Abstract
Evidence from observational studies and randomized trials suggests that prediabetes and type 2 diabetes mellitus (T2DM) can develop in genetically susceptible individuals in parallel with weight (that is, fat) gain. Accordingly, studies show that weight loss can produce remission of T2DM in a dose-dependent manner. A weight loss of ~15 kg, achieved by calorie restriction as part of an intensive management programme, can lead to remission of T2DM in ~80% of patients with obesity and T2DM. However, long-term weight loss maintenance is challenging. Obesity and T2DM are associated with diminished glucose uptake in the brain that impairs the satiating effect of dietary carbohydrate; therefore, carbohydrate restriction might help maintain weight loss and maximize metabolic benefits. Likewise, increases in physical activity and fitness are an important contributor to T2DM remission when combined with calorie restriction and weight loss. Preliminary studies suggest that a precision dietary management approach that uses pretreatment glycaemic status to stratify patients can help optimize dietary recommendations with respect to carbohydrate, fat and dietary fibre. This approach might lead to improved weight loss maintenance and glycaemic control. Future research should focus on better understanding the individual response to dietary treatment and translating these findings into clinical practice.
Key points
-
Studies show that weight loss can produce remission of type 2 diabetes mellitus (T2DM) in a dose-dependent manner.
-
In patients with T2DM and obesity, weight loss of ~15 kg, achieved by an intensive management programme involving calorie restriction, can lead to remission of T2DM in ~80% of individuals.
-
Long-term maintenance of weight loss and metabolic health in people who have undergone intensive lifestyle intervention is challenging.
-
Carbohydrate restriction might help maintain weight loss and maximize metabolic benefits.
-
When combined with calorie restriction and weight loss, increases in physical activity and fitness are an important contributor to T2DM remission.
-
Preliminary work suggests that pretreatment glycaemic status could be used to stratify patients in order to optimize dietary recommendations.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
International Diabetes Federation. IDF Diabetes Atlas 9th edn (International Diabetes Federation, 2019).
Zhu, Y. et al. Racial/ethnic disparities in the prevalence of diabetes and prediabetes by BMI: patient outcomes research to advance learning (PORTAL) multisite cohort of adults in the U.S. Diabetes Care 42, 2211–2219 (2019).
Magkos, F. Metabolically healthy obesity: what’s in a name? Am. J. Clin. Nutr. 110, 533–539 (2019). A review of the dissociation between excess body weight and metabolic dysfunction.
Willett, W. C., Dietz, W. H. & Colditz, G. A. Guidelines for healthy weight. N. Engl. J. Med. 341, 427–434 (1999).
Prospective Studies Collaboration. Body-mass index and cause-specific mortality in 900,000 adults: collaborative analyses of 57 prospective studies. Lancet 373, 1083–1096 (2009).
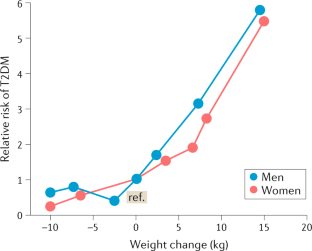
Chan, J. M., Rimm, E. B., Colditz, G. A., Stampfer, M. J. & Willett, W. C. Obesity, fat distribution, and weight gain as risk factors for clinical diabetes in men. Diabetes Care 17, 961–969 (1994).
Colditz, G. A., Willett, W. C., Rotnitzky, A. & Manson, J. E. Weight gain as a risk factor for clinical diabetes mellitus in women. Ann. Intern. Med. 122, 481–486 (1995).
Hu, F. B. et al. Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. N. Engl. J. Med. 345, 790–797 (2001).
Kendall, D. M., Cuddihy, R. M. & Bergenstal, R. M. Clinical application of incretin-based therapy: therapeutic potential, patient selection and clinical use. Am. J. Med. 122, S37–S50 (2009).
Mittendorfer, B., Magkos, F., Fabbrini, E., Mohammed, B. S. & Klein, S. Relationship between body fat mass and free fatty acid kinetics in men and women. Obesity 17, 1872–1877 (2009).
Conte, C. et al. Multiorgan insulin sensitivity in lean and obese subjects. Diabetes Care 35, 1316–1321 (2012).
Wilman, H. R. et al. Characterisation of liver fat in the UK Biobank cohort. PLoS One 12, e0172921 (2017).
Pienkowska, J. et al. MRI assessment of ectopic fat accumulation in pancreas, liver and skeletal muscle in patients with obesity, overweight and normal BMI in correlation with the presence of central obesity and metabolic syndrome. Diabetes Metab. Syndr. Obes. 12, 623–636 (2019).
Tabak, A. G. et al. Trajectories of glycaemia, insulin sensitivity, and insulin secretion before diagnosis of type 2 diabetes: an analysis from the Whitehall II study. Lancet 373, 2215–2221 (2009). A prospective study of the temporal changes in metabolic function and glucose control along the natural history of T2DM.
Weir, G. C. & Bonner-Weir, S. Five stages of evolving beta-cell dysfunction during progression to diabetes. Diabetes 53 (Suppl. 3), 16–21 (2004).
Astrup, A. & Finer, N. Redefining type 2 diabetes: ‘diabesity’ or ‘obesity dependent diabetes mellitus’? Obes. Rev. 1, 57–59 (2000).
Leitner, D. R. et al. Obesity and type 2 diabetes: two diseases with a need for combined treatment strategies — EASO can lead the way. Obes. Facts 10, 483–492 (2017).
Sjostrom, L. Review of the key results from the Swedish Obese Subjects (SOS) trial — a prospective controlled intervention study of bariatric surgery. J. Intern. Med. 273, 219–234 (2013).
Jans, A. et al. Duration of type 2 diabetes and remission rates after bariatric surgery in Sweden 2007–2015: a registry-based cohort study. PLoS Med. 16, e1002985 (2019).
Davies, M. J. et al. Efficacy of liraglutide for weight loss among patients with type 2 diabetes: the SCALE diabetes randomized clinical trial. JAMA 314, 687–699 (2015).
Madsbad, S. & Holst, J. J. GLP-1 as a mediator in the remission of type 2 diabetes after gastric bypass and sleeve gastrectomy surgery. Diabetes 63, 3172–3174 (2014).
MacDonald, P. E. et al. The multiple actions of GLP-1 on the process of glucose-stimulated insulin secretion. Diabetes 51 (Suppl. 3), 434–442 (2002).
Magkos, F. et al. Effects of moderate and subsequent progressive weight loss on metabolic function and adipose tissue biology in humans with obesity. Cell Metab. 23, 591–601 (2016). A randomized controlled trial of the effects of progressive diet-induced weight loss on body composition and metabolic function.
Wing, R. R. et al. Long-term effects of modest weight loss in type II diabetic patients. Arch. Intern. Med. 147, 1749–1753 (1987).
Henry, R. R., Wallace, P. & Olefsky, J. M. Effects of weight loss on mechanisms of hyperglycemia in obese non-insulin-dependent diabetes mellitus. Diabetes 35, 990–998 (1986).
Markovic, T. P. et al. The determinants of glycemic responses to diet restriction and weight loss in obesity and NIDDM. Diabetes Care 21, 687–694 (1998).
Henry, R. R., Scheaffer, L. & Olefsky, J. M. Glycemic effects of intensive caloric restriction and isocaloric refeeding in noninsulin-dependent diabetes mellitus. J. Clin. Endocrinol. Metab. 61, 917–925 (1985).
Hughes, T. A., Gwynne, J. T., Switzer, B. R., Herbst, C. & White, G. Effects of caloric restriction and weight loss on glycemic control, insulin release and resistance, and atherosclerotic risk in obese patients with type II diabetes mellitus. Am. J. Med. 77, 7–17 (1984).
Steven, S. & Taylor, R. Restoring normoglycaemia by use of a very low calorie diet in long- and short-duration type 2 diabetes. Diabet. Med. 32, 1149–1155 (2015).
Lim, E. L. et al. Reversal of type 2 diabetes: normalisation of beta cell function in association with decreased pancreas and liver triacylglycerol. Diabetologia 54, 2506–2514 (2011).
Taylor, R. et al. Remission of human type 2 diabetes requires decrease in liver and pancreas fat content but is dependent upon capacity for beta cell recovery. Cell Metab. 28, 547–556.e3 (2018).
Al-Mrabeh, A. et al. Hepatic lipoprotein export and remission of human type 2 diabetes after weight loss. Cell Metab. 31, 233–249 (2020). A prospective study evaluating the potential mechanisms of T2DM remission and relapse following lifestyle modification.
Taylor, R. Pathogenesis of type 2 diabetes: tracing the reverse route from cure to cause. Diabetologia 51, 1781–1789 (2008).
Taylor, R. & Barnes, A. C. Can type 2 diabetes be reversed and how can this best be achieved? James Lind Alliance research priority number one. Diabet. Med. 36, 308–315 (2019).
Brown, A. et al. Low-energy total diet replacement intervention in patients with type 2 diabetes mellitus and obesity treated with insulin: a randomized trial. BMJ Open Diabetes Res. Care 8, e001012 (2020).
Gregg, E. W. et al. Association of an intensive lifestyle intervention with remission of type 2 diabetes. JAMA 308, 2489–2496 (2012).
Annuzzi, G., Rivellese, A. A., Bozzetto, L. & Riccardi, G. The results of Look AHEAD do not row against the implementation of lifestyle changes in patients with type 2 diabetes. Nutr. Metab. Cardiovasc. Dis. 24, 4–9 (2014).
Raynor, H. A. et al. Partial meal replacement plan and quality of the diet at 1 year: action for health in diabetes (Look AHEAD) trial. J. Acad. Nutr. Diet. 115, 731–742 (2015).
Lean, M. E. et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial. Lancet 391, 541–551 (2018).
Lean, M. E. J. et al. Durability of a primary care-led weight-management intervention for remission of type 2 diabetes: 2-year results of the DiRECT open-label, cluster-randomised trial. Lancet Diabetes Endocrinol. 7, 344–355 (2019). A randomized controlled trial of diet-induced weight loss demonstrating that remission of T2DM depends on the amount of weight loss.
Heymsfield, S. B., Gonzalez, M. C., Shen, W., Redman, L. & Thomas, D. Weight loss composition is one-fourth fat-free mass: a critical review and critique of this widely cited rule. Obes. Rev. 15, 310–321 (2014).
DeFronzo, R. A. et al. The effect of insulin on the disposal of intravenous glucose. Results from indirect calorimetry and hepatic and femoral venous catheterization. Diabetes 30, 1000–1007 (1981).
Ferrannini, E. et al. The disposal of an oral glucose load in healthy subjects. A quantitative study. Diabetes 34, 580–588 (1985).
American Diabetes Association. Standards of medical care in diabetes — 2020. Diabetes Care 43, S1–S212 (2020).
Ajala, O., English, P. & Pinkney, J. Systematic review and meta-analysis of different dietary approaches to the management of type 2 diabetes. Am. J. Clin. Nutr. 97, 505–516 (2013).
Hjorth, M. F., Zohar, Y., Hill, J. O. & Astrup, A. Personalized dietary management of overweight and obesity based on measures of insulin and glucose. Annu. Rev. Nutr. 38, 245–272 (2018). A review of evidence supporting baseline glycaemia as a major predictor of weight loss success in response to dietary interventions.
Snorgaard, O., Poulsen, G. M., Andersen, H. K. & Astrup, A. Systematic review and meta-analysis of dietary carbohydrate restriction in patients with type 2 diabetes. BMJ Open. Diabetes Res. Care 5, e000354 (2017).
Kirk, E. et al. Dietary fat and carbohydrates differentially alter insulin sensitivity during caloric restriction. Gastroenterology 136, 1552–1560 (2009).
Wing, R. R. et al. Caloric restriction per se is a significant factor in improvements in glycemic control and insulin sensitivity during weight loss in obese NIDDM patients. Diabetes Care 17, 30–36 (1994).
Look Ahead Research Group. Cardiovascular effects of intensive lifestyle intervention in type 2 diabetes. N. Engl. J. Med. 369, 145–154 (2013).
Sjostrom, L. et al. Association of bariatric surgery with long-term remission of type 2 diabetes and with microvascular and macrovascular complications. JAMA 311, 2297–2304 (2014).
Wing, R. R., Blair, E., Marcus, M., Epstein, L. H. & Harvey, J. Year-long weight loss treatment for obese patients with type II diabetes: does including an intermittent very-low-calorie diet improve outcome? Am. J. Med. 97, 354–362 (1994).
Samkani, A. et al. A carbohydrate-reduced high-protein diet acutely decreases postprandial and diurnal glucose excursions in type 2 diabetes patients. Br. J. Nutr. 119, 910–917 (2018).
Skytte, M. J. et al. A carbohydrate-reduced high-protein diet improves HbA1c and liver fat content in weight stable participants with type 2 diabetes: a randomised controlled trial. Diabetologia 62, 2066–2078 (2019). A cross-over study showing that low-carbohydrate diets can improve metabolic risk factors in patients with T2DM without much weight loss.
Taylor, R., Al-Mrabeh, A. & Sattar, N. Understanding the mechanisms of reversal of type 2 diabetes. Lancet Diabetes Endocrinol. 7, 726–736 (2019). A review of the mechanisms of T2DM remission.
Hellerstein, M. K. De novo lipogenesis in humans: metabolic and regulatory aspects. Eur. J. Clin. Nutr. 53 (Suppl. 1), 53–65 (1999).
van Wyk, H. J., Davis, R. E. & Davies, J. S. A critical review of low-carbohydrate diets in people with type 2 diabetes. Diabet. Med. 33, 148–157 (2016).
Kodama, S. et al. Influence of fat and carbohydrate proportions on the metabolic profile in patients with type 2 diabetes: a meta-analysis. Diabetes Care 32, 959–965 (2009).
Hamdy, O. et al. Fat versus carbohydrate-based energy-restricted diets for weight loss in patients with type 2 diabetes. Curr. Diab Rep. 18, 128 (2018).
Forouhi, N. G., Misra, A., Mohan, V., Taylor, R. & Yancy, W. Dietary and nutritional approaches for prevention and management of type 2 diabetes. BMJ 361, k2234 (2018).
Shan, Z., Guo, Y., Hu, F. B., Liu, L. & Qi, Q. Association of low-carbohydrate and low-fat diets with mortality among US adults. JAMA Intern. Med. 180, 513–523 (2020).
Livesey, G. et al. Dietary glycemic index and load and the risk of type 2 diabetes: a systematic review and updated meta-analyses of prospective cohort studies. Nutrients 11, 1280 (2019).
Livesey, G. et al. Dietary glycemic index and load and the risk of type 2 diabetes: assessment of causal relations. Nutrients 11, 1436 (2019).
Hwang, J. J. et al. Blunted rise in brain glucose levels during hyperglycemia in adults with obesity and T2DM. JCI Insight 2, e95913 (2017). A study showing that patients with obesity and T2DM have a blunted rise in brain blood glucose levels in response to carbohydrate ingestion, and this associates with their feelings of appetite and hunger.
Astrup, A. & Hjorth, M. F. Classification of obesity targeted personalized dietary weight loss management based on carbohydrate tolerance. Eur. J. Clin. Nutr. 72, 1300–1304 (2018).
Frost, G. et al. The short-chain fatty acid acetate reduces appetite via a central homeostatic mechanism. Nat. Commun. 5, 3611 (2014).
Trajkovski, M. & Wollheim, C. B. Physiology: microbial signals to the brain control weight. Nature 534, 185–187 (2016).
Hjorth, M. F. et al. Pretreatment prevotella-to-bacteroides ratio and salivary amylase gene copy number as prognostic markers for dietary weight loss. Am. J. Clin. Nutr. 111, 1079–1086 (2020).
Sanna, S. et al. Causal relationships among the gut microbiome, short-chain fatty acids and metabolic diseases. Nat. Genet. 51, 600–605 (2019).
Yamada, Y. et al. A non-calorie-restricted low-carbohydrate diet is effective as an alternative therapy for patients with type 2 diabetes. Intern. Med. 53, 13–19 (2014).
Tay, J. et al. A very low-carbohydrate, low-saturated fat diet for type 2 diabetes management: a randomized trial. Diabetes Care 37, 2909–2918 (2014).
Balducci, S. et al. Physical exercise as therapy for type 2 diabetes mellitus. Diabetes Metab. Res. Rev. 30 (Suppl 1), 13–23 (2014).
Boule, N. G., Haddad, E., Kenny, G. P., Wells, G. A. & Sigal, R. J. Effects of exercise on glycemic control and body mass in type 2 diabetes mellitus: a meta-analysis of controlled clinical trials. JAMA 286, 1218–1227 (2001).
Snowling, N. J. & Hopkins, W. G. Effects of different modes of exercise training on glucose control and risk factors for complications in type 2 diabetic patients: a meta-analysis. Diabetes Care 29, 2518–2527 (2006).
Balducci, S. et al. Effect of an intensive exercise intervention strategy on modifiable cardiovascular risk factors in subjects with type 2 diabetes mellitus: a randomized controlled trial: the Italian Diabetes and Exercise Study (IDES). Arch. Intern. Med. 170, 1794–1803 (2010).
Di Loreto, C. et al. Make your diabetic patients walk: long-term impact of different amounts of physical activity on type 2 diabetes. Diabetes Care 28, 1295–1302 (2005).
Balducci, S. et al. Changes in physical fitness predict improvements in modifiable cardiovascular risk factors independently of body weight loss in subjects with type 2 diabetes participating in the Italian Diabetes and Exercise Study (IDES). Diabetes Care 35, 1347–1354 (2012).
Balducci, S. et al. Effect of high- versus low-intensity supervised aerobic and resistance training on modifiable cardiovascular risk factors in type 2 diabetes: the Italian Diabetes and Exercise Study (IDES). PLoS One 7, e49297 (2012).
Eriksson, K. F. & Lindgarde, F. Prevention of type 2 (non-insulin-dependent) diabetes mellitus by diet and physical exercise. The 6-year Malmo feasibility study. Diabetologia 34, 891–898 (1991).
Saltin, B. et al. Physical training and glucose tolerance in middle-aged men with chemical diabetes. Diabetes 28 (Suppl. 1), 30–32 (1979).
Nagi, D. Diabetes in Practice 2nd edn (John Wiley & Sons, 2005).
Ades, P. A., Savage, P. D., Marney, A. M., Harvey, J. & Evans, K. A. Remission of recently diagnosed type 2 diabetes mellitus with weight loss and exercise. J. Cardiopulm. Rehabil. Prev. 35, 193–197 (2015).
Ried-Larsen, M. et al. Type 2 diabetes remission 1 year after an intensive lifestyle intervention: a secondary analysis of a randomized clinical trial. Diabetes Obes. Metab. 21, 2257–2266 (2019).
Johansen, M. Y. et al. Effect of an intensive lifestyle intervention on glycemic control in patients with type 2 diabetes: a randomized clinical trial. JAMA 318, 637–646 (2017).
Vetter, M. L., Ritter, S., Wadden, T. A. & Sarwer, D. B. Comparison of bariatric surgical procedures for diabetes remission: efficacy and mechanisms. Diabetes Spectr. 25, 200–210 (2012).
Bray, G. A., Krauss, R. M., Sacks, F. M. & Qi, L. Lessons learned from the POUNDS Lost Study: genetic, metabolic, and behavioral factors affecting changes in body weight, body composition, and cardiometabolic risk. Curr. Obes. Rep. 8, 262–283 (2019).
Franz, M. J. & Evert, A. B. American Diabetes Association Guide to Nutrition Therapy for Diabetes 2 edn (American Diabetes Association, 2012).
Rowley, W. R., Bezold, C., Arikan, Y., Byrne, E. & Krohe, S. Diabetes 2030: insights from yesterday, today, and future trends. Popul. Health Manag. 20, 6–12 (2017).
Gillies, C. L. et al. Pharmacological and lifestyle interventions to prevent or delay type 2 diabetes in people with impaired glucose tolerance: systematic review and meta-analysis. BMJ 334, 299 (2007).
Knowler, W. C. et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 346, 393–403 (2002).
Lindstrom, J. et al. Sustained reduction in the incidence of type 2 diabetes by lifestyle intervention: follow-up of the Finnish Diabetes Prevention study. Lancet 368, 1673–1679 (2006).
Pan, X. R. et al. Effects of diet and exercise in preventing NIDDM in people with impaired glucose tolerance. The Da Qing IGT and Diabetes study. Diabetes Care 20, 537–544 (1997).
Li, G. et al. The long-term effect of lifestyle interventions to prevent diabetes in the China Da Qing Diabetes Prevention study: a 20-year follow-up study. Lancet 371, 1783–1789 (2008).
Poulsen, S. K. et al. Health effect of the New Nordic Diet in adults with increased waist circumference: a 6-mo randomized controlled trial. Am. J. Clin. Nutr. 99, 35–45 (2014).
Hjorth, M. F. et al. Pretreatment fasting plasma glucose and insulin modify dietary weight loss success: results from 3 randomized clinical trials. Am. J. Clin. Nutr. 106, 499–505 (2017).
Ritz, C., Astrup, A., Larsen, T. M. & Hjorth, M. F. Weight loss at your fingertips: personalized nutrition with fasting glucose and insulin using a novel statistical approach. Eur. J. Clin. Nutr. 73, 1529–1535 (2019). This article uses a novel statistical approach to model and estimate diet-induced weight loss according to baseline levels of glycaemia.
Due, A. et al. Comparison of 3 ad libitum diets for weight-loss maintenance, risk of cardiovascular disease, and diabetes: a 6-mo randomized, controlled trial. Am. J. Clin. Nutr. 88, 1232–1241 (2008).
Hjorth, M. F., Due, A., Larsen, T. M. & Astrup, A. Pretreatment fasting plasma glucose modifies dietary weight loss maintenance success: results from a stratified RCT. Obesity 25, 2045–2048 (2017).
Larsen, T. M. et al. Diets with high or low protein content and glycemic index for weight-loss maintenance. N. Engl. J. Med. 363, 2102–2113 (2010).
Greenway, F. L. et al. A randomized, double-blind, placebo-controlled study of Gelesis100: a novel nonsystemic oral hydrogel for weight loss. Obesity 27, 205–216 (2019).
Dansinger, M. L., Gleason, J. A., Griffith, J. L., Selker, H. P. & Schaefer, E. J. Comparison of the Atkins, Ornish, Weight Watchers, and Zone diets for weight loss and heart disease risk reduction: a randomized trial. JAMA 293, 43–53 (2005).
Greenberg, I., Stampfer, M. J., Schwarzfuchs, D., Shai, I. & Group, D. Adherence and success in long-term weight loss diets: the dietary intervention randomized controlled trial (DIRECT). J. Am. Coll. Nutr. 28, 159–168 (2009).
Sacks, F. M. et al. Comparison of weight-loss diets with different compositions of fat, protein, and carbohydrates. N. Engl. J. Med. 360, 859–873 (2009). The largest and longest (to date) randomized study comparing the weight loss effectiveness of diets differing in macronutrient composition shows no differences among diets.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
M.F.H. and A.A. are co-inventors on a pending provisional patent application on the use of biomarkers for prediction of weight loss responses and co-founders/owners of the University of Copenhagen spin-out company Personalized Weight Management Research Consortium ApS (Gluco-diet.dk). A.A. is a consultant or advisory board member for Basic Research, USA, Beachbody, USA, BioCare Copenhagen, Denmark, Gelesis, USA, Groupe Éthique et Santé, France, McCain Foods Limited, USA, Nestlé Research Center, Switzerland, and Weight Watchers, USA. A.A. and M.F.H. are co-authors of a number of diet/cookery books, including personalized nutrition for weight loss, published in several languages. F.M. declares no competing interests.
Additional information
Peer review information
Nature Reviews Endocrinology thanks P. Clifton, R. Taylor and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Prediabetes
-
An intermediate condition between normoglycaemia and type 2 diabetes mellitus, characterized by moderately elevated fasting or postprandial blood glucose or HbA1c.
- Glycaemic index
-
A relative ranking of foods according to their ability to increase blood glucose levels relative to a reference food (glucose or white bread) for the same amount of bioavailable carbohydrate.
- Glycaemic load
-
An extension of the glycaemic index that takes into account the actual amount of available carbohydrate present in one serving of a food or in the whole diet.
Rights and permissions
About this article
Cite this article
Magkos, F., Hjorth, M.F. & Astrup, A. Diet and exercise in the prevention and treatment of type 2 diabetes mellitus. Nat Rev Endocrinol 16, 545–555 (2020). https://doi.org/10.1038/s41574-020-0381-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-020-0381-5
This article is cited by
-
Hypothalamic POMC neuron-specific knockout of MC4R affects insulin sensitivity by regulating Kir2.1
Molecular Medicine (2024)
-
Knowledge, attitude, and practice toward weight management among diabetic patients in Qidong City, Jiangsu Province
BMC Public Health (2024)
-
Advances in secondary prevention mechanisms of macrovascular complications in type 2 diabetes mellitus patients: a comprehensive review
European Journal of Medical Research (2024)
-
Assessment of the impact of a personalised nutrition intervention in impaired glucose regulation over 26 weeks: a randomised controlled trial
Scientific Reports (2024)
-
Association of low-carbohydrate diet scores and type 2 diabetes in Chinese rural adults: The Henan Rural Cohort Study
Endocrine (2024)