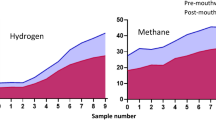
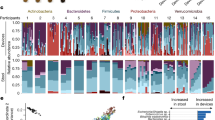
Abstract
The inner workings of the intestines, in which the body and microbiome intersect to influence gut function and systemic health, remain elusive. Carbon dioxide, hydrogen, methane and hydrogen sulfide, as well as a variety of trace gases, are generated by the chemical interactions and microbiota within the gut. Profiling of these intestinal gases and their responses to dietary changes can reveal the products and functions of the gut microbiota and their influence on human health. Indeed, different tools for measuring these intestinal gases have been developed, including newly developed gas-sensing capsule technology. Gases can, according to their type, concentration and volume, induce or relieve abdominal symptoms, and might also have physiological, pathogenic and therapeutic effects. Thus, profiling and modulating intestinal gases could be powerful tools for disease prevention and/or therapy. As the interactions between the microbiota, chemical constituents and fermentative substrates of the gut are principally influenced by dietary intake, altering the diet, which, in turn, changes gas profiles, is the main therapeutic approach for gastrointestinal disorders. An improved understanding of the complex interactions within the intestines that generate gases will enhance our ability to prevent, diagnose, treat and monitor many gastrointestinal disorders.
Key points
-
Profiling intestinal gases enables assessment of the functions of the gut microbiome.
-
Intestinal gas composition can influence gut physiology and generate abdominal symptoms in patients with gastrointestinal disorders such as IBS and IBD.
-
Multiple different techniques have been developed for assessing intestinal gases, all of which are limited by the fact that they either measure intestinal gases indirectly or are highly invasive.
-
Ingestible gas-sensing capsules seem to be a very promising alternative to indirect or invasive techniques as they provide direct gas concentration measurements and are minimally invasive.
-
Intestinal gas profiles are predominantly influenced by the composition of the luminal microbiota and by consumed dietary substrates.
-
Dietary manipulations readily alter intestinal gas production and composition and are, therefore, attractive tools in the management of patients with gas-associated gastrointestinal disorders.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Zmora, N., Suez, J. & Elinav, E. You are what you eat: diet, health and the gut microbiota. Nat. Rev. Gastroenterol. Hepatol. 16, 35–56 (2019).
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, 559–563 (2014).
Newlove-Delgado, T. V. et al. Dietary interventions for recurrent abdominal pain in childhood. Cochrane Database Syst. Rev. 3, CD010972 (2017).
Bohn, L. et al. Diet low in FODMAPs reduces symptoms of irritable bowel syndrome as well as traditional dietary advice: a randomized controlled trial. Gastroenterology 149, 1399–1407 (2015).
Carbonero, F., Benefiel, A. C. & Gaskins, H. R. Contributions of the microbial hydrogen economy to colonic homeostasis. Nat. Rev. Gastroenterol. Hepatol. 9, 504–518 (2012).
Topping, D. L. & Clifton, P. M. Short-chain fatty acids and human colonic function: roles of resistant starch and nonstarch polysaccharides. Physiol. Res. 81, 1031–1064 (2001).
Arasaradnam, R. P., Covington, J. A., Harmston, C. & Nwokolo, C. U. Review article: next generation diagnostic modalities in gastroenterology – gas phase volatile compound biomarker detection. Aliment. Pharmacol. Ther. 39, 780–789 (2014).
Berean, K. J. et al. The safety and sensitivity of a telemetric capsule to monitor gastrointestinal hydrogen production in vivo in healthy subjects: a pilot trial comparison to concurrent breath analysis. Aliment. Pharmacol. Ther. 48, 646–654 (2018).
Pillai, N. et al. An in-vitro upper gut simulator for assessing continuous gas production: a proof-of-concept using milk digestion. J. Funct. Foods 47, 200–210 (2018).
Lacy, B. E., Gabbard, S. L. & Crowell, M. D. Pathophysiology, evaluation, and treatment of bloating: hope, hype, or hot air? Gastroenterol. Hepatol. 7, 729–739 (2011).
Suarez, F., Furne, J., Springfield, J. & Levitt, M. Insights into human colonic physiology obtained from the study of flatus composition. Am. J. Physiol. Gastrointest. Liver Physiol. 272, G1028–G1033 (1997).
Sahakian, A. B., Jee, S. R. & Pimentel, M. Methane and the gastrointestinal tract. Dig. Liver Dis. 55, 2135–2143 (2010).
Levitt, M. D. Volume and composition of human intestinal gas determined by means of an intestinal washout technic. N. Engl. J. Med. 284, 1394–1398 (1971).
Levitt, M. D. & Bond, J. H. Volume, composition, and source of intestinal gas. Gastroenterology 59, 921–929 (1970).
Bettinger, C. J. Materials advances for next-generation ingestible electronic medical devices. Trends Biotechnol. 33, 575–585 (2015).
Konturek, P. C., Konturek, S. J. & Ochmański, W. Neuroendocrinology of gastric H+ and duodenal HCO3 − secretion: the role of brain–gut axis. Eur. J. Pharmacol. 499, 15–27 (2004).
Yao, C. K., Muir, J. G. & Gibson, P. R. Review article: insights into colonic protein fermentation, its modulation and potential health implications. Aliment. Pharmacol. Ther. 43, 181–196 (2016).
Marchesi, J. R. et al. The gut microbiota and host health: a new clinical frontier. Gut 65, 330–339 (2016).
El Kaoutari, A., Armougom, F., Gordon, J. I., Raoult, D. & Henrissat, B. The abundance and variety of carbohydrate-active enzymes in the human gut microbiota. Nat. Rev. Microbiol. 11, 497–504 (2013).
Eckburg, P. B. et al. Diversity of the human intestinal microbial flora. Science 308, 1635–1638 (2005).
Ze, X., Duncan, S. H., Louis, P. & Flint, H. J. Ruminococcus bromii is a keystone species for the degradation of resistant starch in the human colon. ISME J. 6, 1535–1543 (2012).
Mowat, A. M. & Agace, W. W. Regional specialization within the intestinal immune system. Nat. Rev. Immunol. 14, 667–685 (2014).
Simren, M. et al. Intestinal microbiota in functional bowel disorders: a Rome Foundation report. Gut 62, 159–176 (2013).
Kalantar-Zadeh, K. et al. A human pilot trial of ingestible electronic capsules capable of sensing different gases in the gut. Nat. Electron. 1, 79–87 (2018).
Gill, P. A., van Zelm, M. C., Muir, J. G. & Gibson, P. R. Short chain fatty acids as potential therapeutic agents in human gastrointestinal and inflammatory disorders. Aliment. Pharmacol. Ther. 48, 15–34 (2018).
Scott, K. P., Gratz, S. W., Sheridan, P. O., Flint, H. J. & Duncan, S. H. The influence of diet on the gut microbiota. Pharmacol. Res. 69, 52–60 (2013).
Ong, A. M. L. et al. Diaphragmatic breathing reduces belching and proton pump inhibitor refractory gastroesophageal reflux symptoms. Clin. Gastroenterol. Hepatol. 16, 407–416.e2 (2018).
Gasbarrini, A. et al. Methodology and indications of H2-breath testing in gastrointestinal diseases: the Rome Consensus Conference. Aliment. Pharmacol. Ther. 29, 1–3 (2009).
Vernia, P., Di Camillo, M. & Marinaro, V. Lactose malabsorption, irritable bowel syndrome and self-reported milk intolerance. Dig. Liver Dis. 33, 234–239 (2001).
Drossman, D. A. The functional gastrointestinal disorders and the Rome II process. Gut 45, 1–5 (1999).
Basilisco, G., Marino, B., Passerini, L. & Ogliari, C. Abdominal distension after colonic lactulose fermentation recorded by a new extensometer. J. Neurogastroenterol. Motil. 15, 427–433 (2003).
Attar, A. et al. Comparison of a low dose polyethylene glycol electrolyte solution with lactulose for treatment of chronic constipation. Gut 44, 226–230 (1999).
Wang, R. Physiological implications of hydrogen sulfide: a whiff exploration that blossomed. Physiol. Res. 92, 791–896 (2012).
Medani, M. et al. Emerging role of hydrogen sulfide in colonic physiology and pathophysiology. Inflamm. Bowel Dis. 17, 1620–1625 (2011).
Matsunami, M. et al. Luminal hydrogen sulfide plays a pronociceptive role in mouse colon. Gut 58, 751–761 (2009).
Li, H., Wang, Y. J., Wang, S. R. & Chen, O. Y. Comments and hypotheses on the mechanism of methane against ischemia/reperfusion injury. Med. Gas Res. 7, 120–123 (2017).
Naito, Y., Uchiyama, K. & Takagi, T. Redox-related gaseous mediators in the gastrointestinal tract. J. Clin. Biochem. Nutr. 63, 1–4 (2018).
Dupont, A. W. & Dupont, H. L. The intestinal microbiota and chronic disorders of the gut. Nat. Rev. Gastroenterol. Hepatol. 8, 523–531 (2011).
Laine, L., Takeuchi, K. & Tarnawski, A. Gastric mucosal defense and cytoprotection: bench to bedside. Gastroenterology 135, 41–60 (2008).
Abdel-Salam, O. M. E., Czimmer, J., Debreceni, A., Szolcsányi, J. & Mózsik, G. Gastric mucosal integrity: gastric mucosal blood flow and microcirculation. An overview. J. Physiol. Paris 95, 105–127 (2001).
Ou, J. Z. et al. Potential of in vivo real-time gastric gas profiling: a pilot evaluation of heat-stress and modulating dietary cinnamon effect in an animal model. Sci. Rep. 6, 33387 (2016).
Gäbel, G., Vogler, S. & Martens, H. Short-chain fatty acids and CO2 as regulators of Na+ and Cl− absorption in isolated sheep rumen mucosa. J. Comp. Physiol. B 161, 419–426 (1991).
Kurada, S., Alkhouri, N., Fiocchi, C., Dweik, R. & Rieder, F. Review article: breath analysis in inflammatory bowel diseases. Aliment. Pharmacol. Ther. 41, 329–341 (2016).
Ma, N., Tian, Y., Wu, Y. & Ma, X. Contributions of the interaction between dietary protein and gut microbiota to intestinal health. Curr. Protein Pept. Sci. 18, 795–808 (2017).
Yao, C. K. et al. Modulation of colonic hydrogen sulfide production by diet and mesalazine utilizing a novel gas-profiling technology. Gut Microbes 9, 510–522 (2018).
Ahlquist, D. A. Universal cancer screening: revolutionary, rational, and realizable. NPJ Precis. Oncol. 2, 23 (2018).
Altomare, D. F. et al. Exhaled volatile organic compounds identify patients with colorectal cancer. Br. J. Surg. 100, 144–150 (2013).
Bond, A. et al. Volatile organic compounds emitted from faeces as a biomarker for colorectal cancer. Aliment. Pharmacol. Ther. 49, 1005–1012 (2019).
Krilaviciute, A., Stock, C., Leja, M. & Brenner, H. Potential of non-invasive breath tests for preselecting individuals for invasive gastric cancer screening endoscopy. J. Breath Res. 12, 036009 (2018).
Modak, A. S. Stable isotope breath tests in clinical medicine: a review. J. Breath Res. 1, 014003 (2007).
Kolkman, J. J., Otte, J. A. & Groeneveld, A. B. J. Gastrointestinal luminal P(CO2) tonometry: an update on physiology, methodology and clinical applications. Br. J. Anaesth. 84, 74–86 (2000).
Ghoos, Y. F. et al. Measurement of gastric emptying rate of solids by means of a carbon-labeled octanoic acid breath test. Gastroenterology 104, 1640–1647 (1993).
Modak, A. Breath tests with 13C substrates. J. Breath Res. 3, 040201 (2009).
Louis, P., Hold, G. L. & Flint, H. J. The gut microbiota, bacterial metabolites and colorectal cancer. Nat. Rev. Microbiol. 12, 661–672 (2014).
Nakamura, N., Lin, H. C., McSweeney, C. S., MacKie, R. I. & Rex Gaskins, H. Mechanisms of microbial hydrogen disposal in the human colon and implications for health and disease. Annu. Rev. Food Sci. Technol. 1, 363–395 (2010).
Levitt, M. D. Production and excretion of hydrogen gas in man. N. Engl. J. Med. 281, 122–127 (1969).
Christl, S. U., Murgatroyd, P. R., Gibson, G. R. & Cummings, J. H. Production, metabolism, and excretion of hydrogen in the large-intestine. Gastroenterology 102, 1269–1277 (1992).
Chassard, C. et al. Assessment of metabolic diversity within the intestinal microbiota from healthy humans using combined molecular and cultural approaches. FEMS Microbiol. Ecol. 66, 496–504 (2008).
Nava, G. M., Carbonero, F., Croix, J. A., Greenberg, E. & Gaskins, H. R. Abundance and diversity of mucosa-associated hydrogenotrophic microbes in the healthy human colon. ISME J. 6, 57–70 (2012).
Duncan, S. H., Hold, G. L., Barcenilla, A., Stewart, C. S. & Flint, H. J. Roseburia intestinalis sp nov., a novel saccharolytic, butyrate-producing bacterium from human faeces. Int. J. Syst. Evol. Microbiol. 52, 1615–1620 (2002).
Simmering, R. et al. Ruminococcus luti sp nov., isolated from a human faecal sample. Syst. Appl. Microbiol. 25, 189–193 (2002).
Chassard, C., Delmas, E., Lawson, P. A. & Bernalier-Donadille, A. Bacteroides xylanisolvens sp nov., a xylan-degrading bacterium isolated from human faeces. Int. J. Syst. Evol. Microbiol. 58, 1008–1013 (2008).
Cummings, J. H. & Englyst, H. N. Measurement of starch fermentation in the human large-intestine. Can. J. Physiol. Pharmacol. 69, 121–129 (1991).
Skoog, S. M., Bharucha, A. E. & Zinsmeister, A. R. Comparison of breath testing with fructose and high fructose corn syrups in health and IBS. J. Neurogastroenterol. Motil. 20, 505–511 (2008).
Marthinsen, D. & Fleming, S. E. Excretion of breath and flatus gases by humans consuming high-fiber diets. J. Nutr. 112, 1133–1143 (1982).
Miller, T. L. & Wolin, M. J. Methanosphaera-stadtmaniae gen. nov., sp. nov.: a species that forms methane by reducing methanol with hydrogen. Arch. Microbiol. 141, 116–122 (1985).
Gottlieb, K., Wacher, V., Sliman, J. & Pimentel, M. Review article: inhibition of methanogenic archaea by statins as a targeted management strategy for constipation and related disorders. Aliment. Pharmacol. Ther. 43, 197–212 (2016).
Triantafyllou, K., Chang, C. & Pimentel, M. Methanogens, methane and gastrointestinal motility. J. Neurogastroenterol. Motil. 20, 31–40 (2014).
Pochart, P., Dore, J., Lemann, F., Goderel, I. & Rambaud, J. C. Interrelations between populations of methanogenic archaea and sulfate-reducing bacteria in the human colon. FEMS Microbiol. Lett. 98, 225–228 (1992).
Miller, T. L. & Wolin, M. J. Methanogens in human and animal intestinal tracts. Syst. Appl. Microbiol. 7, 223–229 (1986).
Ong, D. K. et al. Manipulation of dietary short chain carbohydrates alters the pattern of gas production and genesis of symptoms in irritable bowel syndrome. J. Gastroenterol. Hepatol. 25, 1366–1373 (2010).
Bond, J. H., Engel, R. R. & Levitt, M. D. Factors influencing pulmonary methane excretion in man – indirect method of studying in-situ metabolism of methane-producing colonic bacteria. J. Exp. Med. 133, 572–588 (1971).
Peled, Y., Gilat, T., Liberman, E. & Bujanover, Y. The development of methane production in childhood and adolescence. J. Pediatr. Gastroenterol. Nutr. 4, 575–579 (1985).
Chatterjee, S., Park, S., Low, K., Kong, Y. & Pimentel, M. The degree of breath methane production in IBS correlates with the severity of constipation. Am. J. Gastroenterol. 102, 837–841 (2007).
Houben, E., De Preter, V., Billen, J., Van Ranst, M. & Verbeke, K. Additional value of CH4 measurement in a combined C-13/H2 lactose malabsorption breath test: a retrospective analysis. Nutrients 7, 7469–7485 (2015).
Gibson, G. R., Macfarlane, S. & Macfarlane, G. T. Metabolic interactions involving sulfate-reducing and methanogenic bacteria in the human large-intestine. FEMS Microbiol. Ecol. 12, 117–125 (1993).
Wang, R. Two’s company, three’s a crowd: can H2S be the third endogenous gaseous transmitter? FASEB J. 16, 1792–1798 (2002).
Roediger, W. E. W. Review article: nitric oxide from dysbiotic bacterial respiration of nitrate in the pathogenesis and as a target for therapy of ulcerative colitis. Aliment. Pharmacol. Ther. 27, 531–541 (2008).
Tomasova, L., Konopelski, P. & Ufnal, M. Gut bacteria and hydrogen sulfide: the new old players in circulatory system homeostasis. Molecules 21, E1558 (2016).
Lefebvre, R. A. Non-adrenergic non-cholinergic neurotransmission in the proximal stomach. Gen. Pharmacol. 24, 257–266 (1993).
Stark, M. E. & Szurszewski, J. H. Role of nitric oxide in gastrointestinal and hepatic function and disease. Gastroenterology 103, 1928–1949 (1992).
Kalff, J. C., Schraut, W. H., Billiar, T. R., Simmons, R. L. & Bauer, A. J. Role of inducible nitric oxide synthase in postoperative intestinal smooth muscle dysfunction in rodents. Gastroenterology 118, 316–327 (2000).
Magierowski, M., Magierowska, K., Kwiecien, S. & Brzozowski, T. Gaseous mediators nitric oxide and hydrogen sulfide in the mechanism of gastrointestinal integrity, protection and ulcer healing. Molecules 20, 9099–9123 (2015).
Reade, S. et al. Potential role of fecal volatile organic compounds as biomarkers of chemically induced intestinal inflammation in mice. FASEB J. 33, 3129–3136 (2018).
Rossi, M. et al. Volatile organic compounds in feces associate with response to dietary intervention in patients with irritable bowel syndrome. Clin. Gastroenterol. Hepatol. 16, 385–391.e1 (2018).
Japikse, C. (ed) Fart Proudly: Writings of Benjamin Franklyn You Never Read in School (Frog Books, 2003).
Kim, K. H., Jahan, S. A. & Kabir, E. A review of breath analysis for diagnosis of human health. TrAC Trends Anal. Chem. 33, 1–8 (2012).
Kalantar-Zadeh, K., Ha, N., Ou, J. Z. & Berean, K. J. Ingestible sensors. ACS Sens. 2, 468–483 (2017).
Zhang, S., Wang, H. & Zhu, M. J. A sensitive GC/MS detection method for analyzing microbial metabolites short chain fatty acids in fecal and serum samples. Talanta 196, 249–254 (2019).
Wu, H. et al. Metabolomic study for diagnostic model of oesophageal cancer using gas chromatography/mass spectrometry. J. Chromatogr. B 877, 3111–3117 (2009).
Braden, B. Methods and functions: breath tests. Best Pract. Res. Clin. Gastroenterol. 23, 337–352 (2009).
Sivieri, K. et al. Prebiotic effect of fructooligosaccharide in the simulator of the human intestinal microbial ecosystem (SHIME(®) model). J. Med. Food 17, 894–901 (2014).
Venema, K. & van den Abbeele, P. Experimental models of the gut microbiome. Best Pract. Res. Clin. Gastroenterol. 27, 115–126 (2013).
Aguirre, M. et al. Evaluation of an optimal preparation of human standardized fecal inocula for in vitro fermentation studies. J. Microbiol. Methods 117, 78–84 (2015).
Dura, A., Rose, D. J. & Rosell, C. M. Enzymatic modification of corn starch influences human fecal fermentation profiles. J. Agric. Food Chem. 65, 4651–4657 (2017).
Moon, J. S., Li, L., Bang, J. & Han, N. S. Application of in vitro gut fermentation models to food components: a review. Food Sci. Biotechnol. 25, 1–7 (2016).
Wang, X. & Gibson, G. R. Effects of the in vitro fermentation of oligofructose and inulin by bacteria growing in the human large intestine. J. Appl. Bacteriol. 75, 373–380 (1993).
Rotbart, A. et al. Designing an in-vitro gas profiling system for human faecal samples. Sens. Actuators B Chem. 238, 754–764 (2017).
Hernot, D. C. et al. In vitro fermentation profiles, gas production rates, and microbiota modulation as affected by certain fructans, galactooligosaccharides, and polydextrose. J. Agric. Food Chem. 57, 1354–1361 (2009).
Riviere, A., Selak, M., Lantin, D., Leroy, F. & De Vuyst, L. Bifidobacteria and butyrate-producing colon bacteria: importance and strategies for their stimulation in the human gut. Front. Microbiol. 7, 979 (2016).
Williams, B. A., Grant, L. J., Gidley, M. J. & Mikkelsen, D. Gut fermentation of dietary fibres: physico-chemistry of plant cell walls and implications for health. Int. J. Mol. Sci. 18, E2203 (2017).
Dear, K. L. E., Elia, M. & Hunter, J. O. Do interventions which reduce colonic bacterial fermentation improve symptoms of irritable bowel syndrome? Dig. Liver Dis. 50, 758–766 (2005).
Sonko, B. J., Prentice, A. M., Coward, W. A., Murgatroyd, P. R. & Goldberg, G. R. Dose-response relationship between fat ingestion and oxidation: quantitative estimation using whole-body calorimetry and C-13 isotope ratio mass spectrometry. Dig. Liver Dis. 55, 10–18 (2001).
King, T. S., Elia, M. & Hunter, J. O. Abnormal colonic fermentation in irritable bowel syndrome. Lancet 352, 1187–1189 (1998).
Hanf, S., Bogozi, T., Keiner, R., Frosch, T. & Popp, J. Fast and highly sensitive fiber-enhanced raman spectroscopic monitoring of molecular H2 and CH4 for point-of-care diagnosis of malabsorption disorders in exhaled human breath. Analyt. Chem. 87, 982–988 (2015).
Rezaie, A. et al. Hydrogen and methane-based breath testing in gastrointestinal disorders: the North American consensus. Am. J. Gastroenterol. 112, 775–784 (2017).
Ghoshal, U. C. How to interpret hydrogen breath tests. J. Neurogastroenterol. Motil. 17, 312–317 (2011).
Lin, E. C. & Massey, B. T. Scintigraphy demonstrates high rate of false-positive results from glucose breath tests for small bowel bacterial overgrowth. Clin. Gastroenterol. Hepatol. 14, 203–208 (2016).
Nucera, G. et al. Abnormal breath tests to lactose, fructose and sorbitol in irritable bowel syndrome may be explained by small intestinal bacterial overgrowth. Aliment. Pharmacol. Ther. 21, 1391–1395 (2005).
Szilagyi, A. et al. Comparison of a real-time polymerase chain reaction assay for lactase genetic polymorphism with standard indirect tests for lactose maldigestion. Clin. Gastroenterol. Hepatol. 5, 192–196 (2007).
Ghoshal, U. C., Kumar, S., Misra, A. & Mittal, B. Lactose malabsorption diagnosed by 50-g dose is inferior to assess clinical intolerance and to predict response to milk withdrawal than 25-g dose in an endemic area. J. Gastroenterol. Hepatol. 28, 1462–1468 (2013).
Perez, F., Accarino, A., Azpiroz, F., Quiroga, S. & Malagelada, J. R. Gas distribution within the human gut: effect of meals. Am. J. Gastroenterol. 102, 842–849 (2007).
Mc Williams, S. R. et al. Computed tomography assessment of intestinal gas volumes in functional gastrointestinal disorders. J. Neurogastroenterol. Motil. 18, 419–425 (2012).
Murray, K. et al. Differential effects of FODMAPs (fermentable oligo-, di-, mono-saccharides and polyols) on small and large intestinal contents in healthy subjects shown by MRI. Am. J. Gastroenterol. 109, 110–119 (2014).
Park, S. Y., Khemani, D., Nelson, A. D., Eckert, D. & Camilleri, M. Rectal gas volume measured by computerized tomography identifies evacuation disorders in patients with constipation. Clin. Gastroenterol. Hepatol. 15, 543–552 (2017).
Major, G. et al. Colon hypersensitivity to distension, rather than excessive gas production, produces carbohydrate-related symptoms in individuals with irritable bowel syndrome. Gastroenterology 152, 124–133 (2017).
Furne, J. K. & Levitt, M. D. Factors influencing frequency of flatus emission by healthy subjects. Dig. Dis. Sci. 41, 1631–1635 (1996).
Dickson, I. Gas-sensing gut capsules. Nat. Rev. Gastroenterol. Hepatol. 15, 130–131 (2018).
Steiger, C. et al. Ingestible electronics for diagnostics and therapy. Nat. Rev. Mater. 4, 83–98 (2019).
Tang, T. B. et al. Toward a miniature wireless integrated multisensor microsystem for industrial and biomedical applications. IEEE Sens. J. 2, 628–635 (2002).
Kalantar-Zadeh, K. et al. Intestinal gas capsules: a proof-of-concept demonstration. Gastroenterology 150, 37–39 (2016).
Ou, J. Z. et al. Human intestinal gas measurement systems: in vitro fermentation and gas capsules. Trends Biotechnol. 33, 208–213 (2015).
Wu, G. D. et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 334, 105–108 (2011).
Gibson, P. R., Varney, J., Malakar, S. & Muir, J. G. Food components and irritable bowel syndrome. Gastroenterology 148, 1158–1174 (2015).
Burri, E. et al. Mechanisms of postprandial abdominal bloating and distension in functional dyspepsia. Gut 63, 395–400 (2014).
Eswaran, S., Tack, J. & Chey, W. D. Food: the forgotten factor in the irritable bowel syndrome. Gastroenterol. Clin. North Am. 40, 141–162 (2011).
Gibson, P. R., Newnham, E., Barrett, J. S., Shepherd, S. J. & Muir, J. G. Review article: fructose malabsorption and the bigger picture. Aliment. Pharmacol. Ther. 25, 349–363 (2007).
Halmos, E. P., Power, V. A., Shepherd, S. J., Gibson, P. R. & Muir, J. G. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology 146, 67–75 (2014).
Manichanh, C. et al. Anal gas evacuation and colonic microbiota in patients with flatulence: effect of diet. Gut 63, 401–408 (2014).
Tomlin, J., Lowis, C. & Read, N. W. Investigation of normal flatus production in healthy-volunteers. Gut 32, 665–669 (1991).
Cummings, J. H. & Englyst, H. N. What is dietary fibre? Trends Food Sci. Technol. 2, 99–103 (1991).
Swallow, D. M., Poulter, M. & Hollox, E. J. Intolerance to lactose and other dietary sugars. Drug Metab. Dispos. 29, 513–516 (2001).
Shepherd, S. J., Parker, F. C., Muir, J. G. & Gibson, P. R. Dietary triggers of abdominal symptoms in patients with irritable bowel syndrome: randomized placebo-controlled evidence. Clin. Gastroenterol. Hepatol. 6, 765–771 (2008).
De Preter, V. et al. Baseline microbiota activity and influence responses to prebiotic initial bifidobacteria counts dosing in healthy subjects. Aliment. Pharmacol. Ther. 27, 504–513 (2008).
Barrett, J. S. & Gibson, P. R. Fermentable oligosaccharides, disaccharides, monosaccharides and polyols (FODMAPs) and nonallergic food intolerance: FODMAPs or food chemicals? Ther. Adv. Gastroenterol. 5, 261–268 (2012).
Flint, H. J., Scott, K. P., Duncan, S. H., Louis, P. & Forano, E. Microbial degradation of complex carbohydrates in the gut. Gut Microbes 3, 289–306 (2012).
Tran, T. H. T. et al. Adding mucins to an in vitro batch fermentation model of the large intestine induces changes in microbial population isolated from porcine feces depending on the substrate. FEMS Microbiol. Ecol. 92, fiv165 (2016).
Roberfroid, M. et al. Prebiotic effects: metabolic and health benefits. Br. J. Nutr. 104, S1–S63 (2010).
Shepherd, S. J., Lomer, M. C. E. & Gibson, P. R. Short-chain carbohydrates and functional gastrointestinal disorders. Am. J. Gastroenterol. 108, 707–717 (2013).
Eswaran, S., Muir, J. & Chey, W. D. Fiber and functional gastrointestinal disorders. Am. J. Gastroenterol. 108, 718–727 (2013).
Gibson, G. R. et al. Expert consensus document: the International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 14, 491–502 (2017).
Cummings, J. H. & Macfarlane, G. T. The control and consequences of bacterial fermentation in the human colon. J. Appl. Bacteriol. 70, 443–459 (1991).
Davila, A. M. et al. Intestinal luminal nitrogen metabolism: role of the gut microbiota and consequences for the host. Pharmacol. Res. 68, 95–107 (2013).
Rawlings, N. D., Barrett, A. J. & Finn, R. Twenty years of the MEROPS database of proteolytic enzymes, their substrates and inhibitors. Nucleic Acids Res. 44, D343–D350 (2016).
Windey, K., de Preter, V. & Verbeke, K. Relevance of protein fermentation to gut health. Mol. Nutr. Food Res. 56, 184–196 (2012).
Kim, J., Hetzel, M., Boiangiu, C. D. & Buckel, W. Dehydration of (R)-2-hydroxyacyl-CoA to enoyl-CoA in the fermentation of alpha-amino acids by anaerobic bacteria. FEMS Microbiol. Rev. 28, 455–468 (2004).
Portune, K. J. et al. Gut microbiota role in dietary protein metabolism and health-related outcomes: the two sides of the coin. Trends Food Sci. Technol. 57, 213–232 (2016).
Hughes, R., Magee, E. A. M. & Bingham, S. Protein degradation in the large intestine: relevance to colorectal cancer. Curr. Issues Intest. Microbiol. 1, 51–58 (2000).
Magee, E. A., Richardson, C. J., Hughes, R. & Cummings, J. H. Contribution of dietary protein to sulfide production in the large intestine: an in vitro and a controlled feeding study in humans. Am. J. Gastroenterol. 72, 1488–1494 (2000).
Lust, M., Nandurkar, S. & Gibson, P. R. Measurement of faecal fat excretion: an evaluation of attitudes and practices of Australian gastroenterologists. J. Gen. Intern. Med. 36, 77–85 (2006).
Brinkworth, G. D., Noakes, M., Clifton, P. M. & Bird, A. R. Comparative effects of very low-carbohydrate, high-fat and high-carbohydrate, low-fat weight-loss diets on bowel habit and faecal short-chain fatty acids and bacterial populations. Br. J. Nutr. 101, 1493–1502 (2009).
Zhang, C. H. et al. Structural resilience of the gut microbiota in adult mice under high-fat dietary perturbations. ISME J. 6, 1848–1857 (2012).
Cani, P. D. et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 56, 1761–1772 (2007).
Masset, J. et al. Fermentative hydrogen production from glucose and starch using pure strains and artificial co-cultures of Clostridium spp. Biotechnol. Biofuels 5, 35 (2012).
Calusinska, M., Happe, T., Joris, B. & Wilmotte, A. The surprising diversity of clostridial hydrogenases: a comparative genomic perspective. Microbiology 156, 1575–1588 (2010).
Molly, K., Woestyne, M. V., Smet, I. D. & Verstraete, W. Validation of the simulator of the human intestinal microbial ecosystem (SHIME) reactor using microorganism-associated activities. Microb. Ecol. Health Dis. 7, 191–200 (1994).
Acknowledgements
The authors acknowledge financial support from the National Health and Medical Research Council (NHMRC) of Australia (Development Grant, APP1154969).
Author information
Authors and Affiliations
Contributions
K.K.-Z., K.J.B., R.E.B. and P.R.G. researched data for the article. K.K.-Z. and P.R.G. made substantial contributions to discussion of the article contents. K.K.-Z., R.E.B. and P.R.G. wrote the manuscript. All authors reviewed and/or edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
K.K.-Z, P.R.G. and K.J.B. are the lead scientific advisor, lead medical advisor and chief technical officer for Atmo Biosciences, respectively, a company that owns the patents related to swallowable capsules for profiling gases along the gut. R.E.B., J.G.M. and P.R.G. declare that their affiliation, Monash University, financially gains from the sales of digital applications, booklets and education tools associated with the low FODMAP diet.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Fermentation
-
The chemical breakdown of a substance by microorganisms, typically involving effervescence and the production of heat.
- Volatile organic compounds
-
(VOCs). Organic chemicals that have a high vapour pressure at near room temperature.
- Flatus
-
Gas that is expelled from the colon via the anus.
- Headspace gas
-
The gas that occupies the volume of the gut that is not filled with the gut liquid.
- Short-chain fatty acids
-
(SCFAs). Fatty acids with fewer than six carbon atoms.
- Visceral sensitivity
-
A response to stretching of the intestinal wall by distension.
- Prebiotic
-
A substrate that is selectively utilized by host microorganisms conferring a health benefit.
- Gasotransmitter effect
-
Whereby certain gases, termed gasotransmitters, exert specific physiological functions through interaction with cells expressing specific target chemical components.
- Probiotics
-
Live microorganisms intended to provide health benefits when consumed, generally by improving gut microbiota composition.
- Heat stress
-
A situation in which too much heat is absorbed by the organ or tissue, causing stress, pathological processes and/or illness to occur.
- Colonic compliance
-
The ability of the colon to yield elastically when a force is applied.
- Carbohydrate malabsorption
-
The passage of dietary carbohydrates to the colon, as the result of a failure to completely absorb monosaccharides during passage through the small intestine, or of a lack of hydrolases in the small intestine to digest the carbohydrates into absorbable monosaccharides.
- Small-intestinal bacterial overgrowth
-
(SIBO). Excessive bacterial growth in the small intestine (greater than considered normal).
- Nonadrenergic noncholinergic inhibition
-
The inhibition of nerve cells in which epinephrine (adrenaline), norepinephrine (noradrenaline), a similar adrenergic substance or acetylcholine (a ‘cholinergic’) functions as a neurotransmitter.
Rights and permissions
About this article
Cite this article
Kalantar-Zadeh, K., Berean, K.J., Burgell, R.E. et al. Intestinal gases: influence on gut disorders and the role of dietary manipulations. Nat Rev Gastroenterol Hepatol 16, 733–747 (2019). https://doi.org/10.1038/s41575-019-0193-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41575-019-0193-z
This article is cited by
-
Effects of dietary fibre on metabolic health and obesity
Nature Reviews Gastroenterology & Hepatology (2024)
-
End-to-end design of ingestible electronics
Nature Electronics (2024)
-
Dichotomous effect of dietary fiber in pediatrics: a narrative review of the health benefits and tolerance of fiber
European Journal of Clinical Nutrition (2024)
-
Prospects of molecular hydrogen in cancer prevention and treatment
Journal of Cancer Research and Clinical Oncology (2024)
-
Gut microbiota in COVID-19: key microbial changes, potential mechanisms and clinical applications
Nature Reviews Gastroenterology & Hepatology (2023)