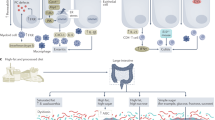
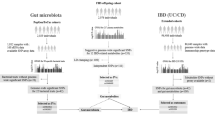
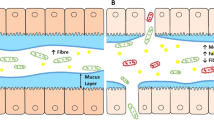
Abstract
Crohn’s disease and ulcerative colitis, collectively known as IBD, are chronic inflammatory disorders of the gastrointestinal tract. Although the aetiopathogenesis of IBD is largely unknown, it is widely thought that diet has a crucial role in the development and progression of IBD. Indeed, epidemiological and genetic association studies have identified a number of promising dietary and genetic risk factors for IBD. These preliminary studies have led to major interest in investigating the complex interaction between diet, host genetics, the gut microbiota and immune function in the pathogenesis of IBD. In this Review, we discuss the recent epidemiological, gene–environment interaction, microbiome and animal studies that have explored the relationship between diet and the risk of IBD. In addition, we highlight the limitations of these prior studies, in part by explaining their contradictory findings, and review future directions.
Key points
-
Epidemiological studies have identified a number of potential dietary risk factors for Crohn’s disease and ulcerative colitis.
-
Early results from dietary intervention studies in Crohn’s disease and ulcerative colitis have been promising, particularly in paediatric patients, but high-quality randomized trials are needed to assess efficacy.
-
Preliminary gene–environment and microbiome studies have demonstrated an interaction between diet, host genetics and the gut microbiota in the aetiopathogenesis of Crohn’s disease and ulcerative colitis.
-
Large-scale studies are needed to prospectively examine the role of diet in the aetiopathogenesis of Crohn’s disease and ulcerative colitis in the context of host genetics and the gut microbiota.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Molodecky, N. A. et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 142, 46–54.e2 (2012).
Burisch, J., Jess, T., Martinato, M., Lakatos, P. L. & EpiCom, E. The burden of inflammatory bowel disease in Europe. J. Crohns Colitis 7, 322–337 (2013).
Kappelman, M. D. et al. The prevalence and geographic distribution of Crohn’s disease and ulcerative colitis in the United States. Clin. Gastroenterol. Hepatol. 5, 1424–1429 (2007).
Ferguson, A., Sedgwick, D. M. & Drummond, J. Morbidity of juvenile onset inflammatory bowel disease: effects on education and employment in early adult life. Gut 35, 665–668 (1994).
Kappelman, M. D. et al. Direct health care costs of Crohn’s disease and ulcerative colitis in US children and adults. Gastroenterology 135, 1907–1913 (2008).
Longobardi, T., Jacobs, P. & Bernstein, C. N. Work losses related to inflammatory bowel disease in the United States: results from the National Health Interview Survey. Am. J. Gastroenterol. 98, 1064–1072 (2003).
Franke, A. et al. Genome-wide meta-analysis increases to 71 the number of confirmed Crohn’s disease susceptibility loci. Nat. Genet. 42, 1118–1125 (2010).
Kenny, E. E. et al. A genome-wide scan of Ashkenazi Jewish Crohn’s disease suggests novel susceptibility loci. PLoS Genet. 8, e1002559 (2012).
Kugathasan, S. & Amre, D. Inflammatory bowel disease — environmental modification and genetic determinants. Pediatr. Clin. North Amer. 53, 727–749 (2006).
Tysk, C., Lindberg, E., Jarnerot, G. & Floderus-Myrhed, B. Ulcerative colitis and Crohn’s disease in an unselected population of monozygotic and dizygotic twins. A study of heritability and the influence of smoking. Gut 29, 990–996 (1988).
Ng, S. C. et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet 390, 2769–2778 (2018).
Williams, C. N. Does the incidence of IBD increase when persons move from a low- to a high-risk area? Inflamm. Bowel Dis. 14 (Suppl. 2), S41–S42 (2008).
Probert, C. S., Jayanthi, V., Pinder, D., Wicks, A. C. & Mayberry, J. F. Epidemiological study of ulcerative proctocolitis in Indian migrants and the indigenous population of Leicestershire. Gut 33, 687–693 (1992).
Benchimol, E. I. et al. Inflammatory bowel disease in immigrants to Canada and their children: a population-based cohort study. Am. J. Gastroenterol. 110, 553–563 (2015).
Li, X., Sundquist, J., Hemminki, K. & Sundquist, K. Risk of inflammatory bowel disease in first- and second-generation immigrants in Sweden: a nationwide follow-up study. Inflamm. Bowel Dis. 17, 1784–1791 (2011).
Albenberg, L. G. & Wu, G. D. Diet and the intestinal microbiome: associations, functions, and implications for health and disease. Gastroenterology 146, 1564–1572 (2014).
Poullis, A., Foster, R., Shetty, A., Fagerhol, M. K. & Mendall, M. A. Bowel inflammation as measured by fecal calprotectin: a link between lifestyle factors and colorectal cancer risk. Cancer Epidemiol. Biomarkers Prev. 13, 279–284 (2004).
Martinez-Medina, M. et al. Western diet induces dysbiosis with increased E coli in CEABAC10 mice, alters host barrier function favouring AIEC colonisation. Gut 63, 116–124 (2014).
Stenman, L. K., Holma, R., Eggert, A. & Korpela, R. A novel mechanism for gut barrier dysfunction by dietary fat: epithelial disruption by hydrophobic bile acids. Am. J. Physiol. Gastrointest. Liver Physiol. 304, G227–234 (2013).
Halmos, E. P. & Gibson, P. R. Dietary management of IBD — insights and advice. Nat. Rev. Gastroenterol. Hepatol. 12, 133–146 (2015).
Borrelli, O. et al. Polymeric diet alone versus corticosteroids in the treatment of active pediatric Crohn’s disease: a randomized controlled open-label trial. Clin. Gastroenterol. Hepatol. 4, 744–753 (2006).
Zachos, M., Tondeur, M. & Griffiths, A. M. Enteral nutritional therapy for induction of remission in Crohn’s disease. Cochrane Database Syst. Rev. 1, CD000542 (2007).
Charlebois, A., Rosenfeld, G. & Bressler, B. The impact of dietary interventions on the symptoms of inflammatory bowel disease: a systematic review. Crit. Rev. Food Sci. Nutr. 56, 1370–1378 (2016).
Persson, P. G., Ahlbom, A. & Hellers, G. Diet and inflammatory bowel disease: a case-control study. Epidemiology 3, 47–52 (1992).
Chapman-Kiddell, C. A., Davies, P. S., Gillen, L. & Radford-Smith, G. L. Role of diet in the development of inflammatory bowel disease. Inflamm. Bowel Dis. 16, 137–151 (2010).
Tragnone, A. et al. Dietary habits as risk factors for inflammatory bowel disease. Eur. J. Gastroenterol. Hepatol. 7, 47–51 (1995).
Willett, W. C., Reynolds, R. D., Cottrell-Hoehner, S., Sampson, L. & Browne, M. L. Validation of a semi-quantitative food frequency questionnaire: comparison with a 1-year diet record. J. Am. Diet Assoc. 87, 43–47 (1987).
Colditz, G. A., Manson, J. E. & Hankinson, S. E. The Nurses’ Health Study: 20-year contribution to the understanding of health among women. J. Womens Health 6, 49–62 (1997).
Ananthakrishnan, A. N. et al. A prospective study of long-term intake of dietary fiber and risk of Crohn’s disease and ulcerative colitis. Gastroenterology 145, 970–977 (2013).
Ananthakrishnan, A. N. et al. Long-term intake of dietary fat and risk of ulcerative colitis and Crohn’s disease. Gut 63, 776–784 (2014).
Khalili, H. et al. Dietary iron and heme iron consumption, genetic susceptibility, and risk of crohn’s disease and ulcerative colitis. Inflamm. Bowel Dis. 23, 1088–1095 (2017).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238 (2006).
Eil, R. et al. Ionic immune suppression within the tumour microenvironment limits T cell effector function. Nature 537, 539–543 (2016).
Kleinewietfeld, M. et al. Sodium chloride drives autoimmune disease by the induction of pathogenic TH17 cells. Nature 496, 518–522 (2013).
Wu, C. et al. Induction of pathogenic TH17 cells by inducible salt-sensing kinase SGK1. Nature 496, 513–517 (2013).
Yosef, N. et al. Dynamic regulatory network controlling TH17 cell differentiation. Nature 496, 461–468 (2013).
Bao, S. et al. Zinc modulates the innate immune response in vivo to polymicrobial sepsis through regulation of NF-kappaB. Am. J. Physiol. Lung Cell. Mol. Physiol. 298, L744–L754 (2010).
Finamore, A., Massimi, M., Conti Devirgiliis, L. & Mengheri, E. Zinc deficiency induces membrane barrier damage and increases neutrophil transmigration in Caco-2 cells. J. Nutr. 138, 1664–1670 (2008).
Prasad, A. S. Zinc: role in immunity, oxidative stress and chronic inflammation. Curr. Opin. Clin. Nutr. Metab. Care 12, 646–652 (2009).
Khalili, H. et al. Identification and characterization of a novel association between dietary potassium and risk of crohn’s disease and ulcerative colitis. Front. Immunol. 7, 554 (2016).
Ananthakrishnan, A. N. et al. Zinc intake and risk of Crohn’s disease and ulcerative colitis: a prospective cohort study. Int. J. Epidemiol. 44, 1995–2005 (2015).
Riboli, E. et al. European Prospective Investigation into Cancer and Nutrition (EPIC): study populations and data collection. Publ. Health Nutr. 5, 1113–1124 (2002).
Ferrari, P. et al. Within- and between-cohort variation in measured macronutrient intakes, taking account of measurement errors, in the European Prospective Investigation into Cancer and Nutrition study. Am. J. Epidemiol. 160, 814–822 (2004).
Hart, A. R. et al. Diet in the aetiology of ulcerative colitis: a European prospective cohort study. Digestion 77, 57–64 (2008).
IBD in EPIC Study Investigators et al. Linoleic acid, a dietary n-6 polyunsaturated fatty acid, and the aetiology of ulcerative colitis: a nested case-control study within a European prospective cohort study. Gut 58, 1606–1611 (2009).
Calder, P. C. Fatty acids and inflammation: the cutting edge between food and pharma. Eur. J. Pharmacol. 668 (Suppl. 1), S50–S58 (2011).
Simopoulos, A. P. Evolutionary aspects of diet, the omega-6/omega-3 ratio and genetic variation: nutritional implications for chronic diseases. Biomed. Pharmacother. 60, 502–507 (2006).
Chan, S. S. et al. Association between high dietary intake of the n-3 polyunsaturated fatty acid docosahexaenoic acid and reduced risk of Crohn’s disease. Aliment. Pharmacol. Ther. 39, 834–842 (2014).
Chan, S. S. et al. Carbohydrate intake in the etiology of Crohn’s disease and ulcerative colitis. Inflamm. Bowel Dis. 20, 2013–2021 (2014).
Racine, A. et al. Dietary patterns and risk of inflammatory bowel disease in Europe: results from the EPIC study. Inflamm. Bowel Dis. 22, 345–354 (2016).
Opstelten, J. L. et al. Dairy products, dietary calcium, and risk of inflammatory bowel disease: results from a European prospective cohort investigation. Inflamm. Bowel Dis. 22, 1403–1411 (2016).
Bergmann, M. M. et al. No association of alcohol use and the risk of ulcerative colitis or Crohn’s disease: data from a European Prospective cohort study (EPIC). Eur. J. Clin. Nutr. 71, 512–518 (2017).
Gavaler, J. S., Rosenblum, E. R., Deal, S. R. & Bowie, B. T. The phytoestrogen congeners of alcoholic beverages: current status. Proc. Soc. Exp. Biol. Med. 208, 98–102 (1995).
Seifried, H. E., Anderson, D. E., Fisher, E. I. & Milner, J. A. A review of the interaction among dietary antioxidants and reactive oxygen species. J. Nutr. Biochem. 18, 567–579 (2007).
Yamamoto, T., Nakahigashi, M. & Saniabadi, A. R. Review article: diet and inflammatory bowel disease — epidemiology and treatment. Aliment. Pharmacol. Ther. 30, 99–112 (2009).
Barton, L. L., Ritz, N. L., Fauque, G. D. & Lin, H. C. Sulfur cycling and the intestinal microbiome. Dig. Dis. Sci. 62, 2241–2257 (2017).
Kim, I. W. et al. Western-style diets induce macrophage infiltration and contribute to colitis-associated carcinogenesis. J. Gastroenterol. Hepatol. 25, 1785–1794 (2010).
Breton, J. et al. Intrinsic immunomodulatory effects of low-digestible carbohydrates selectively extend their anti-inflammatory prebiotic potentials. Biomed. Res. Int. 2015, 162398 (2015).
Le Leu, R. K., Young, G. P., Hu, Y., Winter, J. & Conlon, M. A. Dietary red meat aggravates dextran sulfate sodium-induced colitis in mice whereas resistant starch attenuates inflammation. Dig. Dis. Sci. 58, 3475–3482 (2013).
Degagne, E. et al. Sphingosine-1-phosphate lyase downregulation promotes colon carcinogenesis through STAT3-activated microRNAs. J. Clin. Invest. 124, 5368–5384 (2014).
Jantchou, P., Morois, S., Clavel-Chapelon, F., Boutron-Ruault, M. C. & Carbonnel, F. Animal protein intake and risk of inflammatory bowel disease: the E3N prospective study. Am. J. Gastroenterol. 105, 2195–2201 (2010).
Liang, J. et al. Sphingosine-1-phosphate links persistent STAT3 activation, chronic intestinal inflammation, and development of colitis-associated cancer. Cancer Cell 23, 107–120 (2013).
Hernandez, A. L. et al. Sodium chloride inhibits the suppressive function of FOXP3+ regulatory T cells. J. Clin. Invest. 125, 4212–4222 (2015).
Zhang, W. C. et al. High salt primes a specific activation state of macrophages, M(Na). Cell Res. 25, 893–910 (2015).
Hunter, D. J. Gene-environment interactions in human diseases. Nat. Rev. Genet. 6, 287–298 (2005).
Khor, B., Gardet, A. & Xavier, R. J. Genetics and pathogenesis of inflammatory bowel disease. Nature 474, 307–317 (2011).
Leone, V. A., Cham, C. M. & Chang, E. B. Diet, gut microbes, and genetics in immune function: can we leverage our current knowledge to achieve better outcomes in inflammatory bowel diseases? Curr. Opin. Immunol. 31, 16–23 (2014).
Van Der Sloot, K. W. et al. Visceral adiposity, genetic susceptibility, and risk of complications among individuals with Crohn’s disease. Inflamm. Bowel Dis. 23, 82–88 (2017).
Ananthakrishnan, A. N. et al. Genetic polymorphisms in fatty acid metabolism modify the association between dietary n3: n6 intake and risk of ulcerative colitis: a prospective cohort study. Inflamm. Bowel Dis. 23, 1898–1904 (2017).
Costea, I. et al. Interactions between the dietary polyunsaturated fatty acid ratio and genetic factors determine susceptibility to pediatric Crohn’s disease. Gastroenterology 146, 929–931 (2014).
Hargreaves, C. E. et al. Fcgamma receptors: genetic variation, function, and disease. Immunol. Rev. 268, 6–24 (2015).
Stein, M. P. et al. C-reactive protein binding to FcγRIIa on human monocytes and neutrophils is allele-specific. J. Clin. Invest. 105, 369–376 (2000).
Stein, M. P., Mold, C. & Du Clos, T. W. C-reactive protein binding to murine leukocytes requires Fc gamma receptors. J. Immunol. 164, 1514–1520 (2000).
Bharadwaj, D. et al. The major receptor for C-reactive protein on leukocytes is Fcγ receptor II. J. Exp. Med. 190, 585–590 (1999).
Zhou, L. et al. IL-6 programs T(H)-17 cell differentiation by promoting sequential engagement of the IL-21 and IL-23 pathways. Nat. Immunol. 8, 967–974 (2007).
Fantini, M. C. et al. IL-21 regulates experimental colitis by modulating the balance between Treg and Th17 cells. Eur. J. Immunol. 37, 3155–3163 (2007).
Caspi, A. et al. Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science 301, 386–389 (2003).
Risch, N. et al. Interaction between the serotonin transporter gene (5-HTTLPR), stressful life events, and risk of depression: a meta-analysis. JAMA 301, 2462–2471 (2009).
Kau, A. L., Ahern, P. P., Griffin, N. W., Goodman, A. L. & Gordon, J. I. Human nutrition, the gut microbiome and the immune system. Nature 474, 327–336 (2011).
Brown, K., DeCoffe, D., Molcan, E. & Gibson, D. L. Diet-induced dysbiosis of the intestinal microbiota and the effects on immunity and disease. Nutrients 4, 1095–1119 (2012).
D’Haens, G. R. et al. Early lesions of recurrent Crohn’s disease caused by infusion of intestinal contents in excluded ileum. Gastroenterology 114, 262–267 (1998).
Sellon, R. K. et al. Resident enteric bacteria are necessary for development of spontaneous colitis and immune system activation in interleukin-10-deficient mice. Infect. Immun. 66, 5224–5231 (1998).
Hudcovic, T., Stepankova, R., Cebra, J. & Tlaskalova-Hogenova, H. The role of microflora in the development of intestinal inflammation: acute and chronic colitis induced by dextran sulfate in germ-free and conventionally reared immunocompetent and immunodeficient mice. Folia Microbiol. 46, 565–572 (2001).
Morgan, X. C. et al. Dysfunction of the intestinal microbiome in inflammatory bowel disease and treatment. Genome Biol. 13, R79 (2012).
Nagalingam, N. A. & Lynch, S. V. Role of the microbiota in inflammatory bowel diseases. Inflamm. Bowel Dis. 18, 968–984 (2012).
Spor, A., Koren, O. & Ley, R. Unravelling the effects of the environment and host genotype on the gut microbiome. Nat. Rev. Microbiol. 9, 279–290 (2011).
Wu, G. D. et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 334, 105–108 (2011).
De Filippo, C. et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl Acad. Sci. USA 107, 14691–14696 (2010).
Ijssennagger, N. et al. Gut microbiota facilitates dietary heme-induced epithelial hyperproliferation by opening the mucus barrier in colon. Proc. Natl Acad. Sci. USA 112, 10038–10043 (2015).
Ijssennagger, N., de Wit, N., Muller, M. & van der Meer, R. Dietary heme-mediated PPARalpha activation does not affect the heme-induced epithelial hyperproliferation and hyperplasia in mouse colon. PLoS ONE 7, e43260 (2012).
N., I. J. et al. Dietary heme alters microbiota and mucosa of mouse colon without functional changes in host-microbe cross-talk. PLoS ONE 7, e49868 (2012).
Seregin, S. S. et al. NLRP6 protects Il10(−/−) mice from colitis by limiting colonization of Akkermansia muciniphila. Cell Rep. 19, 733–745 (2017).
Werner, T. et al. Depletion of luminal iron alters the gut microbiota and prevents Crohn’s disease-like ileitis. Gut 60, 325–333 (2011).
Turnbaugh, P. J., Backhed, F., Fulton, L. & Gordon, J. I. Diet-induced obesity is linked to marked but reversible alterations in the mouse distal gut microbiome. Cell Host Microbe 3, 213–223 (2008).
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, 559–563 (2014).
Devkota, S. et al. Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10−/− mice. Nature 487, 104–108 (2012).
Kevans, D. et al. IBD genetic risk profile in healthy first-degree relatives of crohn’s disease patients. J. Crohns Colitis 10, 209–215 (2016).
Kevans, D. et al. Determinants of intestinal permeability in healthy first-degree relatives of individuals with Crohn’s disease. Inflamm. Bowel Dis. 21, 879–887 (2015).
Zhernakova, A. et al. Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Science 352, 565–569 (2016).
Sokol, H. et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl Acad. Sci. USA 105, 16731–16736 (2008).
Imhann, F. et al. Interplay of host genetics and gut microbiota underlying the onset and clinical presentation of inflammatory bowel disease. Gut https://doi.org/10.1136/gutjnl-2016-312135 (2016).
Knights, D. et al. Complex host genetics influence the microbiome in inflammatory bowel disease. Genome Med. 6, 107 (2014).
Turpin, W. et al. Association of host genome with intestinal microbial composition in a large healthy cohort. Nat. Genet. 48, 1413–1417 (2016).
Bonder, M. J. et al. The effect of host genetics on the gut microbiome. Nat. Genet. 48, 1407–1412 (2016).
Feskanich, D. et al. Reproducibility and validity of food intake measurements from a semiquantitative food frequency questionnaire. J. Am. Diet Assoc. 93, 790–796 (1993).
Rimm, E. et al. Reproducibility and validity of an expanded self-administered semiquantitative food questionnaire among health professionals. Am. J. Epidemiol. 135, 1114–1126 (1992).
Rockett, H. R. et al. Validation of a youth/adolescent food frequency questionnaire. Prev. Med. 26, 808–816 (1997).
Rockett, H. R., Wolf, A. M. & Colditz, G. A. Development and reproducibility of a food frequency questionnaire to assess diets of older children and adolescents. J. Am. Diet Assoc. 95, 336–340 (1995).
Ascherio, A. et al. Correlations of vitamin A and E intakes with the plasma concentrations of carotenoids and tocopherols among American men and women. J. Nutr. 122, 1792–1801 (1992).
Romieu, I. et al. Food predictors of plasma beta-carotene and alpha-tocopherol: validation of a food frequency questionnaire. Am. J. Epidemiol. 131, 864–876 (1990).
Willett, W. C. et al. Validation of dietary questionnaire with plasma carotenoid and alpha-tocopherol level. Am. J. Clin. Nutr. 38, 631–639 (1983).
Giovannucci, E. et al. Folate, methionine, and alcohol intake and risk of colorectal adenoma. J. Natl Cancer Inst. 85, 875–884 (1993).
Hunter, D. J. et al. Comparison of measures of fatty acid intake by subcutaneous fat aspirate, food frequency questionnaire, and diet records in a free-living population of US men. Am. J. Epidemiol. 135, 418–427 (1992).
London, S. J. et al. Fatty acid composition of subcutaneous adipose tissue and diet in postmenopausal US women. Am. J. Clin. Nutr. 54, 340–345 (1991).
Yuan, C. et al. Validity of a dietary questionnaire assessed by comparison with multiple weighed dietary records or 24-hour recalls. Am. J. Epidemiol. 185, 570–584 (2017).
Ordás, I., Eckmann, L., Talamini, M., Baumgart, D. C. & Sandborn, W. J. Ulcerative colitis. Lancet 380, 1606–1619 (2012).
Ananthakrishnan, A. N. et al. High school diet and risk of Crohn’s disease and ulcerative colitis. Inflamm. Bowel Dis. 21, 2311–2319 (2015).
Bergmann, M. M. et al. No association of alcohol use and the risk of ulcerative colitis or Crohn’s disease: data from a European Prospective cohort study (EPIC). Eur. J. Clin. Nutr. 71, 566 (2017).
Acknowledgements
H.K. is funded by the US National Institute of Diabetes and Digestive and Kidney Diseases (K23 DK099681 and R03). A.T.C. is funded by the US National Institutes of Health (NIH) grant K24 DK098311, a Stuart and Suzanne Steele Massachusetts General Hospital (MGH) Research Scholar Award and a Senior Investigator Award from the Crohn’s and Colitis Foundation.
Author information
Authors and Affiliations
Contributions
H.K., S.S.M.C. and A.N.A. made substantial contributions to discussion of the content. H.K., P.L. and A.R.H. wrote the article. All authors reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
H.K. has received consulting fees from AbbVie and Samsung Bioepis. H.K. also receives funding from Takeda. S.S.M.C. has received consulting fees from AbbVie and Ferring Pharmaceuticals. P.L. is supported by a career development grant by the Crohn’s and Colitis Foundation (CCF). A.N.A. is a member of the scientific advisory board for Exact Sciences, AbbVie and Cubist Pharmaceuticals. A.T.C. has served as a consultant for Bayer Healthcare, Pfizer and Takeda. A.R.H. declares no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khalili, H., Chan, S.S.M., Lochhead, P. et al. The role of diet in the aetiopathogenesis of inflammatory bowel disease. Nat Rev Gastroenterol Hepatol 15, 525–535 (2018). https://doi.org/10.1038/s41575-018-0022-9
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41575-018-0022-9
This article is cited by
-
Diet prevents the expansion of segmented filamentous bacteria and ileo-colonic inflammation in a model of Crohn’s disease
Microbiome (2023)
-
The Prophylactic Protection of Salmonella Typhimurium Infection by Lentilactobacillus buchneri GX0328-6 in Mice
Probiotics and Antimicrobial Proteins (2023)
-
Perianal Fistulas Are Associated with Persistently Higher Direct Health Care Costs in Crohn’s Disease: A Population-Based Study
Digestive Diseases and Sciences (2023)
-
Lifestyle factors associated with inflammatory bowel disease: data from the Swiss IBD cohort study
BMC Gastroenterology (2023)
-
Chronic exposure to synthetic food colorant Allura Red AC promotes susceptibility to experimental colitis via intestinal serotonin in mice
Nature Communications (2022)