Abstract
The incidence of thyroid cancer is on the rise, and this disease is projected to become the fourth leading type of cancer across the globe. From 1990 to 2013, the global age-standardized incidence rate of thyroid cancer increased by 20%. This global rise in incidence has been attributed to several factors, including increased detection of early tumours, the elevated prevalence of modifiable individual risk factors (for example, obesity) and increased exposure to environmental risk factors (for example, iodine levels). In this Review, we explore proven and novel hypotheses for how modifiable risk factors and environmental exposures might be driving the worldwide increase in the incidence of thyroid cancer. Although overscreening and the increased diagnosis of possibly clinically insignificant disease might have a role in certain parts of the world, other areas could be experiencing a true increase in incidence due to elevated exposure risks. In the current era of personalized medicine, national and international registry data should be applied to identify populations who are at increased risk for the development of thyroid cancer.
Key points
The incidence of thyroid cancer in higher-income countries has been rising; the causes appear to be complex and multifactorial.
Several upper middle-income countries have regional cancer registries that help to better understand cancer incidence in the context of environmental influences, such as iodine supplementation.
In lower middle-income and low-income countries, understanding thyroid cancer incidence has been predominantly limited to single-centre studies.
Proposed influences in the development of thyroid cancer include dietary changes and environmental exposures, which vary based on region.
The development of regional or national cancer registries in low-income countries would provide a rich repository to study thyroid cancer in regions with varying environmental and social exposures.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Fitzmaurice, C. et al. The global burden of cancer 2013. JAMA Oncol. 1, 505–527 (2015).
Ferlay, J. et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 136, E359–E386 (2015).
La Vecchia, C. et al. Thyroid cancer mortality and incidence: a global overview. Int. J. Cancer 136, 2187–2195 (2014). This study uses data from the WHO and Cancer Incidence in Five Continents to describe recent worldwide trends in thyroid cancer.
Lim, H., Devesa, S. S., Sosa, J. A., Check, D. & Kitahara, C. M. Trends in thyroid cancer incidence and mortality in the United States, 1974–2013. JAMA 317, 1338–1348 (2017). This study of the Surveillance, Epidemiology, and End Results-9 cancer registry in the USA finds that the incidence of thyroid cancer has increased from 1974 to 2013.
Lee, T.-J., Kim, S., Cho, H.-J. & Lee, J.-H. The incidence of thyroid cancer is affected by the characteristics of a healthcare system. J. Korean Med. Sci. 27, 1491–1498 (2012).
Liu, Y., Su, L. & Xiao, H. Review of factors related to the thyroid cancer epidemic. Int. J. Endocrinol. 2017, 5308635 (2017).
World Bank Country and Lending Groups – World Bank Data Help Desk. Available at: https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups. (Accessed: February 2019).
Vaccarella, S. et al. The impact of diagnostic changes on the rise in thyroid cancer incidence: a population-based study in selected high-resource countries. Thyroid 25, 1127–1136 (2015).
Enewold, L. et al. Rising thyroid cancer incidence in the United States by demographic and tumor characteristics, 1980–2005. Cancer Epidemiol. Biomark. Prev. 18, 784–791 (2009).
Ogden, C. L., Carroll, M. D., Kit, B. K. & Flegal, K. M. Prevalence of childhood and adult obesity in the United States, 2011–2012. JAMA 311, 806–814 (2014).
Kitahara, C. M. et al. Obesity and thyroid cancer risk among US men and women: a pooled analysis of five prospective studies. Cancer Epidemiol. Biomark. Prev. 20, 464–472 (2011).
Malik, V. S., Willett, W. C. & Hu, F. B. Global obesity: trends, risk factors and policy implications. Nat. Rev. Endocrinol. 9, 13–27 (2013).
Colonna, M. et al. Incidence of thyroid cancer in adults recorded by French cancer registries (1978–1997). Eur. J. Cancer 38, 1762–1768 (2002).
Forside - DTU Fødevareinstituttet. http://www.food.dtu.dk Available at: http://www.food.dtu.dk/. (Accessed: February 2019).
Laurberg, P. et al. Implementation and monitoring of iodine supplementation in Denmark: the Danthyr program. IDD Newsletter 19(4), (2003).
Rasmussen, L. B. et al. Iodine intake before and after mandatory iodization in Denmark: results from the Danish investigation of iodine intake and thyroid diseases (DanThyr) study. Br. J. Nutr. 100, 166–173 (2008).
Blomberg, M., Feldt-Rasmussen, U., Andersen, K. K. & Kjaer, S. K. Thyroid cancer in Denmark 1943–2008, before and after iodine supplementation. Int. J. Cancer 131, 2360–2366 (2012).
Bülow Pedersen, I. et al. An increased incidence of overt hypothyroidism after iodine fortification of salt in Denmark: a prospective population study. J. Clin. Endocrinol. Metab. 92, 3122–3127 (2007).
Carle, A. et al. Epidemiology of subtypes of hypothyroidism in Denmark. Eur. J. Endocrinol. 154, 21–28 (2006).
Møllehave, L. T. et al. Trends in treatments of thyroid disease following iodine fortification in Denmark: a nationwide register-based study. Clin. Epidemiol. 10, 763–770 (2018).
Hori, M. et al. Cancer incidence and incidence rates in Japan in 2009: a study of 32 population-based cancer registries for the monitoring of cancer incidence in Japan (MCIJ) project. Jpn. J. Clin. Oncol. 45, 884–891 (2015).
Jung, K.-W. et al. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2012. Cancer Res. Treat. 47, 127–141 (2015).
Lee, J. W. et al. Cancer screening using 18F-FDG PET/CT in Korean asymptomatic volunteers: a preliminary report. Ann. Nucl. Med. 23, 685–691 (2009).
Tsuda, T., Tokinobu, A., Yamamoto, E. & Suzuki, E. Thyroid cancer detection by ultrasound among residents ages 18 years and younger in Fukushima, Japan: 2011 to 2014. Epidemiology 27, 316–322 (2016).
Hiranuma, Y. Misrepresented risk of thyroid cancer in Fukushima. Lancet Diabetes Endocrinol. 4, 970 (2016).
Sadkowsky, K. et al. Epidemiology research using the national cancer statistics clearing house and the national death index databases. Australas. Epidemiol. 8, 29 (2001).
Burgess, J. R., Dwyer, T., McArdle, K., Tucker, P. & Shugg, D. The changing incidence and spectrum of thyroid carcinoma in Tasmania (1978–1998) during a transition from iodine sufficiency to iodine deficiency. J. Clin. Endocrinol. Metab. 85, 1513–1517 (2000).
Richards, P. A. Iodine nutrition in two Tasmanian cultures. Med. J. Aust. 163, 628–630 (1995).
Melick R. & Van Middlesworth L. Radio-iodine fallout in Australian sheep. Med. J. Aust. 2, 930–932 (1966).
Burgess, J. R. Temporal trends for thyroid carcinoma in Australia: an increasing incidence of papillary thyroid carcinoma (1982–1997). Thyroid 12, 141–149 (2002).
Wang, Y. & Wang, W. Increasing incidence of thyroid cancer in Shanghai, China, 1983–2007. Asia Pac. J. Public Health 27, NP223–NP229 (2015).
Liu, Y. et al. The prevalence of thyroid nodules in northwest China and its correlation with metabolic parameters and uric acid. Oncotarget 8, 41555–41562 (2017).
Yao, Y. et al. Thyroid nodules in centenarians: prevalence and relationship to lifestyle characteristics and dietary habits. Clin. Interv. Aging 13, 515–522 (2018).
Tazi, M. A., Er-Raki, A. & Benjaafar, N. Cancer incidence in Rabat, Morocco: 2006–2008. Ecancermedicalscience 7, (338 (2013).
Jagathnath Krishna, K. M. & Sebastian, P. Cancer incidence and mortality: district cancer registry, Trivandrum, South India. Asian Pac. J. Cancer Prev. 18, 1485–1491 (2017).
Lo, T. E., Uy, A. & Maningat, P. D. Well-differentiated thyroid cancer: the Philippine general hospital experience. Endocrine Abstracts 37, EP838, https://doi.org/10.1530/endoabs.37.EP838 (2015).
Ukekwe, F. I., Olusina, D. B. & Okere, P. C. N. Patterns of thyroid cancers in Southeastern Nigeria: a 15 year histopathologic review (2000–2014). J. Clin. Diagn. Res. 11, EC16–EC19 (2017).
Bukhari, U., Sadiq, S., Memon, J. & Baig, F. Thyroid carcinoma in Pakistan: a retrospective review of 998 cases from an academic referral center. Hematol. Oncol. Stem Cell Ther. 2, 345–348 (2009).
Lortet-Tieulent, J. & Vaccarella, S. International and subnational variation thyroid cancer incidence and mortality over 2008–2012. Rev. d’Épidémiologie et de Santé Publique 66, S254 (2018).
James, B. C. et al. An update in international trends in incidence rates of thyroid cancer, 1973–2007. Cancer Causes Control 29, 465–473 (2018).
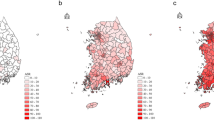
Ahn, H. S., Kim, H. J. & Welch, H. G. Korea’s thyroid-cancer ‘epidemic’ — screening and overdiagnosis. N. Engl. J. Med. 371, 1765–1767 (2014). This perspective piece reviews trends in thyroid cancer incidence in South Korea following the initiation of a government-led national cancer screening program.
Vaccarella, S. et al. Worldwide thyroid-cancer epidemic? The increasing impact of overdiagnosis. N. Engl. J. Med. 375, 614–617 (2016).
D’Avanzo, B., La Vecchia, C., Franceschi, S., Negri, E. & Talamini, R. History of thyroid diseases and subsequent thyroid cancer risk. Cancer Epidemiol. Biomark. Prev. 4, 193–199 (1995).
Balasubramaniam, S., Ron, E., Gridley, G., Schneider, A. B. & Brenner, A. V. Association between benign thyroid and endocrine disorders and subsequent risk of thyroid cancer among 4.5 million U.S. male veterans. J. Clin. Endocrinol. Metab. 97, 2661–2669 (2012).
Nosé, V. Familial thyroid cancer: a review. Mod. Pathol. 24, S19–S33 (2011).
Xu, L., Li, G., Wei, Q., El-Naggar, A. K. & Sturgis, E. M. Family history of cancer and risk of sporadic differentiated thyroid carcinoma. Cancer 118, 1228–1235 (2012).
Azar, F. K., Lee, S. L. & Rosen, J. E. Medullary thyroid cancer: an update for surgeons. Am. Surg. 81, 1–8 (2015).
Kouvaraki, M. A. et al. RET proto-oncogene: a review and update of genotype–phenotype correlations in hereditary medullary thyroid cancer and associated endocrine tumors. Thyroid 15, 531–544 (2005).
Moses, W., Weng, J. & Kebebew, E. Prevalence, clinicopathologic features, and somatic genetic mutation profile in familial versus sporadic nonmedullary thyroid cancer. Thyroid 21, 367–371 (2011).
Xing, M. The T1799A BRAF mutation is not a germline mutation in familial nonmedullary thyroid cancer. Clin. Endocrinol. 63, 263–266 (2005).
Cavaco, B. M. et al. Familial non-medullary thyroid carcinoma (FNMTC): analysis of fPTC/PRN, NMTC1, MNG1 and TCO susceptibility loci and identification of somatic BRAF and RAS mutations. Endocr. Relat. Cancer 15, 207–215 (2008).
Nosé, V. Familial non-medullary thyroid carcinoma: an update. Endocr. Pathol. 19, 226–240 (2008).
El Lakis, M. et al. Do patients with familial nonmedullary thyroid cancer present with more aggressive disease? Implications for initial surgical treatment. Surgery 165, 50–57 (2019).
Peiling Yang, S. & Ngeow, J. Familial non-medullary thyroid cancer: unraveling the genetic maze. Endocr. Relat. Cancer 23, R577–R595 (2016).
Robinson, D. W. & Orr, T. G. Carcinoma of the thyroid and other diseases of the thyroid in identical twins. AMA Arch. Surg. 70, 923–928 (1955).
McKay, J. D. et al. At least three genes account for familial papillary thyroid carcinoma: TCO and MNG1 excluded as susceptibility loci from a large Tasmanian family. Eur. J. Endocrinol. 141, 122–125 (1999).
Tsilchorozidou, T. et al. A Greek family with a follicular variant of familial papillary thyroid carcinoma: TCO, MNG1, fPTC/PRN, and NMTC1 excluded as susceptibility loci. Thyroid 15, 1349–1354 (2005).
Loh, K. C. Familial nonmedullary thyroid carcinoma: a meta-review of case series. Thyroid 7, 107–113 (1997).
Fallah, M. et al. Risk of thyroid cancer in first-degree relatives of patients with non-medullary thyroid cancer by histology type and age at diagnosis: a joint study from five Nordic countries. J. Med. Genet. 50, 373–382 (2013).
Roti, E., degli Uberti, E. C., Bondanelli, M. & Braverman, L. E. Thyroid papillary microcarcinoma: a descriptive and meta-analysis study. Eur. J. Endocrinol. 159, 659–673 (2008).
Fukunaga, F. H. & Yatani, R. Geographic pathology of occult thyroid carcinomas. Cancer 36, 1095–1099 (1975).
Sampson, R. J., Woolner, L. B., Bahn, R. C. & Kurland, L. T. Occult thyroid carcinoma in Olmsted county, Minnesota: prevalence at autopsy compared with that in Hiroshima and Nagasaki, Japan. Cancer 34, 2072–2076 (1974).
Harach, H. R., Franssila, K. O. & Wasenius, V.-M. Occult papillary carcinoma of the thyroid. A ‘normal’ finding in Finland. A systematic autopsy study. Cancer 56, 531–538 (1985).
Lim, D.-J. et al. Clinical, histopathological, and molecular characteristics of papillary thyroid microcarcinoma. Thyroid 17, 883–888 (2007).
Yamashita, H. et al. Extracapsular invasion of lymph node metastasis. A good indicator of disease recurrence and poor prognosis in patients with thyroid microcarcinoma. Cancer 86, 842–849 (1999).
Pelizzo, M. R. et al. High prevalence of occult papillary thyroid carcinoma in a surgical series for benign thyroid disease. Tumori 76, 255–257 (1990).
Pelizzo, M. R. et al. Papillary thyroid microcarcinoma. Long-term outcome in 587 cases compared with published data. Minerva Chir. 62, 315–325 (2007).
Shin, H.-R. et al. Nationwide cancer incidence in Korea, 1999–2001: first result using the national cancer incidence database. Cancer Res. Treat. 37, 325–331 (2005).
Leenhardt, L., Grosclaude, P. & Chérié-Challine, L. Increased incidence of thyroid carcinoma in France: a true epidemic or thyroid nodule management effects? report from the French thyroid cancer committee. Thyroid 14, 1056–1060 (2004).
Leenhardt, L., Grosclaude, P., Chérié-Challine, L. & Others. Guidelines for a national epidemiological surveillance system of thyroid cancer in France. Paris: Public Health Agency 1–211 (2003).
US Preventive Services Task Force. et al. Screening for thyroid cancer: US preventive services task force recommendation statement. JAMA 317, 1882–1887 (2017).
Harris, R. Don’t screen for thyroid cancer, task force says. NPR https://www.npr.org/sections/health-shots/2017/05/09/527569291/dont-screen-for-thyroid-cancer-task-force-says?t=1568120751891 (2017).
Kitahara, C. M. et al. Anthropometric factors and thyroid cancer risk by histological subtype: pooled analysis of 22 prospective studies. Thyroid 26, 306–318 (2016). This pooled analysis of 22 prospective studies including over 2 million patients finds that adulthood obesity is associated with higher incidence of papillary, follicular, and anaplastic thyroid cancer.
Kitahara, C. M. & Sosa, J. A. The changing incidence of thyroid cancer. Nat. Rev. Endocrinol. 12, 646–653 (2016).
Ng, M. et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: a systematic analysis for the global burden of disease study 2013. Lancet 384, 766–781 (2014).
Bianchini, F., Kaaks, R. & Vainio, H. Overweight, obesity, and cancer risk. Lancet Oncol. 3, 565–574 (2002).
Han, J. M. et al. Obesity is a risk factor for thyroid cancer in a large, ultrasonographically screened population. Eur. J. Endocrinol. 168, 879–886 (2013).
Rinaldi, S. et al. Body size and risk of differentiated thyroid carcinomas: findings from the EPIC study. Int. J. Cancer 131, E1004–E1014 (2012).
Engeland, A., Tretli, S., Akslen, L. A. & Bjørge, T. Body size and thyroid cancer in two million Norwegian men and women. Br. J. Cancer 95, 366–370 (2006).
Trésallet, C. et al. The incidence of papillary thyroid carcinoma and outcomes in operative patients according to their body mass indices. Surgery 156, 1145–1152 (2014).
Meinhold, C. L. et al. Nonradiation risk factors for thyroid cancer in the US Radiologic Technologists Study. Am. J. Epidemiol. 171, 242–252 (2010).
Rossing, M. A., Cushing, K. L., Voigt, L. F., Wicklund, K. G. & Daling, J. R. Risk of papillary thyroid cancer in women in relation to smoking and alcohol consumption. Epidemiology 11, 49–54 (2000).
Kitahara, C. M. et al. Cigarette smoking, alcohol intake, and thyroid cancer risk: a pooled analysis of five prospective studies in the United States. Cancer Causes Control 23, 1615–1624 (2012). A pooled analysis of five prospective studies conducted in the USA shows that active smoking and alcohol use are associated with a reduced risk of thyroid cancer.
Mack, W. J. et al. A pooled analysis of case–control studies of thyroid cancer: cigarette smoking and consumption of alcohol, coffee, and tea. Cancer Causes Control 14, 773–785 (2003).
Henderson, B. E., Ross, R. K., Pike, M. C. & Casagrande, J. T. Endogenous hormones as a major factor in human cancer. Cancer Res. 42, 3232–3239 (1982).
Williams, E. D. TSH and thyroid cancer. Horm. Metab. Res. Suppl. 23, 72–75 (1990).
Soldin, O. P., Goughenour, B. E., Gilbert, S. Z., Landy, H. J. & Soldin, S. J. Thyroid hormone levels associated with active and passive cigarette smoking. Thyroid 19, 817–823 (2009).
Belin, R. M., Astor, B. C., Powe, N. R. & Ladenson, P. W. Smoke exposure is associated with a lower prevalence of serum thyroid autoantibodies and thyrotropin concentration elevation and a higher prevalence of mild thyrotropin concentration suppression in the third National Health and Nutrition Examination Survey (NHANES III). J. Clin. Endocrinol. Metab. 89, 6077–6086 (2004).
Piirtola, M. et al. Association of current and former smoking with body mass index: a study of smoking discordant twin pairs from 21 twin cohorts. PLOS One 13, e0200140 (2018).
Rasmussen, F., Tynelius, P. & Kark, M. Importance of smoking habits for longitudinal and age-matched changes in body mass index: a cohort study of Swedish men and women. Prev. Med. 37, 1–9 (2003).
IARC Working Group on the Evaluation of Carcinogenic Risks to Humans & International Agency for Research on Cancer. Betel-quid and Areca-nut Chewing and Some Areca-nut-derived Nitrosamines. (IARC, 2004).
Dasgupta, R. et al. Ultrastructural and hormonal modulations of the thyroid gland following arecoline treatment in albino mice. Mol. Cell. Endocrinol. 319, 1–7 (2010).
Bouvard, V. et al. Carcinogenicity of consumption of red and processed meat. Lancet Oncol. 16, 1599–1600 (2015).
Wie, G.-A. et al. Red meat consumption is associated with an increased overall cancer risk: a prospective cohort study in Korea. Br. J. Nutr. 112, 238–247 (2014).
Refetoff, S. et al. Continuing occurrence of thyroid carcinoma after irradiation to the neck in infancy and childhood. N. Engl. J. Med. 292, 171–175 (1975).
Heidenreich, W. F. et al. Time trends of thyroid cancer incidence in Belarus after the Chernobyl accident. Radiat. Res. 151, 617–625 (1999).
Mathews, J. D. et al. Cancer risk in 680,000 people exposed to computed tomography scans in childhood or adolescence: data linkage study of 11 million Australians. BMJ 346, f2360 (2013).
Schonfeld, S. J., Lee, C. & Berrington de González, A. Medical exposure to radiation and thyroid cancer. Clin. Oncol. 23, 244–250 (2011).
Lubin, J. H. et al. Thyroid cancer following childhood low-dose radiation exposure: a pooled analysis of nine cohorts. J. Clin. Endocrinol. Metab. 102, 2575–2583 (2017).
Brenner, D. J. & Hall, E. J. Computed tomography — an increasing source of radiation exposure. N. Engl. J. Med. 357, 2277–2284 (2007).
United Nations Scientific Committee on the Effects of Atomic Radiation. Sources and Effects of Ionizing Radiation. (United Nations Publications, 2000).
Konoplya, E. F. & Rolevich, I. V. The Chernobyl catastrophe consequences in the Republic of Belarus. Natl. Rep. https://inis.iaea.org/search/search.aspx?orig_q=RN:27042492 (1996).
Mahoney, M. C. et al. Thyroid cancer incidence trends in Belarus: examining the impact of Chernobyl. Int. J. Epidemiol. 33, 1025–1033 (2004).
Goldman, M. The Russian radiation legacy: its integrated impact and lessons. Environ. Health Perspect. 105, 1385–1391 (1997).
Stsjazhko, V. A., Tsyb, A. F., Tronko, N. D., Souchkevitch, G. & Baverstock, K. F. Childhood thyroid cancer since accident at Chernobyl. BMJ 310, 801–801 (1995).
Buglova, E. E., Kenigsberg, J. E. & Sergeeva, N. V. Cancer risk estimation in Belarussian children due to thyroid irradiation as a consequence of the Chernobyl nuclear accident. Health Phys. 71, 45–49 (1996).
Furukawa, K. et al. Long-term trend of thyroid cancer risk among Japanese atomic-bomb survivors: 60 years after exposure. Int. J. Cancer 132, 1222–1226 (2013). In this analysis of the Life Span Study cohort of Japanese atomic bomb survivors, childhood exposure to radiation continues to be associated with higher thyroid cancer risk, even decades after exposure.
Mughal, B. B. & Demeneix, B. A. Endocrine disruptors: flame retardants and increased risk of thyroid cancer. Nat. Rev. Endocrinol. 13, 627–628 (2017).
Hoffman, K., Sosa, J. A. & Stapleton, H. M. Do flame retardant chemicals increase the risk for thyroid dysregulation and cancer? Curr. Opin. Oncol. 29, 7–13 (2017).
Meeker, J. D., Johnson, P. I., Camann, D. & Hauser, R. Polybrominated diphenyl ether (PBDE) concentrations in house dust are related to hormone levels in men. Sci. Total Environ. 407, 3425–3429 (2009).
Slagle, M. Greater exposure to flame retardants might be associated with thyroid cancer — DCRI. Available at: https://dcri.org/flame-retardants-thyroid-cancer/ (2017). (Accessed: August 2019).
Betts, K. S. Unwelcome guest: PBDEs in indoor dust. Environ. Health Perspect. 116, A202–A208 (2008).
National Toxicology Program. NTP toxicology and carcinogenesis studies of decabromodiphenyl oxide (CAS No. 1163-19-5) in F344/N rats and B6C3F1 mice (feed studies). Natl Toxicol. Program Tech. Rep. Ser. 309, 1–242 (1986).
Turyk, M. E. et al. Hormone disruption by PBDEs in adult male sport fish consumers. Environ. Health Perspect. 116, 1635–1641 (2008).
Johnson, P. I., Stapleton, H. M., Mukherjee, B., Hauser, R. & Meeker, J. D. Associations between brominated flame retardants in house dust and hormone levels in men. Sci. Total Environ. 445–446, 177–184 (2013).
Chevrier, J. et al. Polybrominated diphenyl ether (PBDE) flame retardants and thyroid hormone during pregnancy. Environ. Health Perspect. 118, 1444–1449 (2010).
Stapleton, H. M., Eagle, S., Anthopolos, R., Wolkin, A. & Miranda, M. L. Associations between polybrominated diphenyl ether (PBDE) flame retardants, phenolic metabolites, and thyroid hormones during pregnancy. Environ. Health Perspect. 119, 1454–1459 (2011).
Aschebrook-Kilfoy, B. et al. Polybrominated diphenyl ethers and thyroid cancer risk in the prostate, colorectal, lung, and ovarian cancer screening trial cohort. Am. J. Epidemiol. 181, 883–888 (2015). This nested, case-control study in the Prostate, Lung, Colorectal, and Ovarian Cancer screening trial examines serum levels of PBDEs and finds no association between PBDEs and thyroid cancer.
Hoffman, K. et al. Exposure to flame retardant chemicals and occurrence and severity of papillary thyroid cancer: a case–control study. Environ. Int. 107, 235–242 (2017).
Woodruff, S. L., Arowolo, O. A., Akute, O. O., Afolabi, A. O. & Nwariaku, F. Global variation in the pattern of differentiated thyroid cancer. Am. J. Surg. 200, 462–466 (2010).
Wiltshire, J. J., Drake, T. M., Uttley, L. & Balasubramanian, S. P. Systematic review of trends in the incidence rates of thyroid cancer. Thyroid 26, 1541–1552 (2016).
Regional Committee for Africa. Iodine deficiency disorders in the WHO African region: situation analysis and way forward. https://apps.who.int/iris/handle/10665/19986 (2008).
Kalk, W. J., Sitas, F. & Patterson, A. C. Thyroid cancer in South Africa — an indicator of regional iodine deficiency. S. Afr. Med. J. 87, 735–738 (1997).
Kung, T. M., Ng, W. L. & Gibson, J. B. Volcanoes and carcinoma of the thyroid: a possible association. Arch. Environ. Health 36, 265–267 (1981).
Paksoy, N., Montaville, B. & McCarthy, S. W. Cancer occurrence in Vanuatu in the South Pacific, 1980–86. Asia Pac. J. Public Health 3, 231–236 (1989).
Truong, T. et al. Time trends and geographic variations for thyroid cancer in New Caledonia, a very high incidence area (1985–1999). Eur. J. Cancer Prev. 16, 62 (2007).
Pellegriti, G. et al. Papillary thyroid cancer incidence in the volcanic area of Sicily. J. Natl Cancer Inst. 101, 1575–1583 (2009).
Malandrino, P. et al. Descriptive epidemiology of human thyroid cancer: experience from a regional registry and the ‘volcanic factor’. Front. Endocrinol. 4, 65 (2013).
Vigneri, R., Malandrino, P., Gianì, F., Russo, M. & Vigneri, P. Heavy metals in the volcanic environment and thyroid cancer. Mol. Cell. Endocrinol. 457, 73–80 (2017).
Tchounwou, P. B., Yedjou, C. G., Patlolla, A. K. & Sutton, D. J. Heavy metal toxicity and the environment. Experientia Suppl. 101, 133–164 (2012).
Ferlay J., et al Global cancer observatory: cancer today. Available at: https://gco.iarc.fr/. (Accessed: August 2019).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Endocrinology thanks J. Brito, D. Carneiro-Pla and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Papillary thyroid cancer
-
(PTC). The most common type of thyroid cancer, which tends to grow slowly and can spread to lymph nodes in the neck.
- Thyroid nodules
-
Abnormal growths of thyroid cells that can form a mass within the thyroid.
- Betel quid
-
A mix of betel nut, tobacco and spices, which is used like chewing tobacco.
- Fine-needle aspiration
-
A biopsy procedure in which a thin needle is used collect a sample of tissue for evaluation.
- Occult thyroid cancers
-
Thyroid cancers that are not detectable by contemporary clinical methods.
- Microcarcinomas
-
Tumours less than or equal to 1 cm in size.
- Anaplastic thyroid cancer
-
A rare but aggressive type of thyroid cancer.
Rights and permissions
About this article
Cite this article
Kim, J., Gosnell, J.E. & Roman, S.A. Geographic influences in the global rise of thyroid cancer. Nat Rev Endocrinol 16, 17–29 (2020). https://doi.org/10.1038/s41574-019-0263-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-019-0263-x
This article is cited by
-
Effects of altitude on thyroid disorders according to Chinese three-rung, ladder-like topography: national cross-sectional study
BMC Public Health (2024)
-
Genetic alterations and allele frequency of BRAF V600E and TERT mutation in papillary thyroid carcinoma with intermediate-to-high recurrence risk: a retrospective study
Clinical and Experimental Medicine (2024)
-
Tnseg: adversarial networks with multi-scale joint loss for thyroid nodule segmentation
The Journal of Supercomputing (2024)
-
A Cuproptosis-Related gene Signature as a Prognostic Biomarker in Thyroid Cancer Based on Transcriptomics
Biochemical Genetics (2024)
-
MiR-138-5p Inhibits Thyroid Cancer Cell Growth and Stemness by Targeting TRPC5/Wnt/β-Catenin Pathway
Molecular Biotechnology (2024)