Abstract
The liver is often thought of as a single functional unit, but both its structural and functional architecture make it highly multivalent and adaptable. In any given physiological situation, the liver can maintain metabolic homeostasis, conduct appropriate inflammatory responses, carry out endobiotic and xenobiotic transformation and synthesis reactions, as well as store and release multiple bioactive molecules. Moreover, the liver is a very resilient organ. This resilience means that chronic liver diseases can go unnoticed for decades, yet culminate in life-threatening clinical complications once the adaptive capacity of the liver is overwhelmed. Non-alcoholic fatty liver disease (NAFLD) predisposes individuals to cirrhosis and increases liver-related and cardiovascular disease-related mortality. This Review discusses the accumulating evidence of sexual dimorphism in NAFLD, which is currently rarely considered in preclinical and clinical studies. Increased awareness of the mechanistic causes of hepatic sexual dimorphism could lead to improved understanding of the biological processes that are dysregulated in NAFLD, to the identification of relevant therapeutic targets and to improved risk stratification of patients with NAFLD undergoing therapeutic intervention.
Key points
-
Liver pathophysiology is sexually dimorphic.
-
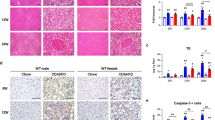
Male individuals predominantly show more severe stages of non-alcoholic fatty liver, non-alcoholic steatohepatitis (NASH) and fibrosis than do female individuals.
-
Individual variations in humans require large cohorts to identify sex-specific features of non-alcoholic fatty liver disease (NAFLD).
-
Preclinical and clinical investigations of NAFLD and of fibrosis rarely consider sex as a biological variable.
-
The time (and technologies) are ripe for investigating the sexual dimorphism of liver diseases in time and space.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Schork, N. J. Personalized medicine: time for one-person trials. Nature 520, 609–611 (2015).
Prendergast, B. J., Onishi, K. G. & Zucker, I. Female mice liberated for inclusion in neuroscience and biomedical research. Neurosci. Biobehav. Rev. 40, 1–5 (2014).
Woitowich, N. C. & Woodruff, T. K. Opinion: research community needs to better appreciate the value of sex-based research. Proc. Natl Acad. Sci. USA 116, 7154–7156 (2019).
Lam, C. S. P. et al. Sex differences in heart failure. Eur. Heart J. 40, 3859–3868 (2019).
Gannon, M., Kulkarni, R. N., Tse, H. M. & Mauvais-Jarvis, F. Sex differences underlying pancreatic islet biology and its dysfunction. Mol. Metab. 15, 82–91 (2018).
Rubin, J. B. et al. Sex differences in cancer mechanisms. Biol. Sex. Differ. 11, 17–46 (2020).
Kokturk, N., Kilic, H., Baha, A., Lee, S. D. & Jones, P. W. Sex difference in chronic obstructive lung disease. Does it matter? A concise review. COPD 13, 799–806 (2016).
Mauvais-Jarvis, F. et al. Sex and gender: modifiers of health, disease, and medicine. Lancet 396, 565–582 (2020).
Lonardo, A. & Suzuki, A. Sexual dimorphism of NAFLD in adults. Focus on clinical aspects and implications for practice and translational research. J. Clin. Med. 9, 1278–1307 (2020).
Ludwig, J., Viggiano, T. R., McGill, D. B. & Oh, B. J. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin. Proc. 55, 434–438 (1980).
Younossi, Z. M. et al. From NAFLD to MAFLD: implications of a premature change in terminology. Hepatology https://doi.org/10.1002/hep.31420 (2020).
Eslam, M. et al. MAFLD: a consensus-driven proposed nomenclature for metabolic associated fatty liver disease. Gastroenterology 158, 1999–2014 (2020).
James, O. F. & Day, C. P. Non-alcoholic steatohepatitis (NASH): a disease of emerging identity and importance. J. Hepatol. 29, 495–501 (1998).
James, O. & Day, C. Non-alcoholic steatohepatitis: another disease of affluence. Lancet 353, 1634–1636 (1999).
Dowman, J. K., Tomlinson, J. W. & Newsome, P. N. Pathogenesis of non-alcoholic fatty liver disease. QJM 103, 71–83 (2010).
Tilg, H., Adolph, T. E. & Moschen, A. R. Multiple parallel hits hypothesis in NAFLD — revisited after a decade. Hepatology 73, 833–842 (2021).
Kolodziejczyk, A. A., Zheng, D., Shibolet, O. & Elinav, E. The role of the microbiome in NAFLD and NASH. EMBO Mol. Med. 11, e9302 (2019).
Eslam, M., Valenti, L. & Romeo, S. Genetics and epigenetics of NAFLD and NASH: clinical impact. J. Hepatol. 68, 268–279 (2018).
Younossi, Z. et al. Global burden of NAFLD and NASH: trends, predictions, risk factors and prevention. Nat. Rev. Gastroenterol. Hepatol. 15, 11–20 (2018).
Sheka, A. C. et al. Nonalcoholic steatohepatitis: a review. JAMA 323, 1175–1183 (2020).
Ye, Q. et al. Global prevalence, incidence, and outcomes of non-obese or lean non-alcoholic fatty liver disease: a systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 5, 739–752 (2020).
Chen, F. et al. Lean NAFLD: a distinct entity shaped by differential metabolic adaptation. Hepatology 71, 1213–1227 (2019).
Chang, Y. et al. Metabolically healthy obesity and the development of nonalcoholic fatty liver disease. Am. J. Gastroenterol. 111, 1133–1140 (2016).
Younossi, Z. M. et al. The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: a systematic review and meta-analysis. J. Hepatol. 71, 793–801 (2019).
Jarvis, H. et al. Metabolic risk factors and incident advanced liver disease in non-alcoholic fatty liver disease (NAFLD): a systematic review and meta-analysis of population-based observational studies. PLoS Med. 17, e1003100 (2020).
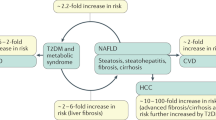
Targher, G., Byrne, C. D. & Tilg, H. NAFLD and increased risk of cardiovascular disease: clinical associations, pathophysiological mechanisms and pharmacological implications. Gut 69, 1691–1705 (2020).
Hagstrom, H. et al. Fibrosis stage but not NASH predicts mortality and time to development of severe liver disease in biopsy-proven NAFLD. J. Hepatol. 67, 1265–1273 (2017).
Deprince, A., Haas, J. T. & Staels, B. Dysregulated lipid metabolism links NAFLD to cardiovascular disease. Mol. Metab. 42, 101092 (2020).
Haas, J. T., Francque, S. & Staels, B. Pathophysiology and mechanisms of nonalcoholic fatty liver disease. Annu. Rev. Physiol. 78, 181–205 (2016).
Schwabe, R. F., Tabas, I. & Pajvani, U. B. Mechanisms of fibrosis development in NASH. Gastroenterology 158, 1913–1928 (2020).
Smith, G. I. et al. Insulin resistance drives hepatic de novo lipogenesis in nonalcoholic fatty liver disease. J. Clin. Invest. 130, 1453–1460 (2019).
Goossens, G. H., Jocken, J. W. E. & Blaak, E. E. Sexual dimorphism in cardiometabolic health: the role of adipose tissue, muscle and liver. Nat. Rev. Endocrinol. 17, 47–66 (2021).
Romero, F. A., Jones, C. T., Xu, Y., Fenaux, M. & Halcomb, R. L. The race to bash NASH: emerging targets and drug development in a complex liver disease. J. Med. Chem. 63, 5031–5073 (2020).
Jensen, V. S. et al. Variation in diagnostic NAFLD/NASH read-outs in paired liver samples from rodent models. J. Pharmacol. Toxicol. Methods 101, 106651–106660 (2020).
Rinella, M. E. et al. Report on the AASLD/EASL joint workshop on clinical trial endpoints in NAFLD. J. Hepatol. 71, 823–833 (2019).
Ren, C. & Sylvia, K. E. Sexual dimorphism in the gut microbiome. IU J. Undergrad. Res. 4, 12–16 (2018).
Jaillon, S., Berthenet, K. & Garlanda, C. Sexual dimorphism in innate immunity. Clin. Rev. Allergy Immunol. 56, 308–321 (2019).
Roy, A. K. & Chatterjee, B. Sexual dimorphism in the liver. Annu. Rev. Physiol. 45, 37–50 (1983).
Klaassen, C. D. & Aleksunes, L. M. Xenobiotic, bile acid, and cholesterol transporters: function and regulation. Pharmacol. Rev. 62, 1–96 (2010).
Della Torre, S. & Maggi, A. Sex differences: a resultant of an evolutionary pressure? Cell Metab. 25, 499–505 (2017).
Maggi, A. & Della Torre, S. Sex, metabolism and health. Mol. Metab. 15, 3–7 (2018).
Justo, R. et al. Gender dimorphism in rat liver mitochondrial oxidative metabolism and biogenesis. Am. J. Physiol. Cell Physiol. 289, C372–C378 (2005).
Weger, B. D. et al. The mouse microbiome is required for sex-specific diurnal rhythms of gene expression and metabolism. Cell Metab. 29, 1–21 (2018).
Jansson, J. O., Edén, S. & Isaksson, O. Sexual dimorphism in the control of growth hormone secretion. Endocr. Rev. 6, 128–150 (1985).
Waxman, D. J. & Holloway, M. G. Sex differences in the expression of hepatic drug metabolizing enzymes. Mol. Pharmacol. 76, 215–228 (2009).
Davey, H. W., Wilkins, R. J. & Waxman, D. J. STAT5 signaling in sexually dimorphic gene expression and growth patterns. Am. J. Hum. Genet. 65, 959–965 (1999).
Waxman, D. J. & O’Connor, C. Growth hormone regulation of sex-dependent liver gene expression. Mol. Endocrinol. 20, 2613–2629 (2006).
Zheng, D., Wang, X., Antonson, P., Gustafsson, J. & Li, Z. Genomics of sex hormone receptor signaling in hepatic sexual dimorphism. Mol. Cell Endocrinol. 471, 33–41 (2018).
Rando, G. & Wahli, W. Sex differences in nuclear receptor-regulated liver metabolic pathways. Biochim. Biophys. Acta 1812, 964–973 (2011).
Yang, X., Zhang, Y. K., Esterly, N., Klaassen, C. D. & Wan, Y. J. Gender disparity of hepatic lipid homoeostasis regulated by the circadian clock. J. Biochem. 145, 609–623 (2009).
Qian, J. et al. Sex differences in the circadian misalignment effects on energy regulation. Proc. Natl Acad. Sci. USA 116, 23806–23812 (2019).
Sheng, L. et al. Gender differences in bile acids and microbiota in relationship with gender dissimilarity in steatosis induced by diet and FXR inactivation. Sci. Rep. 7, 1748 (2017).
Chiang, J. Y. L. Linking sex differences in non-alcoholic fatty liver disease to bile acid signaling, gut microbiota, and high fat diet. Am. J. Pathol. 187, 1658–1659 (2017).
Jena, P. K. et al. Western diet-induced dysbiosis in farnesoid X receptor knockout mice causes persistent hepatic inflammation after antibiotic treatment. Am. J. Pathol. 187, 1800–1813 (2017).
Giles, D. A. et al. Thermoneutral housing exacerbates nonalcoholic fatty liver disease in mice and allows for sex-independent disease modeling. Nat. Med. 23, 829–838 (2017).
Nobs, S. P., Tuganbaev, T. & Elinav, E. Microbiome diurnal rhythmicity and its impact on host physiology and disease risk. EMBO Rep. 20, e47129 (2019).
Fernandez-Perez, L. et al. in Chemistry and Biological Activity of Steroids (eds Ribeiro Salvador, J. A. & Cruz Silva, M. M.) Ch.4 (Intechopen, 2019).
Jansson, J. O. & Frohman, L. A. Differential effects of neonatal and adult androgen exposure on the growth hormone secretory pattern in male rats. Endocrinology 120, 1551–1557 (1987).
Reizel, Y. et al. Gender-specific postnatal demethylation and establishment of epigenetic memory. Genes Dev. 29, 923–933 (2015).
Hosui, A. & Hennighausen, L. Genomic dissection of the cytokine-controlled STAT5 signaling network in liver. Physiol. Genomics 34, 135–143 (2008).
Clodfelter, K. H. et al. Sex-dependent liver gene expression is extensive and largely dependent upon signal transducer and activator of transcription 5b (STAT5b): STAT5b-dependent activation of male genes and repression of female genes revealed by microarray analysis. Mol. Endocrinol. 20, 1333–1351 (2006).
Lau-Corona, D., Suvorov, A. & Waxman, D. J. Feminization of male mouse liver by persistent growth hormone stimulation: activation of sex-biased transcriptional networks and dynamic changes in chromatin states. Mol. Cell Biol. 37, e00301–e00317 (2017).
Palmisano, B. T., Zhu, L. & Stafford, J. M. Role of estrogens in the regulation of liver lipid metabolism. Adv. Exp. Med. Biol. 1043, 227–256 (2017).
Mittendorfer, B. Sexual dimorphism in human lipid metabolism. J. Nutr. 135, 681–686 (2005).
Phelps, T., Snyder, E., Rodriguez, E., Child, H. & Harvey, P. The influence of biological sex and sex hormones on bile acid synthesis and cholesterol homeostasis. Biol. Sex. Differ. 10, 52–64 (2019).
Koulouri, O., Ostberg, J. & Conway, G. S. Liver dysfunction in Turner’s syndrome: prevalence, natural history and effect of exogenous oestrogen. Clin. Endocrinol. 69, 306–310 (2008).
Tramunt, B. et al. Sex differences in metabolic regulation and diabetes susceptibility. Diabetologia 63, 453–461 (2020).
Shen, M. & Shi, H. Sex hormones and their receptors regulate liver energy homeostasis. Int. J. Endocrinol. 2015, 294278 (2015).
Grossmann, M., Wierman, M. E., Angus, P. & Handelsman, D. J. Reproductive endocrinology of nonalcoholic fatty liver disease. Endocr. Rev. 40, 417–446 (2019).
Sharma, G. & Prossnitz, E. R. G-protein-coupled estrogen receptor (GPER) and sex-specific metabolic homeostasis. Adv. Exp. Med. Biol. 1043, 427–453 (2017).
Meda, C. et al. Hepatic ERα accounts for sex differences in the ability to cope with an excess of dietary lipids. Mol. Metab. 32, 97–108 (2020).
Anderson, S. T. & Fitzgerald, G. A. Sexual dimorphism in body clocks. Science 369, 1164–1165 (2020).
Saran, A. R., Dave, S. & Zarrinpar, A. Circadian rhythms in the pathogenesis and treatment of fatty liver disease. Gastroenterology 158, 1948–1966 (2020).
Mukherji, A., Bailey, S. M., Staels, B. & Baumert, T. F. The circadian clock and liver function in health and disease. J. Hepatol. 71, 200–211 (2019).
Stols-Goncalves, D., Hovingh, G. K., Nieuwdorp, M. & Holleboom, A. G. NAFLD and atherosclerosis: two sides of the same dysmetabolic coin? Trends Endocrinol. Metab. 30, 891–902 (2019).
Anstee, Q. M., Reeves, H. L., Kotsiliti, E., Govaere, O. & Heikenwalder, M. From NASH to HCC: current concepts and future challenges. Nat. Rev. Gastroenterol. Hepatol. 16, 411–428 (2019).
Victor, R. G. et al. The Dallas Heart Study: a population-based probability sample for the multidisciplinary study of ethnic differences in cardiovascular health. Am. J. Cardiol. 93, 1473–1480 (2004).
Browning, J. D. et al. Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity. Hepatology 40, 1387–1395 (2004).
Guerrero, R., Vega, G. L., Grundy, S. M. & Browning, J. D. Ethnic differences in hepatic steatosis: an insulin resistance paradox? Hepatology 49, 791–801 (2009).
Romeo, S. et al. Genetic variation in PNPLA3 confers susceptibility to nonalcoholic fatty liver disease. Nat. Genet. 40, 1461–1465 (2008).
Mashek, D. G. Hepatic lipid droplets: a balancing act between energy storage and metabolic dysfunction in NAFLD. Mol. Metab. https://doi.org/10.1016/j.molmet.2020.101115 (2020).
Trepo, E. & Valenti, L. Update on NAFLD genetics: from new variants to the clinic. J. Hepatol. 72, 1196–1209 (2020).
Loomba, R. et al. Heritability of hepatic fibrosis and steatosis based on a prospective twin study. Gastroenterology 149, 1784–1793 (2015).
Verrijken, A. et al. A gene variant of PNPLA3, but not of APOC3, is associated with histological parameters of NAFLD in an obese population. Obesity 21, 2138–2145 (2013).
Parisinos, C. A. et al. Genome-wide and Mendelian randomisation studies of liver MRI yield insights into the pathogenesis of steatohepatitis. J. Hepatol. 73, 241–325 (2020).
Petaja, E. M. & Yki-Jarvinen, H. Definitions of normal liver fat and the association of insulin sensitivity with acquired and genetic NAFLD—a systematic review. Int. J. Mol. Sci. 17, 633–649 (2016).
Yki-Jarvinen, H. & Luukkonen, P. K. Heterogeneity of non-alcoholic fatty liver disease. Liver Int. 35, 2498–2500 (2015).
Stender, S. et al. Adiposity amplifies the genetic risk of fatty liver disease conferred by multiple loci. Nat. Genet. 49, 842–847 (2017).
Ballestri, S. et al. NAFLD as a sexual dimorphic disease: role of gender and reproductive status in the development and progression of nonalcoholic fatty liver disease and inherent cardiovascular risk. Adv. Ther. 34, 1291–1326 (2017).
Beaudry, K. M. & Devries, M. C. Sex-based differences in hepatic and skeletal muscle triglyceride storage and metabolism. Appl. Physiol. Nutr. Metab. 44, 805–813 (2019).
Tian, G. X. et al. Oestradiol is a protective factor for non-alcoholic fatty liver disease in healthy men. Obes. Rev. 13, 381–387 (2012).
Cai, M. J., Kong, X. N. & Zhao, X. Y. Influences of gender and age on the prevalence and complications of nonalcoholic fatty liver disease. Zhongguo Yi Xue Ke Xue Yuan Xue Bao 39, 499–505 (2017).
Younossi, Z. M. et al. Global epidemiology of nonalcoholic fatty liver disease—meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 64, 73–84 (2016).
Ribeiro, A., Igual-Perez, M. J., Santos Silva, E. & Sokal, E. M. Childhood fructoholism and fructoholic liver disease. Hepatol. Commun. 3, 44–51 (2019).
Nobili, V., Alisi, A., Newton, K. P. & Schwimmer, J. B. Comparison of the phenotype and approach to pediatric vs adult patients with nonalcoholic fatty liver disease. Gastroenterology 150, 1798–1810 (2016).
Mueller, N. T. et al. Sex hormone relations to histologic severity of pediatric nonalcoholic fatty liver disease. J. Clin. Endocrinol. Metab. 105, 3496–3504 (2020).
Hatton, G., Alterio, T., Nobili, V. & Mann, J. P. Unmet needs in pediatric NAFLD research: what do we need to prioritize for the future? Expert. Rev. Gastroenterol. Hepatol. 12, 961–967 (2018).
Arun, J., Clements, R. H., Lazenby, A. J., Leeth, R. R. & Abrams, G. A. The prevalence of nonalcoholic steatohepatitis is greater in morbidly obese men compared to women. Obes. Surg. 16, 1351–1358 (2006).
Skubic, C., Drakulić, Ž. & Rozman, D. Personalized therapy when tackling nonalcoholic fatty liver disease: a focus on sex, genes, and drugs. Expert Opin. Drug Metab. Toxicol. 14, 831–841 (2018).
Lonardo, A. et al. Sex differences in NAFLD: state of the art and identification of research gaps. Hepatology 70, 1457–1469 (2019).
Petrick, J. L. et al. International trends in hepatocellular carcinoma incidence, 1978–2012. Int. J. Cancer 147, 317–330 (2020).
El-Serag, H. B. & Rudolph, K. L. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology 132, 2557–2576 (2007).
Durazzo, M. et al. Gender specific medicine in liver diseases: a point of view. World J. Gastroenterol. 20, 2127–2135 (2014).
Yang, J. D. et al. Gender and menopause impact severity of fibrosis among patients with nonalcoholic steatohepatitis. Hepatology 59, 1406–1414 (2014).
Klair, J. S. et al. A longer duration of estrogen deficiency increases fibrosis risk among postmenopausal women with nonalcoholic fatty liver disease. Hepatology 64, 85–91 (2016).
Turola, E. et al. Ovarian senescence increases liver fibrosis in humans and zebrafish with steatosis. Dis. Model. Mech. 8, 1037–1046 (2015).
Lee, C., Kim, J. & Jung, Y. Potential therapeutic application of estrogen in gender disparity of nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. Cells 8, 1259–1279 (2019).
Kumarendran, B. et al. Polycystic ovary syndrome, androgen excess, and the risk of nonalcoholic fatty liver disease in women: a longitudinal study based on a United Kingdom primary care database. PLoS Med. 15, e1002542–e1002562 (2018).
Ali, M. A. et al. Nonalcoholic steatohepatitis-related hepatocellular carcinoma: is there a role for the androgen receptor pathway? Onco Targets Ther. 10, 1403–1412 (2017).
Negro, F. Natural history of NASH and HCC. Liver Int. 40 (Suppl. 1), 72–76 (2020).
Altayar, O., Noureddin, N., Thanda Han, M. A., Murad, M. H. & Noureddin, M. Fibrosis changes in the placebo arm of NASH clinical trials. Clin. Gastroenterol. Hepatol. 17, 2387 (2019).
Singh, S. et al. Fibrosis progression in nonalcoholic fatty liver vs nonalcoholic steatohepatitis: a systematic review and meta-analysis of paired-biopsy studies. Clin. Gastroenterol. Hepatol. 13, 643–654 (2015).
Angulo, P. et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology 149, 389–397 (2015).
Ekstedt, M. et al. Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology 61, 1547–1554 (2015).
Dulai, P. S. et al. Increased risk of mortality by fibrosis stage in nonalcoholic fatty liver disease: systematic review and meta-analysis. Hepatology 65, 1557–1565 (2017).
Ramachandran, P. et al. Resolving the fibrotic niche of human liver cirrhosis at single-cell level. Nature 575, 512–518 (2019).
Moylan, C. A. et al. Hepatic gene expression profiles differentiate presymptomatic patients with mild versus severe nonalcoholic fatty liver disease. Hepatology 59, 471–482 (2014).
Arendt, B. M. et al. Altered hepatic gene expression in nonalcoholic fatty liver disease is associated with lower hepatic ω-3 and ω-6 polyunsaturated fatty acids. Hepatology 61, 1565–1578 (2015).
Lefebvre, P. et al. Interspecies NASH disease activity whole-genome profiling identifies a fibrogenic role of PPARα-regulated dermatopontin. JCI Insight 2, 92264–92281 (2017).
Haas, J. T. et al. Transcriptional network analysis implicates altered hepatic immune function in NASH development and resolution. Nat. Metab. 1, 604–614 (2019).
Suppli, M. P. et al. Hepatic transcriptome signatures in patients with varying degrees of nonalcoholic fatty liver disease compared with healthy normal-weight individuals. Am. J. Physiol. Gastrointest. Liver Physiol. 316, G462–G472 (2019).
Gerhard, G. S. et al. Transcriptomic profiling of obesity-related nonalcoholic steatohepatitis reveals a core set of fibrosis-specific genes. J. Endocr. Soc. 2, 710–726 (2018).
Ratziu, V. Back to Byzance: querelles byzantines over NASH and fibrosis. J. Hepatol. 67, 1134–1136 (2017).
Chen, W. et al. Multi-transcriptome analyses reveal prioritized genes specifically associated with liver fibrosis progression independent of etiology. Am. J. Physiol. Gastrointest. Liver Physiol. 316, G744–G754 (2019).
Vandel, J. et al. Hepatic molecular signatures highlight the sexual dimorphism of non-alcoholic steatohepatitis (NASH). Hepatology 73, 920–936 (2020).
Zhang, Y. et al. Transcriptional profiling of human liver identifies sex-biased genes associated with polygenic dyslipidemia and coronary artery disease. PLoS ONE 6, e23506–e23522 (2011).
Puri, P. et al. The plasma lipidomic signature of nonalcoholic steatohepatitis. Hepatology 50, 1827–1838 (2009).
Loomba, R., Quehenberger, O., Armando, A. & Dennis, E. A. Polyunsaturated fatty acid metabolites as novel lipidomic biomarkers for noninvasive diagnosis of nonalcoholic steatohepatitis. J. Lipid Res. 56, 185–192 (2015).
Gaggini, M. et al. Altered amino acid concentrations in NAFLD: impact of obesity and insulin resistance. Hepatology 67, 145–158 (2017).
van den Berg, E. H. et al. Non-alcoholic fatty liver disease and risk of incident type 2 diabetes: role of circulating branched-chain amino acids. Nutrients 11, 705–720 (2019).
Lake, A. D. et al. Branched chain amino acid metabolism profiles in progressive human nonalcoholic fatty liver disease. Amino Acids 47, 603–615 (2015).
Hoyles, L. et al. Molecular phenomics and metagenomics of hepatic steatosis in non-diabetic obese women. Nat. Med. 24, 1070–1080 (2018).
Grzych, G. et al. Plasma BCAA changes in patients with NAFLD are sex dependent. J. Clin. Endocrinol. Metab. 105, 2311–2321 (2020).
Terakura, D. et al. Preventive effects of branched-chain amino acid supplementation on the spontaneous development of hepatic preneoplastic lesions in C57BL/KsJ-db/db obese mice. Carcinogenesis 33, 2499–2506 (2012).
Miyake, T. et al. Long-term branched-chain amino acid supplementation improves glucose tolerance in patients with nonalcoholic steatohepatitis-related cirrhosis. Int. Med. 51, 2151–2155 (2012).
Canfora, E. E., Meex, R. C. R., Venema, K. & Blaak, E. E. Gut microbial metabolites in obesity, NAFLD and T2DM. Nat. Rev. Endocrinol. 15, 261–273 (2019).
Pietrangelo, A. Iron in NASH, chronic liver diseases and HCC: how much iron is too much? J. Hepatol. 50, 249–251 (2009).
Handa, P. et al. Iron overload results in hepatic oxidative stress, immune cell activation, and hepatocellular ballooning injury, leading to nonalcoholic steatohepatitis in genetically obese mice. Am. J. Physiol. Gastrointest. Liver Physiol. 310, G117–G127 (2016).
Margerie, D. et al. Hepatic transcriptomic signatures of statin treatment are associated with impaired glucose homeostasis in severely obese patients. BMC Med. Genomics 12, 80 (2019).
Clocchiatti, A., Cora, E., Zhang, Y. & Dotto, G. P. Sexual dimorphism in cancer. Nat. Rev. Cancer 16, 330–339 (2016).
Natri, H. M., Wilson, M. A. & Buetow, K. H. Distinct molecular etiologies of male and female hepatocellular carcinoma. BMC Cancer 19, 951–963 (2019).
Forner, A., Reig, M. & Bruix, J. Hepatocellular carcinoma. Lancet 391, 1301–1314 (2018).
Reddy, S. K. et al. Outcomes of curative treatment for hepatocellular cancer in nonalcoholic steatohepatitis versus hepatitis C and alcoholic liver disease. Hepatology 55, 1809–1819 (2012).
Yasui, K. et al. Characteristics of patients with nonalcoholic steatohepatitis who develop hepatocellular carcinoma. Clin. Gastroenterol. Hepatol. 9, 428–433 (2011).
Gupta, R. K. & Kaestner, K. H. HNF-4α: from MODY to late-onset type 2 diabetes. Trends Mol. Med. 10, 521–524 (2004).
Lau, H. H., Ng, N. H. J., Loo, L. S. W., Jasmen, J. B. & Teo, A. K. K. The molecular functions of hepatocyte nuclear factors — in and beyond the liver. J. Hepatol. 68, 1033–1048 (2018).
Fekry, B. et al. HNF4α-deficient fatty liver provides a permissive environment for sex-independent hepatocellular carcinoma. Cancer Res. 79, 5860–5873 (2019).
Varlamov, O., Bethea, C. L. & Roberts, C. T. Jr. Sex-specific differences in lipid and glucose metabolism. Front. Endocrinol. 5, 241–248 (2014).
Aldhoon-Hainerova, I. et al. Glucose homeostasis and insulin resistance: prevalence, gender differences and predictors in adolescents. Diabetol. Metab. Syndr. 6, 100–109 (2014).
Weger, B. D., Rawashdeh, O. & Gachon, F. At the intersection of microbiota and circadian clock: are sexual dimorphism and growth hormones the missing link to pathology? Circadian clock and microbiota: potential effect on growth hormone and sexual development. Bioessays 41, e1900059 (2019).
Bloor, I. D. & Symonds, M. E. Sexual dimorphism in white and brown adipose tissue with obesity and inflammation. Horm. Behav. 66, 95–103 (2014).
Moreira-Pais, A. et al. Sex differences on adipose tissue remodeling: from molecular mechanisms to therapeutic interventions. J. Mol. Med. 98, 483–493 (2020).
Korf, H., Boesch, M., Meelberghs, L. & van der Merwe, S. Macrophages as key players during adipose tissue-liver crosstalk in nonalcoholic fatty liver disease. Semin. Liver Dis. 39, 291–300 (2019).
Van Herck, M. A. et al. The differential roles of T cells in non-alcoholic fatty liver disease and obesity. Front. Immunol. 10, 82–102 (2019).
Vasanthakumar, A. et al. Sex-specific adipose tissue imprinting of regulatory T cells. Nature 579, 581–585 (2020).
Remmerie, A., Martens, L. & Scott, C. L. Macrophage subsets in obesity, aligning the liver and adipose tissue. Front. Endocrinol. 11, 259–264 (2020).
Brier, A. B. et al. The KDM5 family is required for activation of pro-proliferative cell cycle genes during adipocyte differentiation. Nucleic Acids Res. 45, 1743–1759 (2016).
Link, J. C. et al. X chromosome dosage of histone demethylase KDM5C determines sex differences in adiposity. J. Clin. Invest. 130, 5688–5701 (2020).
Kaltenecker, D. et al. Hepatic growth hormone–JAK2–STAT5 signalling: metabolic function, non-alcoholic fatty liver disease and hepatocellular carcinoma progression. Cytokine 124, 154569–154581 (2019).
Wattacheril, J. et al. Non-alcoholic fatty liver disease phosphoproteomics: a functional piece of the precision puzzle. Hepatol. Res. 47, 1469–1483 (2017).
Kurt, Z. et al. Tissue-specific pathways and networks underlying sexual dimorphism in non-alcoholic fatty liver disease. Biol. Sex. Differ. 9, 46–60 (2018).
Cvitanović Tomaš, T., Urlep, Ž., Moškon, M., Mraz, M. & Rozman, D. LiverSex computational model: sexual aspects in hepatic metabolism and abnormalities. Front. Physiol. 9, 360–372 (2018).
Haczeyni, F. et al. Mouse models of non-alcoholic steatohepatitis: a reflection on recent literature. J. Gastroenterol. Hepatol. 33, 1312–1320 (2018).
Acknowledgements
The authors’ research work was supported by grants from Agence Nationale pour la Recherche (ANR-16-RHUS-0006-PreciNASH and ANR-10-LBEX-46 to P.L. and B.S.), the European Union (FP6 Hepadip FP6-018734 and FP7 Resolve FP7-305707 to B.S.), Fondation de France (grant 2014 00047965 to P.L.), Fondation pour la Recherche Médicale (Equipe labellisée DEQ20150331724 to P.L.). B.S. is a recipient of an Advanced ERC Grant (694717).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Endocrinology thanks L. Valenti, J. Sethi, who co-reviewed with J. Bilson, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Expandability
-
The capacity of adipocytes to proliferate and differentiate, particularly in a metabolically challenging environment.
- Partially refractory
-
A general phenomenon in receptor biology whereby continuous activation of a signalling pathway renders it less active and resistant to further stimulation.
- Pioneer transcription factors
-
Transcription factors that are able to install an open chromatin state, thus opening new methods or ways to regulate transcription.
- X-escape genes
-
Genes on the inactivated X chromosome that are nonetheless transcribed to produce RNA levels ≥10% of the expression of their counterparts on the active X chromosome.
Rights and permissions
About this article
Cite this article
Lefebvre, P., Staels, B. Hepatic sexual dimorphism — implications for non-alcoholic fatty liver disease. Nat Rev Endocrinol 17, 662–670 (2021). https://doi.org/10.1038/s41574-021-00538-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-021-00538-6
This article is cited by
-
A genetic mouse model of lean-NAFLD unveils sexual dimorphism in the liver-heart axis
Communications Biology (2024)
-
The hepato-ovarian axis: genetic evidence for a causal association between non-alcoholic fatty liver disease and polycystic ovary syndrome
BMC Medicine (2023)
-
Sex differences in glycolipidic disorders after exposure to maternal hyperglycemia during early development
Journal of Endocrinological Investigation (2023)
-
The cross-talk between leptin and circadian rhythm signaling proteins in physiological processes: a systematic review
Molecular Biology Reports (2023)
-
Impact of metabolic syndrome and metabolic dysfunction-associated fatty liver disease on cardiovascular risk by the presence or absence of type 2 diabetes and according to sex
Cardiovascular Diabetology (2022)