fatty liver disease: a phase 2 randomized, placebo-controlled trial [abstract LB-4]. Hepatology 68, 1448A (2018).
Loomba, R. et al. VK2809, a novel liver-directed thyroid receptor beta agonist, significantly reduces liver fat with both low and high doses in patients with non-alcoholic fatty liver disease: a phase 2 randomized, placebo-controlled trial [abstract]. J. Hepatol. 70, E150–E151 (2019).
Harrison, S. A. et al. Resmetirom (MGL-3196) for the treatment of non-alcoholic steatohepatitis: a multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 394, 2012–2024 (2019).
Madrigal Pharmaceuticals. Madrigal Pharmaceuticals announces first patient dosed in MAESTRO-NAFLD-1, a second phase 3 multi-center, double-blind, randomized, placebo-controlled study of resmetirom (MGL-3196) in patients with non-alcoholic steatohepatitis (NASH) and presumed NASH (NASH/NAFLD (non-alcoholic fatty liver disease)). Madrigal Pharmaceuticals https://ir.madrigalpharma.com/news-releases/news-release-details/madrigal-pharmaceuticals-announces-first-patient-dosed-maestro (2019).
Safadi, R. et al. The fatty acid-bile acid conjugate Aramchol reduces liver fat content in patients with nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 12, 2085–2091.e1 (2014).
Ratziu, V. et al. One-year results of the global phase 2b randomized placebo-controlled ARREST trial of aramchol, a stearoyl CoA desaturase inhibitor, in patients with NASH [abstract]. Hepatology 68, LB-5 (2018).
Lefebvre, E. et al. Antifibrotic effects of the dual CCR2/CCR5 antagonist cenicriviroc in animal models of liver and kidney fibrosis. PLoS ONE 11, e0158156 (2016).
Lawitz, E. et al. A combination of the ACC inhibitor GS-0976 and the nonsteroidal FXR agonist GS-9674 improves hepatic steatosis, biochemistry, and stiffness in patients with non-alcoholic steatohepatitis [abstract]. J. Hepatol. 70, E794 (2019).
Chen, W., Zhang, J., Fan, H. N. & Zhu, J. S. Function and therapeutic advances of chemokine and its receptor in nonalcoholic fatty liver disease. Ther. Adv. Gastroenterol. 11, 1756284818815184 (2018).
Miura, K., Yang, L., van Rooijen, N., Ohnishi, H. & Seki, E. Hepatic recruitment of macrophages promotes nonalcoholic steatohepatitis through CCR2. Am. J. Physiol. Gastrointest. Liver Physiol. 302, G1310–G1321 (2012).
Friedman, S. L. et al. A randomized, placebo-controlled trial of cenicriviroc for treatment of nonalcoholic steatohepatitis with fibrosis. Hepatology 67, 1754–1767 (2018).
Ratziu, V. et al. Cenicriviroc treatment for adults with nonalcoholic steatohepatitis and fibrosis: final analysis of the phase 2b CENTAUR study. Hepatology 72, 892–905 (2020).
Harrison, S. A. et al. NGM282 for treatment of non-alcoholic steatohepatitis: a multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 391, 1174–1185 (2018).
Harrison, S. A. et al. Efficacy and safety of aldafermin, an engineered FGF19 analog, in a randomized, double-blind, placebo-controlled trial of patients with nonalcoholic steatohepatitis. Gastroenterology 160, 219–231.e1 (2020).
Harrison, S. A. et al. NGM282 improves liver fibrosis and histology in 12 weeks in patients with nonalcoholic steatohepatitis. Hepatology 71, 1198–1212 (2020).
Harrison, S. A. et al. Insulin sensitizer MSDC-0602K in non-alcoholic steatohepatitis: a randomized, double-blind, placebo-controlled phase IIb study. J. Hepatol. 72, 613–626 (2020).
Potts, J. E. et al. The effect of glucagon-like peptide 1 receptor agonists on weight loss in type 2 diabetes: a systematic review and mixed treatment comparison meta-analysis. PLoS ONE 10, e0126769 (2015).
Htike, Z. Z. et al. Efficacy and safety of glucagon-like peptide-1 receptor agonists in type 2 diabetes: a systematic review and mixed-treatment comparison analysis. Diabetes Obes. Metab. 19, 524–536 (2017).
Armstrong, M. J. et al. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): a multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet 387, 679–690 (2016).
Petit, J. M. et al. Effect of liraglutide therapy on liver fat content in patients with inadequately controlled type 2 diabetes: the Lira-NAFLD study. J. Clin. Endocrinol. Metab. 102, 407–415 (2017).
Boyle, J. G., Livingstone, R. & Petrie, J. R. Cardiovascular benefits of GLP-1 agonists in type 2 diabetes: a comparative review. Clin. Sci. 132, 1699–1709 (2018).
Brown, J. M. & Everett, B. M. Cardioprotective diabetes drugs: what cardiologists need to know. Cardiovasc. Endocrinol. Metab. 8, 96–105 (2019).
Marso, S. P. et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N. Engl. J. Med. 375, 311–322 (2016).
Marso, S. P. et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N. Engl. J. Med. 375, 1834–1844 (2016).
Clegg, L. E. et al. Effects of exenatide and open-label SGLT2 inhibitor treatment, given in parallel or sequentially, on mortality and cardiovascular and renal outcomes in type 2 diabetes: insights from the EXSCEL trial. Cardiovasc. Diabetol. 18, 138 (2019).
Husain, M. et al. Semaglutide (SUSTAIN and PIONEER) reduces cardiovascular events in type 2 diabetes across varying cardiovascular risk. Diabetes Obes. Metab. 22, 442–451 (2020).
Newsome, P. N. et al. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N. Engl. J. Med. https://doi.org/10.1056/NEJMoa2028395 (2020).
Harrison, S. A. et al. Semaglutide for the treatment of non-alcoholic steatohepatitis: trial design and comparison of non-invasive biomarkers. Contemp. Clin. Trials 97, 106174 (2020).
Flint, A. et al. Semaglutide treatment in subjects with NAFLD: effects assessed by magnetic resonance elastography and magnetic resonance imaging proton density fat fraction [abstract]. Hepatology 72, 1036 (2020).
Ahren, B. et al. Efficacy and safety of once-weekly semaglutide versus once-daily sitagliptin as an add-on to metformin, thiazolidinediones, or both, in patients with type 2 diabetes (SUSTAIN 2): a 56-week, double-blind, phase 3a, randomised trial. Lancet Diabetes Endocrinol. 5, 341–354 (2017).
Warren, M. et al. Semaglutide as a therapeutic option for elderly patients with type 2 diabetes: pooled analysis of the SUSTAIN 1-5 trials. Diabetes Obes. Metab. 20, 2291–2297 (2018).
Frias, J. P. et al. Efficacy and tolerability of tirzepatide, a dual glucose-dependent insulinotropic peptide and glucagon-like peptide-1 receptor agonist in patients with type 2 diabetes: a 12-week, randomized, double-blind, placebo-controlled study to evaluate different dose-escalation regimens. Diabetes Obes. Metab. 22, 938–946 (2020).
Coskun, T. et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: from discovery to clinical proof of concept. Mol. Metab. 18, 3–14 (2018).
Frias, J. P. et al. Efficacy and safety of LY3298176, a novel dual GIP and GLP-1 receptor agonist, in patients with type 2 diabetes: a randomised, placebo-controlled and active comparator-controlled phase 2 trial. Lancet 392, 2180–2193 (2018).
Hartman, M. L. et al. Effects of novel dual GIP and GLP-1 receptor agonist tirzepatide on biomarkers of nonalcoholic steatohepatitis in patients with type 2 diabetes. Diabetes Care 43, 1352–1355 (2020).
Chalasani, N. et al. Effects of belapectin, an inhibitor of galectin-3, in patients with nonalcoholic steatohepatitis with cirrhosis and portal hypertension. Gastroenterology 158, 1334–1345.e5 (2019).
Harrison, S. A. et al. Randomised clinical study: GR-MD-02, a galectin-3 inhibitor, vs. placebo in patients having non-alcoholic steatohepatitis with advanced fibrosis. Aliment. Pharmacol. Ther. 44, 1183–1198 (2016).
Frenette, C. T. et al. Emricasan (IDN-6556) orally for 6 months in patients with cirrhosis and elevated MELD score improves liver function [abstract]. Hepatology 64, 1042A (2016).
Garcia-Tsao, G. et al. Randomized placebo-controlled trial of emricasan for non-alcoholic steatohepatitis-related cirrhosis with severe portal hypertension. J. Hepatol. 72, 885–895 (2020).
Harrison, S. A. et al. A randomized, placebo-controlled trial of emricasan in patients with NASH and F1-F3 fibrosis. J. Hepatol. 72, 816–827 (2020).
Mehta, G. et al. A placebo-controlled, multicenter, double-blind, phase 2 randomized trial of the pan-caspase inhibitor emricasan in patients with acutely decompensated cirrhosis. J. Clin. Exp. Hepatol. 8, 224–234 (2018).
Shiffman, M. et al. Randomised clinical trial: emricasan versus placebo significantly decreases ALT and caspase 3/7 activation in subjects with non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 49, 64–73 (2019).
Trauner, M. et al. The nonsteroidal farnesoid X receptor agonist cilofexor (GS-9674) improves markers of cholestasis and liver injury in patients with primary sclerosing cholangitis. Hepatology 70, 788–801 (2019).
Patel, K. et al. Cilofexor, a nonsteroidal FXR agonist, in patients with noncirrhotic NASH: a phase 2 randomized controlled trial. Hepatology 72, 58–71 (2020).
Badman, M. K. et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of the novel non-bile acid FXR agonist tropifexor (LJN452) in healthy volunteers. Clin. Pharmacol. Drug Dev. 9, 395–410 (2020).
Tully, D. C. et al. Discovery of tropifexor (LJN452), a highly potent non-bile acid FXR agonist for the treatment of cholestatic liver diseases and nonalcoholic steatohepatitis (NASH). J. Med. Chem. 60, 9960–9973 (2017).
Sanyal, A. et al. Tropifexor, a farnesoid X receptor agonist for the treatment of non-alcoholic steatohepatitis: interim results based on baseline body mass index from first two parts of phase 2b study FLIGHT-FXR [abstract]. J. Hepatol. 70, E796–E797 (2019).
Lucas, K. J. et al. Tropifexor, a highly potent FXR agonist, produces robust and dose-dependent reductions in hepatic fat and serum alanine aminotransferase in patients with fibrotic nash after 12 weeks of therapy: FLIGHT-FXR part C interim results [abstract L04]. Hepatology 70, 1479A–1480A (2019).
Lucas, K. J. et al. Safety and efficacy of tropifexor in patients with fibrotic nonalcoholic steatohepatitis: 48-week results from part C of the phase 2 FLIGHT-FXR study [abstract 139]. Hepatology 72, 101A–102A (2020).
Ratziu, V. et al. EDP-305, a non-bile acid farnesoid X receptor (FXR) agonist, showed statistically significant improvements in liver biochemistry and hepatic steatosis in the phase 2a ARGON-1 study [abstract AS078]. J. Hepatol. 73, S56–S57 (2020).
ENANTA Pharmaceuticals. ENANTA announces positive results of ARGON-1 study of its lead FXR agonist, EDP-305, for the treatment of NASH. ENANTA Pharmaceuticals https://www.enanta.com/investors/news-releases/press-release/2019/Enanta-Announces-Positive-Results-of-ARGON-1-Study-of-its-lead-FXR-Agonist-EDP-305-for-the-Treatment-of-NASH/default.aspx (2019).
Metacrine. Metacrine demonstrates best-in-class FXR drug program with positive clinical results in NASH patients. Metacrine https://www.metacrine.com/metacrine-demonstrates-best-in-class-fxr-drug-program-with-positive-clinical-results-in-nash-patients/ (2020).
Lawitz E, B. M. et al. MET409, an optimized farnesoid X receptor agonist, decreased liver fat and improved liver enzymes in patients with nonalcoholic steatohepatitis: a 12-week, randomized, placebocontrolled study [abstract LB16]. J. Hepatol. 73, S132 (2020).
Loomba, R. et al. GS-0976 reduces hepatic steatosis and fibrosis markers in patients with nonalcoholic fatty liver disease. Gastroenterology 155, 1463–1473.e6 (2018).
Bergman, A. et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of a liver-targeting acetyl-CoA carboxylase inhibitor (PF-05221304): a three-part randomized phase 1 study. Clin. Pharmacol. Drug Dev. 9, 514–526 (2020).
Huard, K. et al. Optimizing the benefit/risk of acetyl-CoA carboxylase (ACC) inhibitors through liver targeting. J. Med. Chem. 63, 10879–10896 (2020).
Amin, N. et al. PF-05221304 (PF’1304), a liver-targeted acetyl-CoA carboxylase inhibitor (ACCI), in adults with nonalcoholic fatty liver disease (NAFLD) demonstrates robust reductions in liver fat and alt - phase 2a, dose-ranging study [abstract]. Hepatology 70, 21A–22A (2019).
Jain, M. R. et al. Dual PPARα/γ agonist saroglitazar improves liver histopathology and biochemistry in experimental NASH models. Liver Int. 38, 1084–1094 (2018).
Kaul, U. et al. New dual peroxisome proliferator activated receptor agonist–Saroglitazar in diabetic dyslipidemia and non-alcoholic fatty liver disease: integrated analysis of the real world evidence. Cardiovasc. Diabetol. 18, 80 (2019).
Gawrieh, S. et al. A phase 2, prospective, multicenter, double-blind, randomized study of saroglitazar magnesium 1 mg, 2 mg or 4 mg versus placebo in patients with nonalcoholic fatty liver disease and/or nonalcoholic steatohepatitis (EVIDENCES IV) [abstract L010]. Hepatology 70, 1484A–1485A (2019).
Boubia, B. et al. Design, synthesis, and evaluation of a novel series of indole sulfonamide peroxisome proliferator activated receptor (PPAR) α/γ/δ triple activators: discovery of lanifibranor, a new antifibrotic clinical candidate. J. Med. Chem. 61, 2246–2265 (2018).
Inventiva. Inventiva’s lanifibranor meets the primary and key secondary endpoints in the Phase IIb NATIVE clinical trial in non-alcoholic steatohepatitis (NASH). Inventiva https://inventivapharma.com/inventivas-lanifibranor-meets-the-primary-and-key-secondary-endpoints-in-the-phase-iib-native-clinical-trial-in-non-alcoholic-steatohepatitis-nash/ (2020).
Francque, S. M. et al. The pan-PPAR agonist lanifibranor induces both resolution of NASH and regression of fibrosis after 24 weeks of treatment in non-cirrhotic NASH: results of the NATIVE phase 2b trial [Abstract]. Hepatology 72 (Suppl. S1), 9A (2020).
Charles, E. D. et al. Pegbelfermin (BMS-986036), PEGylated FGF21, in patients with obesity and type 2 diabetes: results from a randomized phase 2 study. Obesity 27, 41–49 (2019).
Verzijl, C. R. C., Van de Peppel, I. P., Struik, D. & Jonker, J. W. Pegbelfermin (BMS-986036): an investigational PEGylated fibroblast growth factor 21 analogue for the treatment of nonalcoholic steatohepatitis. Expert Opin. Investig. Drugs 29, 125–133 (2020).
Sanyal, A. et al. Pegbelfermin (BMS-986036), a PEGylated fibroblast growth factor 21 analogue, in patients with non-alcoholic steatohepatitis: a randomised, double-blind, placebo-controlled, phase 2a trial. Lancet 392, 2705–2717 (2019).
Harrison, S. A. et al. Efruxifermin (EFX), a long-acting Fc-FGF21 fusion protein, administered for 16 weeks to patients with NASH substantially reduces liver fat and ALT, and improves liver histology: analysis of a randomized, placebo-controlled, phase 2a study (balanced) [abstract]. Hepatology 72, 6A–7A (2020).
Corey, K. E. & Chalasani, N. Should combination therapy be the paradigm for future nonalcoholic steatohepatitis clinical trials? Hepatology 54, 1503–1505 (2011).
Sanyal, A. J. Past, present and future perspectives in nonalcoholic fatty liver disease. Nat. Rev. Gastroenterol. Hepatol. 16, 377–386 (2019).
Loomba, R. et al. Safety and efficacy of combination therapies including cilofexor/firsocostat in patients with bridging fibrosis and cirrhosis due to NASH: results of the phase 2b ATLAS trial [abstract LB004]. J. Hepatol. 73, S116–S117 (2020).
Gilead. Gilead announces topline results from phase 2 ATLAS study in patients with bridging fibrosis (F3) and compensated cirrhosis (F4) due to nonalcoholic steatohepatitis (NASH). Gilead https://www.gilead.com/news-and-press/press-room/press-releases/2019/12/gilead-announces-topline-results-from-phase-2-atlas-study-in-patients-with-bridging-fibrosis-f3-and-compensated-cirrhosis-f4-due-to-nonalcoholic-s (2019).
Alkhouri, N. et al. Safety and efficacy of combination therapies including semaglutide, cilofexor, and firsocostat in patients with NASH [Abstract LO2]. Presented at The Liver Meeting (2020).
Pedrosa, M. et al. A randomized, double-blind, multicenter, phase 2b study to evaluate the safety and efficacy of a combination of tropifexor and cenicriviroc in patients with nonalcoholic steatohepatitis and liver fibrosis: study design of the TANDEM trial. Contemp. Clin. Trials 88, 105889 (2020).
CymaBay Therapeutics. CymaBay therapeutics halts clinical development of seladelpar. CymaBay Therapeutics https://ir.cymabay.com/press-releases/detail/476/cymabay-therapeutics-halts-clinical-development-of-seladelpar (2019).
CymaBay Therapeutics. FDA lifts all clinical holds on seladelpar. CymaBay Therapeutics https://ir.cymabay.com/press-releases/detail/485/fda-lifts-all-clinical-holds-on-seladelpar (2020).
Loomba, R. et al. The ASK1 inhibitor selonsertib in patients with nonalcoholic steatohepatitis: a randomized, phase 2 trial. Hepatology 67, 549–559 (2018).
Caiazzo, R. et al. Roux-en-Y gastric bypass versus adjustable gastric banding to reduce nonalcoholic fatty liver disease: a 5-year controlled longitudinal study. Ann. Surg. 260, 893–898; discussion 898–899 (2014).
Fakhry, T. K. et al. Bariatric surgery improves nonalcoholic fatty liver disease: a contemporary systematic review and meta-analysis. Surg. Obes. Relat. Dis. 15, 502–511 (2019).
Davison, B. A. et al. Suboptimal reliability of liver biopsy evaluation has implications for randomized clinical trials. J. Hepatol. 73, 1322–1332 (2020).
Vanderbeck, S., Bockhorst, J., Komorowski, R., Kleiner, D. E. & Gawrieh, S. Automatic classification of white regions in liver biopsies by supervised machine learning. Hum. Pathol. 45, 785–792 (2014).
Vanderbeck, S. et al. Automatic quantification of lobular inflammation and hepatocyte ballooning in nonalcoholic fatty liver disease liver biopsies. Hum. Pathol. 46, 767–775 (2015).
Hagstrom, H. et al. Fibrosis stage but not NASH predicts mortality and time to development of severe liver disease in biopsy-proven NAFLD. J. Hepatol. 67, 1265–1273 (2017).
Younossi, Z. M., Otgonsuren, M., Venkatesan, C. & Mishra, A. In patients with non-alcoholic fatty liver disease, metabolically abnormal individuals are at a higher risk for mortality while metabolically normal individuals are not. Metabolism 62, 352–360 (2013).
Golabi, P. et al. Components of metabolic syndrome increase the risk of mortality in nonalcoholic fatty liver disease (NAFLD). Medicine 97, e0214 (2018).
Paik, J. M. et al. The impact of modifiable risk factors on the long-term outcomes of non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 51, 291–304 (2020).
Paik, J. M. et al. Mortality related to nonalcoholic fatty liver disease is increasing in the United States. Hepatol. Commun. 3, 1459–1471 (2019).
Musso, G. et al. Fatty liver and chronic kidney disease: novel mechanistic insights and therapeutic opportunities. Diabetes Care 39, 1830–1845 (2016).
Musso, G. et al. Association of non-alcoholic fatty liver disease with chronic kidney disease: a systematic review and meta-analysis. PLoS Med. 11, e1001680 (2014).
Brzozowska, M. M., Ostapowicz, G. & Weltman, M. D. An association between non-alcoholic fatty liver disease and polycystic ovarian syndrome. J. Gastroenterol. Hepatol. 24, 243–247 (2009).
Chen, M. J. & Ho, H. N. Hepatic manifestations of women with polycystic ovary syndrome. Best Pract. Res. Clin. Obstet. Gynaecol. 37, 119–128 (2016).
Kim, S. et al. A low level of serum total testosterone is independently associated with nonalcoholic fatty liver disease. BMC Gastroenterol. 12, 69 (2012).
Macut, D. et al. Non-alcoholic fatty liver disease is associated with insulin resistance and lipid accumulation product in women with polycystic ovary syndrome. Hum. Reprod. 31, 1347–1353 (2016).
Seo, N. K. et al. Prediction of prevalent but not incident non-alcoholic fatty liver disease by levels of serum testosterone. J. Gastroenterol. Hepatol. 30, 1211–1216 (2015).
Yim, J. Y., Kim, J., Kim, D. & Ahmed, A. Serum testosterone and non-alcoholic fatty liver disease in men and women in the US. Liver Int. 38, 2051–2059 (2018).
Zhang, J. et al. Analyses of risk factors for polycystic ovary syndrome complicated with non-alcoholic fatty liver disease. Exp. Ther. Med. 15, 4259–4264 (2018).
Wang, T., Yang, W., Karakas, S. & Sarkar, S. NASH in nondiabetic endocrine disorders. Metab. Syndr. Relat. Disord. 16, 315–320 (2018).
Sarkar, M. et al. Low testosterone is associated with nonalcoholic steatohepatitis (NASH) and severity of NASH fibrosis in men with NAFLD. Clin. Gastroenterol. Hepatol. https://doi.org/10.1016/j.cgh.2019.11.053 (2019).
Asfari, M. M. et al. Association of non-alcoholic fatty liver disease and polycystic ovarian syndrome. BMJ Open Gastroenterol. 7, e000352 (2020).
Ramezani-Binabaj, M., Motalebi, M., Karimi-Sari, H., Rezaee-Zavareh, M. S. & Alavian, S. M. Are women with polycystic ovarian syndrome at a high risk of non-alcoholic fatty liver disease; a meta-analysis. Hepat. Mon. 14, e23235 (2014).
Younossi, Z. M. et al. The impact of pruritus on patient-reported outcomes (Pros) in patients with non-alcoholic steatohepatitis (NASH) treated with obeticholic acid (OCA) [abstract]. Hepatology 70, 39A–40A (2019).
Anstee, Q. M., Reeves, H. L., Kotsiliti, E., Govaere, O. & Heikenwalder, M. From NASH to HCC: current concepts and future challenges. Nat. Rev. Gastroenterol. Hepatol. 16, 411–428 (2019).
Noureddin, M. & Rinella, M. E. Nonalcoholic fatty liver disease, diabetes, obesity, and hepatocellular carcinoma. Clin. Liver Dis. 19, 361–379 (2015).
Sadler, E. M. et al. Liver transplantation for NASH-related hepatocellular carcinoma versus non-NASH etiologies of hepatocellular carcinoma. Transplantation 102, 640–647 (2018).
Younossi, Z. et al. Nonalcoholic steatohepatitis is the fastest growing cause of hepatocellular carcinoma in liver transplant candidates. Clin. Gastroenterol. Hepatol. 17, 748–755.e3 (2019).
Kwak, M. et al. Bariatric surgery is associated with reduction in non-alcoholic steatohepatitis and hepatocellular carcinoma: a propensity matched analysis. Am. J. Surg. 219, 504–507 (2020).
Takahashi, S. et al. Role of farnesoid X receptor and bile acids in hepatic tumor development. Hepatol. Commun. 2, 1567–1582 (2018).
Deuschle, U. et al. FXR controls the tumor suppressor NDRG2 and FXR agonists reduce liver tumor growth and metastasis in an orthotopic mouse xenograft model. PLoS ONE 7, e43044 (2012).
Attia, Y. M., Tawfiq, R. A., Ali, A. A. & Elmazar, M. M. The FXR agonist, obeticholic acid, suppresses HCC proliferation & metastasis: role of IL-6/STAT3 signalling pathway. Sci. Rep. 7, 12502 (2017).
Anakk, S. et al. Bile acids activate YAP to promote liver carcinogenesis. Cell Rep. 5, 1060–1069 (2013).
Su, H. et al. Downregulation of nuclear receptor FXR is associated with multiple malignant clinicopathological characteristics in human hepatocellular carcinoma. Am. J. Physiol. Gastrointest. Liver Physiol 303, G1245–G1253 (2012).
Sawey, E. T. et al. Identification of a therapeutic strategy targeting amplified FGF19 in liver cancer by oncogenomic screening. Cancer Cell 19, 347–358 (2011).
Wang, K. et al. Genomic landscape of copy number aberrations enables the identification of oncogenic drivers in hepatocellular carcinoma. Hepatology 58, 706–717 (2013).
Avila, M. A. & Moschetta, A. The FXR-FGF19 gut-liver axis as a novel “hepatostat”. Gastroenterology 149, 537–540 (2015).
Piglionica, M., Cariello, M. & Moschetta, A. The gut-liver axis in hepatocarcinoma: a focus on the nuclear receptor FXR and the enterokine FGF19. Curr. Opin. Pharmacol. 43, 93–98 (2018).
US Food and Drug Administration. Expedited programs for serious conditions — drugs and biologics (FDA, 2014).
Alonso, R., Cuevas, A. & Cafferata, A. Diagnosis and management of statin intolerance. J. Atheroscler. Thromb. 26, 207–215 (2019).
Brown, A. S. & Watson, K. E. Statin intolerance. Rev. Cardiovasc. Med. 19, S9–S19 (2018).
Penson, P. E. et al. Introducing the ‘drucebo’ effect in statin therapy: a systematic review of studies comparing reported rates of statin-associated muscle symptoms, under blinded and open-label conditions. J. Cachexia Sarcopenia Muscle 9, 1023–1033 (2018).
Robinson, J. G. New insights into managing symptoms during statin therapy. Prog. Cardiovasc. Dis. 62, 390–394 (2019).
Hanf, R. et al. Assessment of NIS4 clinical utility for identification of patients with active NASH (NAS ≥ 4) and significant fibrosis (F ≥ 2) in patients at risk of NASH [abstract SAT-299]. J. Hepatology 70, E770 (2019).
Suh, C. H. et al. Shear wave elastography as a quantitative biomarker of clinically significant portal hypertension: a systematic review and meta-analysis. AJR Am. J. Roentgenol. 210, W185–W195 (2018).
Vilar-Gomez, E. et al. Long-term metformin use may improve clinical outcomes in diabetic patients with non-alcoholic steatohepatitis and bridging fibrosis or compensated cirrhosis. Aliment. Pharmacol. Ther. 50, 317–328 (2019).
Vilar-Gomez, E. et al. Vitamin E improves transplant-free survival and hepatic decompensation among patients with nonalcoholic steatohepatitis and advanced fibrosis. Hepatology 71, 495–509 (2020).
Armstrong, M. J. et al. Liraglutide efficacy and action in non-alcoholic steatohepatitis (LEAN): study protocol for a phase II multicentre, double-blinded, randomised, controlled trial. BMJ Open 3, e003995 (2013).
Noureddin, M. Muthiah, M. D. & Sanyal, A. J. Drug discovery and treatment paradigms in nonalcoholic steatohepatitis. Endocrinol. Diabetes Metab. 3, e00105 (2019).
Acknowledgements
We acknowledge N. Chalasani for his thoughtful review and suggestions. We thank Y. Rahimi for sharing the detailed list of drug candidates currently under development for NASH and their timelines as listed on ClinicalTrials.gov.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to substantial discussion of content, writing and reviewing/editing the manuscript before submission. R.V., M.N. and N.A. researched data for the article.
Corresponding author
Ethics declarations
Competing interests
R.V. has received consulting fees for serving on the Data Safety Monitoring Boards for Covance, Enyio and Enanta; R.V. also received research grant support from Gilead Sciences, Zydus Discovery, Cara Therapeutics, Novo Nordisk, Eli Lilly, Astra Zeneca, Terns Pharmaceuticals and Intercept where his institution receives the funding; M.N. has been on the advisory board or a speaker for Allergan, Gilead, Intercept, Pfizer, Novartis, Blade, EchoSens North America, OWL, Simply Speaking and Abbott; M.N. has received research support from Allergan, BMS, Gilead, Galmed, Galectin, Genfit, Conatus, Enanta, Novartis, Shire and Zydus; M.N. is a minor shareholder or has stocks in Anaetos and Viking. N.A. has received research funding from Albireo, Akero, Allergan, Boehringer Ingelheim, Bristol-Myers Squibb, Galmed, Genfit, Gilead, Intercept, Madrigal, MedImmune, Novartis, Novo Nordisk, Pfizer, Poxel and Zydus, and has acted as a speaker for AbbVie, Alexion, Allergan, Eisai, Exelixis, Gilead, Intercept and Salix and as a consultant for Allergan, Gilead and Intercept. A.J.S. is President of Sanyal Biotechnology and has stock options in Genfit, Akarna, Tiziana, Indalo, Durect, Exalenz and Hemoshear; A.J.S. has served as a consultant to AstraZeneca, Nitto Denko, Ardelyx, Conatus, Nimbus, Amarin, Salix, Tobira, Takeda, Fibrogen, Jannsen, Gilead, Lilly, Poxel, Artham, Cymabay, Boehringer Ingelheim, Novo Nordisk, Bird Rock Bio, Novartis, Pfizer, Jannsen and Genfit; A.J.S. has been an unpaid consultant to Intercept, Echosens, Immuron, Galectin, Fractyl, Syntlogic, Afimmune, ChemomAb, Nordic Bioscience and Bristol Myers Squibb; his institution has received grant support from Gilead, Salix, Tobira, Bristol Myers, Shire, Intercept, Merck, AstraZeneca, Mallinckrodt, Cumberland and Novartis; A.J.S. receives royalties from Elsevier and UptoDate.
Additional information
Peer review information
Nature Reviews Gastroenterology & Hepatology thanks H. Cortez-Pinto, V. Ratziu and V.S. Wong for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
ClinicalTrials.gov: https://clinicaltrials.gov/
Liver Investigation: Testing Marker Utility in Steatohepatitis (LITMUS): https://litmus-project.eu/
Non-Invasive Biomarkers of Metabolic Liver Disease (NIMBLE): https://fnih.org/what-we-do/biomarkers-consortium/programs/nimble
Supplementary information
Rights and permissions
About this article
Cite this article
Vuppalanchi, R., Noureddin, M., Alkhouri, N. et al. Therapeutic pipeline in nonalcoholic steatohepatitis. Nat Rev Gastroenterol Hepatol 18, 373–392 (2021). https://doi.org/10.1038/s41575-020-00408-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41575-020-00408-y
This article is cited by
-
Huanglian-Hongqu herb pair improves nonalcoholic fatty liver disease via NF-κB/NLRP3 pathway in mice: network pharmacology, molecular docking and experimental validation
Hereditas (2024)
-
Recompensation in cirrhosis: unravelling the evolving natural history of nonalcoholic fatty liver disease
Nature Reviews Gastroenterology & Hepatology (2024)
-
Emerging Drug Therapies for Metabolic Dysfunction-Associated Steatotic Liver Disease: A Glimpse into the Horizon
Current Hepatology Reports (2024)
-
Prediction of outcomes in patients with metabolic dysfunction-associated steatotic liver disease based on initial measurements and subsequent changes in magnetic resonance elastography
Journal of Gastroenterology (2024)
-
Systematic analysis of randomised controlled trials of Chinese herb medicine for non-alcoholic steatohepatitis (NASH): implications for future drug development and trial design
Chinese Medicine (2023)