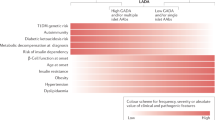
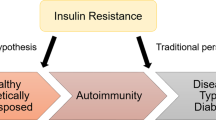
Abstract
Type 1 diabetes mellitus (T1DM) is a progressive autoimmune disease that starts long before a clinical diagnosis is made. The American Diabetes Association recognizes three stages: stage 1 (normoglycaemic and positive for autoantibodies to β-cell antigens); stage 2 (asymptomatic with dysglycaemia); and stage 3, which is defined by glucose levels consistent with the definition of diabetes mellitus. This Perspective focuses on the management of the proportion of individuals with early stage 3 T1DM who do not immediately require insulin; a stage we propose should be termed stage 3a. To date, this period of non-insulin-dependent T1DM has been largely unrecognized. Importantly, it represents a window of opportunity for intervention, as remaining at this stage might delay the need for insulin by months or years. Extending the insulin-free period and/or avoiding unnecessary insulin therapy are important goals, as there is no risk of hypoglycaemia during this period and the adherence burden on patients of glycaemic monitoring and daily adjustments for diet and exercise is substantially reduced. Recognizing the pressing need for guidance on adequate management of children and adults with stage 3a T1DM, we present our perspective on the subject, which needs to be tested in formal and adequately powered clinical trials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
03 April 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41574-023-00837-0
References
Greenbaum, C. J. et al. Fall in C-peptide during first 2 years from diagnosis: evidence of at least two distinct phases from composite Type 1 Diabetes TrialNet data. Diabetes 61, 2066–2073 (2012).
Oram, R. A., Sims, E. K. & Evans-Molina, C. Beta cells in type 1 diabetes: mass and function; sleeping or dead? Diabetologia 62, 567–577 (2019).
Powers, A. C. Type 1 diabetes mellitus: much progress, many opportunities. J. Clin. Invest. 131, e142242 (2021).
Carr, A. L. J. et al. Circulating C-peptide levels in living children and young people and pancreatic β-cell loss in pancreas donors across type 1 diabetes disease duration. Diabetes 71, 1591–1596 (2022).
Tatovic, D. & Dayan, C. M. Replacing insulin with immunotherapy: time for a paradigm change in type 1 diabetes. Diabet. Med. 38, e14696 (2021).
Mathieu, C., Martens, P. J. & Vangoitsenhoven, R. One hundred years of insulin therapy. Nat. Rev. Endocrinol. 17, 715–725 (2021).
Russell-Jones, D. & Herring, R. 100 years of physiology, discrimination and wonder. Diabet. Med. 38, e14642 (2021).
Sims, E. K., Carr, A. L. J., Oram, R. A., DiMeglio, L. A. & Evans-Molina, C. 100 years of insulin: celebrating the past, present and future of diabetes therapy. Nat. Med. 27, 1154–1164 (2021).
Miller, K. M. et al. Current state of type 1 diabetes treatment in the US: updated data from the T1D Exchange clinic registry. Diabetes Care 38, 971–978 (2015).
McKnight, J. A. et al. Glycaemic control of type 1 diabetes in clinical practice early in the 21st century: an international comparison. Diabet. Med. 32, 1036–1050 (2015).
Wasag, D. R., Gregory, J. W., Dayan, C., Harvey, J. N. & Brecon, G. Excess all-cause mortality before age 30 in childhood onset type 1 diabetes: data from the Brecon Group Cohort in Wales. Arch. Dis. Child. 103, 44–48 (2018).
Foster, N. C. et al. State of type 1 diabetes management and outcomes from the T1D Exchange in 2016–2018. Diabetes Technol. Ther. 21, 66–72 (2019).
Anderzen, J. et al. International benchmarking in type 1 diabetes: large difference in childhood HbA1c between eight high-income countries but similar rise during adolescence – a quality registry study. Pediatr. Diabetes 21, 621–627 (2020).
Prigge, R. et al. International comparison of glycaemic control in people with type 1 diabetes: an update and extension. Diabet. Med. 39, e14766 (2022).
Jones, A. G. & Hattersley, A. T. The clinical utility of C-peptide measurement in the care of patients with diabetes. Diabet. Med. 30, 803–817 (2013).
Tatovic, D. et al. Diagnosing type 1 diabetes in adults: guidance from the UK T1D immunotherapy consortium. Diabet. Med. 39, e14862 (2022).
Food and Drug Administration. FDA approves first drug that can delay onset of type 1 diabetes. FDA https://www.fda.gov/news-events/press-announcements/fda-approves-first-drug-can-delay-onset-type-1-diabetes (2022).
Couper, J. J. et al. ISPAD clinical practice consensus guidelines 2018: stages of type 1 diabetes in children and adolescents. Pediatr. Diabetes 19, 20–27 (2018).
American Diabetes Association. 2. Classification and diagnosis of diabetes: Standards of Medical Care in Diabetes — 2021. Diabetes Care 44, S15–S33 (2021).
Ziegler, A. G. et al. Seroconversion to multiple islet autoantibodies and risk of progression to diabetes in children. JAMA 309, 2473–2479 (2013).
Insel, R. A. et al. Staging presymptomatic type 1 diabetes: a scientific statement of JDRF, the Endocrine Society, and the American Diabetes Association. Diabetes Care 38, 1964–1974 (2015).
Cherubini, V. et al. Temporal trends in diabetic ketoacidosis at diagnosis of paediatric type 1 diabetes between 2006 and 2016: results from 13 countries in three continents. Diabetologia 63, 1530–1541 (2020).
Ng, S. M. et al. Presentation of newly diagnosed type 1 diabetes in children and young people during COVID-19: a national UK survey. BMJ Paediatr. Open 4, e000884 (2020).
Karges, B. et al. A comparison of familial and sporadic type 1 diabetes among young patients. Diabetes Care 44, 1116–1124 (2021).
Atkinson, M. A., Campbell-Thompson, M., Kusmartseva, I. & Kaestner, K. H. Organisation of the human pancreas in health and in diabetes. Diabetologia 63, 1966–1973 (2020).
Bingley, P. J. & Williams, A. J. Islet autoantibody testing: an end to the trials and tribulations? Diabetes 62, 4009–4011 (2013).
Wyatt, R. & Williams, A. J. Islet autoantibody analysis: radioimmunoassays. Methods Mol. Biol. 1433, 57–83 (2016).
Lampasona, V. & Liberati, D. Islet autoantibodies. Curr. Diab Rep. 16, 53 (2016).
Williams, C. L. & Long, A. E. What has zinc transporter 8 autoimmunity taught us about type 1 diabetes? Diabetologia 62, 1969–1976 (2019).
So, M. et al. Advances in type 1 diabetes prediction using islet autoantibodies: beyond a simple count. Endocr. Rev. 42, 584–604 (2021).
Besser, R. E. J., Ng, S. M. & Robertson, E. J. Screening children for type 1 diabetes. BMJ 375, e067937 (2021).
Besser, R. E. J. et al. General population screening for childhood type 1 diabetes: is it time for a UK strategy? Arch. Dis. Child. 107, 790–795 (2022).
Sims, E. K. et al. Screening for type 1 diabetes in the general population: a status report and perspective. Diabetes 71, 610–623 (2022).
Quinn, L. M. et al. EarLy surveillance for autoimmune diabetes: protocol for a qualitative study of general population and stakeholder perspectives on screening for type 1 diabetes in the UK (ELSA 1). BMJ Open Diabetes Res. Care 10, e002750 (2022).
Ziegler, A. G. et al. Yield of a public health screening of children for islet autoantibodies in Bavaria, Germany. JAMA 323, 339–351 (2020).
Diabetes Prevention Trial – Type 1 Diabetes Study Group. Effects of insulin in relatives of patients with type 1 diabetes mellitus. N. Engl. J. Med. 346, 1685–1691 (2002).
Mahon, J. L. et al. The TrialNet natural history study of the development of type 1 diabetes: objectives, design, and initial results. Pediatr. Diabetes 10, 97–104 (2009).
Gale, E. A., Bingley, P. J., Emmett, C. L. & Collier, T. European Nicotinamide Diabetes Intervention Trial (ENDIT) Group.European Nicotinamide Diabetes Intervention Trial (ENDIT): a randomised controlled trial of intervention before the onset of type 1 diabetes. Lancet 363, 925–931 (2004).
Malkani, S. & Kotwal, A. Frequency and predictors of self-reported hypoglycemia in insulin-treated diabetes. J. Diabetes Res. 2017, 7425925 (2017).
Flatt, A. J. S., Greenbaum, C. J., Shaw, J. A. M. & Rickels, M. R. Pancreatic islet reserve in type 1 diabetes. Ann. NY Acad. Sci. 1495, 40–54 (2021).
Barker, J. M. et al. Clinical characteristics of children diagnosed with type 1 diabetes through intensive screening and follow-up. Diabetes Care 27, 1399–1404 (2004).
Steck, A. K. et al. Predictors of progression from the appearance of islet autoantibodies to early childhood diabetes: the environmental determinants of diabetes in the young (TEDDY). Diabetes Care 38, 808–813 (2015).
Steck, A. K. et al. Residual beta-cell function in diabetes children followed and diagnosed in the TEDDY study compared to community controls. Pediatr. Diabetes 18, 794–802 (2017).
Herold, K. C. et al. An anti-CD3 antibody, teplizumab, in relatives at risk for type 1 diabetes. N. Engl. J. Med. 381, 603–613 (2019).
Foteinopoulou, E. et al. Impact of routine clinic measurement of serum C-peptide in people with a clinician-diagnosis of type 1 diabetes. Diabet. Med. 38, e14449 (2021).
Greenbaum, C. J. et al. Mixed-meal tolerance test versus glucagon stimulation test for the assessment of β-cell function in therapeutic trials in type 1 diabetes. Diabetes Care 31, 1966–1971 (2008).
Willemsen, R. H. et al. Frequent monitoring of C-peptide levels in newly diagnosed type 1 subjects using dried blood spots collected at home. J. Clin. Endocrinol. Metab. 103, 3350–3358 (2018).
Marren, S. M. et al. Persistent C-peptide is associated with reduced hypoglycaemia but not HbA1c in adults with longstanding type 1 diabetes: evidence for lack of intensive treatment in UK clinical practice? Diabet. Med. 36, 1092–1099 (2019).
Taylor, G. S. et al. Capturing the real-world benefit of residual β-cell function during clinically important time-periods in established type 1 diabetes. Diabet. Med. 39, e14814 (2022).
Buzzetti, R. et al. Management of latent autoimmune diabetes in adults: a consensus statement from an international expert panel. Diabetes 69, 2037–2047 (2020).
Jeyam, A. et al. Clinical impact of residual C-peptide secretion in type 1 diabetes on glycemia and microvascular complications. Diabetes Care 44, 390–398 (2021).
Yki-Jarvinen, H. & Koivisto, V. A. Natural course of insulin resistance in type I diabetes. N. Engl. J. Med. 315, 224–230 (1986).
Sosenko, J. M. et al. Glucose and C-peptide changes in the perionset period of type 1 diabetes in the diabetes prevention trial-type 1. Diabetes Care 31, 2188–2192 (2008).
Hao, W. et al. Fall in C-peptide during first 4 years from diagnosis of type 1 diabetes: variable relation to age, HbA1c, and insulin dose. Diabetes Care 39, 1664–1670 (2016).
Weiss, A. et al. Progression likelihood score identifies substages of presymptomatic type 1 diabetes in childhood public health screening. Diabetologia 65, 2121–2131 (2022).
Mortensen, H. B. et al. Multinational study in children and adolescents with newly diagnosed type 1 diabetes: association of age, ketoacidosis, HLA status, and autoantibodies on residual beta-cell function and glycemic control 12 months after diagnosis. Pediatr. Diabetes 11, 218–226 (2010).
Ludvigsson, J. et al. C-peptide in the classification of diabetes in children and adolescents. Pediatr. Diabetes 13, 45–50 (2012).
Lee, T. H. et al. The clinical measures associated with C-peptide decline in patients with type 1 diabetes over 15 years. J. Korean Med. Sci. 28, 1340–1344 (2013).
Govan, L. et al. Achieved levels of HbA1c and likelihood of hospital admission in people with type 1 diabetes in the Scottish population: a study from the Scottish Diabetes Research Network Epidemiology Group. Diabetes Care 34, 1992–1997 (2011).
Thunander, M. et al. Levels of C-peptide, body mass index and age, and their usefulness in classification of diabetes in relation to autoimmunity, in adults with newly diagnosed diabetes in Kronoberg, Sweden. Eur. J. Endocrinol. 166, 1021–1029 (2012).
Wang, Z. H., Kihl-Selstam, E. & Eriksson, J. W. Ketoacidosis occurs in both type 1 and type 2 diabetes – a population-based study from Northern Sweden. Diabet. Med. 25, 867–870 (2008).
Fourlanos, S., Narendran, P., Byrnes, G. B., Colman, P. G. & Harrison, L. C. Insulin resistance is a risk factor for progression to type 1 diabetes. Diabetologia 47, 1661–1667 (2004).
Besser, R. E. J. et al. ISPAD clinical practice consensus guidelines 2022: stages of type 1 diabetes in children and adolescents. Pediatr. Diabetes 23, 1175–1187 (2022).
Ooi, E. et al. Clinical and biochemical profile of 786 sequential episodes of diabetic ketoacidosis in adults with type 1 and type 2 diabetes mellitus. BMJ Open Diabetes Res. Care 9, e002451 (2021).
Cavero-Redondo, I., Peleteiro, B., Alvarez-Bueno, C., Rodriguez-Artalejo, F. & Martinez-Vizcaino, V. Glycated haemoglobin A1c as a risk factor of cardiovascular outcomes and all-cause mortality in diabetic and non-diabetic populations: a systematic review and meta-analysis. BMJ Open 7, e015949 (2017).
Steck, A. K. et al. CGM metrics predict imminent progression to type 1 diabetes: autoimmunity screening for kids (ASK) study. Diabetes Care 45, 365–371 (2022).
Dhatariya, K. K., Joint British Diabetes Societies for Inpatient Care. The management of diabetic ketoacidosis in adults – an updated guideline from the Joint British Diabetes Society for Inpatient Care. Diabet. Med. 39, e14788 (2022).
Garg, S. K., Peters, A. L., Buse, J. B. & Danne, T. Strategy for mitigating DKA risk in patients with type 1 diabetes on adjunctive treatment with SGLT inhibitors: a STICH protocol. Diabetes Technol. Ther. 20, 571–575 (2018).
Goldenberg, R. M., Gilbert, J. D., Hramiak, I. M., Woo, V. C. & Zinman, B. Sodium-glucose co-transporter inhibitors, their role in type 1 diabetes treatment and a risk mitigation strategy for preventing diabetic ketoacidosis: the STOP DKA protocol. Diabetes Obes. Metab. 21, 2192–2202 (2019).
Danne, T. et al. International consensus on risk management of diabetic ketoacidosis in patients with type 1 diabetes treated with sodium-glucose cotransporter (SGLT) inhibitors. Diabetes Care 42, 1147–1154 (2019).
Teng, R. et al. Comparison of protocols to reduce diabetic ketoacidosis in patients with type 1 diabetes prescribed a sodium-glucose cotransporter 2 inhibitor. Diabetes Spectr. 34, 42–51 (2021).
Dashora, U. et al. Association of British Clinical Diabetologists (ABCD) and Diabetes UK joint position statement and recommendations on the use of sodium-glucose cotransporter inhibitors with insulin for treatment of type 1 diabetes (updated October 2020). Diabet. Med. 38, e14458 (2021).
Biolo, G., Declan Fleming, R. Y. & Wolfe, R. R. Physiologic hyperinsulinemia stimulates protein synthesis and enhances transport of selected amino acids in human skeletal muscle. J. Clin. Invest. 95, 811–819 (1995).
Knowler, W. C. et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 346, 393–403 (2002).
Narendran, P. et al. Exercise to preserve β-cell function in recent-onset type 1 diabetes mellitus (EXTOD) – a randomized controlled pilot trial. Diabet. Med. 34, 1521–1531 (2017).
Chetan, M. R. et al. The type 1 diabetes ‘honeymoon’ period is five times longer in men who exercise: a case-control study. Diabet. Med. 36, 127–128 (2019).
Carr, A. L. J. et al. Measurement of peak C-peptide at diagnosis informs glycemic control but not hypoglycemia in adults with type 1 diabetes. J. Endocr. Soc. 5, bvab127 (2021).
Chimen, M. et al. What are the health benefits of physical activity in type 1 diabetes mellitus? A literature review. Diabetologia 55, 542–551 (2012).
Musil, F. et al. Effect of low calorie diet and controlled fasting on insulin sensitivity and glucose metabolism in obese patients with type 1 diabetes mellitus. Physiol. Res. 62, 267–276 (2013).
Schmidt, S. et al. Low versus high carbohydrate diet in type 1 diabetes: a 12-week randomized open-label crossover study. Diabetes Obes. Metab. 21, 1680–1688 (2019).
Seckold, R., Fisher, E., de Bock, M., King, B. R. & Smart, C. E. The ups and downs of low-carbohydrate diets in the management of type 1 diabetes: a review of clinical outcomes. Diabet. Med. 36, 326–334 (2019).
Ma, M. et al. Triglyceride is independently correlated with insulin resistance and islet beta cell function: a study in population with different glucose and lipid metabolism states. Lipids Health Dis. 19, 121 (2020).
Pursey, K. M., Hart, M., Jenkins, L., McEvoy, M. & Smart, C. E. Screening and identification of disordered eating in people with type 1 diabetes: a systematic review. J. Diabetes Complicat. 34, 107522 (2020).
Cree-Green, M. et al. Metformin improves peripheral insulin sensitivity in youth with type 1 diabetes. J. Clin. Endocrinol. Metab. 104, 3265–3278 (2019).
Vella, S. et al. The use of metformin in type 1 diabetes: a systematic review of efficacy. Diabetologia 53, 809–820 (2010).
Tandon, S., Ayis, S., Hopkins, D., Harding, S. & Stadler, M. The impact of pharmacological and lifestyle interventions on body weight in people with type 1 diabetes: a systematic review and meta-analysis. Diabetes Obes. Metab. 23, 350–362 (2021).
von Herrath, M. et al. Anti-interleukin-21 antibody and liraglutide for the preservation of β-cell function in adults with recent-onset type 1 diabetes: a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Diabetes Endocrinol. 9, 212–224 (2021).
Wentworth, J. M., Fourlanos, S., Colman, P. G. & Harrison, L. C. A pilot study of the feasibility of empagliflozin in recent-onset type 1 diabetes. Metab. Open 5, 100021 (2020).
Garg, S. K. et al. Effects of sotagliflozin added to insulin in patients with type 1 diabetes. N. Engl. J. Med. 377, 2337–2348 (2017).
Rosenstock, J. et al. Empagliflozin as adjunctive to insulin therapy in type 1 diabetes: the EASE trials. Diabetes Care 41, 2560–2569 (2018).
Buse, J. B. et al. Sotagliflozin in combination with optimized insulin therapy in adults with type 1 diabetes: the North American inTandem1 Study. Diabetes Care 41, 1970–1980 (2018).
Perkins, B. A. et al. Exploring patient preferences for adjunct-to-insulin therapy in type 1 diabetes. Diabetes Care 42, 1716–1723 (2019).
Ervin, C. et al. Insights into patients’ experience with type 1 diabetes: exit interviews from phase III studies of sotagliflozin. Clin. Ther. 41, 2219–2230.e6 (2019).
Ehrmann, D. et al. Risk factors and prevention strategies for diabetic ketoacidosis in people with established type 1 diabetes. Lancet Diabetes Endocrinol. 8, 436–446 (2020).
Rao, L., Ren, C., Luo, S., Huang, C. & Li, X. Sodium-glucose cotransporter 2 inhibitors as an add-on therapy to insulin for type 1 diabetes mellitus: meta-analysis of randomized controlled trials. Acta Diabetol. 58, 869–880 (2021).
Phillip, M. et al. Long-term efficacy and safety of dapagliflozin in patients with inadequately controlled type 1 diabetes: pooled 52-week outcomes from the DEPICT-1 and -2 studies. Diabetes Obes. Metab. 23, 549–560 (2021).
Seufert, J. et al. Real-world data of 12-month adjunct sodium-glucose co-transporter-2 inhibitor treatment in type 1 diabetes from the German/Austrian DPV registry: improved HbA1c without diabetic ketoacidosis. Diabetes Obes. Metab. 24, 742–746 (2022).
Herring, R. A. et al. Metabolic effects of an SGLT2 inhibitor (dapagliflozin) during a period of acute insulin withdrawal and development of ketoacidosis in people with type 1 diabetes. Diabetes Care 43, 2128–2136 (2020).
Hampp, C. et al. Use of sodium-glucose cotransporter 2 inhibitors in patients with type 1 diabetes and rates of diabetic ketoacidosis. Diabetes Care 43, 90–97 (2020).
Musso, G., Sircana, A., Saba, F., Cassader, M. & Gambino, R. Assessing the risk of ketoacidosis due to sodium-glucose cotransporter (SGLT)-2 inhibitors in patients with type 1 diabetes: a meta-analysis and meta-regression. PLoS Med. 17, e1003461 (2020).
Shimoda, M. et al. Efficacy and safety of adding ipragliflozin to insulin in Japanese patients with type 1 diabetes mellitus: a retrospective study. Endocr. J. 68, 1455–1461 (2021).
Curran, M. et al. The benefits of physical exercise for the health of the pancreatic β-cell: a review of the evidence. Exp. Physiol. 105, 579–589 (2020).
Choi, S. B., Jang, J. S., Hong, S. M., Jun, D. W. & Park, S. Exercise and dexamethasone oppositely modulate β-cell function and survival via independent pathways in 90% pancreatectomized rats. J. Endocrinol. 190, 471–482 (2006).
Kiraly, M. A. et al. Attenuation of type 2 diabetes mellitus in the male Zucker diabetic fatty rat: the effects of stress and non-volitional exercise. Metabolism 56, 732–744 (2007).
Paula, F. M. M. et al. Exercise training protects human and rodent β cells against endoplasmic reticulum stress and apoptosis. FASEB J. 32, 1524–1536 (2018).
Choi, S. B., Jang, J. S. & Park, S. Estrogen and exercise may enhance β-cell function and mass via insulin receptor substrate 2 induction in ovariectomized diabetic rats. Endocrinology 146, 4786–4794 (2005).
Huang, H. H. et al. Exercise increases insulin content and basal secretion in pancreatic islets in type 1 diabetic mice. Exp. Diabetes Res. 2011, 481427 (2011).
AbouAssi, H. et al. The effects of aerobic, resistance, and combination training on insulin sensitivity and secretion in overweight adults from STRRIDE AT/RT: a randomized trial. J. Appl. Physiol. 118, 1474–1482 (2015).
Coomans de Brachène, A. et al. Exercise as a non-pharmacological intervention to protect pancreatic beta cells in individuals with type 1 and type 2 diabetes. Diabetologia 66, 450–460 (2022).
Jackness, C. et al. Very low-calorie diet mimics the early beneficial effect of Roux-en-Y gastric bypass on insulin sensitivity and β-cell function in type 2 diabetic patients. Diabetes 62, 3027–3032 (2013).
Allen, L. A. & Dayan, C. M. Immunotherapy for type 1 diabetes. Br. Med. Bull. 140, 76–90 (2021).
Jacobsen, L. M. et al. Comparing beta cell preservation across clinical trials in recent-onset type 1 diabetes. Diabetes Technol. Ther. 22, 948–953 (2020).
Sims, E. K. et al. Teplizumab improves and stabilizes beta cell function in antibody-positive high-risk individuals. Sci. Transl Med. 13, eabc8980 (2021).
Orban, T. et al. Costimulation modulation with abatacept in patients with recent-onset type 1 diabetes: follow-up 1 year after cessation of treatment. Diabetes Care 37, 1069–1075 (2014).
Ovalle, F. et al. Verapamil and beta cell function in adults with recent-onset type 1 diabetes. Nat. Med. 24, 1108–1112 (2018).
Xu, G. et al. Exploratory study reveals far reaching systemic and cellular effects of verapamil treatment in subjects with type 1 diabetes. Nat. Commun. 13, 1159 (2022).
Quattrin, T. et al. Golimumab and beta-cell function in youth with new-onset type 1 diabetes. N. Engl. J. Med. 383, 2007–2017 (2020).
Hopkinson, H. E., White, A. D., Nightingale, P. & Narendran, P. A novel approach to basal-bolus insulin initiation in adults with newly diagnosed type 1 diabetes: an observational cohort study of a service redesign. Br. J. Diabetes 18, 71–75 (2018).
Boughton, C. K. et al. Closed-loop therapy and preservation of C-peptide secretion in type 1 diabetes. N. Engl. J. Med. 387, 882–893 (2022).
Roberts, C. K., Hevener, A. L. & Barnard, R. J. Metabolic syndrome and insulin resistance: underlying causes and modification by exercise training. Compr. Physiol. https://doi.org/10.1002/cphy.c110062 (2013).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Endocrinology thanks Olga Kordonouri and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tatovic, D., Narendran, P. & Dayan, C.M. A perspective on treating type 1 diabetes mellitus before insulin is needed. Nat Rev Endocrinol 19, 361–370 (2023). https://doi.org/10.1038/s41574-023-00816-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-023-00816-5