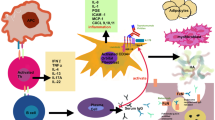
Abstract
Graves orbitopathy, also known as thyroid eye disease or thyroid-associated orbitopathy, is visually disabling, cosmetically disfiguring and has a substantial negative impact on a patient’s quality of life. There is increasing awareness of the need for early diagnosis and rapid specialist input from endocrinologists and ophthalmologists. Glucocorticoids are the mainstay of treatment; however, recurrence occurs frequently once these are withdrawn. Furthermore, in >60% of cases, normal orbital anatomy is not restored, and skilled rehabilitative surgery is required. Clinical trials have shown that considerable benefit can be derived from the addition of antiproliferative agents (such as mycophenolate or azathioprine) in preventing deterioration after steroid cessation. In addition, targeted biologic therapies have shown promise, including teprotumumab, which reduces proptosis, rituximab (anti-CD20), which reduces inflammation, and tocilizumab, which potentially benefits both of these parameters. Other strategies such as orbital radiotherapy have had their widespread role in combination therapy called into question. The pathophysiology of Graves orbitopathy has also been revised with identification of new potential therapeutic targets. In this Review we provide an up-to-date overview of the field, outline the optimal management of Graves orbitopathy and summarize the research developments in this area to highlight future research questions and direct future clinical trials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Smith, T. J. & Hegedus, L. Graves’ disease. N. Engl. J. Med. 375, 1552–1565 (2016).
Bahn, R. S. Graves’ ophthalmopathy. N. Engl. J. Med. 362, 726–738 (2010).
Ponto, K. A. et al. Quality of life in a German Graves orbitopathy population. Am. J. Ophthalmol. 152, 483–490 (2011).
Kahaly, G. J., Petrak, F., Hardt, J., Pitz, S. & Egle, U. T. Psychosocial morbidity of Graves’ orbitopathy. Clin. Endocrinol. (Oxf.) 63, 395–402 (2005).
Wiersinga, W. M. & Kahaly, G. J. Graves’ orbitopathy: a multidisciplinary approach. 3rd edn, (Karger, 2017).
Ponto, K. A. et al. Public health relevance of Graves’ orbitopathy. J. Clin. Endocrinol. Metab. 98, 145–152 (2013).
De Leo, S., Lee, S. Y. & Braverman, L. E. Hyperthyroidism. Lancet 388, 906–918 (2016).
Taylor, P. N. et al. Global epidemiology of hyperthyroidism and hypothyroidism. Nat. Rev. Endocrinol. 14, 301–316 (2018).
Perros, P. et al. Graves’ orbitopathy as a rare disease in Europe: a European Group on Graves’ Orbitopathy (EUGOGO) position statement. Orphanet J. Rare Dis. 12, 72 (2017).
Wiersinga, W. et al. Predictive score for the development or progression of Graves’ orbitopathy in patients with newly diagnosed Graves’ hyperthyroidism. Eur. J. Endocrinol. 178, 635–643 (2018).
Perros, P. & Wiersinga, W. M. The Amsterdam declaration on Graves’ orbitopathy. Thyroid 20, 245–246 (2010).
Hansen, C., Rouhi, R., Forster, G. & Kahaly, G. J. Increased sulfatation of orbital glycosaminoglycans in Graves’ ophthalmopathy. J. Clin. Endocrinol. Metab. 84, 1409–1413 (1999).
Zang, S. & Kahaly, G. Steroids and the immune response in Graves orbitopathy. Immunol. Endocr. Metab. Agents Med. Chem. 11, 90–98 (2011).
Lee, R. A., Harris, C. A. & Wang, J. C. Glucocorticoid receptor and adipocyte biology. Nucl. Receptor Res. 5, 101373 (2018).
Tomlinson, J. W. et al. The role of 11beta-hydroxysteroid dehydrogenase 1 in adipogenesis in thyroid-associated ophthalmopathy. J. Clin. Endocrinol. Metab. 95, 398–406 (2010).
Bujalska, I. J. et al. Characterisation of 11beta-hydroxysteroid dehydrogenase 1 in human orbital adipose tissue: a comparison with subcutaneous and omental fat. J. Endocrinol. 192, 279–288 (2007).
Bartalena, L. et al. Consensus statement of the European Group on Graves’ orbitopathy (EUGOGO) on management of GO. Eur. J. Endocrinol. 158, 273–285 (2008).
Dolman, P. J. & Rootman, J. VISA classification for Graves orbitopathy. Ophthalmic Plast. Reconstr. Surg. 22, 319–324 (2006).
Mourits, M. P., Prummel, M. F., Wiersinga, W. M. & Koornneef, L. Clinical activity score as a guide in the management of patients with Graves’ ophthalmopathy. Clin. Endocrinol. (Oxf.) 47, 9–14 (1997).
Werner, S. C. Classification of the eye changes of Graves’ disease. Am. J. Ophthalmol. 68, 646–648 (1969).
Werner, S. C. Modification of the classification of the eye changes of Graves’ disease: recommendations of the Ad Hoc Committee of the American Thyroid Association. J. Clin. Endocrinol. Metab. 44, 203–204 (1977).
Perros, P., Crombie, A. L., Matthews, J. N. & Kendall-Taylor, P. Age and gender influence the severity of thyroid-associated ophthalmopathy: a study of 101 patients attending a combined thyroid-eye clinic. Clin. Endocrinol. (Oxf.) 38, 367–372 (1993).
Bartalena, L. et al. The 2016 European Thyroid Association/European Group on Graves’ Orbitopathy guidelines for the management of Graves’ orbitopathy. Eur. Thyroid. J. 5, 9–26 (2016).
Barrio-Barrio, J., Sabater, A. L., Bonet-Farriol, E., Velazquez-Villoria, A. & Galofre, J. C. Graves’ ophthalmopathy: VISA versus EUGOGO classification, assessment, and management. J. Ophthalmol. 2015, 249125 (2015).
Bartalena, L. et al. Consensus statement of the European group on Graves’ orbitopathy (EUGOGO) on management of Graves’ orbitopathy. Thyroid 18, 333–346 (2008).
Terwee, C. B. et al. Interpretation and validity of changes in scores on the Graves’ ophthalmopathy quality of life questionnaire (GO-QOL) after different treatments. Clin. Endocrinol. (Oxf.) 54, 391–398 (2001).
Terwee, C. B., Gerding, M. N., Dekker, F. W., Prummel, M. F. & Wiersinga, W. M. Development of a disease specific quality of life questionnaire for patients with Graves’ ophthalmopathy: the GO-QOL. Br. J. Ophthalmol. 82, 773–779 (1998).
Dietrich, A. et al. Establishing the usefulness of the GO-QOL in a UK hospital-treated population with thyroid eye disease in the CIRTED trial. Psychol. Health Med 23, 1341–1355 (2018).
Rajendram, R. et al. Combined immunosuppression and radiotherapy in thyroid eye disease (CIRTED): a multicentre, 2 × 2 factorial, double-blind, randomised controlled trial. Lancet Diabetes Endocrinol 6, 299–309 (2018).
Kahaly, G. J. et al. Mycophenolate plus methylprednisolone versus methylprednisolone alone in active, moderate-to-severe Graves’ orbitopathy (MINGO): a randomised, observer-masked, multicentre trial. Lancet. Diabetes Endocrinol. 6, 287–298 (2018).
Perros, P. et al. PREGO (presentation of Graves’ orbitopathy) study: changes in referral patterns to European Group On Graves’ Orbitopathy (EUGOGO) centres over the period from 2000 to 2012. Br. J. Ophthalmol. 99, 1531–1535 (2015).
British Thyroid Foundation. TEAMeD-5, http://www.btf-thyroid.org/TEAMeD-5 (2018).
Marcocci, C., Bartalena, L., Bogazzi, F., Panicucci, M. & Pinchera, A. Studies on the occurrence of ophthalmopathy in Graves’ disease. Acta Endocrinol. 120, 473–478 (1989).
Bartalena, L. The dilemma of how to manage Graves’ hyperthyroidism in patients with associated orbitopathy. J. Clin. Endocrinol. Metab. 96, 592–599 (2011).
Bartalena, L. et al. More on smoking habits and Graves’ ophthalmopathy. J. Endocrinol. Invest. 12, 733–737 (1989).
Wiersinga, W. M. Smoking and thyroid. Clin. Endocrinol. (Oxf.) 79, 145–151 (2013).
Prummel, M. F. & Wiersinga, W. M. Smoking and risk of Graves’ disease. Jama 269, 479–482 (1993).
Bartalena, L. et al. Cigarette smoking and treatment outcomes in Graves ophthalmopathy. Ann. Intern. Med. 129, 632–635 (1998).
Eckstein, A. et al. Impact of smoking on the response to treatment of thyroid associated ophthalmopathy. Br. J. Ophthalmol. 87, 773–776 (2003).
Pfeilschifter, J. & Ziegler, R. Smoking and endocrine ophthalmopathy: impact of smoking severity and current vs lifetime cigarette consumption. Clin. Endocrinol. (Oxf.) 45, 477–481 (1996).
Cawood, T. J., Moriarty, P., O’Farrelly, C. & O’Shea, D. Smoking and thyroid-associated ophthalmopathy: a novel explanation of the biological link. J. Clin. Endocrinol. Metab. 92, 59–64 (2007).
Regensburg, N. I., Wiersinga, W. M., Berendschot, T. T., Saeed, P. & Mourits, M. P. Effect of smoking on orbital fat and muscle volume in Graves’ orbitopathy. Thyroid 21, 177–181 (2011).
Marcocci, C. et al. Selenium and the course of mild Graves’ orbitopathy. N. Engl. J. Med. 364, 1920–1931 (2011).
Marcocci, C. et al. Comparison of the effectiveness and tolerability of intravenous or oral glucocorticoids associated with orbital radiotherapy in the management of severe Graves’ ophthalmopathy: results of a prospective, single-blind, randomized study. J. Clin. Endocrinol. Metab. 86, 3562–3567 (2001).
Kahaly, G. J., Pitz, S., Hommel, G. & Dittmar, M. Randomized, single blind trial of intravenous versus oral steroid monotherapy in Graves’ orbitopathy. J. Clin. Endocrinol. Metab. 90, 5234–5240 (2005).
Bartalena, L., Pinchera, A. & Marcocci, C. Management of Graves’ ophthalmopathy: reality and perspectives. Endocr. Rev. 21, 168–199 (2000).
Bartalena, L. et al. Efficacy and safety of three different cumulative doses of intravenous methylprednisolone for moderate to severe and active Graves’ orbitopathy. J. Clin. Endocrinol. Metab. 97, 4454–4463 (2012).
Ye, X. et al. Efficacy and safety of mycophenolate mofetil in patients with active moderate-to-severe Graves’ orbitopathy. Clin. Endocrinol. (Oxf.) 86, 247–255 (2017).
Prummel, M. F. et al. Prednisone and cyclosporine in the treatment of severe Graves’ ophthalmopathy. N. Engl. J. Med. 321, 1353–1359 (1989).
Kahaly, G. J. et al. 2018 European Thyroid Association guideline for the management of Graves’ hyperthyroidism. Eur. Thyroid. J. 7, 167–186 (2018).
Wiersinga, W. M. Advances in treatment of active, moderate-to-severe Graves’ ophthalmopathy. lancet. Diabetes & Endocrinol. 5, 134–142 (2017).
Salvi, M. & Campi, I. Medical treatment of Graves’ orbitopathy. Horm. Metab. Res. 47, 779–788 (2015).
Zang, S., Ponto, K. A. & Kahaly, G. J. Clinical review: intravenous glucocorticoids for Graves’ orbitopathy: efficacy and morbidity. J. Clin. Endocrinol. Metab. 96, 320–332 (2011).
Matheis, N. et al. Proteomics of orbital tissue in thyroid-associated orbitopathy. J. Clin. Endocrinol. Metab. 100, E1523–E1530 (2015).
Kahaly, G. J., Rosler, H. P., Pitz, S. & Hommel, G. Low- versus high-dose radiotherapy for Graves’ ophthalmopathy: a randomized, single blind trial. J. Clin. Endocrinol. Metab. 85, 102–108 (2000).
Prummel, M. F. et al. A randomized controlled trial of orbital radiotherapy versus sham irradiation in patients with mild Graves’ ophthalmopathy. J. Clin. Endocrinol. Metab. 89, 15–20 (2004).
Prummel, M. F. et al. Randomized double-blind trial of prednisone versus radiotherapy in Graves’ ophthalmopathy. Lancet 342, 949–954 (1993).
Christiansen, E. & Kofoed-Enevoldsen, A. Graves’ ophthalmopathy. J. Clin. Endocrinol. Metab. 86, 2327–2328 (2001).
Gorman, C. A. et al. A prospective, randomized, double-blind, placebo-controlled study of orbital radiotherapy for Graves’ ophthalmopathy. Ophthalmology 108, 1523–1534 (2001).
Mourits, M. P. et al. Radiotherapy for Graves’ orbitopathy: randomised placebo-controlled study. Lancet 355, 1505–1509 (2000).
Godfrey, K. J. & Kazim, M. Radiotherapy for active thyroid eye disease. Ophthalmic Plast. Reconstr. Surg. 34, S98–S104 (2018).
Verity, D. H. & Rose, G. E. Acute thyroid eye disease (TED): principles of medical and surgical management. Eye (Lond.) 27, 308–319 (2013).
Eugui, E. M. & Allison, A. C. Immunosuppressive activity of mycophenolate mofetil. Ann. NY Acad. Sci. 685, 309–329 (1993).
Allison, A. C. Mechanisms of action of mycophenolate mofetil in preventing chronic rejection. Transplant. Proc. 34, 2863–2866 (2002).
Riedl, M., Kuhn, A., Kramer, I., Kolbe, E. & Kahaly, G. J. Prospective, systematically recorded mycophenolate safety data in Graves’ orbitopathy. J. Endocrinol. Invest. 39, 687–694 (2016).
Eugui, E. M. & Allison, A. C. Immunosuppressive activity of mycophenolate mofetil. Ann. NY Acad. Sci. 685, 309–329 (1993).
Mazumder, A. G., Patial, V. & Singh, D. Mycophenolate mofetil contributes to downregulation of the hippocampal interleukin type 2 and 1beta mediated PI3K/AKT/mTOR pathway hyperactivation and attenuates neurobehavioral comorbidities in a rat model of temporal lobe epilepsy. Brain, Behav. Immun. 75, 84–93 (2019).
Perros, P., Weightman, D. R., Crombie, A. L. & Kendall-Taylor, P. Azathioprine in the treatment of thyroid-associated ophthalmopathy. Acta Endocrinol. 122, 8–12 (1990).
Smith, T. J. & Janssen, J. A. Building the case for insulin-like growth factor receptor-1 involvement in thyroid-associated ophthalmopathy. Front. Endocrinol. 7, 167 (2016).
Smith, T. J. & Janssen, J. Insulin-like growth factor-1 receptor and thyroid-associated ophthalmopathy. Endocr. Rev. 40, 236–267 (2016).
Pritchard, J., Han, R., Horst, N., Cruikshank, W. W. & Smith, T. J. Immunoglobulin activation of T cell chemoattractant expression in fibroblasts from patients with Graves’ disease is mediated through the insulin-like growth factor 1 receptor pathway. J. Immunol. 170, 6348–6354 (2003).
Smith, T. J., Hegedus, L. & Douglas, R. S. Role of insulin-like growth factor-1 (IGF-1) pathway in the pathogenesis of Graves’ orbitopathy. Best. Pract. Clin. Endocrinol. Metab. 26, 291–302 (2012).
Smith, T. J. et al. Teprotumumab for thyroid-associated ophthalmopathy. N. Engl. J. Med. 376, 1748–1761 (2017).
Piro, L. D. et al. Extended rituximab (anti-CD20 monoclonal antibody) therapy for relapsed or refractory low-grade or follicular non-Hodgkin’s lymphoma. Ann. Oncol. 10, 655–661 (1999).
Stan, M. N. et al. Randomized controlled trial of rituximab in patients with Graves’ orbitopathy. J. Clin. Endocrinol. Metab. 100, 432–441 (2015).
Salvi, M. et al. Efficacy of B-cell targeted therapy with rituximab in patients with active moderate to severe Graves’ orbitopathy: a randomized controlled study. J. Clin. Endocrinol. Metab. 100, 422–431 (2015).
Stan, M. N. & Salvi, M. Management of endocrine disease: rituximab therapy for Graves’ orbitopathy — lessons from randomized control trials. Eur. J. Endocrinol. 176, R101–R109 (2017).
Salvi, M. & Covelli, D. B cells in Graves’ orbitopathy: more than just a source of antibodies? Eye (Lond.) 33, 230–234 (2019).
Perez-Moreiras, J. V., Alvarez-Lopez, A. & Gomez, E. C. Treatment of active corticosteroid-resistant graves’ orbitopathy. Ophthalmic Plast. Reconstr. Surg. 30, 162–167 (2014).
Russell, D. J., Wagner, L. H. & Seiff, S. R. Tocilizumab as a steroid sparing agent for the treatment of Graves’ orbitopathy. Am. J. Ophthalmol. Case Rep. 7, 146–148 (2017).
Perez-Moreiras, J. V. et al. Efficacy of tocilizumab in patients with moderate-to-severe corticosteroid-resistant graves orbitopathy: a randomized clinical trial. Am. J. Ophthalmol. 195, 181–190 (2018).
Stohl, W. Inhibition of B cell activating factor (BAFF) in the management of systemic lupus erythematosus (SLE). Expert. Rev. Clin. Immunol. 13, 623–633 (2017).
Draman, M. S. et al. Effects of prostaglandin F2alpha on adipocyte biology relevant to graves’ orbitopathy. Thyroid 23, 1600–1608 (2013).
Draman, M. S. et al. Prostaglandin F2-alpha eye drops (bimatoprost) in Graves’ orbitopathy: a randomized controlled double-masked crossover trial (BIMA trial). Thyroid 29, 563–572 (2019).
Kozdon, K., Fitchett, C., Rose, G. E., Ezra, D. G. & Bailly, M. Mesenchymal stem cell-like properties of orbital fibroblasts in Graves’ orbitopathy. Invest. Ophthalmol. Vis. Sci. 56, 5743–5750 (2015).
Brandau, S. et al. Orbital fibroblasts from Graves’ orbitopathy patients share functional and immunophenotypic properties with mesenchymal stem/stromal cells. Investig. Ophthalmol. Vis. Sci. 56, 6549–6557 (2015).
Starkey, K. J. et al. Adipose thyrotrophin receptor expression is elevated in Graves’ and thyroid eye diseases ex vivo and indicates adipogenesis in progress in vivo. J. Mol. Endocrinol. 30, 369–380 (2003).
Potgieser, P. W., Wiersinga, W. M., Regensburg, N. I. & Mourits, M. P. Some studies on the natural history of Graves’ orbitopathy: increase in orbital fat is a rather late phenomenon. Eur. J. Endocrinol. 173, 149–153 (2015).
Smith, T. J. Insights into the role of fibroblasts in human autoimmune diseases. Clin. Exp. Immunol. 141, 388–397 (2005).
Lehmann, G. M., Garcia-Bates, T. M., Smith, T. J., Feldon, S. E. & Phipps, R. P. Regulation of lymphocyte function by PPARgamma: relevance to thyroid eye disease-related inflammation. PPAR Res. 2008, 895901 (2008).
Kahaly, G. J. et al. Regulatory T-cells in Graves’ orbitopathy: baseline findings and immunomodulation by anti-T lymphocyte globulin. J. Clin. Endocrinol. Metab. 96, 422–429 (2011).
Douglas, R. S. et al. Increased generation of fibrocytes in thyroid-associated ophthalmopathy. J. Clin. Endocrinol. Metab. 95, 430–438 (2010).
Lu, Y. et al. CD34– orbital fibroblasts from patients with thyroid-associated ophthalmopathy modulate TNF-alpha expression in CD34+ fibroblasts and fibrocytes. Investig. Ophthalmol. Vis. Sci. 59, 2615–2622 (2018).
Fernando, R. et al. Human fibrocytes coexpress thyroglobulin and thyrotropin receptor. Proc. Natl Acad. Sci. USA 109, 7427–7432 (2012).
Rotondo Dottore, G. et al. Association of T and B cells infiltrating orbital tissues with clinical features of graves orbitopathy. JAMA Ophthalmol. 136, 613–619 (2018).
Pawlowski, P. et al. Markers of inflammation and fibrosis in the orbital fat/connective tissue of patients with Graves’ orbitopathy: clinical implications. Mediators. Inflamm. 2014, 412158 (2014).
van Steensel, L. et al. Orbit-infiltrating mast cells, monocytes, and macrophages produce PDGF isoforms that orchestrate orbital fibroblast activation in Graves’ ophthalmopathy. J. Clin. Endocrinol. Metab. 97, E400–E408 (2012).
Draman, M. S. & Ludgate, M. Thyroid eye disease — an update. Expert. Rev. Ophthalmol. 11, 273–284 (2016).
Tsui, S. et al. Evidence for an association between thyroid-stimulating hormone and insulin-like growth factor 1 receptors: a tale of two antigens implicated in Graves’ disease. J. Immunol. 181, 4397–4405 (2008).
Weightman, D. R., Perros, P., Sherif, I. H. & Kendall-Taylor, P. Autoantibodies to IGF-1 binding sites in thyroid associated ophthalmopathy. Autoimmunity 16, 251–257 (1993).
Moshkelgosha, S., So, P. W., Deasy, N., Diaz-Cano, S. & Banga, J. P. Cutting edge: retrobulbar inflammation, adipogenesis, and acute orbital congestion in a preclinical female mouse model of Graves’ orbitopathy induced by thyrotropin receptor plasmid-in vivo electroporation. Endocrinology 154, 3008–3015 (2013).
Berchner-Pfannschmidt, U. et al. Comparative assessment of female mouse model of Graves’ orbitopathy under different environments, accompanied by proinflammatory cytokine and T-cell responses to thyrotropin hormone receptor antigen. Endocrinology 157, 1673–1682 (2016).
Feliciello, A. et al. Expression of thyrotropin-receptor mRNA in healthy and Graves’ disease retro-orbital tissue. Lancet 342, 337–338 (1993).
Crisp, M. S., Lane, C., Halliwell, M., Wynford-Thomas, D. & Ludgate, M. Thyrotropin receptor transcripts in human adipose tissue. J. Clin. Endocrinol. Metab. 82, 2003–2005 (1997).
Boschi, A. et al. Quantification of cells expressing the thyrotropin receptor in extraocular muscles in thyroid associated orbitopathy. Br. J. Ophthalmol. 89, 724–729 (2005).
Ludgate, M. et al. The thyrotropin receptor in thyroid eye disease. Thyroid 8, 411–413 (1998).
Zhang, L. et al. Biological effects of thyrotropin receptor activation on human orbital preadipocytes. Investig. Ophthalmol. Vis. Sci. 47, 5197–5203 (2006).
Lu, M. & Lin, R. Y. TSH stimulates adipogenesis in mouse embryonic stem cells. J. Endocrinol. 196, 159–169 (2008).
Kumar, S., Nadeem, S., Stan, M. N., Coenen, M. & Bahn, R. S. A stimulatory TSH receptor antibody enhances adipogenesis via phosphoinositide 3-kinase activation in orbital preadipocytes from patients with Graves’ ophthalmopathy. J. Mol. Endocrinol. 46, 155–163 (2011).
Zhang, L. et al. Thyrotropin receptor activation increases hyaluronan production in preadipocyte fibroblasts: contributory role in hyaluronan accumulation in thyroid dysfunction. J. Biol. Chem. 284, 26447–26455 (2009).
Morshed, S. A., Ando, T., Latif, R. & Davies, T. F. Neutral antibodies to the TSH receptor are present in Graves’ disease and regulate selective signaling cascades. Endocrinology 151, 5537–5549 (2010).
Zhang, L. et al. Adipose tissue depot-specific differences in the regulation of hyaluronan production of relevance to Graves’ orbitopathy. J. Clin. Endocrinol. Metab. 97, 653–662 (2012).
Billon, N. & Dani, C. Developmental origins of the adipocyte lineage: new insights from genetics and genomics studies. Stem Cell Rev. 8, 55–66 (2012).
Gesta, S., Tseng, Y. H. & Kahn, C. R. Developmental origin of fat: tracking obesity to its source. Cell 131, 242–256 (2007).
DeGroot, L. J. in Endotext (eds K. R. Feingold et al.) 1–135 (MDText.com, Inc., 2000).
Peyster, R. G., Ginsberg, F., Silber, J. H. & Adler, L. P. Exophthalmos caused by excessive fat: CT volumetric analysis and differential diagnosis. AJR. Am. J. Roentgenol. 146, 459–464 (1986).
Smolders, M. H. et al. Exophthalmos in obesity. Ophthalmic. Res. 36, 78–81 (2004).
Zhang, L. et al. Possible targets for nonimmunosuppressive therapy of Graves’ orbitopathy. J. Clin. Endocrinol. Metab. 99, E1183–E1190 (2014).
Krieger, C. C. et al. TSH/IGF-1 receptor cross talk in Graves’ ophthalmopathy pathogenesis. J. Clin. Endocrinol. Metab. 101, 2340–2347 (2016).
Krieger, C. C., Neumann, S., Place, R. F., Marcus-Samuels, B. & Gershengorn, M. C. Bidirectional TSH and IGF-1 receptor cross talk mediates stimulation of hyaluronan secretion by Graves’ disease immunoglobins. J. Clin. Endocrinol. Metab. 100, 1071–1077 (2015).
Zhang, L. et al. Reversal of pathological features of Graves’ orbitopathy by activation of forkhead transcription factors, FOXOs. J. Clin. Endocrinol. Metab. 101, 114–122 (2016).
Kumar, S., Coenen, M., Iyer, S. & Bahn, R. S. Forkhead transcription factor FOXO1 is regulated by both a stimulatory thyrotropin receptor antibody and insulin-like growth factor-1 in orbital fibroblasts from patients with Graves’ ophthalmopathy. Thyroid 25, 1145–1150 (2015).
Tardy, M., Dold, M., Engel, R. R. & Leucht, S. Trifluoperazine versus low-potency first-generation antipsychotic drugs for schizophrenia. Cochrane Database Syst. Rev. https://doi.org/10.1002/14651858.CD009396.pub2 (2014).
Eckstein, A. K. et al. Patients with severe Graves’ ophthalmopathy have a higher risk of relapsing hyperthyroidism and are unlikely to remain in remission. Clin. Endocrinol. (Oxf.) 67, 607–612 (2007).
Khoo, D. H. et al. The combination of absent thyroid peroxidase antibodies and high thyroid-stimulating immunoglobulin levels in Graves’ disease identifies a group at markedly increased risk of ophthalmopathy. Thyroid 9, 1175–1180 (1999).
Kahaly, G. J., Wuster, C., Olivo, P. D. & Diana, T. High titers of thyrotropin receptor antibodies are associated with orbitopathy in patients with Graves disease. J. Clin. Endocrinol. Metab. 104, 2561–2568 (2019).
Ponto, K. A. et al. Clinical relevance of thyroid-stimulating immunoglobulins in Graves’ ophthalmopathy. Ophthalmology 118, 2279–2285 (2011).
Metcalfe, R. et al. Demonstration of immunoglobulin G, A, and E autoantibodies to the human thyrotropin receptor using flow cytometry. J. Clin. Endocrinol. Metab. 87, 1754–1761 (2002).
Minich, W. B. et al. Autoantibodies to the IGF1 receptor in Graves’ orbitopathy. J. Clin. Endocrinol. Metab. 98, 752–760 (2013).
Marino, M. et al. Serum antibodies against the insulin-like growth factor-1 receptor (IGF-1R) in Graves’ disease and Graves’ orbitopathy. J. Endocrinol. Invest. 42, 471–480 (2019).
Fang, S. et al. IL-17A exacerbates fibrosis by promoting the proinflammatory and profibrotic function of orbital fibroblasts in TAO. J. Clin. Endocrinol. Metab. 101, 2955–2965 (2016).
Fang, S. et al. Regulation of orbital fibrosis and adipogenesis by pathogenic Th17 cells in graves orbitopathy. J. Clin. Endocrinol. Metab. 102, 4273–4283 (2017).
Banga, J. P., Moshkelgosha, S., Berchner-Pfannschmidt, U. & Eckstein, A. Modeling Graves’ orbitopathy in experimental Graves’ disease. Horm. Metab. Res. 47, 797–803 (2015).
McLachlan, S. M. & Rapoport, B. Breaking tolerance to thyroid antigens: changing concepts in thyroid autoimmunity. Endocr. Rev. 35, 59–105 (2014).
Ludgate, M. Animal models of Graves’ disease. Eur. J. Endocrinol. 142, 1–8 (2000).
Many, M. C. et al. Development of an animal model of autoimmune thyroid eye disease. J. Immunol. 162, 4966–4974 (1999).
Baker, G., Mazziotti, G., von Ruhland, C. & Ludgate, M. Reevaluating thyrotropin receptor-induced mouse models of graves’ disease and ophthalmopathy. Endocrinology 146, 835–844 (2005).
Zhao, S. X. et al. Orbital fibrosis in a mouse model of Graves’ disease induced by genetic immunization of thyrotropin receptor cDNA. J. Endocrinol. 210, 369–377 (2011).
Masetti, G. et al. Gut microbiota in experimental murine model of Graves’ orbitopathy established in different environments may modulate clinical presentation of disease. Microbiome 6, 97 (2018).
Eckstein, A., Esser, J., Oeverhaus, M., Saeed, P. & Jellema, H. M. Surgical treatment of diplopia in Graves orbitopathy patients. Ophthalmic Plast. Reconstr. Surg. 34, S75–S84 (2018).
Barker, L., Mackenzie, K., Adams, G. G. & Hancox, J. Long-term surgical outcomes for vertical deviations in thyroid eye disease. Strabismus 25, 67–72 (2017).
Mourits, M. P. & Sasim, I. V. A single technique to correct various degrees of upper lid retraction in patients with Graves’ orbitopathy. Br. J. Ophthalmol. 83, 81–84 (1999).
Author information
Authors and Affiliations
Contributions
All authors equally contributed to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The Johannes Gutenberg University (JGU) Medical Center and the JGU Thyroid Laboratory received research grants from Novartis, Germany, and River Vision, USA, when performing the MINGO and teprotumumab trials.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Proptosis
-
Abnormal protrusion of the eyeball.
- Diplopia
-
Double vision.
- Observer masked
-
The observer assessing outcomes does not know the treatment allocation (the same as “blinded” in clinical trials); masked is used instead of blinded in ophthalmology trials to not alarm patients.
Rights and permissions
About this article
Cite this article
Taylor, P.N., Zhang, L., Lee, R.W.J. et al. New insights into the pathogenesis and nonsurgical management of Graves orbitopathy. Nat Rev Endocrinol 16, 104–116 (2020). https://doi.org/10.1038/s41574-019-0305-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-019-0305-4
This article is cited by
-
Combined glucocorticoids and cyclophosphamide in the treatment of Graves’ ophthalmopathy: a systematic review and meta-analysis
BMC Endocrine Disorders (2024)
-
Whole-orbit radiomics: machine learning-based multi- and fused- region radiomics signatures for intravenous glucocorticoid response prediction in thyroid eye disease
Journal of Translational Medicine (2024)
-
Artificial Intelligence-based quantitative evaluation of retinal vascular parameters in thyroid-associated ophthalmopathy
Endocrine (2024)
-
Alemtuzumab induces severe orbitopathy in relapsing–remitting multiple sclerosis
Journal of Neurology (2024)
-
Blocking insulin-like growth factor 1 receptor signaling pathway inhibits neuromuscular junction regeneration after botulinum toxin-A treatment
Cell Death & Disease (2023)